Reprogramming Human Adipocytes to Enhance Glucose Utilization
Cellular Reprogramming of Human Primary Adipocytes into Brown Adipose Tissue-Like Cells Preferentially Utilize Glucose
Taylor SRT1, Cartwright KM1, Sorrells AM1, and Harding P A1,*
Department of Biology, Cell Molecular and Structural Biology Program, Miami University, Oxford, OH 45056, United States
OPEN ACCESS
PUBLISHED:25, July 2025
CITATION:SR, Taylor et al. Cellular Reprogramming of Human Primary Adipocytes into Brown Adipose Tissue-Like Cells Preferentially Utilize Glucose. Medical Research Archives, [S.l.], v. 13, n. 7, july 2025. ISSN 2375-1924. Available at: <https://esmed.org/MRA/mra/article/view/6709>. Date accessed: 14 sep. 2025.
COPYRIGHT:©2025 European society of medicine. This is an open- access article distributed under the term of creative commons attribution license,which permit unrestricted use, distribution, and reproduction in any meduim provided the original authors and source are credited.
DOI: https://doi.org/10.18103/mra.v13i7.6709.
ISSN 2375-1924
ABSTRACT
Brown adipose tissue has emerged as a potential therapeutic target for the treatment of type 2 diabetes and obesity. This aim of this study is to investigate how the expression of a disintegrin and metalloproteinase 12 (ADAM 12) expression alone in human primary adipocytes can drive the reprogramming of white adipocytes into metabolically active BAT-like cells through endogenous HB-EGF signaling. Such reprogramming could lay the groundwork for novel therapeutic strategies aimed at increasing energy expenditure, reducing fat mass, and addressing the metabolic conditions associated with obesity and type 2 diabetes.
Keywords: brown adipose tissue, adipocytes, glucose utilization, ADAM 12, metabolic disorders
Introduction
Obesity remains one of the most significant public health challenges, with rising prevalence rates where the increased availability of calorie-dense foods and a sedentary lifestyle have contributed to its rising prevalence. Defined by a body mass index (BMI) of 30 kg/m2 or higher, obesity is strongly associated with serious health conditions such as type 2 diabetes, cardiovascular diseases, and certain cancers. This growing epidemic not only impacts individual health but also places a tremendous burden on healthcare systems worldwide, resulting in escalating healthcare costs.
This study investigates the role of ADAM 12 in the reprogramming of human primary adipocytes into metabolically active BAT-like cells, independent of HB-EGF co-expression, and aims to elucidate the underlying mechanisms that drive this process.
Materials and Methods
Human primary adipocytes used in this study was generated using the AdAM12 adenovirus (Agilent Technologies, Santa Clara, CA). The AdAM12 expression vector (2.1 kb) was cloned into the pShuttle-1/Ad5-NGFP1.1 plasmid and transfected into HEK 293 cells, and resulted in non-infectious adenovirus.
qPCR IN HUMAN PRIMARY PREADIPOCYTES
For quantitative PCR (qPCR), total RNA from AdAM 12S and Ad-MOCK infected cells was reverse-transcribed into cDNA using a high-capacity cDNA reverse transcription kit (Applied Biosystems). qPCR was conducted using a SYBR Green RT-PCR Kit (Quantabio) on a CFX Connect Real-Time PCR Detection System (BioRad). The relative expression of brown adipose tissue (BAT) marker genes PRDM16, PGC-1α, UCP-1, and GAPDH was analyzed.
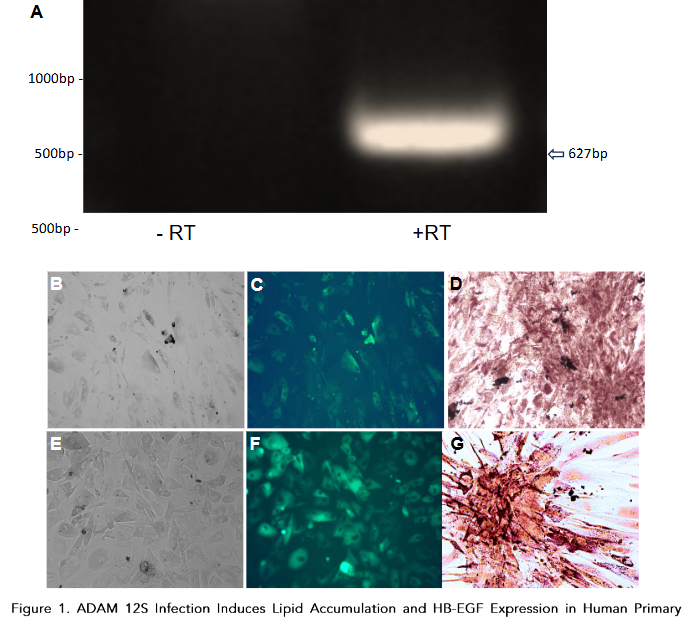
Table 1. Human primer sequences for all PCR and qPCR analyses.
