Role of Sleep in Epileptogenesis and Brain Networks
Sleep is a Major Participant of Epileptogenesis in Physiological Brain Networks
Péter Halász1 and Anna Szűcs2
- Szentágothai Doctoral School, Semmelweis University, Budapest, Hungary
- Institute of Behavioral Sciences, Semmelweis University, Budapest, Hungary
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Halász, P., and Szűcs, A., 2024. Sleep is a Major Participant of Epileptogenesis in Physiological Brain Networks. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.5953
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.5953
ISSN: 2375-1924
ABSTRACT
We aimed to provide a comprehensive review of the system epilepsy concept and adapt its principles to major epilepsy syndromes. We present our views on epileptogenesis: NREM sleep and especially sleep homeostatic plasticity are essential contributors of the epileptic transformation, upgrading and derailing normal brain networks. We follow the common process and specific features epileptic transformation in major epilepsies.
Keywords
sleep homeostatic power; plasticity; epileptogenesis; interictal epileptiform discharges; system-epilepsy
Introduction
Epilepsy is a „secondary disease”. A „first hit”, be it a brain lesion or a gene mutation, sets in motion the silent and years-lasting process of epileptogenesis with structural and functional changes of the affected brain network. Age and the developmental stage determine epileptogenesis that may have a common mechanism in different brain systems.
The role of plasticity in epileptogenesis has been raised for a long time. Plasticity is defined as an “activity-dependent alteration of the strength of inter-neuronal connections, through which information is stored”. First Goddard et al. supposed that the plastic process of engram-formation and epileptogenesis were similar. Bliss and Lomo demonstrated that the repetitive stimulation of a neuron may generate long-term potentiation (LTP) in another one in synaptic connection, by increasing the strength of the synapse. LTP has become the elementary model of plasticity. Kindling is similar; daily electrical stimulation may result in an epileptic process leading to spontaneous seizures. Accordingly, experimental epileptic foci that bombard distant regions with spikes, may establish secondary interictal spike foci becoming independent later. Those secondary cortical spots “learn to be epileptic” due to a progressive plastic process induced by recurrent stimulation.
Since the eighties of the past century, sleep research has penetrated into epilepsy research. NREM sleep homeostasis has been shown to be related to synaptic plasticity; NREM sleep slow wave-power seems to regulate synaptic “re-charging” as a general regulating factor of neural excitability.
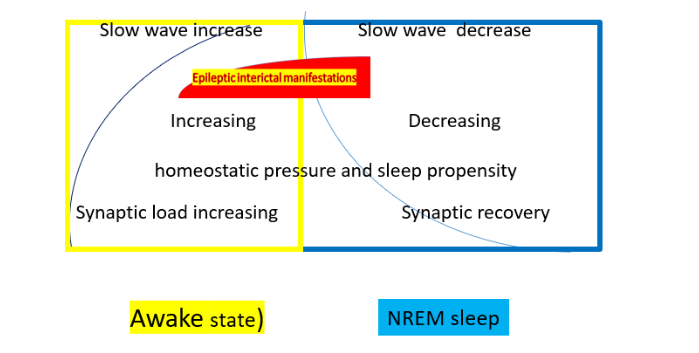
In waking, the homeostatic pressure increases due to synaptic use. Slow waves and sleep propensity increase as well. During sleep, the homeostatic pressure, slow waves and sleep propensity decrease and the synapses recover. The red highlight shows the increase of interictal epileptic discharges (IEDs) during the first sleep cycles. Sleep and waking interdigitate through antagonistic twin working modes. Non-REM (NREM) sleep is the strongest multilevel activator of ictal and interictal epileptic symptoms, while also sleep-deprivation activates seizures. NREM sleep and its deprivation countermarch, but the augmentation of homeostatic pressure (increasing sleep need) after sleep deprivation evidenced by exponentially increasing delta-power in post-deprivation sleep, renders it epileptogenic, even more than NREM sleep itself.
The epoch-making work of Beenhakker and Huguenard highlighted the crucial facts of the sleep-epilepsy relationship. They have shown in two epilepsy prone circuits (the hippocampo-frontal memory-network and the thalamocortical network) the basic features of a normal networks` epileptic transformation (Box 1) in a circuit-specific way (sleep spindle transforms to bifrontal spike-wave pattern in idiopathic „generalized„ epilepsy; and sharp wave-ripple to spike-fast ripple in medial temporal lobe epilepsy). It has become clear that normal networks provide sleep-„templates” for the epileptic variants.
Box 1
Epileptic transformation
The process of ‘epileptic transformation’ involves the following changes in a brain-system: an increase of excitability manifested in seizure-proneness; the transformation of sleep-, (sometimes also waking) EEG patterns to interictal epileptiform discharges (IEDs) in a system-specific way; and variable-degree permanent dysfunction of the system likely caused by IEDs including high frequency oscillations.
We follow Beenhakker’s and Huguenard`s way showing epileptogenesis in additional epilepsies such as the self-limited focal childhood epilepsy (SeLFE) spectrum – resulting from the transformation of the perisylvian network (PN); and frontal lobe epilepsies building upon the transformation of the arousal-system. We extend the neural network-concept (substituting the focal concept linking functions to single foci) to epilepsies, considering them as „system epilepsies”.
