Reprogramming Human Adipocytes to Enhance Metabolism
Cellular Reprogramming of Human Primary Adipocytes into Brown Adipose Tissue-Like Cells Preferentially Utilize Glucose
Taylor SR¹, Cartwright KM¹, Sferrella AM¹, and Harding PA¹
- Department of Biology, Cell Molecular and Structural Biology Program, Miami University, Oxford, OH 45056, United States
OPEN ACCESS
PUBLISHED: 31 July 2025
CITATION:Taylor, SR., et al., 2025. Cellular Reprogramming of Human Primary Adipocytes into Brown Adipose Tissue-Like Cells Preferentially Utilize Glucose. Medical Research Archives, [online] 13(7).
https://doi.org/10.18103/mra.v13i7.6709
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6709
ISSN 2375-1924
Abstract
Brown adipose tissue has emerged as a potential therapeutic target for the treatment of type 2 diabetes and obesity. This aim of this study is to investigate the role of a disintegrin and metalloproteinase 12 expression alone in human primary adipocytes to induce a brown adipose tissue-like phenotype. The results indicate that ADAM 12S infection induces endogenous HB-EGF in primary human preadipocytes, which promotes the formation of brown adipose tissue-like cells. ADAM 12S-infected cells preferentially utilize glucose.
Keywords
- Brown adipose tissue
- Human primary adipocytes
- ADAM 12S
- HB-EGF
- Glucose utilization
Introduction
Obesity remains one of the most significant public health challenges globally, especially in developed nations where the increased availability of calorie-dense foods and a sedentary lifestyle have contributed to its rising prevalence¹. Defined by a body mass index (BMI) of 30 kg/m² or higher, obesity is strongly associated with serious health conditions such as type 2 diabetes, cardiovascular diseases, and certain cancers¹. This growing epidemic not only impacts individual health but also places a tremendous burden on healthcare systems worldwide, resulting in escalating healthcare costs. Current treatment strategies focusing on dietary modifications, increased physical activity, and lifestyle interventions often demonstrate limited long-term efficacy, underscoring the urgent need for innovative therapeutic approaches.
In recent years, brown adipose tissue (BAT) has garnered considerable interest as a potential target for obesity treatment and related metabolic disorders. Unlike white adipose tissue (WAT), which primarily stores energy, BAT dissipates energy through non-shivering thermogenesis by oxidizing glucose and fatty acids. This process is mediated by uncoupling protein-1 (UCP-1), which enables BAT to uncouple oxidative phosphorylation from ATP production, thereby generating heat². BAT’s capacity to burn energy and improve glucose metabolism, reduce insulin resistance, and promote lipid catabolism makes it a promising candidate for therapeutic strategies aimed at increasing energy expenditure and reducing obesity-related comorbidities³,⁴. Additionally, the role of BAT in thermogenesis has been supported by studies demonstrating its activation by various stimuli, including cold exposure and β-adrenergic agonists, which promote mitochondrial biogenesis and the expression of thermogenic genes⁵,⁶.
Although BAT is abundant in newborns, where it plays a key role in thermoregulation, its presence in adults was historically thought to be negligible. However, recent discoveries have demonstrated that functional BAT depots persist in adults, particularly in regions such as the neck and upper thorax². The identification of beige or brite cells, which are induced within WAT in response to stimuli like cold exposure or β-adrenergic activation, has expanded the therapeutic potential of BAT. These cells exhibit a BAT-like phenotype, suggesting that the reprogramming of WAT into metabolically active BAT could serve as an innovative approach to treating obesity and metabolic diseases⁷.
Our laboratory has previously demonstrated that the co-expression of heparin-binding epidermal growth factor-like growth factor (HB-EGF) and the soluble form of a disintegrin and metalloproteinase 12 (ADAM 12S) induces the reprogramming of cells into a BAT-like phenotype⁸. HB-EGF, a transmembrane protein that undergoes proteolytic cleavage, generates both a soluble form (sHB-EGF) and a carboxy-terminal fragment (HB-EGF C), each with mitogenic activity⁹. HB-EGF is involved in numerous physiological processes, including wound healing, cell proliferation, and tissue remodeling, and has been linked to the expansion of WAT and enhanced adipogenesis in obesity¹⁰,¹¹.