| Gene Name | Primer Sequence for all PCR and qPCR |
|---|---|
| PRDM16 | GGAAGTGCCTGACCTGAC |
| UCP-1 | CTGTTCCATCAGCCTGAC |
| β-actin | TCCTCCTGAGCGCAAGT |
| GAPDH | CCCTGTTGCTGTAGCCAA |
| PPARγ | CCAGATGTTGCCCTGCTG |
| HB-EGF | ACGATGTCGCCCTGCGC |
Table 2. Differential Gene Expression in Human Primary Preadipocytes.
| Gene Name | Fold Change | P-value |
|---|---|---|
| PRDM16 | -2.40 | 0.0008 |
| UCP-1 | -2.31 | 0.0010 |
| PGC-1α | -2.10 | 0.0018 |
Discussion
According to the World Health Organization, 1 in 8 people in the world were living with obesity in 2015. The lipid accumulation is a characteristic feature of brown adipocytes, which store fuel for thermogenesis in the form of multilocular triglyceride droplets. Oil Red O staining confirmed the presence of lipids in ADAM 12S-infected cells, while mock-infected cells showed no such accumulation, consistent with previous findings in transgenic mice expressing ADAM 12S.
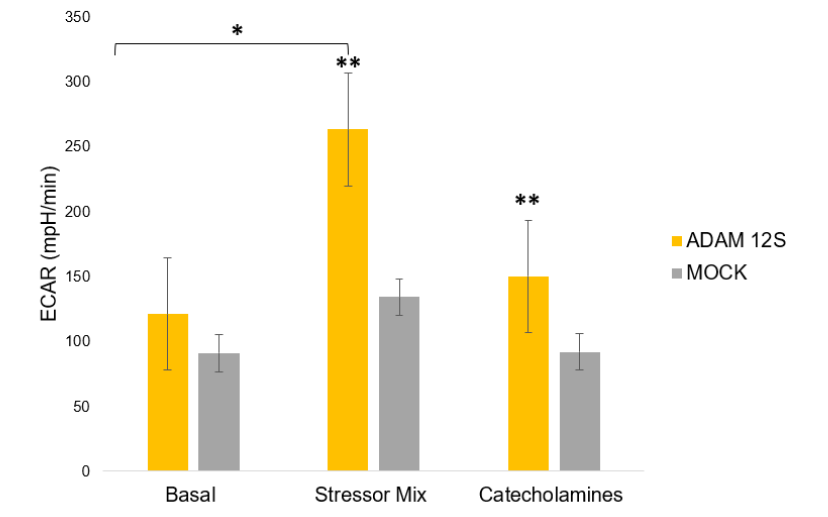
The ability of ADAM 12S to reprogram human primary preadipocytes into metabolically active BAT-like cells has significant therapeutic potential. Future research will further explore the mechanisms underlying adipocyte reprogramming and explore the therapeutic efficacy of this approach.
References
1. Poirier P, Giles TD, Bray GA, et al. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss. Arterioscler Thromb Vasc Biol. 2006;26(5):968-976. doi:10.1161/01.ATV.000 0216787.85457.
2. Harms M, Seale P. Brown and beige fat: development, function and therapeutic potential. Nat Med. 2013;19(10):1252-1263. doi:10.1038/n m.3361
3. Nedergaard J, Bengtsson T, Cannon B. Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab. 2007;293(2):E444-E452. doi:10.1152/ajpendo.006 91.2006
4. Peirce V, Vidal-Puig A. Regulation of glucose homoeostasis by brown adipose tissue. Lancet Diabetes Endocrinol. 2013;1(4):353-360. doi:10.10 16/S2213-8587(13)70055-X
5. no M, Raab G, Lau K, Abraham JA, Klagsbrun M. Purification and characterization of transmembrane forms of heparin-binding EGF-like growth factor. J Biol Chem. 1994;269(49):31315-31321
6. Suzuki M, Raab G, Moses MA, Fernandez CA, Klagsbrun M. Matrix metalloproteinase-3 releases active heparin-binding EGF-like growth factor by cleavage at a specific juxtamembrane site. J Biol Chem. 1997;272(50):31730-31737. doi:10.1074/jb c.272.50.31730
7. Ghorbani M, Claus TH, Himms-Hagen J. Hypertrophy of brown adipocytes in brown and white adipose tissues and reversal of diet-induced obesity in rats treated with a beta3-adrenoceptor agonist. Biochem Pharmacol. 1997;54(1):121-131. doi:10.1016/s0006-2952(97)00162-7
8. Zhou Z, Darwal MA, Cheng EA, Taylor SR, Duan E, Harding PA. Cellular reprogramming into a brown adipose tissue-like phenotype by co-expression of HB-EGF and ADAM 12S. Growth Factors. 2013;31 (6):185-198. doi:10.3109/08977194.2013.84029
9. Higashiyama S, Nanba D. ADAM-mediated ectodomain shedding of HB-EGF in receptor cross-talk. Biochim Biophys Acta. 2005;1751(1):110-117. doi:10.1016/j.bbapap.2004.11.009
10. Nanba D, Mammoto A, Hashimoto K, Higashiyama S. Proteolytic release of the carboxy-terminal fragment of proHB-EGF causes nuclear export of PLZF. J Cell Biol. 2003;163(3):489-502. doi:10.1083/jcb.200303017