Box 2
System epilepsy
Seizures produced by a network, manifest its distorted functions. The idea that epilepsy ”grabs” the hosting brain-systems, has led to the concept of system epilepsy with the following implications: 1) Both the seizure-triggers and the ictal symptoms identify the hosting system; 2) The epileptic transformation favors developmental periods; and 3) it prefers plastic brain-structures such as the corticothalamic, the fronto-hippocampal and the visual system; the perisylvian network and the systems of vigilance-regulation; 4) A “second disease” character: an early precipitator injury is followed by a hidden, long-lasting ripening-period with synaptic reorganization.
We deal with ictogenesis, i.e. the issue of spontaneous versus triggered seizures. While the classic definition of epilepsies requires spontaneous seizures, in several epilepsies not belonging to the ‘reflex-epilepsy’ group, careful analysis reveals a consistent seizure-trigger, which is a certain activity or process that evokes the function of the affected brain network. Out of many examples, a simple one is a tumor in the motor strip causing symptomatic epilepsy, in which focal motor seizures are triggered by flexing the paralyzed limb. In other words, apparently spontaneous seizures are actually triggered by the activation of the affected brain network; constituting an extended reflex epilepsy concept.
In this sense, absence epilepsy (AE) would be the ‘reflex’-epilepsy of sleep-promotion, sleep related hypermotor epilepsy (SHE) would be that of the arousal-system.
Postinjury epilepsies implement an “experiment of nature” and model the essential trajectories of epileptogenesis in any brain-localization. We devote a separate section to this group.
Box 3
The hippocampo-frontal declarative memory system
The role of the hippocampus in memory formation is now evident. In the early fifties, when bilateral hippocampal resections of MTLE patients have led to catastrophic permanent amnestic states, neurology learned a big lesson about hippocampal memory functions; a whole generation of neuropsychologist has grown up with the new knowledge about memory-organization.
The team of Ian Born and the Buzsáki-school presented experimental data on the process – encoding, consolidation and recall – of memory-formation evidencing the two-steps memory model. In waking, information streams through the entorhinal cortex to the hippocampus, where transient memory-traces (engrams) are made. During the subsequent NREM sleep, those engrams are replayed and transmitted to the frontal lobe where they get consolidated in a permanent “store”.
Thalamocortical oscillations contribute to the memory process. Sleep spindles are “enveloped” by the up states of slow oscillations, and couple with ripples in learning. The fine-tuning of these interlinked oscillations render the cortex receptive for plastic changes and orchestrate the hippocampo-cortical dialogue. Spindle-density and the length of phase 2 NREM sleep increased after learning, evidencing the involvement of those oscillations in the memory-process.
Evidence linking MTLE with the mediotemporal-hippocampal network.
Hughlings Jackson has early recognized the psychic or intellectual aura” phenomenon, also termed “dreamy state”. The patient has a delusive experience (e.g. childhood scenes), while in parallel, they are aware of the reality. Jackson precisely localized this peculiar state to the uncus hippocampi by the post-mortem study of his famous patient Z, revealing a small cystic lesion in the uncus hippocampi. Later Penfield elicited memory-fragments by electric stimulation of the exposed temporal cortex of epileptic patients during surgery. Stimulating certain neocortical spots during stereotactic epilepsy-surgery later, a neuronal network far beyond the uncus hippocampi could be shown to operate behind epileptic delusion.
One of the most studied epilepsies is MTLE. It develops from an early damage of anterior limbic structures and mainly affects the CA 3 sector of the hippocampus leading to hippocampal sclerosis through synaptic reorganization and sprouting of mossy fibers. The hidden histological transformation lasts several years before the first seizure.
How does the declarative memory-function and memory-network transform to medial temporal lobe epilepsy?
While interictal spikes are eminent epileptic patterns, their role in epileptogenesis and the course epilepsy is not clear. Their hippocampal origin in (MTLE) had remained hidden until the use of foramen ovale and intracranial electrodes. Due to the ‘hiding’ of hippocampal spikes and the lack of systematic neuropsychology assessments, IEDs’ deleterious effect on memory in MTLE, had passed undetected for a long time. The close connection between hippocampal spikes and memory disturbances has been recognized late. IEDs’ cognitive harm in MTLE patients has been confirmed by sleep studies. Later studies found that hippocampal spindles correlated negatively with interictal spike-counts, suggesting that spiking lowered the rate of spindling, like the supposed swap of spindles to spike wave discharges (SWs) in AE.
The epileptic transformation renders the hippocampus a “discharging body” ejecting spikes and seizures, which impair memory especially in NREM sleep, the period of intense spiking in animals and humans. IEDs have been assumed to interfere (as „dummies”) with memory-consolidation.
The transformation of sharp wave-ripple (SpW-R) to spikes and pathological (fast, 200-500 Hz) ripples.
The most important link between the MTLE and the memory system is the transformation of SpW-R, a key graphoelement of the memory-system, to epileptic spike and pathological ripple pattern. SpW-Rs “teeter on the edge” of epileptic over-excitation wherever plastic changes occur; making the hippocampus a most epilepsy-prone structure. Physiological SpW-R’s epileptic counterpart is spike-pathological ripple. The pathological ripple is considered the essential marker of epilepsy. The transformation of physiological SpW-R (with <200 Hz ripples) to spikes and pathologic ripples marks the epileptic transformation.