ADAM 12, a member of the ADAM family of metalloproteinases, plays a crucial role in the proteolytic processing of HB-EGF and is involved in extracellular matrix remodeling, myogenesis, and adipogenesis¹². ADAM 12 exists in both a membrane-bound form (ADAM 12L) and an alternatively spliced soluble form (ADAM 12S), with ADAM 12S shown to induce adipogenesis in transgenic mice¹³. Previous work from our lab demonstrated that the co-expression of HB-EGF and ADAM 12S in murine fibroblasts, human epidermoid carcinoma cells, and preadipocytes resulted in lipid accumulation, upregulation of BAT-specific genes such as PGC-1α, and increased mitochondrial density, all characteristic features of BAT-like cells¹⁴.
Given the metabolic potential of brown and beige adipose tissues to enhance energy expenditure and improve metabolic health, this study seeks to expand on our previous findings by examining the effects of ADAM 12S expression in human primary
adipocytes that are representative of a wide variety of patients from all levels of adiposity and age including both male and female donors. We hypothesize that ADAM 12S expression alone, independent of HB-EGF co-expression, can drive the reprogramming of white adipocytes into metabolically active BAT-like cells through endogenous HB-EGF signaling. Such reprogramming could lay the groundwork for novel therapeutic strategies aimed at increasing energy expenditure, reducing fat mass, and addressing the metabolic dysfunctions associated with obesity and type 2 diabetes.
Materials and Methods
GENERATION OF ADAM12S ADENOVIRUS
The ADAM12S adenovirus used in this study was generated using the AdEasy adenoviral system (Agilent Technologies) as previously described¹⁴. Briefly, the ADAM12S cDNA (2.1 kb) was cloned into the pShuttle-IRES-hrGFP-1 plasmid for co-expression with a humanized recombinant green fluorescent protein (hrGFP), allowing visual confirmation of infection. The recombinant plasmid was electroporated into BJ5183-AD-1 cells, followed by recombination with the pAdEasy-1 backbone. High-titer viral stocks were produced in AD-293 cells, and viral titers were determined by serial dilution as described in our prior work¹⁴.
GENERATION OF ADAM 12S EXPRESSING HUMAN PRIMARY PREADIPOCYTES
Human primary subcutaneous preadipocytes (ZenBio Inc.) were cultured in DMEM supplemented with 10% fetal bovine serum (FBS), penicillin (100 μg/mL), streptomycin (100 μg/mL), and amphotericin B (2.5 μg/mL) until they reached 40% confluence. Cells were then infected with either 15 μL of Ad-ADAM 12S or Ad-MOCK viral particles in the same media, as previously described¹⁵. The primary human cultured preadipocytes and adipocytes are pooled lots of cells derived from a wide variety of patients with representation from all levels of adiposity and age including both male and female donors appropriate for basic research in the field of adipogenesis.
