Responses of Renal Cell Carcinoma to 29 Therapies
Responses of Renal Cell Carcinomas to 29 Different Systemic Anti-Cancer Therapies
Ofer N. Gofrit¹, Liat Appelbaum², Vladimir Yutkin¹, Mordechai Duvdevani¹, Guy Hidas¹, Steve Frank³, Yakir Rottenberg³
- Department of Urology, Hebrew University of Jerusalem, Hadassah Medical Center, Faculty of Medicine
- Department of Radiology, Hebrew University of Jerusalem, Hadassah Medical Center, Faculty of Medicine
- Department of Oncology, Hebrew University of Jerusalem, Hadassah Medical Center, Faculty of Medicine
OPEN ACCESS
PUBLISHED: 31 July 2025
CITATION: Gofrit, O. N., et al., 2025. Responses of Renal Cell Carcinomas to 29 Different Systemic Anti-Cancer Therapies. Medical Research Archives, [online] 13(7).
https://doi.org/10.18103/mra.v13i7.6752
COPYRIGHT:© 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6752
ISSN 2375-1924
ABSTRACT
Introduction: The prevailing view is that renal cell carcinomas (RCCs) are resistant to chemotherapy. However, it is essential to periodically reassess this perspective, as RCC is diagnosed concurrently with other malignancies, making treatment priority in these patients. The prognosis for these patients is grim.
RCC is considered chemoresistant. In the metastatic setting, its response to single or combined chemotherapy agents has been examined in several phase II studies. Single agents, including vinca alkaloids, gemcitabine, and fluoropyrimidine derivatives, have shown an objective response rate (ORR) of 0-20%. One study reported an ORR of 26% with acceptable. Combination chemotherapy, particularly involving gemcitabine and fluorouracil, has not been shown to provide a meaningful response in this setting. 4 Immunotherapy, including anti-PD-1 agents, has also shown promise in this regard. The aim of this study was to evaluate the response of RCC to various systemic anti-cancer therapies.
Methods: A retrospective review of patients with renal cell carcinoma (RCC) present at our institution was conducted. The largest diameter of the RCC from the initial pathology report was measured and recorded with the measurements taken at the same location at the follow-up period. Renal pathological specimens were evaluated using the 2002 TNM classification, histologic subclassing according to the 1997 UICC classification, and grading based on Fuhrman’s nuclear grading system. The tumor’s linear growth rate was calculated by subtracting the tumor’s largest diameter in the initial CT (mm) from the largest diameter on the last CT (mm) divided by the time elapsed between the studies (in years). Diameter changes within a 3 mm range were classified as “no change”. The research was approved by the institutional review board.
Keywords: renal cell carcinoma, systemic anti-cancer therapies, chemotherapy, immunotherapy
Introduction
RCC is considered chemoresistant. In the metastatic setting, its response to single or combined chemotherapy agents has been examined in several phase II studies. Single agents, including vinca alkaloids, gemcitabine, and fluoropyrimidine derivatives, have shown an objective response rate (ORR) of 0-20%. One study reported an ORR of 26% with acceptable. Combination chemotherapy, particularly involving gemcitabine and fluorouracil, has not been shown to provide a meaningful response in this setting. 4 Immunotherapy, including anti-PD-1 agents, has also shown promise in this regard. The aim of this study was to evaluate the response of RCC to various systemic anti-cancer therapies.
Materials and Methods
The effects of systemic anti-cancer therapies, not targeted at RCC, were retrospectively evaluated through an archival search for patients with diagnosis of RCC (ICD-9-CM 189.0) and another type of cancer (ICD-9-CM codes 140–239). The initial search yielded 746 results. Patients included in the study met all the following criteria:
-
Histologically confirmed non-renal cancer.
-
Histologically confirmed RCC.
-
A CT scan performed before the initiation of systemic therapy, showing measurable RCC.
-
Undergoing chemotherapy or immunotherapy directed at the non-renal cancer, while the RCC received no specific treatment.
Subsequent CT scans conducted at least four months after therapy commencement.
The largest diameter of the RCC from the initial CT scan was recorded and compared with the measurements taken at the same location at the end of the follow-up period. Renal pathological specimens were evaluated using the 2002 TNM classification, histologic subtyping according to the 1997 UICC classification, and grading based on Fuhrman’s nuclear grading system.¹⁹ The tumor’s linear growth rate was calculated by subtracting the tumor’s largest diameter in the initial CT (mm) from the largest diameter on the last CT (mm), then dividing by the time elapsed between the studies (in years). Diameter changes within a 3 mm range were classified as “no change.” The research was approved by the local ethical committee (#302-19-HMO) that waived the requirement for informed consent.