Box 4
Sharp wave-ripple and memory consolidation
SpW-R is a robust, far-reaching population episode, the most synchronized event of the mammalian brain, augmenting the excitability of the hippocampus and connected structures. It originates in the CA1 region of the hippocampus in close relation with the sharp wave emerging in the CA3. It appears relaxed waking and NREM sleep, while active waking inhibits it. In humans, the ripple-component’s frequency is <150 Hz and the sharp wave is a 40-150 milliseconds lasting, ~2.5 mV amplitude pattern involving ~ 50% of the hippocampal pyramidal cells (50-150,000 neurons). The “resection” of SpW-Rs abolished memory-consolidation, evidencing their role. The finding that cellular spike-sequences in NREM sleep were identical with their sequences during SpW-Rs (just faster and compressed), supported this. Memory-consolidation takes place during consummator periods and especially NREM sleep, when recurrent spontaneous SpW-Rs emerge by the iterative potentiation of those synapses, in which the engram was encoded in waking; SpW-Rs transmit the engrams to the neocortex.
Further considerations about MTLE and memory disturbances.
We propose to continue exploring the memory-relations of MTLE seizures. The ictal ‘disturbance of consciousness’ might reflect a transient memory-loss caused by the interference of seizure with memory-recall; the patient cannot recognize self and environment. These seizures may be resulted from an ictal “knock-out” of the hippocampus. Seizure-related acute memory-loss is obvious unlike chronic memory-impairment. In the past, just cross-sectional histology studies were available. The development of neuroimaging has allowed studying structural and brain-connectivity changes, and neuropsychology has provided in vivo follow-up measurements. While a progressive loss of declarative memory affects 25% of MTLE patients, the picture is far from being clear.
Box 5
The discharging damage of the hippocampus interferes with memory consolidation
The memory disturbance in MTLE seems to have a dual nature. The hippocampal damage progressively impairs side-specific memory (reflecting the step-by-step histological transformation detected in resected surgery-samples) on one hand; and the evolving epileptic spiking (sending meaningless information in the frames of the hippocampo-frontal dialogue) obstructing memory consolidation on the other.
The main arguments supporting that MTLE is the epilepsy of the declarative memory-system, are the followings:
- The hippocampus is a protagonist of the memory process; hippocampal sclerosis and other hippocampal lesions are essential substrates of MTLE.
- Clinical and experimental data evidence the existence of memory disturbances in MTLE.
- SpW-R, the key element of the memory-process, is prone to transform to spike-pathological ripple, its epileptic variant.
Absence epilepsy and juvenile myoclonic epilepsy; the spectrum of corticothalamic system-epilepsies
Absence epilepsy
The “sleep working-mode” within the corticothalamic system. A change of the corticothalamic system’s working-mode induces sleep. The decrease of arousing stimuli coming from the ascending reticular system liberates the reticulo-thalamo-cortical loop. It sends then recurrent inhibitory stimuli towards the thalamus and cortex, which in turn produce spindling and 0.75-4.0 Hz slow oscillations (dominating NREM sleep), providing the thalamocortical structures with almost continuous inhibition. When the reticular nucleus (nRE) is destroyed, spindle-production ceases.
The recurrent inhibition from the reticular nuclei creates the so-called „burst-firing” mode throughout the thalamocortical system; manifesting sleep slow waves and coalescent spindles in scalp EEG. Absence epilepsy roots in the NREM sleep working mode of the thalamocortical network; basal ganglia are also involved.
The corticothalamic system produces bilateral 3-4 Hz spike-waves (SW). Due to the diffuse projections of the nRE to thalamic relay cells, a wide inhibitory synchronization comes off, obstructing the corticothalamic communication during SW/absences. This change interrupts the information-flow between the environment and the cortex. The pathological upgrading of the burst-firing mode may lead to the epileptic transformation of the thalamocortical network. The GABAergic neurons in the nRE complex of the thalamus inhibit each other. The strength of the intra-nuclear inhibition determines the characteristics of the thalamic oscillations. A genetic abnormality may increase the strength of inhibition, facilitating a transformation of spindle-production to spike-wave generation. In other words; spindles do transform to spike-waves as supposed by Gloor. The coexistence of spindles with SW-paroxysms in AE, accords with this mechanism.
Steriade and Contreras revealed intermittent transitions of normal NREM sleep-patterns to SW in cats. They showed that “SW originate in the neocortex and are disseminated through mono-, oligo-, and multi-synaptic intracortical circuits, before they spread to the reticular nucleus of the thalamus and exhibit generalized features.” They supported the cortical origin by several ablation experiments and stressed the focal to widespread propagation dynamics of seizures. Out of the numerous factors influencing nRE neurons’ excitability, the most powerful ones are the stimuli originating from the cortex and the substantia nigra.
