Regional Anesthesia in Neonates: Benefits and Safety
The Role of Regional Anesthesia in Neonatal Care: A Review
Balverde Camiño E.1; Terra Laurent F.1; Cristiani Halty F.1
- Anesthesiology Academic Unit -University of the Republic – Pereira Rossell Hospital, Montevideo, Uruguay
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Balverde, EC., Terra, FL., et al., 2024. The Role of Regional Anesthesia in Neonatal Care: A Review. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5927
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5927
ISSN 2375-1924
Abstract
Effective pain management in neonates is critical, as inadequate treatment can result in both immediate complications and long-term cognitive and behavioral issues. Neonates, particularly preterm infants, have heightened sensitivity to pain, which necessitates the use of specialized pain management techniques. Regional anesthesia (RA) offers significant benefits in this patient population, including reducing the need for opioids and their associated risks, minimizing the stress response, and improving postoperative outcomes. Despite its proven efficacy in pediatric and adult populations, there is limited published evidence regarding RA’s use in neonates. This article reviews the fundamental science, clinical applications, and safety considerations of regional anesthesia in neonates, focusing on four modalities: neuraxial blocks, peripheral nerve blocks, fascial plane blocks, and analgesic catheters. While RA has been shown to provide superior pain control and reduce opioid consumption, safety is paramount, especially considering the unique physiological characteristics of neonates.
Keywords
Regional anesthesia, neonatal care, pain management, neuraxial blocks, peripheral nerve blocks, fascial plane blocks, analgesic catheters.
Introduction
Clinical studies have shown that inadequate pain management results in both immediate complications and long-term physiological, behavioral, and cognitive consequences, such as disrupted pain perception, attention deficit disorder, reduced visual-perceptual skills or visual-motor coordination, and impaired executive functions. The American Academy of Pediatrics recognizes that neonates experience pain and that pharmacologic and nonpharmacological interventions are necessary to relieve pain and associated stress. 1,2,3
The use of regional anesthesia techniques has shown to attenuate the stress response, reduce opioid consumption and its associated adverse effects, and provide better pain control. Due to the unique characteristics of newborns, there is less published evidence on the use of regional anesthesia in this patient group.
The aim of this article is to examine the fundamental science and clinical uses of regional anesthesia in neonates, focusing on:
- The importance of pain management in neonates
- The pathophysiology of pain in neonates and infants
- Benefits and risks of regional anesthesia
It is essential for practicing neonatologists to comprehend the available options for regional anesthesia. Four modalities of regional analgesia will be examined:
- Neuraxial blocks: spinal and caudal anesthesia and analgesia
- Peripheral nerve blocks
- Fascial plane blocks
- Analgesia catheters
Importance of pain control in neonates
Neonates are more sensitive to pain than older infants, children, and adults, and this hypersensitivity is exacerbated in preterm neonates. Multiple lines of evidence suggest that repeated and prolonged pain exposure alters their subsequent pain processing, long-term development, and behavior. 4 Such as altered pain perception, cognitive and behavioral issues including attention-deficit/hyperactivity disorder, impaired visual-perceptual ability or visual-motor integration, and diminished executive functioning. 5-6
With enhancements in magnetic resonance imaging techniques, several studies tracking infants subjected to pain have revealed significant and lasting neuroanatomical alterations when assessed during their school years. 7 A study monitoring preterm infants until the age of 8 found that increased exposure to neonatal pain correlated with a thinner cortex across various brain regions, notably in the frontal and parietal lobes. 8 This research group also noted diminished volumes in the limbic system and basal ganglia at eight years of age among those who experienced more pain.
PHYSIOLOGICAL AND ANATOMICAL CONSIDERATIONS OF THE NEWBORN
Given that the pathophysiology of pain is a complex chapter, we will discuss the neonatal and developmental considerations. The nervous system is not fully developed at birth. Some researchers argue that pain perception may begin as early as 12 to 20 weeks of gestation, based on the development of the subplate and observed behavioral responses to pain in fetuses and extremely preterm neonates. 9-10
While transmission through peripheral nerves may be slower due to incomplete myelination in the early stages of life, the majority of peripheral nociceptive afferents, in both children and adults, occur via unmyelinated C fibers. 11
Like all systems, pain pathways are immature at birth and early life stages. It is suggested that the ascending nociceptive pathways from peripheral sensory neurons to the thalamus develop between 20 and 24 weeks of gestation, while the descending inhibitory pathways continue to mature postnatally and during infancy. This favors the conduction of nociceptive afferents. 12
Additionally, the neurons of the dorsal horn of the spinal cord (second-order neurons in pain pathways) have larger nociceptive fields, and their thresholds are lower at birth than in older children and adults. 13 14 15 Central sensitization (with wind-up phenomena) and peripheral sensitization in children from birth, even with a lower threshold for new stimuli, have been demonstrated. 11 16
There are also anatomical differences between neonates and pediatric patients. The spinal canal grows more than the nervous structures during the embryonic and fetal periods, with the conus medullaris at L3 at birth and moving to L1-L2 by one year. Cerebrospinal fluid volume is higher in children under 15 kg (4 ml/kg) than those over 15 kg (2 ml/kg) and has a faster turnover than in adults. The skin-to-dura mater distance in neonates is 6-10 mm, increasing with age. The ligamentum flavum is less dense in children, raising the risk of accidental dura mater puncture during epidurals. 17-18
Incomplete ossification at birth allows for ultrasound visualization of intra-axial nervous structures. Myelination starts at the cervical level during the fetal period, progressing until age 12. In infants, fibers are smaller, the myelin sheath is thinner, and internodal distances are shorter, necessitating lower local anesthetic concentrations for a block compared to adults. 19
BENEFITS OF REGIONAL ANESTHESIA
Regional anesthesia has proven to be the most effective technique for the treatment of acute pain. 20 This allows for a reduction in the use of opioids, which is particularly important in neonates, preterm infants, and former preterm infants, who are especially sensitive to respiratory depression caused by opioids.