Results
PATIENTS
Demographics, types of malignancy, and systemic therapy protocols of the 20 patients that met all criteria are detailed in Table 1. The cohort consisted of 14 men and 6 women, with an average age at diagnosis of 61.4 years (SD 10.0 years). Cancer types included lymphoma in 7 patients, lung adenocarcinoma and breast cancer in 4 patients each, and leukemia in 3 patients. Each of the following was present in one patient: colon cancer, melanoma, gastrointestinal stromal tumor, and carcinoma of the gastroesophageal junction. One patient had three types of malignancies (patient #8). After a median follow-up of 56.5 months (Q1 38.75, Q3 97.75), eleven patients died, all from their non-renal cancers.
SYSTEMATIC TREATMENTS
Patients received a total of 29 preparations, administered either as single agents or in combinations. CHOP or R-CHOP combinations were administered to 4 patients. Other commonly prescribed agents included cyclophosphamide and doxorubicin, each given to 5 patients, and vincristine to 4 patients. The complete list of medications is available in Supplementary Document 1.
RENAL CELL CARCINOMA DIRECTED TREATMENTS
Treatments directed at RCC and the patients’ statuses at the end of the follow-up period are shown in Table 2. By the end of the follow-up, 6 patients had undergone radical nephrectomy, 5 had partial nephrectomy, 3 had percutaneous ablation, and one patient had main renal artery embolization (to control bleeding). Five patients received no specific RCC-directed therapy.