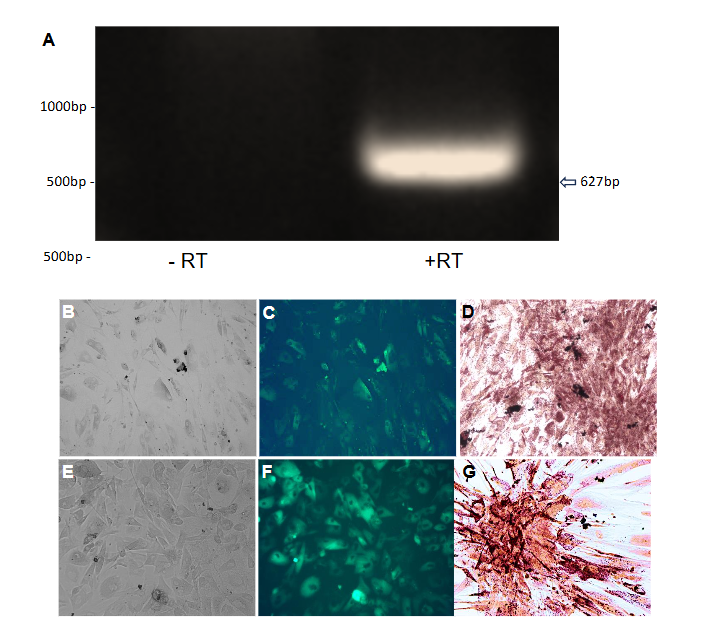
Successful infection was confirmed by observing hrGFP fluorescence 48 hours post-infection using an Olympus fluorescence microscope (Figure 1 panels C and D). To ensure the cells remained healthy and viable for subsequent experiments, the media was replaced with fresh growth media every 72 hours.
Lipid accumulation in infected cells was observed under brightfield microscopy three weeks post-infection. To quantitatively assess lipid content, Oil Red O staining was performed. Cells were washed with PBS, fixed with 10% formalin for 60 minutes at room temperature, and stained with freshly prepared Oil Red O solution for five minutes. Hematoxylin was used to counterstain the nuclei, and images were captured to compare lipid droplet formation between Ad-ADAM 12S and Ad-MOCK infected cells.
RT-PCR IN HUMAN PRIMARY PREADIPOCYTES
Total RNA was extracted from Ad-ADAM 12S and Ad-MOCK infected human primary subcutaneous preadipocytes using TriReagent (Sigma-Aldrich) following the manufacturer’s instructions. RNA was treated with DNase I to remove any contaminating genomic DNA, and concentrations were measured using a NanoDrop spectrophotometer. RT-PCR was performed using the Superscript One-Step RT-PCR kit (Invitrogen) with 1.5 μg of total RNA. The specific primers for human HB-EGF were designed based on previous work⁵ and used to amplify a 627 bp product with and without reverse transcriptase. Reactions were subjected to 30 cycles of denaturation (94°C for 30 seconds), annealing (59°C for one minute), and extension (72°C for 45 seconds), followed by a final extension at 72°C for 15 minutes. PCR products were visualized on a 0.8% agarose gel stained with ethidium bromide.
ADAM 12S INFECTION OF HUMAN PRIMARY PREADIPOCYTES
Adenovirus expressing ADAM 12S or empty (Mock) were prepared as previously described¹⁵. Western blot analysis of ADAM 12S was performed as previously described¹⁴ using cell extracts of Ad-ADAM 12S infected mouse fibroblasts (MCL), human A431 epidermoid carcinoma cells, and human
embryonic kidney cells (HEK 293), and resulted in immunoreactive proteins of 68kDa while no ADAM 12S immunoreactive proteins were observed in Ad-mock infected cells.
qPCR IN HUMAN PRIMARY PREADIPOCYTES
For quantitative PCR (qPCR), total RNA from Ad-ADAM 12S and Ad-MOCK infected cells was reverse-transcribed into cDNA using a high-capacity cDNA reverse transcription kit (Applied Biosystems). qPCR was conducted using a SYBR Green qRT-PCR kit (Quantabio) on a CFX Connect Real-Time PCR Detection System (BioRad). The relative expression of brown adipose tissue (BAT) marker genes—PRDM16, PGC-1α, and UCP-1—was determined using the 2^−ΔΔCt method, normalizing against the housekeeping genes β-actin and GAPDH¹⁶. Each reaction was performed in triplicate, and data were analyzed using CFX Manager software.
