Role of GLP-1 Agonists & SGLT-2 Inhibitors in Kidney Care
The Revolutionary Role of GLP-1 Agonists and SGLT-2 Inhibitors in Managing Diabetes and Chronic Kidney Disease
Shankar Chakkera¹, Elizabeth J. Carey² MD
- Krieger School of Arts and Sciences, Johns Hopkins University, Baltimore. Maryland
- Department of Medicine, Mayo Clinic Arizona, Scottsdale. Arizona
OPEN ACCESS
PUBLISHED:30 October 2024
CITATION:Chakkera, S. and Carey, EJ., 2024.The Revolutionary Role of GLP-1 Agonists and SGLT-2 Inhibitors in Managing Diabetes and Chronic Kidney Disease in Liver Transplant Recipients. Medical Research Archives, [online] 12(10).https://doi.org/10.18103/mra.v12i11.6002
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6002
ISSN 2375-1924
Abstract
Successful liver transplant is the only life-saving intervention for patients with end-stage liver disease (ESLD); however, the success of transplantation is compromised by post-transplant complications, particularly diabetes mellitus and chronic kidney disease (CKD). These complications not only have significant adverse impact both on patient and graft survival but also carry substantial clinical and economic burdens. The growing prevalence of metabolic syndrome has only exacerbated these challenges as our transplant recipients either have pre-existing diabetes mellitus and/or CKD or develop these conditions post-transplantation.
Keywords
- GLP-1 Agonists
- SGLT-2 Inhibitors
- Diabetes Mellitus
- Chronic Kidney Disease
- Liver Transplantation
Introduction
Despite the life-saving intervention of liver transplantation for patients with ESLD (end-stage liver disease), the success of transplantation is compromised often by post-transplant complications, particularly diabetes mellitus and chronic kidney disease (CKD). These complications not only have significant adverse impact both on patient and graft survival but also carry substantial clinical and economic burdens. The growing prevalence of metabolic syndrome has only exacerbated these challenges as our transplant recipients either have pre-existing diabetes mellitus and/or mild CKD or develop these conditions post-transplantation. Additionally, metabolic syndrome milieu is one of the key contributors to development of Metabolic Associated Steatotic Liver Disease (MASLD), previously known as Non-Alcoholic Fatty Liver Disease (NAFLD). Metabolic Associated Steatotic Liver Disease has emerged as the leading cause of liver damage worldwide, affecting approximately 25% of the global adult population (American Liver Foundation, 2022).
The disease spectrum of MASLD ranges from benign hepatic steatosis to more severe forms, including metabolic steatohepatitis (MASH), which can progress to liver fibrosis, cirrhosis, and ultimately, liver failure. In such cases, liver transplantation remains the only definitive cure. In 2023, a record 10,660 liver transplants were performed in the United States alone, with MASH accounting for 27% of these procedures, making it the second leading indication for liver transplantation.
While a successful liver transplantation offers a cure for ESLD, it does not address the underlying metabolic milieu, such as diabetes, that are part of the metabolic syndrome. Additionally, the presence of CKD, whether pre-existing or newly developed post-transplant, compounds the challenges faced by these patients, creating a “double hit” scenario that further complicates their management. This review delves into the pathogenesis of diabetes mellitus and CKD, highlighting the unique challenges these complications present in liver transplant recipients. Moreover, it explores the potential of GLP-1 agonists and SGLT-2 inhibitors—therapeutic agents that have shown promise in non-liver transplant populations—as novel approaches to mitigate the effects of these complications in liver transplant recipients. By addressing these critical issues, this review aims to provide insights into optimizing post-transplant care and improving outcomes of patient and liver allograft survival.