RESPONSE OF RENAL CELL CARCINOMA TO SYSTEMIC THERAPY
RCC diameters, time intervals, and growth rates are detailed in Table 3 and Fig. 1. Representative CT images are presented in Fig. 2. The complete set of photos and case histories is available in Supplementary Material 2. The average RCC diameter on the initial CT was 41.9 mm (SD 25.1 mm). After an average follow-up of 28.9 months (SD 29.6 months), the RCC diameter increased in 15 patients (by an average growth rate of 7.3 mm/year, SD 2.9 mm/year). In patients treated with CHOP or R-CHOP, the average growth rate was 8 mm/year (SD 3.0 mm/year). In those treated with cyclophosphamide or doxorubicin, the average growth rate was 5.9 mm/year (SD 4.5 mm/year), and in patients treated with vincristine, the average growth rate was 6.3 mm/year (SD 5.1 mm/year). The RCC diameter remained stable in four patients and decreased in two patients (by 12 mm/year in a tamoxifen-treated patient and by 3.1 mm/year in a patient treated with a combination of bevacizumab, paclitaxel, and cisplatin).
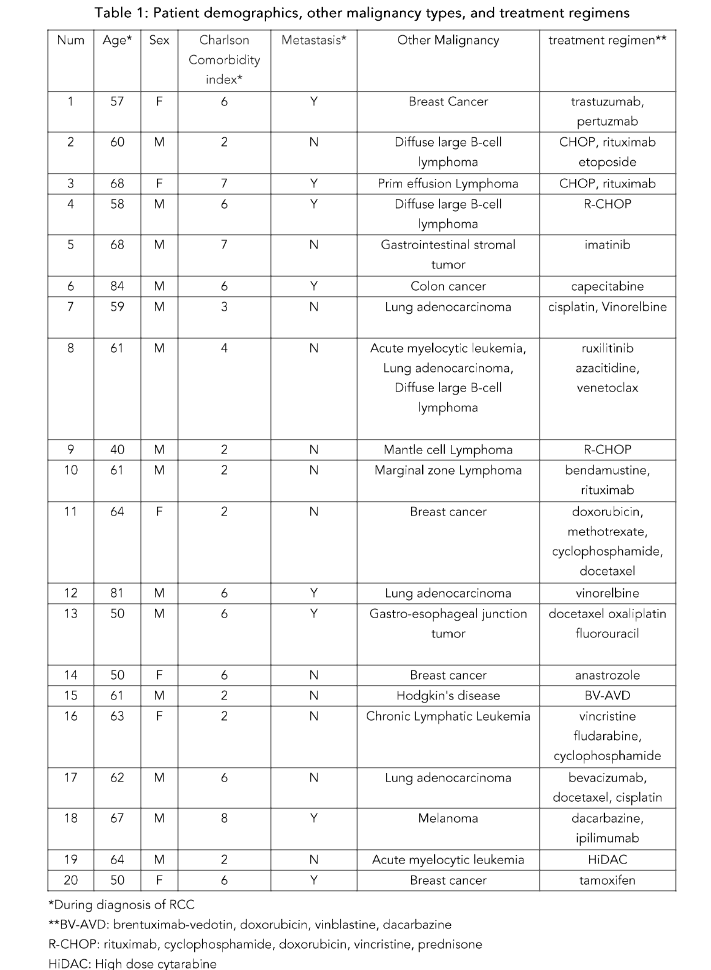
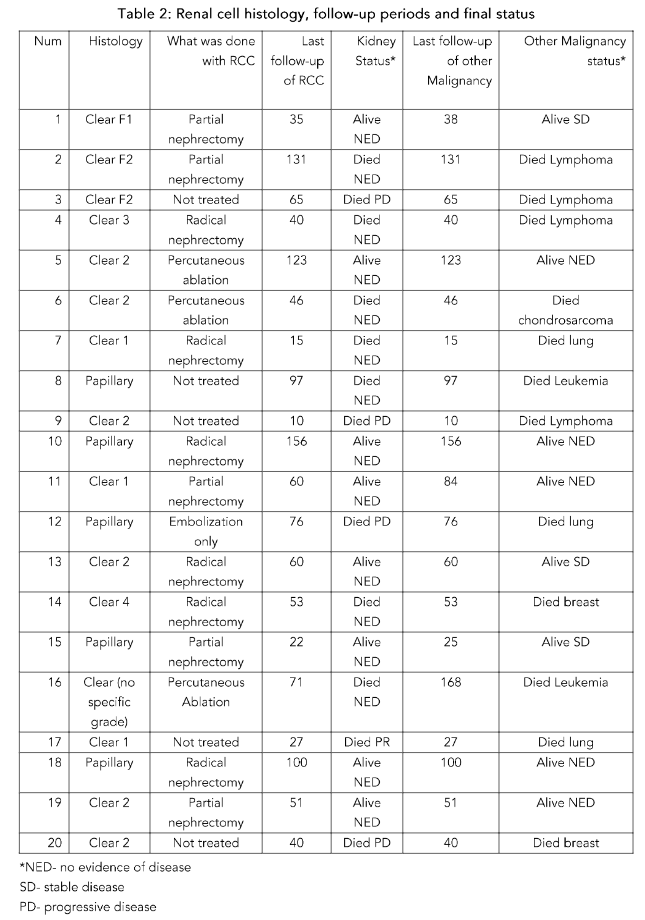
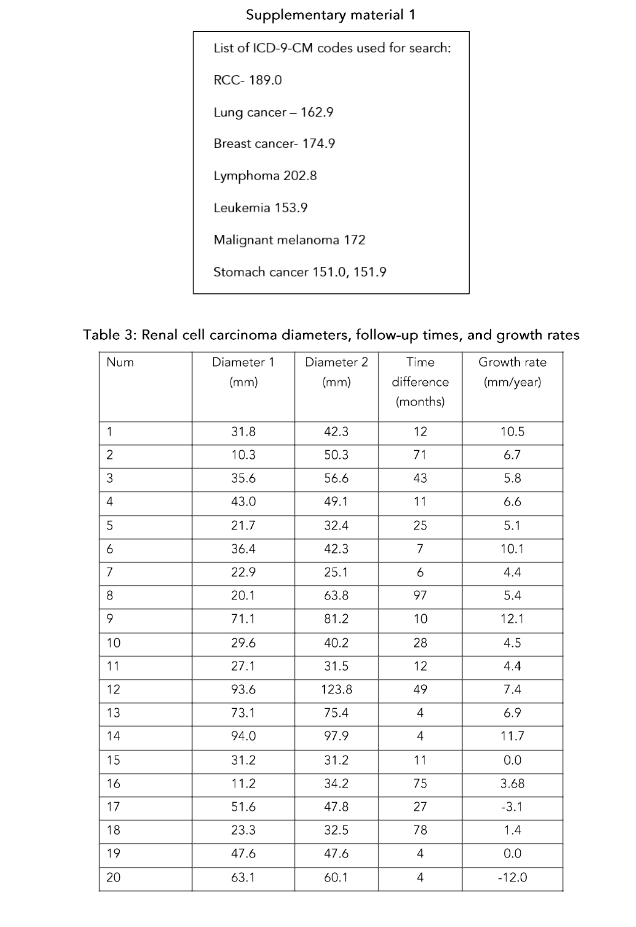
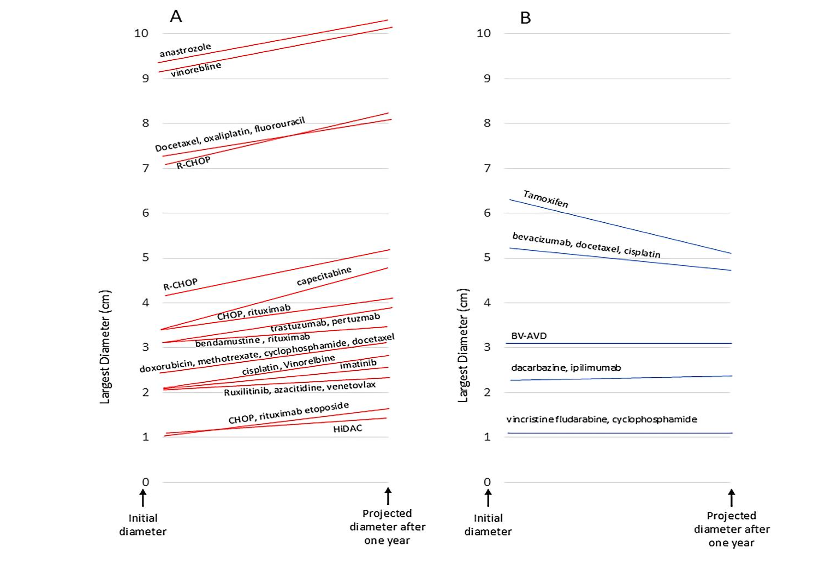