Regional anesthesia finds its primary application in perioperative settings. In postoperative scenarios, it significantly diminishes or eradicates the necessity for opioid analgesics. 21-22 In certain instances, spinal anesthesia can even avoid the requirement for general anesthesia, thereby eliminating exposure to inhalational and other anesthetic agents and the need for intubation. 17
In preterm infants, this approach can assuage concerns regarding postoperative apnea and respiratory complications while enhancing hemodynamic stability. 23 Regional analgesia reduces the need for mechanical ventilation in newborns and infants undergoing abdominal surgery or thoracotomy and reduces postoperative pulmonary complications. 23 24 25 26 Regional techniques reduce the hormonal response to surgical stress. Stress-related hormones (cortisol, ACTH, prolactin) and glucose levels are less elevated when epidural analgesia is used, returning quickly to baseline levels postoperatively. Additionally, local anesthetics stimulate the cellular immune response of T lymphocytes, which have an antitumor role. This is in contrast to opioids, which inhibit the immune response mainly through the inhibition of Natural Killer cells, which have a critical antitumor role. 27 28
Several studies have shown that peristalsis returns more quickly postoperatively when regional anesthesia is used. 29 Animal studies have observed that most agents used in general anesthesia and sedation (GABA agonists and NMDA antagonists) may cause neuronal apoptosis. This in vitro observed effect could have greater consequences during the development of the nervous system. Regional anesthesia as an adjuvant or substitute for general anesthesia is another potential benefit of these techniques. 30
PHARMACOLOGY OF LOCAL ANESTHETICS
Effective and safe drug administration in neonates should be based on an integrated understanding of the evolving physiological characteristics of the patient receiving the drug, along with the pharmacokinetics and pharmacodynamics of the drug. Growth and development throughout pediatric life involve simultaneous physiological events resulting in growth and maturation. 31
Children have peculiar and rapidly changing pharmacokinetic and pharmacodynamic characteristics with growth and development, influencing the efficacy, toxicity, and dosage regimen of medications. Changes in body composition represent substantial individual variation. 18
From a clinical pharmacology perspective, the consequence of such a rapidly changing system is extensive within and between-subject variability in drug disposition and effects. This maturation-related variability is further aggravated by interfering pathological processes (e.g., growth restriction, sepsis, associated cardiopathy, organ failure) or treatment modalities (e.g., co-medication, surgical intervention). Maturational pharmacokinetics considers maturational changes in drug absorption, distribution, metabolism, and elimination, while maturational pharmacodynamics considers changes in the concentration-effect profile, such as differences in receptor expression, function, or specific tissue/organ sensitivity (either more or less vulnerable). 31
PHARMACOLOGY OF LOCAL ANESTHETICS IN CHILDREN
Local anesthetics (LA) interrupt neural conduction by inhibiting the influx of sodium ions through channels or ionophores within neuronal membranes. The amino-amide types are the most commonly used local anesthetics, including lidocaine, bupivacaine, and ropivacaine. Amino-amides are metabolized in the liver by cytochrome P450, while amino-esters are metabolized by plasma cholinesterases. 31
There are several relevant differences in local anesthetic pharmacology between children and adults. Local anesthetic metabolism is affected by age, especially in premature infants and neonates, where the hepatic microsomal enzyme system is not fully developed. Protein binding of local anesthetics is decreased due to the low quantities of albumin and alpha-1-acid glycoprotein produced in the liver during early infancy, resulting in an increased plasma-free fraction of the drug and an increased risk of local anesthetic toxicity. The absorption rate is also essential, as a rapid rise in serum concentration is more likely to result in toxicity. Cardiac output and local blood flow in infants are relatively more significant than in adults, leading to faster systemic absorption of local anesthetics and higher peak plasma concentrations. 31
Anatomic differences between children and adults can also affect local anesthetic pharmacology. The fat within the epidural space is sparse and loose in infants, and the perineural sheath around nerve roots and bundles is more loosely attached to underlying structures in children than in adults. Consequently, injected local anesthetics appear to spread more in children and cover a greater innervation area. Additionally, the endoneurium is relatively loose in young children, allowing rapid exposure of the local anesthetic to the nerve, resulting in a faster onset of anesthesia than in adults. 31
When using any local anesthetic, the anesthesiologist must calculate the total milligram dose on a per-kilogram basis to avoid toxic blood levels by administering less than the recommended maximum doses, especially in neonates and small infants. Even with extreme caution and careful calculation of doses, local anesthetic toxicity can occur. Therefore, the practitioner must be familiar with basic resuscitation strategies and the use of intralipids to rescue from local anesthetic toxicity.