Pathogenesis and Negative Impact of Diabetes Mellitus in Liver Transplant Recipients
The global increase in diabetes mellitus has led to a substantial rise in its prevalence among liver transplant candidates. Studies report that between 31% and 38% of candidates have pre-existing diabetes, while an additional 13% to 28% develop new post-transplant diabetes mellitus (PTDM) within the first three years post-transplant. Consequently, up to 66% of liver transplant recipients are affected by diabetes which has profound impact on liver allograft and patient survival. Post-transplant diabetes mellitus is associated with worse liver allograft outcomes, including an increased risk of rejection and late-onset hepatic artery thrombosis. Diabetes also increases the morbidity and mortality of these patients, by contributing to higher rates of infections, cardiovascular events, and malignancy.
Calcineurin inhibitors (CNIs), the cornerstone of immunosuppression in liver transplant recipients, are a significant contributor to worsening pre-existing diabetes and contributing to development of PTDM. Calcineurin inhibitors exert their immunosuppressive effects by binding to the nuclear factor of activated T-cells, thereby reducing the release of cytokine interleukin-2 from T cells and T proliferation. These drugs are essential to prevention of rejection but come at a metabolic cost, disrupting glucose metabolism through several mechanisms, including downregulation of transcription of the adiponectin gene which can lead to greater risk for glucose intolerance. The adiponectin gene is located on chromosome 3q26, a region highly susceptible to the development of metabolic syndrome and Type 2
diabetes mellitus.⁹ Research conducted on the Pima Indians of Arizona, a population with the highest known prevalence of Type 2 diabetes, has shown low adiponectin levels are associated with insulin resistance, whereas higher concentration of adiponectin helped protect against development of Type 2 diabetes.¹⁰ CNIs in addition to increasing insulin resistance, induces pancreatic beta cell apoptosis if administered in large doses., resulting in decreased insulin secretion.¹¹ Thus CNI’s, while effective in preventing organ rejection, disrupt glucose metabolism by increasing insulin resistance and reducing insulin secretion leading to abnormalities in the glucose homeostasis (Figure 1). Other factors, including patient age, gender, body mass index (BMI), and hepatitis C infection, further exacerbate the risk of PTDM.¹²
Current management of PTDM remains a challenge, often requiring adjustment immunosuppressant regimens and promoting lifestyle changes. However, these strategies often fall short of fully addressing the underlying metabolic challenges faced by transplant recipients. The introduction of GLP-1 agonists and SGLT-2 inhibitors offers a promising therapeutic option. These agents, which have shown efficacy in non-transplant populations, present new opportunities for improving the management of diabetes mellitus in liver transplant recipients, potentially mitigating the adverse effects of PTDM and enhancing long-term outcomes.
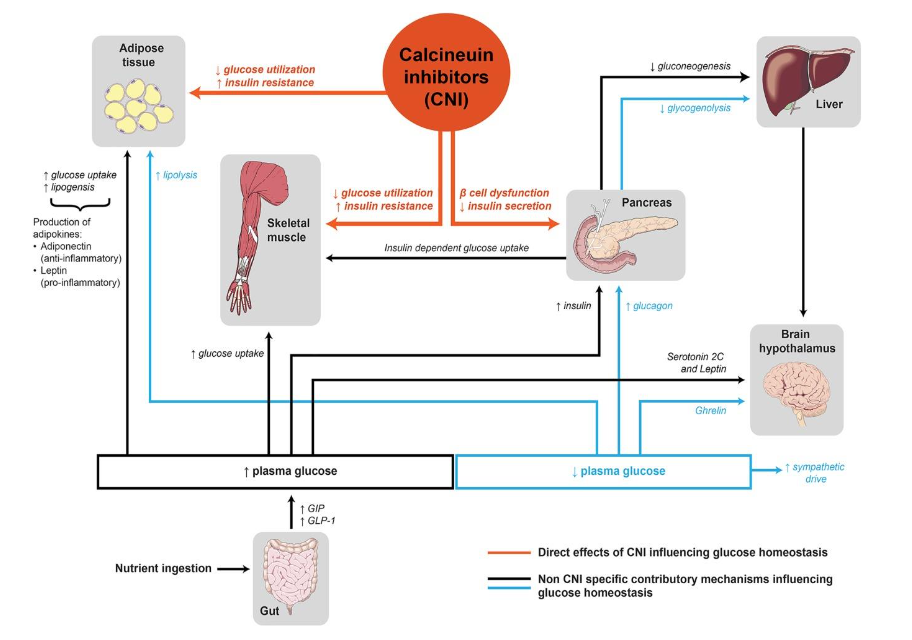
Pathogenesis and Negative Impact of Chronic Kidney Disease in Liver Transplant Recipients
Chronic kidney disease (CKD) is a major comorbidity after liver transplantation, affecting up to 41.5% of recipients.¹³ CKD is defined by a reduction in glomerular filtration rate (GFR) to less than 60 ml/min/1.73 m² for over three months or longer.¹³ It must be recognized that a small percentage of patients have CKD pre-transplant and meet the national UNOS criteria for simultaneous liver-kidney transplant. However, a significant number of patients develop kidney dysfunction from various contributors that impact optimal renal perfusion.