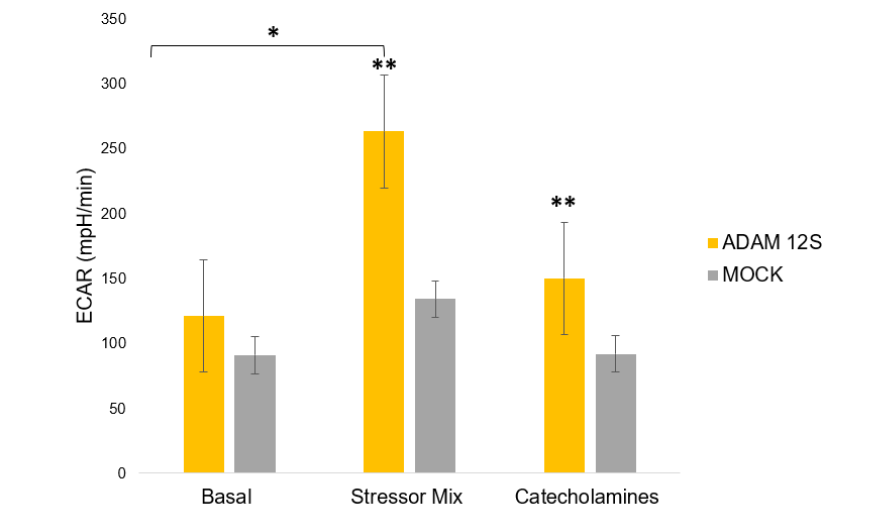
SEAHORSE METABOLIC ANALYSIS
The metabolic activity of Ad-ADAM 12S and Ad-MOCK infected human primary subcutaneous preadipocytes was assessed using the Seahorse XF24 Analyzer (Seahorse Bioscience). Cells were seeded into XF24 plates at a density of 100,000 cells per well and incubated overnight. The following day, the sensor cartridge was hydrated, and assay medium (1 mM pyruvate, 2 mM glutamine, 10 mM glucose, pH 7.4) was prepared. After basal metabolic measurements, cells were exposed to catecholamines (4 μg/mL) to stimulate thermogenesis, followed by a stressor mix containing FCCP (a mitochondrial uncoupler) and Oligomycin (an ATP synthase inhibitor). The extracellular acidification rate (ECAR), which reflects the rate of glycolysis, and oxygen consumption rate (OCR), indicative of mitochondrial respiration, were measured under both basal and stressed conditions.
Results
RT-PCR OF HUMAN PRIMARY PREADIPOCYTES
To confirm the expression of endogenous HB-EGF in human primary preadipocytes, RT-PCR was conducted using primers specific to human HB-EGF (Table 1).
A 627 bp product was observed in reverse-transcriptase-positive (+RT) samples, indicating the presence of HB-EGF (Figure 1 panel A) mRNA in these cells. In contrast, no product was detected in reverse-transcriptase-negative (−RT) samples (Figure 1 panel A), confirming the specificity of the amplification. These results suggest that HB-EGF is endogenously expressed in human primary preadipocytes, potentially playing a role in the cellular reprogramming observed in this study.
LIPID ACCUMULATION AND OIL RED O STAINING IN INFECTED HUMAN PRIMARY PREADIPOCYTES
Human primary preadipocytes infected with Ad-ADAM 12S exhibited significant lipid accumulation three weeks post-infection, as visualized by Oil Red O staining (Figure 1 Panel G). The stained lipid droplets appeared as bright red structures under the microscope, confirming the presence of stored lipids in these cells. Ad-MOCK infected cells, in contrast, showed no visible lipid accumulation (Figure 1 Panel D), indicating that the presence of ADAM 12S promotes a BAT-like phenotype characterized by triglyceride storage. These findings are consistent with previous studies that have shown ADAM 12S’s role in adipogenesis and lipid metabolism¹⁷.
DIFFERENTIAL GENE EXPRESSION IN HUMAN PRIMARY PREADIPOCYTES
qPCR analysis was performed to assess the expression of BAT-specific genes in Ad-ADAM 12S and Ad-MOCK infected cells (Table 2). ADAM 12S infection resulted in a significant upregulation of PGC-1α (p = 0.0018), a master regulator of mitochondrial biogenesis and thermogenesis¹⁸. Although UCP-1, the hallmark gene of BAT thermogenesis, was upregulated, the increase did not reach statistical significance (p = 0.2294). Interestingly, PRDM16, a gene critical for brown fat differentiation, was significantly downregulated in ADAM 12S-infected cells (p = 0.0008). This may suggest that PRDM16 expression is just a molecular switch and not required for maintaining the BAT program¹⁹. In order to assess differential gene expression due to ADAM12S expression, PPARg levels were compared to Ad-mock infected human primary preadipocytes and resulted in no significant difference.