11. Asakura M, Kitakaze M, Takashima S, et al. Cardiac hypertrophy is inhibited by antagonism of ADAM12 processing of HB-EGF: metalloproteinase inhibitors as a new therapy. Nat Med. 2002;8(1):35-40. doi:10.1038/nm0102-35
12. Kurisaki T, Masuda A, Sudo K, et al. Phenotypic analysis of Meltrin alpha (ADAM12)-deficient mice: involvement of Meltrin alpha in adipogenesis and myogenesis. Mol Cell Biol. 2003;23(1):55-61. doi:10.1128/MCB.23.1.55-61.2003
13. Kawaguchi N, Xu X, Tajima R, et al. ADAM 12 protease induces adipogenesis in transgenic mice. Am J Pathol. 2002;160(5):1895-1903. doi:10.101 6/S0002-9440(10)61136-4
14. Taylor, S.R., Klements, J.R., Markesbery, M.G., Johnson, K.D., & Harding, P.A. (2014). Cellular transdifferentiation into brown adipose-like cells by adenoviral-directed expression or stable transfection of HB-EGF and ADAM 12S. J Cell Mol Biol, 10(2): 185-198.
15. Taylor SR, Gemma CA, Cartwright KM, et al. HB-EGF and ADAM 12S directed cellular reprogramming results in metabolically active brown adipose tissue-like cells[J]. AIMS Cell and Tissue Engineering, 2018, 2(4): 203-219. doi: 10.3934/celltissue.2018.4.203
16. Ahfeldt T, Schinzel RT, Lee YK, et al. Programming human pluripotent stem cells into white and brown adipocytes. Nat Cell Biol. 2012;14(2):209-219. Published 2012 Jan 15. doi:10.1038/ncb2411
17. Kawaguchi N, Xu X, Tajima R, et al. ADAM 12 protease induces adipogenesis in transgenic mice. Am J Pathol. 2002;160(5):1895-1903. doi:10.101 6/S0002-9440(10)61136-4
18. Uldry M, Yang W, St-Pierre J, Lin J, Seale P, Spiegelman BM. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 2006;3(5):333-341. doi:10.1016/j.cmet.2006.04.002
19. Seale P, Bjork B, Yang W, et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature. 2008; 454(7207):961-967. doi:10.1038/nature07182
20. Matsumoto S, Kishida K, Shimomura I, et al. Increased plasma HB-EGF associated with obesity and coronary artery disease [published correction appears in Biochem Biophys Res Commun 2002 Jul 12;295(2):571]. Biochem Biophys Res Commun. 2002; 292(3):781-786. doi:10.1006/bbrc.2002.6720
21. Jeong JH, Chang JS, Jo YH. Intracellular glycolysis in brown adipose tissue is essential for optogenetically induced nonshivering thermogenesis in mice. Sci Rep. 2018;8(1):6672. Published 2018 Apr 27. doi:10.1038/s41598-018-252653
22. Barclay JL, Agada H, Jang C, Ward M, Wetzig N, Ho KK. Effects of glucocorticoids on human brown adipocytes. J Endocrinol. 2015;224(2):139-147. doi:10.1530/JOE-14-0538
23. Winther S, Isidor MS, Basse AL, et al. Restricting glycolysis impairs brown adipocyte glucose and oxygen consumption. Am J Physiol Endocrinol Metab. 2018;314(3):E214-E223. doi:10.1152/ajpendo.002 18.2017
24. Inokuma K, Ogura-Okamatsu Y, Toda C, Kimura K, Yamashita H, Saito M. Uncoupling protein 1 is necessary for norepinephrine-induced glucose utilization in brown adipose tissue. Diabetes. 2005; 54(5):1385-1391. doi:10.2337/diabetes.54.5.1385
25. Teperino R, Amann S, Bayer M, et al. Hedgehog partial agonism drives Warburg-like metabolism in muscle and brown fat. Cell. 2012;151(2):414-426. doi:10.1016/j.cell.2012.09.021