According to Cullaro et al., in their complete review of national registry data, there was a 187% increase in the proportion of patients with cirrhosis and CKD at the time of liver transplant between 2002 and 2017.¹² Furthermore, they reported, CKD at transplant was associated with a 16% independent higher risk of mortality post-liver transplant after adjusting for factors such as recipient age, etiology of cirrhosis, ethnicity, hepatic encephalopathy, era of transplant, diabetes mellitus, and donor risk index.¹² This demonstrates the rising prevalence of CKD and its detrimental effects on patients undergoing liver transplantation.
Acute kidney injury (AKI), a common complication with an incidence between 20–57% in patients with decompensated liver disease, is often a precursor to CKD.¹⁴ Early detection and optimal management are crucial since serum creatinine is a late indicator of kidney injury. Biomarkers like neutrophil gelatinase-associated lipocalin (NGAL) have shown promise in predicting AKI in the immediate post-transplant period, offering a potential tool for early intervention. Robertson et al. (2019) reported that urinary NGAL accurately predicted AKI development in 27 subjects, identifying NGAL as a strong early marker of AKI.¹⁵
Despite this, managing CKD in liver transplant recipients remains challenging, with limited options to prevent or mitigate the progression of kidney damage. The emergence of GLP-1 agonists and SGLT-2 inhibitors, with their renal protective effects, may offer new avenues for improving outcomes in this patient population.
Role of GLP-1 Agonists and SGLT-2 on Diabetes and Kidney Disease Post Liver Transplant
The emergence of Glucagon-like peptide-1 receptor (GLP-1) agonists and Sodium-glucose cotransporter-2 (SGLT-2) inhibitors as glucose-lowering therapies has opened new therapeutic avenues for managing patients with diabetic kidney disease. GLP-1 agonists are incretin hormones produced by the enteroendocrine L-cells of the small bowel and colon. A GLP-1 agonist is activated when it binds to its receptor, which activates adenylate cyclase, triggering a rise in cyclic AMP and calcium ion levels. These events stimulate the secretion of insulin, helping to compensate for insulin resistance triggered by new-onset diabetes.¹⁶ The drug also suppresses glucagon release by the pancreas, resulting in glucose-dependent reduction in plasma glucose and postprandial glucose by inhibiting hepatic glucose production and delaying gastric emptying.¹⁷ GLP-1s also act in brain regions associated with appetite and reward, reducing food intake and promoting weight loss.