Results
qPCR in Human Primary Preadipocytes
For quantitative PCR (qPCR), total RNA from AdAM 12S and Ad-MOCK infected cells was reverse-transcribed into cDNA using a high-capacity cDNA reverse transcription kit (Applied Biosystems). qPCR was conducted using a SYBR Green qRT-PCR Kit (Quantabio) on a CFX Connect Real-Time PCR Detection System (BioRad). The relative expression of brown adipose tissue (BAT) marker genes PRDM16, UCP-1, and PGC-1α, along with housekeeping genes GAPDH and β-actin, were analyzed.
Differential Gene Expression in Human Primary Preadipocytes
Analysis of total RNA from human primary preadipocytes infected with Ad-MOCK (n = 3) or Ad-ADAM12S (n = 3) adenoviruses. Analysis of brown adipose tissue (BAT) marker gene expression revealed significant differences in expression between ADAM 12S- and mock-infected cells.
| Gene Name | Fold Change | p-value |
|---|---|---|
| PRDM16 | -2.40 | 0.0008 |
| UCP-1 | -2.01 | 0.0012 |
| β-actin | 1.00 | NA |
| GAPDH | 1.00 | NA |
| PPARg | 0.21 | 0.0018 |
| HB-EGF | 0.40 | 0.0014 |
References
2. Harms M, Seale P. Brown and beige fat: development, function and therapeutic potential. Nat Med. 2013;19(10):1252-1263. doi:10.1038/n m.3361
3. Nedergaard J, Bengtsson T, Cannon B. Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab. 2007;293(2):E444-E452. doi:10.1152/ajpendo.006 91.2006
4. Peirce V, Vidal-Puig A. Regulation of glucose homoeostasis by brown adipose tissue. Lancet Diabetes Endocrinol. 2013;1(4):353-360. doi:10.10 16/S2213-8587(13)70055-X
5. no M, Raab G, Lau K, Abraham JA, Klagsbrun M. Purification and characterization of transmembrane forms of heparin-binding EGF-like growth factor. J Biol Chem. 1994;269(49):31315-31321
6. Suzuki M, Raab G, Moses MA, Fernandez CA, Klagsbrun M. Matrix metalloproteinase-3 releases active heparin-binding EGF-like growth factor by cleavage at a specific juxtamembrane site. J Biol Chem. 1997;272(50):31730-31737. doi:10.1074/jb c.272.50.31730
7. Ghorbani M, Claus TH, Himms-Hagen J. Hypertrophy of brown adipocytes in brown and white adipose tissues and reversal of diet-induced obesity in rats treated with a beta3-adrenoceptor agonist. Biochem Pharmacol. 1997;54(1):121-131. doi:10.1016/s0006-2952(97)00162-7
8. Zhou Z, Darwal MA, Cheng EA, Taylor SR, Duan E, Harding PA. Cellular reprogramming into a brown adipose tissue-like phenotype by co-expression of HB-EGF and ADAM 12S. Growth Factors. 2013;31 (6):185-198. doi:10.3109/08977194.2013.84029
9. Higashiyama S, Nanba D. ADAM-mediated ectodomain shedding of HB-EGF in receptor cross-talk. Biochim Biophys Acta. 2005;1751(1):110-117. doi:10.1016/j.bbapap.2004.11.009
10. Nanba D, Mammoto A, Hashimoto K, Higashiyama S. Proteolytic release of the carboxy-terminal fragment of proHB-EGF causes nuclear export of PLZF. J Cell Biol. 2003;163(3):489-502. doi:10.1083/jcb.200303017
11. Asakura M, Kitakaze M, Takashima S, et al. Cardiac hypertrophy is inhibited by antagonism of ADAM12 processing of HB-EGF: metalloproteinase inhibitors as a new therapy. Nat Med. 2002;8(1):35-40. doi:10.1038/nm0102-35
12. Kurisaki T, Masuda A, Sudo K, et al. Phenotypic analysis of Meltrin alpha (ADAM12)-deficient mice: involvement of Meltrin alpha in adipogenesis and myogenesis. Mol Cell Biol. 2003;23(1):55-61. doi:10.1128/MCB.23.1.55-61.2003
13. Kawaguchi N, Xu X, Tajima R, et al. ADAM 12 protease induces adipogenesis in transgenic mice. Am J Pathol. 2002;160(5):1895-1903. doi:10.101 6/S0002-9440(10)61136-4
14. Taylor, S.R., Klements, J.R., Markesbery, M.G., Johnson, K.D., & Harding, P.A. (2014). Cellular transdifferentiation into brown adipose-like cells by adenoviral-directed expression or stable transfection of HB-EGF and ADAM 12S. J Cell Mol Biol, 10(2): 185-198.