In a study on rat genetic absence-model, cortical seizure spots entrained the frontal cortex, thalamic structures and the whole thalamocortical system culminating in SW-synchronization. Frontal neuroimaging changes preceding ictal SWs, support the cortical initiation of SW blurring the boundaries of focal versus generalized epilepsies; also clinical reports have verified this possibility. The data about cortical high frequency oscillations (HFO) participating in the electrophysiological picture of absences give another support to cortical involvement.
Several interesting molecular features of those driver zones have been described.
For a long period of absence research, there was a contradiction in neuroimaging (fMRI, PET, SPECT) and EEG data. Neuroimaging suggested a lack of activation, while high amplitude epileptic discharges on the EEG pointed to an increase of activity. Recent experimental work has resolved this contradiction revealing a decrease of thalamocortical neuronal activity during absences. In summary, both the focal and generalized concept of AE seems to be challenged by the finding that widespread dynamic networks outside the thalamocortical system involving the cortex and the basal ganglia, initiate the ictal symptoms.
Micro-sleep oscillations towards NREM sleep promote SW / absences. SW occur in a certain vigilance-window called “critical vigilance-level” at the transition-zones towards light NREM sleep as well as other transitional periods. Arousing stimuli and REM sleep inhibit SW/absences in humans, as well as in genetic rat models. While absences can appear in the period of wakefulness, their relation with drops of vigilance-level even within the awake-domain is evident. When arousing stimuli from sleep induce absences, the actual link with reactive sleep-like anti-arousal responses can be shown.
Box 6
The cyclic alternating pattern
The cyclic alternating pattern (CAP) is a micro-cyclicity of NREM sleep. It can appear spontaneously or be elicited by sensory stimuli. In low homeostatic-pressure periods, arousing stimuli evoke the classic EEG-arousal with desynchronization (CAP A3/A2). In periods of high homeostatic pressure, slow wave “antiarousal” responses (CAP A1) appear. This vigilance-level dependence explains absences’ distribution throughout the 24-hour sleep-wake cycle. There is a positive correlation between cyclic CAP A1 and SW. Through vigilance-level oscillations, sensory stimuli may influence the number of SW. More SW occur during fluctuations towards NREM sleep compared to shifts towards waking or REM sleep. There was significantly higher SW-count during CAP A compared to non-CAP, and CAP B inhibited SW.
The link of absences with NREM sleep-promotion is supported by the increased connectivity of the anterior thalamic structures during the falling asleep period of juvenile myoclonic epilepsy (JME) patients. In experimental works, absence seizures can be elicited by the stimulation of the sleep-promoting GABA-ergic hypothalamic ventrolateral preoptic system in WAG/Rij rats (a genetic model of human absence epilepsy).
Therefore, AE can be considered the (reflex) epilepsy of the NREM sleep-promoting system. This accords with the finding that a GABA-ergic gene mutation allows the nRE thalami producing SW/absences instead of spindles.
Absence epilepsy and juvenile myoclonic epilepsy: two members of a spectrum
Although AE is considered an age-dependent condition disappearing in adolescence, this happens only in 58-65% of patients. Fifteen to 44% of AE cases progress to JME between age 11-20 years; “where absence epilepsy decreases, JME starts”. Several novel features of JME have come to light, but its sleep-link has remained: the bilateral synchronous polyspike-waves associate with CAP A, especially in periods of high homeostatic-pressure, like the SW in AE. Generalized tonic-clonic seizures, polyspike-waves in EEG and an enhanced responsivity to transcranial magnetic stimulation (paralleling homeostatic pressure) highlight the augmented excitability in JME compared to AE. Photosensitivity and other seizure-triggers mostly related to the perisylvian network (PN) such as reading, writing, talking, music, calculation and praxis, show JME’s kinship with reflex-epilepsies. More than half of JME patients present reflex-epileptic traits; contrasting focal epilepsies where this is rare. Frontal connectivity-changes and prominent behavioral issues highlight the extension of JME’s network to the frontal lobe; the typical myoclonic jerks suggest an increased motor excitability. Therefore, JME is a multisystem epilepsy with visuo-motor dominance and widespread excitable areas involving the whole cortical mantle.
Summary
The corticothalamic system’s burst-firing working-mode produces NREM sleep. Those rhythmic alternating excitatory-inhibitory cycles of this working-mode generate spindles regulated by the nRe. Frontal cortical drivers can ignite this system, also influenced by the substantia nigra. Experimental data gained on unanaesthetized animal models reveal that during ictal SW paroxysms, the thalamocortical neuronal discharges decrease; resolving the contradictory findings of EEG and neuroimaging studies. The new findings support the unsustainability of the “generalized epilepsy” concept, and provide arguments challenging also the focal/generalized dichotomy introducing the concept of “initiating network” instead of “secondary generalization”. Absences emerge in a critical vigilance-window between wakefulness and superficial NREM sleep; full wakefulness, REM and deep slow wave sleep inhibit them. SW seem to couple with “antiarousal” slow waves (CAP A1) of the sleep microstructure, representing a dynamic shift toward the deepening of NREM sleep. AE and JME constitute the epilepsy-spectrum of the thalamocortical system; sharing absences as endophenotypes. JME involves multiple additional fields.