Seven drugs have currently been approved for the treatment of Type 2 diabetes mellitus, including short-acting agents like exenatide and lixisenatide, as well as long-acting injectable agents such as albiglutide, dulaglutide, liraglutide, and semaglutide.¹⁸
GLP-1s regulate lipid metabolism, reducing the effects of hyperlipidemia and helping to lower the risk for both chronic kidney disease (CKD) and diabetic kidney disease.¹⁹ They have also been shown to reduce oxidative stress, which includes stimulating glomerular filtration and reducing vasoconstriction, slowing the progression of CKD.¹⁷ Limited data from clinical trials have been published on the effects of GLP-1 receptor agonists in individuals with post-liver transplant diabetes mellitus (PTDM). However, a retrospective study by Richardson et al. demonstrated the effectiveness and safety of GLP-1 agonists in managing obesity among liver transplant recipients. The results showed that the GLP-1RA group experienced a significant total body weight loss of 7.87% in one year, compared to 4.24% in the non-GLP-1RA group. Weight loss was consistent and progressive, starting at 3.93% at three months and reaching 7.87% at twelve months.²⁰ In addition, several case series studies have demonstrated benefits of GLP-1s in kidney and pancreas transplant recipients, including lowering blood glucose levels and providing renal protective effects.²¹ GLP-1 RAs would be advantageous in liver transplant recipients due to their renal clearance, avoiding interference with immunosuppressant drug metabolism. However, their use may be limited by gastrointestinal side effects, including pancreatitis, though studies have reported safe co-administration with tacrolimus in kidney transplant recipients with post-transplant diabetes mellitus.¹⁸
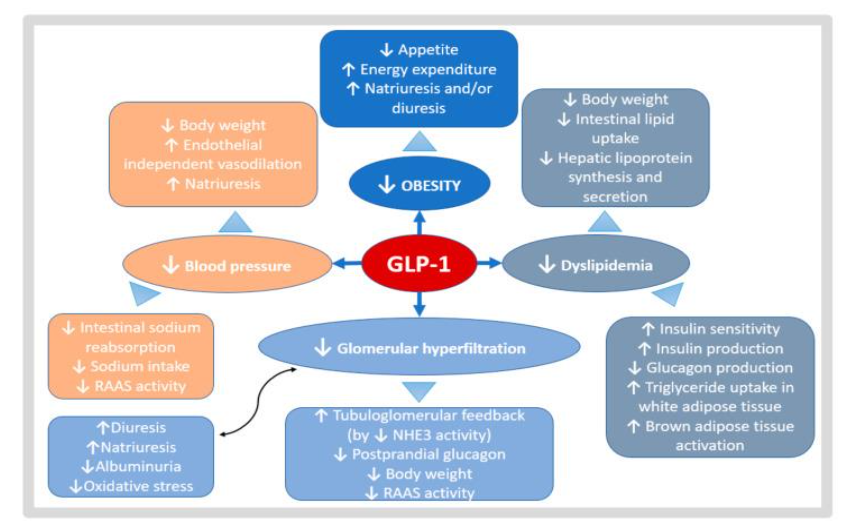
Figure 2: Potential benefits of GLP1-RA in CKD and KTR. GLP1-RA effects act in different metabolic pathways
SGLT-2 drugs have also been effective in reducing the risk of diabetic kidney disease. SGLT-2 inhibitors are a class of drugs that prevent kidney reabsorption of sugar and thus lower blood sugar levels. There are currently four approved SGLT-2 inhibitors (canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin) that have been approved by the FDA for their use in adults. All four of these inhibitors reduce the reabsorption of filtered glucose, decrease the renal threshold for glucose, and promote urinary glucose excretion.²²
Studies show that SGLT-2 use in diabetic kidney transplant recipients resulted in a decrease in hemoglobin A1C.²¹ There were potential side effects including increased infection risk and fluctuating allograft function. However, the beneficial effects of SGLT-2 such as weight loss, renal protection, and metabolic benefit make SGLT-2 a potential therapeutic option post-transplantation among patients with metabolic syndrome, obesity, and kidney dysfunction. While studies have primarily only looked at SGLT-2 in kidney transplant patients, we
can hypothesize comparable results among liver transplant recipients.