Fig. 1. A: Tumors that increased in size; B: Tumors that remained stable or decreased in size.
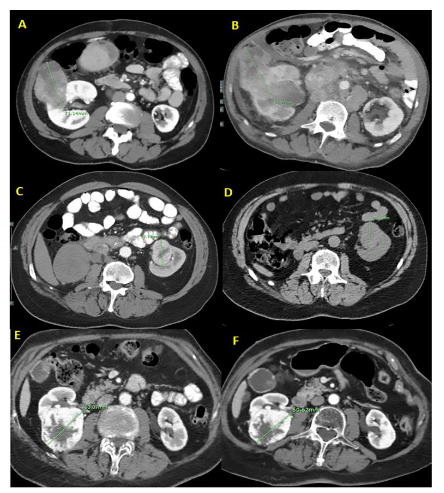 Fig. 2.
Fig. 2.
A: A 40-year-old man diagnosed with mantle cell lymphoma and Fuhrman grade 2 clear cell carcinoma. B: After 10 months and 4 cycles of R-CHOP, the tumor’s diameter increased by 10 mm.
C: A 58-year-old man diagnosed with diffuse large B-cell lymphoma and Fuhrman grade 3 clear cell carcinoma. D: After 11 months and 6 cycles of R-CHOP, the tumor’s diameter increased by 6 mm.
E: A 50-year-old woman with breast cancer and Fuhrman grade 2 clear cell carcinoma. F: After 4 months of tamoxifen, the tumor’s diameter decreased by 2.5 mm.
References
1. Capitanio U, Montorsi F. Renal cancer. Lancet. 2016;387:894-906. 26318520. doi: 10.1016/S0140-6736(15)00046-X. Epub 2015 Aug 25. PMID: 2631 8520.
2. Diamond E, Molina AM, Carbonaro M, Akhtar NH, et al. Cytotoxic chemotherapy in the treatment of advanced renal cell carcinoma in the era of targeted therapy. Crit Rev Oncol Hematol. 2015; 96:518-26. doi: 10.1016/j.critrevonc.2015.08.007. Epub 2015 Aug 13. PMID: 26321263.
3. Tsimafeyeu I, Demidov L, Kharkevich G, et al. Phase II, multicenter, uncontrolled trial of single-agent capecitabine in patients with non-clear cell metastatic renal cell carcinoma. Am J Clin Oncol. 2012;35:251-4. doi: 10.1097/COC.0b013e31820d bc17. PMID: 21358295.