The choice of anesthetic depends on the desired onset of action and duration of the block. See Table 1.
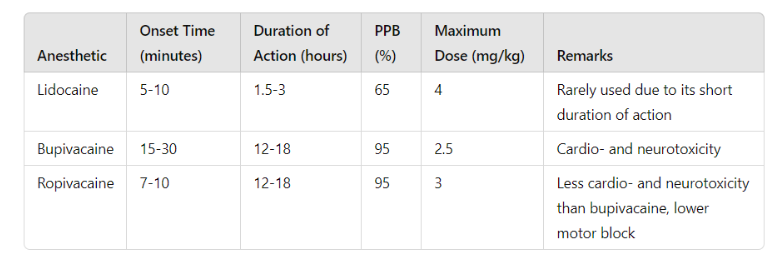
Note: PPB: plasma protein binding. The maximum dose should be halved in children under six months.
TOXICITY OF LOCAL ANESTHETICS
The toxic effects of these drugs occur at the central nervous system and cardiovascular levels. At low concentrations (within the toxicity range), LA crosses the blood-brain barrier and initially produces neurological symptoms such as confusion, perioral paresthesias, and excitation; at higher concentrations, they can cause tonic-clonic seizures, consciousness depression, and cardiorespiratory arrest. Cardiotoxicity appears at concentrations higher than those needed to produce neurotoxicity; however, neurological symptoms can be masked if the block is performed under general anesthesia. The cardiovascular effects of LA toxicity initially include vasodilation with hypotension, direct myocardial depression through action on myocardial sodium channels, progressive bradycardia, and arrest. At high concentrations, ventricular arrhythmias may occur. 32, 33, 34
The primary pharmacological factor involved in LA toxicity is the decrease in α1-acid glycoprotein and albumin. The concentration of these proteins is lower in neonates and infants, which is a risk factor for developing toxicity. Hence, doses must be adjusted in this population, as well as in other types of patients with hypoproteinemia. Acidosis reduces the affinity of these proteins for LA, increasing the free fraction in such cases. Additionally, the hepatic metabolism rate of amides is lower in newborns and infants. 32 The increased volume of distribution in neonates and infants also prolongs the half-life in these patients. The calculated dose should be reduced by 30-50% in patients under six months. Treatment should be immediate. The guidelines from the American Heart Association (AHA) Pediatric Advanced Life Support (PALS) and the American Society for Regional Anesthesia and Pain Medicine (ASRA) for resuscitation from local anesthetic-induced cardiac arrest should be applied:
- Stop administration
- Call for help
- Secure the airway, ensure 100% oxygenation
- Administer lipid emulsion (Intralipid 20%) at a dose of 1.5 mL/kg as a bolus
- Infuse lipid emulsion at 0.25 mL/kg/min
- If there is no return of normal cardiac function within five minutes, repeat the bolus with the same dose at five-minute intervals (maximum three boluses) and continue with an infusion at double the dose. 33
ADJUVANTS
One of the main challenges in regional anesthesia is the limited duration of the effect of LA, which lasts up to 12-18 hours with long-acting LAs such as bupivacaine and ropivacaine. Prolonging the effect can be achieved by placing catheters; however, this requires more equipment, training in their placement, and infrastructure for their safe use. 32 An alternative to achieving prolonged analgesia is to use adjuvant drugs with LAs in single-bolus blocks. 33 The potential benefits of using adjuvants include prolongation of the block effect, improved block quality, reduced LA concentration with less motor block, reduced anesthetic requirements, decreased systemic analgesic use, early discharge, and patient comfort at home in outpatient surgery. 33
In 2018, the European Society of Regional Anaesthesia and Pain Therapy (ESRA) and the American Society of Regional Anesthesia and Pain Medicine (ASRA) published recommendations for dosing local anesthetics and adjuvants in pediatrics. Since the pharmacokinetics of LA vary by the administration site, this guide specifies maximum doses for each block. 32
Preservative-free morphine and clonidine are the only adjuvants approved for spinal/epidural use. Regarding the other drugs mentioned, although there is evidence of their use in neural structures, they are not approved for administration in pediatric regional anesthesia (off-label).