Sleep-related hypermotor epilepsy and the role of the ascending nicotinic acetylcholinergic arousal system
The macro- and microstructure of NREM sleep and the arousal phenomenon. Arousability and reactivity to sensory stimuli are essential features of sleep, discriminating it from coma and keeping the sleeper in contact with the environment. Microarousals (arousals without awakening) serve the reversibility of sleep and carry an alarm-function in danger. Microarousals used to be considered sleep-disturbing/destabilizing factors; however, as inherent parts of the dynamic sleep-microstructure and the CAP-system, they turned out to regulate sleep.
The concepts on the nature of sleep, have been changing. Those data revealing local/regional sleeping and waking areas, have challenged the ‘global’ sleep-idea. The normal, alternating “split-brain” i.e. one hemisphere sleeping, the other one awake; protects deep-water mammals under water and birds of passage during their long flights. There is a (use-dependent) mosaic-like pattern of different vigilance-levels in mammals including humans as well.
Sleep-related hypermotor epilepsy
SHE has an adventurous history reflected by changing denominations. The first one “nocturnal paroxysmal dystonia” was consistent with a concept of sleep-related movement-disorder. The second name ‘idiopathic nocturnal frontal-lobe epilepsy’ (NFLE) suggested the idea of an epileptic disorder as well as the most recent one ‘sleep related hypermotor epilepsy’. SHE incorporates each form of sleep-related epilepsies presenting with hypermotor seizures, irrespectively from etiologies, EEG-features, movement-types and seizure-onset zones.
In most SHE cases, hardly any IEDs occur either in waking or sleep, appearing in ~50% of patients; and a frank ictal EEG-pattern is also rare. The variable projections and seizure-spreads as well as the hidden ictal zones, hamper localization. Those symptomatic cases underlain by cortical dysplasias, seem to make a distinct SHE-population based on the abundance of IEDs appearing in this group.
Sleep related epilepsy and the cholinergic arousal-system. Acetylcholine (Ach) is an important agent of arousals. The thalamus and the frontal cortex are rich in cholinergic fibers originating from the basal nucleus of Meynert. Both trans-synaptic and non-synaptic Ach-release are significant. Most SHE cases are cryptogenic, while in a minority of patients, gain-of-function mutations in the nicotinic acetylcholine-receptor (nAChR) gene sub-unit as well as other mutations have been shown. SHE with mutant nAChR (autosomal dominant sleep related hypermotor epilepsy; ADSHE) is electro-clinically indistinguishable from the cryptogenic forms.
Disorders of arousal and SHE link to arousals. Disorders of arousal (DOA) (first systematized by Broughton) and SHE are parallel spectra of epileptic and non-epileptic “siblings”. Their “increasing-complexity” manifestations are considered as a continuum. Based on video-EEG monitoring by Derry et al, “DOA and SHE episodes present with three fundamental patterns and their composites”: (1) Simple arousal behavior (92%): eye-opening, head-elevation, staring, face-rubbing, yawning, stretching, moaning or mumbling. (2) Non-agitated arousal (72%): sitting forward, manipulating nearby objects, orientating themselves with a passive or perplexed look and motor automatisms originating from the basal ganglia. (3) A pattern with fierce emotional behaviors (51%) facial, verbal and autonomic signs of fear. The last pattern is termed hypermotor seizure in SHE, sleep terror in DOA. In these episodes, patients showed fearful arousal-behaviors, screamed, and behaved frantically. Restraining them evoked aggression. A trigger was more frequent in DOA than in SHE.
The origins of 3rd degree seizures with escape-reactions, panic-like behaviors and hypermotor features are dubious. They might reflect the epileptic activation of automatic movement “conserves” normally suppressed in sub-cortical generators. When the descending frontal inhibition ceases due to a seizure, those atavistic behaviors disengage.
Our retrospective analysis has revealed overlaps of the activated fields in DOA-events and the seizure-onset zones of hypermotor seizures, covering the anterior cingulate cortex, anterior insula and the ventral medio-frontal cortex anterior to the supplementary sensory-motor area. These regions are consistent with the hubs of the salience network (SN; a brain-network, signalizing subjectively meaningful events). Based on the activation of SN and the alarm-like symptoms seen in both conditions’ 3rd degree episodes; an over-activated SN driving an acute stress-response (fight-flight response of Cannon and Selye) has been raised recently. This fight-flight response would manifest a false alarm (escape without a chaser) in a sleeping brain. Sleep dissociation can be traced here as in other DOA episodes as confirmed by neuroimaging.
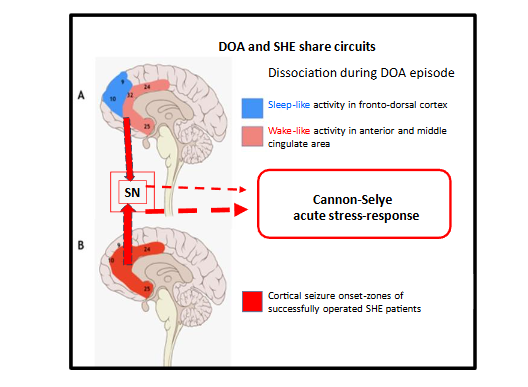
While DOA-events are rare, several SHE seizures/night may emerge. Both conditions’ episodes manifest in the first or second sleep-cycles of night-sleep accumulating in the first one; the episodes’ frequency decreases overnight paralleling the homeostatic decay. Both DOA and SHE patients have a fragmented sleep with frequent microarousals, evidencing the dominance of arousal-forces.