4. Mani S, Vogelzang NJ, Bertucci D, et al. Phase I study to evaluate multiple regimens of intravenous 5-fluorouracil administered in combination with weekly gemcitabine in patients with advanced solid tumors: a potential broadly active regimen for advanced solid tumor malignancies. Cancer. 2001; 92:1567-76. doi: 10.1002/1097-0142(20010915)92 :6<1567::aid-cncr1483>3.0.co;2-l. PMID: 11745236.
5. Ryan CW, Vogelzang NJ, Stadler WM. A phase II trial of intravenous gemcitabine and 5-fluorouracil with subcutaneous interleukin-2 and interferon-alpha in patients with metastatic renal cell carcinoma. Cancer. 2002;94:2602-9. doi: 10.1002/cncr.10528. PMID: 12173327.
6. Posadas EM, Limvorasak S, Figlin RA. Targeted therapies for renal cell carcinoma. Nat Rev Nephrol. 2017;13:496-511. doi: 10.1038/nrneph.2017.82. Epub 2017 Jul 10. PMID: 28691713.
7. Tung I, Sahu A. Immune Checkpoint Inhibitor in First-Line Treatment of Metastatic Renal Cell Carcinoma: A Review of Current Evidence and Future Directions. Front Oncol. 2021;11:707214. doi: 10.3389/fonc.2021.707214. PMID: 34527581; PMCID: PMC8435744.
8. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med. 2018;378:1277-1290. doi: 10.1056/NEJMoa1712126. Epub 2018 Mar 21. PMID: 29562145; PMCID: PMC5972549.
9. Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380:1116-1127. doi: 10.1056/NEJMoa1816714. Epub 2019 Feb 16. PMID: 30779529.
10. Motzer RJ, Escudier B, George Set al. Nivolumab versus everolimus in patients with advanced renal cell carcinoma: Updated results with long-term follow-up of the randomized, open-label, phase 3 CheckMate 025 trial. Cancer. 2020; 126:4156-4167. doi: 10.1002/cncr.33033. Epub 2020 Jul 16. PMID: 32673417; PMCID: PMC8415096.
11. Richard PO, Jewett MA, Bhatt JR, et al. Renal Tumor Biopsy for Small Renal Masses: A Single-center 13-year Experience. Eur Urol. 2015;68:1007-13. doi: 10.1016/j.eururo.2015.04.004. Epub 2015 Apr 18. PMID: 25900781.
12. Chawla SN, Crispen PL, Hanlon AL et al. The natural history of observed enhancing renal masses: meta-analysis and review of the world literature. J Urol. 2006;175: 425-31. doi: 10.1016/S0022-5347 (05)00148-5. PMID: 16406965.
13. Jewett MA, Mattar K, Basiuk J, et al. Active surveillance of small renal masses: progression patterns of early stage kidney cancer. Eur Urol. 2011;60:39-44. doi: 10.1016/j.eururo.2011.03.030. Epub 2011 Apr 1. PMID: 21477920.
14. Mues AC, Haramis G, Badani K et al. Active surveillance for larger (cT1bN0M0 and cT2N0M0) renal cortical neoplasms. Urology;2010;76:620-623. doi: 10.1016/j.urology.2010.04.021. PMID: 20599256.
15. Mehrazin R, Smaldone MC, Kutikov A et al. Growth kinetics and short-term outcomes of cT1b and cT2 renal masses under active surveillance. J Urol;192:659-664. doi: 10.1016/j.juro.2014.03.038. Epub 2014 Mar 15. PMID: 24641909; PMCID: PMC4419692.
16. Staehler M, Haseke N, Stadler T et al. The growth rate of large renal masses opposes active surveillance. BJU Int;105:928-931. doi: 10.1111/j.1464-410X.2009.08840.x. Epub 2009 Sep 14. PMID: 197 51265.
17. Greene FL, Page DL, Flemming ID et al. American Joint Committee on Cancer: AJCC Cancer Staging Manual. 6th ed. New York, NY: Springer:2002:323-5. https://link.springer.com/book/10.1007/978-1-4757-3656-4
18. Störkel S, Eble JN, Adlakha DK, et al. Classification of renal cell carcinoma: Workgroup No. 1. Union Internationale Contre le Cancer (UICC) and the American Joint Committee on Cancer (AJCC). Cancer;1997;80:987-9. doi: 10.1002/(sici)1097-0142(19970901)80:5<987::aid-cncr24>3.0.co;2-r. PMID: 9307203.