NEURAXIAL ADJUVANTS
Clonidine 1-2 µg/kg and morphine 30 µg/kg can be used as adjuvants via spinal route. Given the limited data on neural toxicity, using the lowest possible dose (evidence level A2) is recommended. 33
Ketamine, although used neuraxially in the past, is not currently recommended due to its potential pro-apoptotic effect on the spinal cord (evidence level B3).
There is favorable evidence regarding the use of dexmedetomidine via the epidural route. However, data on its neural toxicity are limited, and low doses are suggested (evidence level A2).
Although dexamethasone has been used neuraxially in small pediatric studies, ASRA/ESRA does not recommend its use by this route. 33
ADJUVANTS IN PERIPHERAL REGIONAL ANESTHESIA
There is evidence for using clonidine and dexmedetomidine as adjuvants in peripheral nerve blocks. A meta-analysis including 283 patients observed a prolongation of block duration as well as a reduction in the use of rescue analgesics. 32 ASRA/ESRA guidelines recommend them with evidence level A1. 33
Ultrasound imaging
Ultrasound imaging for regional anesthesia offers significant benefits in infants and neonates because of their small size and the reduced distance between critical structures and targets. Although ultrasound is less commonly used for neuraxial blocks, its application for peripheral nerve block placement has surged from 0% to 90% over the past decade, with numerous new techniques being introduced for ultrasound guidance 36,37,38,39,40
Before the development of ultrasound-guided techniques, the only regional anesthesia procedures performed in neonates were caudal blocks and spinal anesthesia, typically performed through anatomical landmarks. Ultrasound technology has allowed for the safe advancement of these techniques, including fascial plane blocks and peripheral nerve blocks.
When adjusted for size, ultrasound images of soft tissues (muscles, fascia, subcutaneous tissue) in infants are similar to those in adults and older children. However, infant bones are primarily cartilaginous, and the lack of ossification results in distinct ultrasound images compared to older patients. For instance, when performing a sciatic nerve block, the infant femoral head lacks the characteristic bone echogenicity and may be challenging to identify.
The reduced bone density in infants also creates unique acoustic windows absent in older patients. For example, in neuraxial imaging, the spinal cord and conus medullaris are more easily visualized 41,42 The cartilaginous transverse processes may be incompletely visualized during real-time imaging for a peripheral nerve block (PNB). Still, the structures of the entire neuraxis can be more visible in infants due to the incomplete ossification of the overlying vertebrae. These imaging differences must be considered when performing regional anesthesia (RA) and identifying appropriate landmarks.
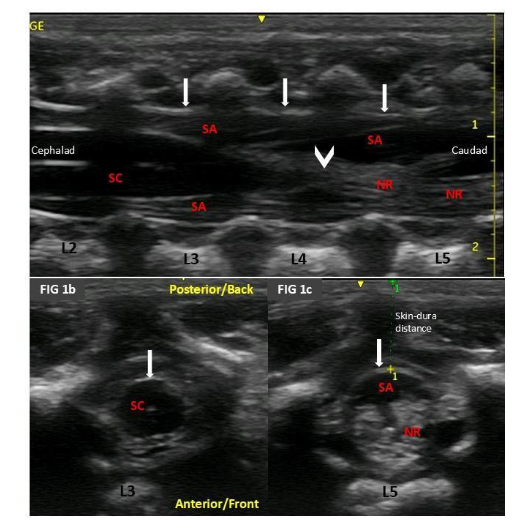
Efficacy
It has been demonstrated in adult and pediatric patients that the best strategy for pain treatment is regional anesthesia techniques. 45-50
Despite this, published evidence on newborns is scarce. Although many case series studies reported that RA has been effective in controlling pain in newborns, few controlled clinical studies compare the effectiveness of regional analgesia techniques with systemic analgesia. A recent study published in 2024 compared opioid consumption in 110 neonates undergoing palate surgery under general anesthesia (n=64) or general plus regional anesthesia (n=46). In this work, it was observed that the consumption of opiates in the postoperative period was lower in patients in whom regional analgesia was administered. 51 A study comparing restoration of gastrointestinal function in neonates receiving general anesthesia (n=25) versus combined spinal epidural anesthesia (n=25) found that restoration of bowel function occurred earlier in the anesthesia and epidural group. Regional analgesia, with less time to mobilize the intestine, for tube feeding and onset time of full enteral nutrition. 52
Multiple studies demonstrate the effectiveness of regional analgesia in pediatric and adult patients. The benefits of these techniques have already been analyzed, and some good-quality studies have been carried out in newborns. Therefore, regional analgesia techniques are a good choice for pain control in children. Furthermore, they are more effective than parenteral analgesia (based mainly on opiates) for pain control and restoration of gastrointestinal physiology.