Genetic relations
In a survey of 100 SHE cases, more than 1/3 of patients had a family history for epilepsy and 1/3 of them had DOA. Bisulli et al. compared 33 SHE patients and their 200 relatives with 31 age, sex, education, and residence-area matched controls and 194 relatives, for the occurrence of DOA. The lifetime prevalence of DOA was more frequent in SHE patients’ relatives compared to the relatives of controls. Some DOA-cases developed SHE later.
Two recent works reviewed the complex genetic relations of SHE, especially ADSHE (having an estimated incidence of 1.8/100000). In a study on 103 SHE patients, 4 kinds of mutation have been identified in altogether 8% of patients; CHRN4 (2.9%), KCTI (1.0%), DEPDC5 (3,9%), NPRL2 (1.0%). The DEPDC5 variant favoured patients with structural lesions. Another study revealed additional gene mutations (CHRNA4, CHRNB2, CHRRNA2). The gain of function mutations affecting the nicotinic acetylcholine receptors (CHRNA4, CHRNB2, CHRRNA2) fit well with the hypersensitivity of the ascending arousal system causing SHE seizures.
While population-genetic findings strongly link SHE and DOA; the molecular genetic relations are less clear. An increased cholinergic excitation leading to epilepsy is evident in ADSHE, but the only feature supporting the cholinergic upregulation in DOA, is the fragmented sleep of those patients. However, the similarity of the two conditions’ episodes, the shared link to NREM sleep microarousals, and similar sleep-distribution, suggest a common origin.
AE and SHE manifest the epileptic transformation of the antagonistic sleep-, and arousal-promoting systems. AE is resulted from the derailment of normal corticothalamic function – producing sleep spindles and slow waves; SHE is built upon the epileptic facilitation of arousal from sleep.
Self-limiting focal childhood epilepsies represent an epilepsy spectrum of the perisylvian human communication network.
Composition of the spectrum. The understanding of self-limiting childhood focal epilepsies (SeLFE) has been changing; the most recent modification was published in 2022. SeLFE accounting for ~20% of childhood epilepsies include Rolandic epilepsy (RE), Panayiotupuolos syndrome (PS), Gastaut’s late childhood occipital epilepsy (GOE) and photosensitive occipital epilepsy (POE).
Rolandic epilepsy. We focus here on one branch of the spectrum from self-limiting epilepsy with centrotemporal spikes (SeLECTS, traditionally termed Rolandic epilepsy – RE – or benign centrotemporal epilepsy), where centrotemporal spikes (CTS, Rolandic spikes) couple with ripples; atypical SeLECTS; and epileptic encephalopathy with spike-wave activation in sleep (EE-SWAS; electrical status epilepticus in sleep -ESES- and its regional variant Landau-Kleffner syndrome – LKS). The sparse CTS appearing without ripples in the sleep of children with autism spectrum- and attention-deficit hyperactivity disorder (ADHD), loosely link also these conditions to the spectrum.
We do not deal here with PS that is an autonomic epilepsy with occipital IEDs, a vague seizure-onset zone; and partially overlapping genetics with SeLECTS. We do not incorporate GOE and POE either, because they don’t show the severity-order as SeLECTS and the related conditions above do.
SeLFE may mutually transform to each other. They occur in a common age-window between 3-10 years with a peak at age 6–7; and typically recover by 15–16. Cognitive impairment is an essential feature of the SeLECTS spectrum.
The host of these epilepsies is the perysilvian network (PN). The perisylvian cortex, widely covering the frontal-prefrontal temporal and temporo-occipital cortices, deserves the name ‘human communication system’ for its key roles in functions related to speaking, reading and writing. Language research has revealed the enormous development of the PN from chimpanzees to humans; associated with an enlargement of that region, most spectacular in higher association cortices.
The human language network interweaves the sensory, motor, auditory and articulatory machinery and occupies the perisylvian cortex with premotor, anterior temporal and, temporo-occipital associative fields. The main information stream is organized through the dorsal and ventral pathways. The dorsal arcuate pathway, bi-directionally connecting the Broca and Wernicke areas, is involved in executive and comprehensive language functions. This bundle has importantly increased during human evolution allowing faster transmission of information. Communication functions developed in close relation to cognitive abilities. They require high-level plasticity, substantially represented by the large perisylvian cortex. It is no wonder that this multifunctional and strategically important territory may harbor several forms of epileptic derailment.
Box 7
The centrotemporal spike
A ~88 milliseconds-duration centro-temporal sharp-wave is termed CTS. Its EEG amplitude-map shows a characteristic frontal positive and temporal negative tangential dipole perpendicular to the Sylvian fissure, followed by a lower amplitude slow wave of generalized SW. Its topography is diverse; preferring the face-field of the somatomotor gyrus. Abundant spiking may emerge from the opposite hemisphere to seizure-onset zones; the spikes may be independent bilateral or poly-focal even within one recording. CTS are not necessarily concordant with the seizure-onset zones and interestingly, they may slide wide around the PN. CTS accumulate during NREM sleep, the strongest activation (highest amplitude, broadest field and bilateralization of potentials) occurring in N3 sleep, on the descending slopes of the first sleep-cycles. Its frequency varies from rare to frequent. Unlike most epilepsies where IEDs couple with CAP A1 slow waves, CTS associate with spindles. The significance of IEDs’ diverse coupling with sleep-components is unclear; it may be related to IEDs’ cognitive relations.