19. Fuhrman SA, Lasky LC. Limas C. Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol;1982;655-653. doi: 10.1097/00000478-198210000-00007. PMID: 71 80965.
20. Mues AC, Landman J. Small renal masses: current concepts regarding the natural history and reflections on the American Urological Association guidelines. Curr Opin Urol. 2010;20:105-10.
doi: 10.1097/MOU.0b013e32833625f8. PMID: 200 75734.
21. Li XS, Yao L, Gong K et al. Growth pattern of renal cell carcinoma (RCC) in patients with delayed surgical intervention. J Cancer Res Clin Oncol. 2013;138:269-274. doi: 10.1007/s00432-011-1083-0. Epub 2011 Nov 22. PMID: 22105897.
22. Walsh N, Larkin A, Kennedy S, et al. Expression of multidrug resistance markers ABCB1 (MDR-1/P-gp) and ABCC1 (MRP-1) in renal cell carcinoma. BMC Urol. 2009;9:6. doi: 10.1186/1471-2490-9-6. PMID: 19552816; PMCID: PMC2723136.
23. McFadyen MC, Melvin WT, Murray GI. Cytochrome P450 CYP1B1 activity in renal cell carcinoma. Br J Cancer. 200491:966-71. doi: 10.103 8/sj.bjc.6602053. PMID: 15280921; PMCID: PMC240 9861.
24. Selvarajah J, Nathawat K, Moumen A, et al. Chemotherapy-mediated p53-dependent DNA damage response in clear cell renal cell carcinoma: role of the mTORC1/2 and hypoxia-inducible factor pathways. Cell Death Dis. 2013: 17;4(10):e865. doi: 10.1038/cddis.2013.395. PMID: 24136229; PMCID: PMC3920935.
25. Aweys H, Lewis D, Sheriff M, et al. Renal Cell Cancer – Insights in Drug Resistance Mechanisms. Anticancer Res. 2023;43:4781-4792. doi: 10.2187 3/anticanres.16675. PMID: 37909991.
26. Mickisch GH, Roehrich K, Koessig J, et al. Mechanisms and modulation of multidrug resistance in primary human renal cell carcinoma. J Urol. 1990; 144:755-9. doi: 10.1016/s0022-5347(17)39586-1. PMID: 1974934.
27. Amin R. Tamoxifen-induced regression of pulmonary metastases from renal cell carcinoma. Br J Radiol. 1983;56:766-7. doi: 10.1259/0007-1285-56-670-766. PMID: 6616143.
28. Atzpodien J, Kirchner H, Illiger HJ, et al. IL-2 in combination with IFN- alpha and 5-FU versus tamoxifen in metastatic renal cell carcinoma: long-term results of a controlled randomized clinical trial. Br J Cancer. 2001;85:1130-6. doi: 10.1054/bjoc.20 01.2076. PMID: 11710825; PMCID: PMC2375150.
29. Al-Sarraf M, Eyre H, Bonnet J, et al. Study of tamoxifen in metastatic renal cell carcinoma and the influence of certain prognostic factors: a Southwest Oncology Group Study. Cancer Treat Rep. 1981; 65:447-51. PMID: 7237466.
30. Bielcikova Z, Werner L, Stursa J, et al. Mitochondrially targeted tamoxifen as anticancer therapy: case series of patients with renal cell carcinoma treated in a phase I/Ib clinical trial. Ther Adv Med Oncol. 2023;30;15:17588359231197957. doi: 10.1177/17588359231197957. PMID: 377865 38; PMCID: PMC10541747.
31. Rixe O, Bukowski RM, Michaelson MD, et al. Axitinib treatment in patients with cytokine-refractory metastatic renal-cell cancer: a phase II study. Lancet Oncol. 2007;8:975-84. doi: 10.1016/S1470-2045(07)70285-1. Epub 2007 Oct 23. PMID: 1795 9415.
32. Johnson L, Bylund J, Strup S, et al. Concomitant Renal Cell Carcinoma and Hematologic Malignancy in Immunosuppressed Patients. Am J Med Sci. 2016; 351:480-4. doi: 10.1016/j.amjms.2016.02.027. Epub 2016 Mar 21. PMID: 27140706.