The ERAS guidelines suggest avoiding caudal blocks; one of the blocks for which positive results are published in the neonatal population is the quadratus lumborum block. 53
Safety
Safety is the most crucial consideration when choosing a pain treatment strategy in neonates. Severe complications of regional anesthesia are systemic/cardiac toxicity, apnoea, visceral puncture, dural puncture, nerve injury, infection requiring antibiotic therapy, and hypotension. The frequency of these complications is low in the pediatric population.
Regional anesthesia is safe, with a low complication rate, so multicenter studies are necessary to evaluate its safety. The Pediatric Regional Anesthesia Network (PRAN) and Association des Anesthésistes Réanimateurs Pédiatriques d’Expression Française (ADARPEF) have carried out studies evaluating complications of regional anesthesia in pediatrics. 54 55 Although there are no published studies whose primary end-point is specifically the study of complications of RA in neonates, these multicenter publications include newborn patients.
The PRAN database included complications on 1082 neonates and 22,624 infants, and the ADARPEF study reports on 596 neonates and 3344 patients younger than six months of age. There were no recorded deaths or permanent injuries. The PRAN study did not observe serious complications in newborns, with an incidence of 0 in 10,000 cases (95% CI 0-46.3). On the other hand, in patients between 1 and 5 months, the highest incidence of local anesthetic toxicity was observed, 5.0 in 10,000 (95% CI 1.5–13.5). This can be explained by the greater vulnerability of the younger patients, as previously analyzed. The fact that no complications were recorded in newborns can be explained by the low number of patients younger than one month. 56
Neonates are at particular risk for local anesthetic toxicity because of hepatic immaturity, and the serum-free fraction of local anesthetic is proportionately greater in neonates and young infants than in older children and adults. As a consequence, the dose of local anesthetic should be reduced to minimize the risk. 56
Techniques under general anesthesia or deep sedation
Regional anesthesia in pediatrics is usually performed under general anesthesia or deep sedation because it is difficult to obtain their cooperation. This contrasts with adults, where regional anesthesia is typically performed while awake or with minimal sedation. Practically, these procedures are safer under general anesthesia than attempting them on a moving, upset, or uncooperative patient. And also, the cognitive inability of the child to relate to symptoms such as paresthesia or pain while the block is performed.
The Pediatric Regional Anesthesia Network (PRAN) database examined nearly 100,000 blocks, followed by focused analyses of specific block types. The data demonstrate that the safety level in pediatric regional anesthesia is comparable to that in adult practice and confirms the safety of placing blocks under general anesthesia in children.
Neuraxial blocks (spinal and caudal)
The first reports of this technique in children date back over 100 years. 57 Its use shows benefits in patients with risk factors for postoperative apnea after general anesthesia, such as former preterm infants with a corrected age of less than 60 weeks and those with respiratory pathology like bronchopulmonary dysplasia and anemia. 18
The GAS study was designed to evaluate the long-term cognitive effects of short general anesthesia (herniorrhaphy). One of the secondary objectives of this study was to assess the presence of postoperative apnea. For this, 722 neonates with a corrected age of less than 60 weeks were randomized to general anesthesia or spinal anesthesia. The spinal anesthesia group observed a reduction in early apneas (first 30 minutes postoperatively), the degree of postoperative desaturation, and the level of interventions for the treatment of apnea. 58 These observations align with findings from previous studies, which showed lower respiratory complications after spinal anesthesia in neonates. 59 60 Regarding the primary objective of the GAS study, no neurocognitive differences were observed at five years between patients undergoing general anesthesia and those undergoing spinal anesthesia. 20 61
Spinal anesthesia is indicated for infra umbilical surgeries in former preterm infants at risk of apnea. The main limitation of this technique is the short duration of anesthesia, approximately 60 minutes. 62
Contraindications include neural tube malformations, coagulation disorders, increased intracranial pressure, and infection at the puncture site.