Box 8
The neuroimaging background of centrotemporal spikes
The development in neuroimaging allowed studying the morphology-features and networks underlying IEDs, including CTS. Several studies revealed deficits of brain-maturation, decreased fractional anisotropies in subcortical structures (e.g. the putamen and caudate nucleus), as well as in the sensorimotor and language-networks of SeLECTS children. There were complex connectivity-changes and disorganization in the small world of the epileptogenic zone and in a widespread related network, preceding and following CTS in a 400ms timespan.
Interictal and ictal symptoms. Most SeLECTS patients experience <10 seizures; 10-20% of them present with just one. The seizures favor NREM sleep or drowsiness; the probability of awake seizures is less than 10%. The CTS is an endophenotype overarching the SeLECTS spectrum, including non-epileptic relatives. CTS occurs in the sleep EEGs of 1-2% of healthy 5-12-year-old children, and only 18-25% of CTS-holders have epilepsy. Rarely, CTS occurs during sleep in autism spectrum and ADHD-children.
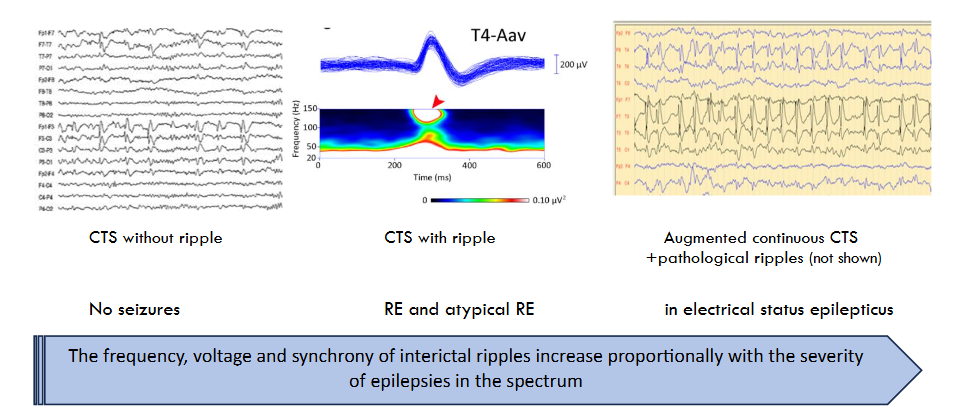
The ‘epileptic’ CTS associates with a HFO (ripple crown), while there is no ripple if CTS doesn’t associate with epilepsy. Most CTS fade away with age. In some CTS-carriers, a peculiar giant middle-latency somatosensory potential (SEP), can be evoked by electrical stimulation of the median nerve or touch; disappearing before age 12 alike CTS. Since the main negative component of the giant SEP resembles CTS, and there are similar giant SEPs in myoclonic syndromes, the possibility of giant SEP’s involvement in epileptogenesis can be raised. The parameters of HFO mark the clinical severity of spectrum-members; the number of ripples (unlike spikes) correlating with the numbers of seizures. Ripples appear on CTS in SeLECTS, more ripples associate in atypical SeLECTS, and when EE-SWAS evolve, the number, voltage and amplitude of ripples augment further.
Atypical Rolandic epilepsy: a missing link of the SeLECTS spectrum.
The warning signs forecasting an encephalopathic progression were first summarized by Fejerman; and confirmed by others. Those warning sings included atypical seizures, oro-motor status epilepticus, a more important cognitive loss, and higher frequency ripples than in the “benign” forms; SW-runs, frequent CTS, and an abnormal EEG-background. The atypical forms may represent transit-stops towards the EE-SWAS or genetically distinct variants.
The progress of SeLECTS to epileptic encephalopathies (electrical status epilepticus in sleep and Landau-Kleffner syndrome). A study on 196 SeLECTS patients suggested that ~7% of them progressed to epileptic encephalopathies (status electricus in sleep-ESES- and Landau-Kleffner syndrome (EE-SWAS) representing 65% of the “atypical” forms. About one-third of ESES cases had a previous diagnosis of SeLECTS.
The history of EE-SWAS has begun with the report on a “peculiar EEG pattern occurring almost continuously during sleep, characterized by apparently subclinical spike-and-waves for variable length of time (months to years)” in six children with cognitive deficits. After this first publication, Tassinari et al introduced the term “encephalopathy related to electrical status epilepticus during slow wave sleep” (ESES). They suggested that “the condition of a protracted (years) status electricus in sleep can be the factor leading to severe mental deterioration and psychic disturbances.” Initially, ESES was deemed rare but more and more cases have been identified later. Due to its broader ‘reputation’ and looser definition, it has become a relatively frequent diagnosis recently. More than 50% of patients have a severe lesional background, and the rest evolve from SeLFE.