15. Taylor SR, Gemma CA, Cartwright KM, et al. HB-EGF and ADAM 12S directed cellular reprogramming results in metabolically active brown adipose tissue-like cells[J]. AIMS Cell and Tissue Engineering, 2018, 2(4): 203-219. doi: 10.3934/celltissue.2018.4.203
16. Ahfeldt T, Schinzel RT, Lee YK, et al. Programming human pluripotent stem cells into white and brown adipocytes. Nat Cell Biol. 2012;14(2):209-219. Published 2012 Jan 15. doi:10.1038/ncb2411
17. Kawaguchi N, Xu X, Tajima R, et al. ADAM 12 protease induces adipogenesis in transgenic mice. Am J Pathol. 2002;160(5):1895-1903. doi:10.101 6/S0002-9440(10)61136-4
18. Uldry M, Yang W, St-Pierre J, Lin J, Seale P, Spiegelman BM. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 2006;3(5):333-341. doi:10.1016/j.cmet.2006.04.002
19. Seale P, Bjork B, Yang W, et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature. 2008; 454(7207):961-967. doi:10.1038/nature07182
20. Matsumoto S, Kishida K, Shimomura I, et al. Increased plasma HB-EGF associated with obesity and coronary artery disease [published correction appears in Biochem Biophys Res Commun 2002 Jul 12;295(2):571]. Biochem Biophys Res Commun. 2002; 292(3):781-786. doi:10.1006/bbrc.2002.6720
21. Jeong JH, Chang JS, Jo YH. Intracellular glycolysis in brown adipose tissue is essential for optogenetically induced nonshivering thermogenesis in mice. Sci Rep. 2018;8(1):6672. Published 2018 Apr 27. doi:10.1038/s41598-018-25265-3
22. Barclay JL, Agada H, Jang C, Ward M, Wetzig N, Ho KK. Effects of glucocorticoids on human brown adipocytes. J Endocrinol. 2015;224(2):139-147. doi:10.1530/JOE-14-0538
23. Winther S, Isidor MS, Basse AL, et al. Restricting glycolysis impairs brown adipocyte glucose and oxygen consumption. Am J Physiol Endocrinol Metab. 2018;314(3):E214-E223. doi:10.1152/ajpendo.002 18.2017
24. Inokuma K, Ogura-Okamatsu Y, Toda C, Kimura K, Yamashita H, Saito M. Uncoupling protein 1 is necessary for norepinephrine-induced glucose utilization in brown adipose tissue. Diabetes. 2005; 54(5):1385-1391. doi:10.2337/diabetes.54.5.1385
25. Teperino R, Amann S, Bayer M, et al. Hedgehog partial agonism drives Warburg-like metabolism in muscle and brown fat. Cell. 2012;151(2):414-426. doi:10.1016/j.cell.2012.09.021