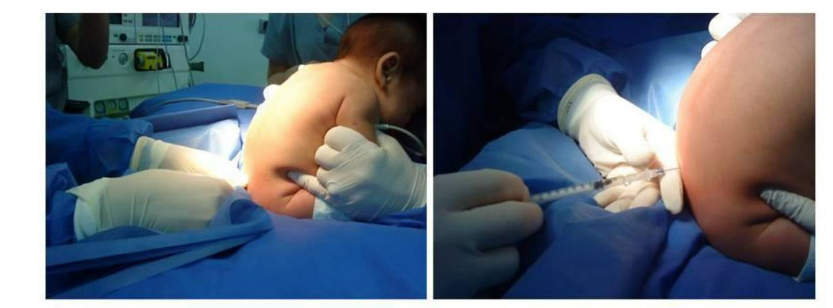
This technique is performed with the patient awake, in a sitting or lateral decubitus position (Figure 2). It is vital to maintain airway patency when sitting the patient. The puncture is made at the L4-L5 or L5-S1 level using a 25 G Quincke needle, 2.5 cm in length. After the cerebrospinal fluid is seen, the LA is administered. Hyperbaric or isobaric bupivacaine at a dose of 0.8-1 mg/kg is used without adjuvants. With isobaric bupivacaine, the duration of the block might be slightly longer. Clonidine can be added to prolong the block, but it also increases the risk of apnea and bradycardia. 18
After administering the LA, the patient is placed in the dorsal decubitus position. If a peripheral IV line has not been placed beforehand, it will be placed in the lower limbs after the block is established. The Trendelenburg position is avoided to prevent high levels of block. 20
Incomplete ossification in this type of patient allows for ultrasound visualization of neural structures. Therefore, it is preferable to perform an ultrasound examination before the procedure to determine the height of the conus medullaris, skin-to-dura distance, and any undiagnosed malformations in the area (Figure 1). 17
CAUDAL EPIDURAL
Caudal epidural anesthesia involves administering LA into the epidural space via the sacral hiatus. It can be used as an analgesic or anesthetic technique, with the latter being the most common indication. It can be performed with a single dose or through catheter placement.
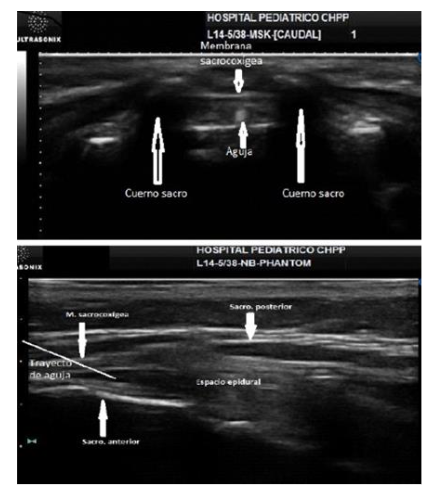
With the patient in dorsal or lateral decubitus, the sacral hiatus is palpated between the sacral cornua and caudal to the sacral crest. After puncturing with a 22 G needle at 45-75° to the skin, a “pop” is felt as the needle traverses the sacrococcygeal membrane. After crossing the membrane, the needle is redirected cephalad at a 30° angle to the skin, advancing a few millimeters to avoid puncturing the dura mater. After negative aspiration of CSF, the LA dose is administered. The distance between the skin and the sacrococcygeal membrane is usually 5 mm, and the distance between this membrane and the dura mater is approximately 10 mm in neonates. 63 The same technique is used for catheter placement. Before inserting the catheter, the necessary insertion depth is measured based on the required level. 18
This block is performed under direct ultrasound guidance (Figure 4). The linear transducer is placed in a short-axis view, where the “frog sign” can be seen, with the frog’s eyes being the sacral cornua. Between them, the sacrococcygeal membrane is visible. Anterior to this, the epidural space is visualized, bordered anteriorly by the anterior wall of the spinal canal. The needle can be inserted out of plane with this view, seen as an echogenic dot. When the in-plane technique is performed, once the sacral hiatus is located, the transducer is rotated to the longitudinal axis, where the anterior and posterior walls of the epidural space and the needle entering the space can be seen. 63
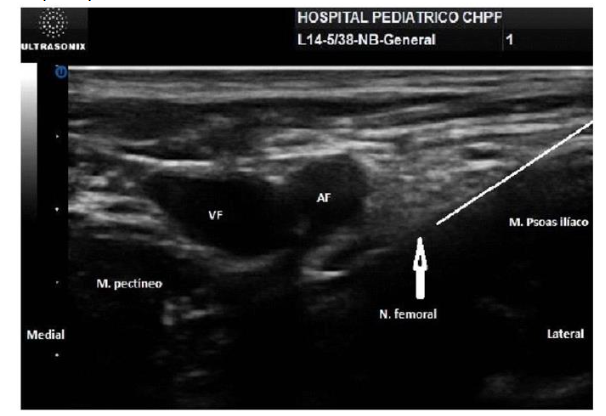
Peripheral nerve blocks
A nerve block is the injection of numbing medication (local anesthetic) near specific nerves to decrease the transmission of pain from certain dermatomes. In pediatrics, these blocks are usually performed under general anesthesia, and their safety under these conditions has been demonstrated in several studies. 55 65 66 67 The technique for the block should be performed aseptically.
A high-frequency ultrasound probe, typically 2.5-5 cm in size, is recommended in neonates, although a low-frequency (curvilinear) probe should be available for situations requiring greater depth (e.g., lumbar plexus block) or in adolescents.