SGLT-2 inhibitors are recommended for diabetes patients with high cardiovascular risk which is a common feature of PTDM. While evidence is limited, animal studies show positive benefits and promises of SGLT-2. In heart and kidney transplant recipients, common FDA-approved SGLT-2 drugs such as empagliflozin and canagliflozin improve weight, blood pressure, and glycemic control. Recent studies in kidney transplant recipients with PTDM found that empagliflozin improves glycemic control and reduces body weight compared to the placebo group. Furthermore, urinary infections are not significantly higher for PTDM patients and can be managed with standard treatments.²²
Combination therapies involving GLP-1 agonists and SGLT-2 inhibitors have shown promise in managing co-morbidities associated with liver transplant recipients. A retrospective study conducted at the University Health Network found that, in liver transplant recipients with diabetes, combination therapy reduced HbA1c and alanine aminotransferase levels more effectively than DPP-4 inhibitors.²³ Additionally, two clinical trials in patients with Type 2 diabetes demonstrated that combination therapy significantly lowered HbA1c by 1.34% and 1.21%, compared to a 0.54% reduction with placebo, starting from a baseline of 8.0%.²⁴ These findings suggest that combination therapy may be especially beneficial in the liver transplant population, where patients often experience weight gain and develop metabolic co-morbidities.
Conclusions
Although studies have demonstrated the efficacy of GLP-1 receptor agonists (GLP-1 RAs) and SGLT-2 inhibitors in improving conditions associated with new-onset diabetes mellitus (DM) and chronic kidney disease (CKD), there is a scarcity of data regarding their use after liver transplantation. While some studies have shown the effectiveness of these drugs in kidney transplantation, there remains a limited number of clinical trials specifically focused on liver transplantation. As PTDM and chronic kidney disease are prevalent comorbidities in liver transplant patients, and with the increasing trend of obesity, it is crucial to further investigate and understand the potential benefits and challenges of utilizing GLP-1 RAs and SGLT-2 inhibitors in this population. Continued research efforts are essential to optimize the management of these complications and enhance the patient survival and graft survival of liver transplant recipients.
Conflict of Interest:
None
Acknowledgements:
None
References
1. American Liver Foundation. Fatty Liver Disease. https://liverfoundation.org/about-your-liver/facts-about-liver-disease/fatty-liver-disease. Accessed October 3, 2024.
2. Yanai H, Adachi H, Hakoshima M, Iida S, Katsuyama H. Metabolic-dysfunction-associated steatotic liver disease—its pathophysiology, association with atherosclerosis and cardiovascular disease, and treatments. Int J Mol Sci. 2023;24 (20):15473. doi:10.3390/ijms242015473
3. United Network for Organ Sharing. Innovations and policy updates drive equity and a record number of lifesaving organ transplants in 2023. UNOS. Published January 11, 2024. https://unos.org/media-resources/releases/innovations-policy-updates-drive-equity-and-record-organ-transplants-2023/. Accessed October 3, 2024.
4. Younossi ZM, Stepanova M, Al Shabeeb R, et al. The changing epidemiology of adult liver transplantation in the United States in 2013-2022: the dominance of metabolic dysfunction–associated steatotic liver disease and alcohol-associated liver disease. Hepatology Commun. 2023;8(1). doi:10.1097/HC9.0000000000000352
5. Burra P, Becchetti C, Germani G. NAFLD and liver transplantation: disease burden, current management and future challenges. JHEP Rep. 2020;2(6):100192. doi:10.1016/j.jhepr.2020.100192
6. Pham P, Sarkar M, Pham P, Pham P. Diabetes mellitus after solid organ transplantation. Endotext. Published July 13, 2022. https://www.ncbi.nlm.nih.gov/books/NBK378977/. Accessed October 3, 2024.