Fascial blocks
Fascial plane blocks (FPBs) are regional anesthesia techniques in which the space (“plane”) between two discrete fascial layers is the target of needle insertion and injection. Compared with a single injection regional block, continuous catheter techniques allow the following:
- Prolonged duration of analgesia
- Reduced opioid requirement and opioid-associated adverse effects
- Shorter hospital stay 68
A PNC can be left in situ for up to 7 days; however, each additional day beyond the fourth day increases the risk of catheter-related infection. 13 When deciding upon the duration of catheter use, the analgesic benefits should be weighed against the risk of infection. Analgesia is primarily achieved by local anesthetic spread to nerves traveling within this plane and adjacent tissues. These blocks are mainly used as an analgesic method, and reducing the concentration of local anesthetics can increase volume. This is particularly important in fascial plane blocks such as the quadratus lumborum and erector spinae blocks. The recommended dose for single-dose peripheral blocks is typically 0.2-0.5 ml/kg of 0.2% ropivacaine or 0.25% bupivacaine. 0.1% ropivacaine or 0.125% bupivacaine is recommended for infusion at 0.1-0.3 ml/kg/h. 69
Catheters
This technique allows us to maintain the level of analgesia for a longer period. The main risks are infection and local anesthetic toxicity due to its accumulation resulting from reduced metabolism and hypoproteinemia. It can be placed at the caudal, peripheral, and fascial levels.
For catheter placement, it is recommended to administer the local anesthetic through the needle to expand the space where the catheter will be placed and insert it 2-3 cm into the space. To prevent leakage of local anesthetic around the catheter and displacement, surgical glue should be used at the insertion site. Por último pondría la figura de la gastrosquisis que presentamos en el congreso con el catéter, y en la nota de la figura un renglón contando algo del paciente, ej: RN con gastrosquisis con altos requerimientos de opiáceos que disminuyeron luego de la colocación de un catéter.
Conclusion
This review provides information on neonatal pain and regional anesthesia, focusing on the importance of pain management in neonates, the pathophysiology of pain in neonates and infants, and the benefits and risks of regional anesthesia.
Neonates exhibit heightened sensitivity to pain compared to older infants, children, and adults, with this hypersensitivity being particularly pronounced in preterm neonates. Evidence suggests that repeated and prolonged pain exposure in this population can significantly alter subsequent pain processing, impacting long-term development and behavior.
It is crucial for practicing neonatologists to understand the available options for regional anesthesia. Four modalities of regional analgesia are examined: neuraxial blocks (spinal and caudal anesthesia and analgesia), peripheral nerve blocks, fascial plane blocks, and analgesia catheters.
Regional anesthesia has proven to be the most effective technique for the treatment of acute pain, particularly in neonates, preterm infants, and former preterm infants who are highly sensitive to respiratory depression caused by opioids. By reducing opioid use, regional anesthesia offers significant benefits in perioperative settings, especially in postoperative scenarios where it can diminish or eradicate the necessity for opioid analgesics. In some cases, spinal anesthesia can even replace the need for general anesthesia, eliminating exposure to inhalational agents and the need for intubation, which is particularly beneficial in preterm infants by reducing the risk of postoperative apnea and respiratory complications while enhancing hemodynamic stability.
References
- Prevention and management of pain and stress in the neonate. American Academy of Pediatrics. Committee on Fetus and Newborn. Committee on Drugs. Section on Anesthesiology. Section on Surgery. Canadian Paediatric Society. Fetus and Newborn Committee. Pediatrics. 2000;105(2):454-461.
- Grunau RE, Holsti L, Peters JW. Long-term consequences of pain in human neonates. Semin Fetal Neonatal Med. 2006;11(4):268-275. doi:10.1016/j.siny.2006.02.007
- Anand KJS, Papanicolaou AC, Palmer FB. Repetitive neonatal pain and neurocognitive abilities in ex-preterm children. Pain. 2013;154(10):1899-1901. doi:10.1016/j.pain.2013.06.027
- Simons SH, van Dijk M, Anand KS, Roofthooft D, van Lingen RA, Tibboel D. Do we still hurt newborn babies? A prospective study of procedural pain and analgesia in neonates. Arch Pediatr Adolesc Med. 2003;157(11):1058-1064. doi:10.1001/archpedi.157.11.1058
- Selvanathan T, Zaki P, McLean MA, et al. Early-life exposure to analgesia and 18-month neurodevelopmental outcomes in very preterm infants. Pediatr Res. 2023;94(2):738-746. doi:10.1038/s41390-023-02536-y
- Ferguson SA, Ward WL, Paule MG, Hall RW, Anand KJ. A pilot study of preemptive morphine analgesia in preterm neonates: effects on head circumference, social behavior, and response latencies in early childhood. Neurotoxicol Teratol. 2012;34(1):47-55. doi:10.1016/j.ntt.2011.10.008
- McPherson C, Miller SP, El-Dib M, Massaro AN, Inder TE. The influence of pain, agitation, and their management on the immature brain. Pediatr Res. 2020;88(2):168-175. doi:10.1038/s41390-019-0744-6
- Reissland N, Francis B, Mason J. Can healthy fetuses show facial expressions of “pain” or “distress”?. PLoS One. 2013;8(6):e65530. Published 2013 Jun 5. doi:10.1371/journal.pone.0065530
- Sekulic S, Gebauer-Bukurov K, Cvijanovic M, et al. Appearance of fetal pain could be associated with maturation of the mesodiencephalic structures. J Pain Res. 2016;9:1031-1038. Published 2016 Nov 11. doi:10.2147/JPR.S117959
- Pattinson D, Fitzgerald M. The neurobiology of infant pain: development of excitatory and inhibitory neurotransmission in the spinal dorsal horn. Reg Anesth Pain Med. 2004;29(1):36-44. doi:10.1016/j.rapm.2003.10.018
- Lowery CL, Hardman MP, Manning N, Hall RW, Anand KJ, Clancy B. Neurodevelopmental changes of fetal pain [published correction appears in Semin Perinatol. 2009 Dec;33(6):410. Clancy, Barbara [added]]. Semin Perinatol. 2007;31(5):275-282. doi:10.1053/j.semperi.2007.07.004
- Torsney C, Fitzgerald M. Spinal dorsal horn cell receptive field size is increased in adult rats following neonatal hindpaw skin injury. J Physiol. 2003;550(Pt 1):255-261. doi:10.1113/jphysiol.2003.043661
- Ririe DG, Bremner LR, Fitzgerald M. Comparison of the immediate effects of surgical incision on dorsal horn neuronal receptive field size and responses during postnatal development. Anesthesiology. 2008;109(4):698-706. doi:10.1097/ALN.0b013e3181870a32
- Walker SM, Tochiki KK, Fitzgerald M. Hindpaw incision in early life increases the hyperalgesic response to repeat surgical injury: critical period and dependence on initial afferent activity. Pain. 2009;147(1-3):99-106. doi:10.1016/j.pain.2009.08.017
- Benzon HT, Strichartz GR, Gissen AJ, Shanks CA, Covino BG, Datta S. Developmental neurophysiology of mammalian peripheral nerves and age-related differential sensitivity to local anaesthetic. Br J Anaesth. 1988;61(6):754-760. doi:10.1093/bja/61.6.75417
- Cristiani F, Henderson R, Lauber C, Boretsky K. Success of bedside ultrasound to identify puncture site for spinal anesthesia in neonates and infants. Reg Anesth Pain Med. Published online July 3, 2019. doi:10.1136/rapm-2019-100672
- Coté CJ, Lerman J, Anderson BJ. A Practice of Anesthesia for Infants and Children. 6th ed. Elsevier; 2019.. 1192 p.
- Benzon HT, Strichartz GR, Gissen AJ, Shanks CA, Covino BG, Datta S. Developmental neurophysiology of mammalian peripheral nerves and age-related differential sensitivity to local anaesthetic. Br J Anaesth. 1988;61(6):754-760. doi:10.1093/bja/61.6.754
- Davidson AJ, Disma N, de Graaff JC, et al. Neurodevelopmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): an international multicentre, randomised controlled trial [published correction appears in Lancet. 2016 Jan 16;387(10015):228. doi: 10.1016/S0140-6736(16)00054-4]. Lancet. 2016;387(10015):239-250. doi:10.1016/S0140-6736(15)00608-X
- Mansfield SA, Woodroof J, Murphy AJ, Davidoff AM, Morgan KJ. Does epidural analgesia really enhance recovery in pediatric surgery patients?. Pediatr Surg Int. 2021;37(9):1201-1206. doi:10.1007/s00383-021-04897-z
- Franz AM, Martin LD, Liston DE, Latham GJ, Richards MJ, Low DK. In Pursuit of an Opioid-Free Pediatric Ambulatory Surgery Center: A Quality Improvement Initiative. Anesth Analg. 2021;132(3):788-797. doi:10.1213/ANE.0000000000004774
- Davidson AJ, Morton NS, Arnup SJ, et al. Apnea after Awake Regional and General Anesthesia in Infants: The General Anesthesia Compared to Spinal Anesthesia Study–Comparing Apnea and Neurodevelopmental Outcomes, a Randomized Controlled Trial. Anesthesiology. 2015;123(1):38-54. doi:10.1097/ALN.0000000000000709
- Bösenberg AT, Hadley GP, Wiersma R. Oesophageal atresia: caudo-thoracic epidural anaesthesia reduces the need for post-operative ventilatory support. Pediatric Surgery International. 1992;7(4):289-291. doi:https://doi.org/10.1007/bf00183983
- Ecoffey C, Dubousset AM, Samii K. Lumbar and thoracic epidural anesthesia for urologic and upper abdominal surgery in infants and children. Anesthesiology. 1986;65(1):87-90. doi:10.1097/00000542-198607000-00017