7. Martín-Timón I. Type 2 diabetes and cardiovascular disease: have all risk factors the same strength? World J Diabetes. 2014;5(4):444-470. doi:10.4239/wjd.v5.i4.444
8. Shivaswamy V, Boerner B, Larsen J. Post-transplant diabetes mellitus: causes, treatment, and impact on outcomes. Endocr Rev. 2016;37(1) :37-61. doi:10.1210/er.2015-1084
9. Florencia Z, Christos M. Adiponectin in insulin resistance: lessons from translational research. Am J Clin Nutr. 2009;91(1):258S-261S. doi:10.3945/ajcn.2009.28449C
10. Serna-Higuita LM, Isaza-López MC, Hernández-Herrera GN, et al. Development and validation of a new score to assess the risk of posttransplantation diabetes mellitus in kidney transplant recipients. Transplantation Direct. 2023; 9(12). doi:10.1097/txd.0000000000001558
11. Lindsay RS, Funahashi T, Hanson RL, et al. Adiponectin and development of type 2 diabetes in the Pima Indian population. Lancet. 2002;360 (9326):57-58. doi:10.1016/s0140-6736(02)09335-2
12. Cullaro G, Verna EC, Lee BP, Lai JC. Chronic kidney disease in liver transplant candidates: a rising burden impacting post–liver transplant outcomes. Liver Transpl. 2020;26(4):498-506. doi:10.1002/lt.25694
13. Ahmed SH, Biddle K, Augustine T, Azmi S. Post-transplantation diabetes mellitus. Diabetes Ther. 2020;11(4):779-801. doi:10.1007/s13300-020-00790-5
14. Poltronieri Pacheco M, Carneiro-D’Albuquerque LA, Ferraz D. Current aspects of renal dysfunction after liver transplantation. World J Hepatol. 2022;14(1):45-61. doi:10.4254/wjh.v14.i1.45
15. Robertson FP, Yeung AC, Male V, et al. Urinary neutrophil gelatinase associated lipocalins (NGALs) predict acute kidney injury post liver transplant. HPB (Oxford). 2019;21(4):473-481. doi:10.1016/j.hpb.2018.09.017
16. Meloni AR, DeYoung MB, Lowe C, Parkes DG. GLP-1 receptor activated insulin secretion from pancreatic β-cells: mechanism and glucose dependence. Diabetes Obes Metab. 2012;15(1): 15-27. doi:10.1111/j.1463-1326.2012.01663.x
17. Anderson JE. Combining glucagon-like peptide 1 receptor agonists and sodium–glucose cotransporter 2 inhibitors to target multiple organ defects in type 2 diabetes. Diabetes Spectr. Published April 3, 2020. doi:10.2337/ds19-0031
18. Yu JH, Park SY, Lee DY, Kim NH, Seo JA. GLP-1 receptor agonists in diabetic kidney disease: current evidence and future directions. Kidney Res Clin Pract. 2022;41(2):136-149. doi:10.23876/j.krcp.22.001
19. Richardson SH, Wong G, Garner E, Izzy M, Srivastava G. Utility of glucagon-like peptide 1 receptor agonists as anti-obesity medications in liver transplant recipients. Liver Transpl. 2024;30 (2):226-228. doi:10.1097/LVT.0000000000000233
20. Pham PTT, Pham PCT. Optimal use of SGLT2 inhibitors in diabetic kidney transplant recipients. Front Nephrol. 2022;2. doi:10.3389/fneph.2022.1014241
21. Padda IS, Mahtani AU, Parmar M. Sodium-glucose transport protein 2 (SGLT2) inhibitors. PubMed. Published 2022. https://www.ncbi.nlm.nih.gov/books/NBK576405/. Accessed October 3, 2024.
22. Berkovic MC, Virovic-Jukic L, Bilic-Curcic I, Mrzljak A. Post-transplant diabetes mellitus and preexisting liver disease – a bidirectional relationship affecting treatment and management. World J Gastroenterol. 2020;26(21):2740-2757. doi:10.3748/wjg.v26.i21.2740
23. Zheng K, Azhie A, You X, et al. Glucagon‐like peptide‐1 receptor agonists and sodium‐glucose cotransporter‐2 inhibitors for the treatment of diabetes mellitus in liver transplant recipients. Diabetes Obes Metab. 2024;26(10):4261-4272. doi:10.1111/dom.15769
24. Baar MJB van, Ruiten CC van, Muskiet MHA, Bloemendaal L van, IJzerman RG, Raalte DH van. SGLT2 Inhibitors in Combination Therapy: From Mechanisms to Clinical Considerations in Type 2 Diabetes Management. Diabetes Care. 2018;41(8): 1543-1556. doi:https://doi.org/10.2337/dc18-0588