First Kellerman documented patients with acquired epileptic aphasia (LKS). The children had localized activation of abundant spike-and-slow wave discharges during NREM sleep consistent with a regional ESES, therefore, several authors have considered LKS an ESES-variant. The evolving aphasia of LKS points to a circumscribed dysfunction of the speech-related perisylvian (Wernicke) network in the posterior part of the first temporal convolution.
SeLECTS and the malignant EE-SWAS used to be considered distinct entities. The supposed big gap between the cognitive outcomes hindered their unification, and also the terminology-chaos caused confusion. However, the recognition of subtle language-dysfunctions in SeLECTS, and even more of them in the atypical forms, facilitated their convergence. Sleep patterns’ epileptic transformation in ESES/LKS and the progress to those severe phenotypes; as well as the genetic relations need further research.
The anatomo-functional background of neuropsychological symptoms in SeLECTS
There are variable neuropsychological symptoms belonging to the self-limited spectrum. They originate in a wide territory of the hosting PN (a rich assembly is given in the exemplary work of Guliyeva et al). Those symptoms may be related to a developmental delay revealed by mild and transient morphological changes (e.g. cortical thinning). CTS emerge early; while seizures may evolve later from the motor strip’s intra-sylvian part; an apparently different area compared to the supposed wide region of neuropsychology symptoms. In view of the rare development of LKS from the milder SeLECTS conditions; a continuity of early and mild language-symptoms and the tragic LKS-symptoms needs to be considered.
Summary
The conditions hosted by the broad PN and association cortices, make a spectrum from typical and atypical SeLECTS to EE-SWAS. These conditions meet the criteria of system epilepsies. The progressive epileptic transformation of the PN may have genetic and developmental origins. The severity of epilepsies within the spectrum is signalized by ripple-parameters. The IEDs of EE-SWAS seem to be consistent with augmented, synchronized and bilateralized CTS with pathological HFO. Each type of ripples enhances in NREM sleep. NREM sleep is a central player or a master-card; participating in each essential symptom of the SeLECTS-spectrum. It seems contradictory, that CTS and seizures localize to the intra-sylvian cortex, while the cognitive loss associates with much wider regions, without consequent traces of epileptic discharges. Those findings showing cognitive deficits before the appearance of epilepsy offer a clue: SeLECTS might emerge in a developmentally compromised brain; a secondary epileptic-derailment of a handicapped PN (as suggested by early cortical thinning) may be assumed.
Reading-, and language-induced epilepsies of the perisylvian network
In addition to reading, there are various seizure-triggers denominated by the term reading epilepsy (EwRIS); including calculation, writing, shorthand, higher cognitive tasks and plays. Therefore, the term “language-induced epilepsy” has been suggested. The PN hosting complex communication-functions is vulnerable to genetic, developmental or early hypoxic damages that may lead to variable epileptic transformations. The individual organization and interconnections of the involved functions, result in colorful syndrome-patterns.
Wolf reviewed 111 EwRIS cases. The seizures have been suggested to occur within a wide-spread neuronal network with no single focus. A polygenic etiology has been assumed. Recently Puteikis et al summarized the demographic, clinical and imaging data of 101 cases collected by PubMed and Web of Science search; published between 1991 and 2022. Reading aloud was a stronger seizure-trigger than silent reading and if reading required concentration, seizure-occurrence was more likely. Seizures followed reading with some delay; the epilepsy-prone system needing to “warm up”. The abnormal interictal and ictal EEGs had various extensions and localizations mostly in the dominant hemisphere. Sleep records were not mentioned in the paper. MRI findings were reported in ~50% of patients; 14% were abnormal. More sophisticated imaging and EEG data were available in 26% of cases. Functional imaging and magnetic source localization studies showed first activation over the frontal lobe and in the supplementary sensory-motor area region, but the results were inconsistent. In addition to typical oro-facial myocloni, other ictal symptoms including visual or dyslexic ones occurred.
The network of juvenile myoclonic epilepsy and reading-induced seizures
Focal triggering (such as reading etc.) of evoked seizures in the frameworks of JME occur, contrasting its supposed “primary generalized” nature. In JME, there is a widespread epileptic network primarily including the visual and motor cortices besides the thalamocortical system. This network of facilitated subsystems accounts for the myoclonic jerks, photosensitivity; as well as the occurrence of reading-induced apparently ‘generalized’ seizures (myoclonic, tonic-clonic or absences) of JME. While JME is held to be a ‘generalized’ epilepsy, many of its seizures might be related to the epileptic hypersensitivity of focal areas entraining the thalamocortical system.
EwRIS differs from SeLFE conditions in its obvious stimulus dependence only. The proximity of SeLECTS and EwRIS is supported by a report on a Rolandic epilepsy patient, who has progressed to EwRIS later.
Commonalities of epileptogenesis related to focal brain injury
In 2018, a group of researchers issued a review „to examine the evidence for possible commonalities in epileptogenic processes”, in different types of brain injuries and acquired epilepsies. They explored, whether evidence from animal-models could be adapted for human epilepsy, finding several shared features of animal and human epileptogenesis as well as of post-injury and other acquired epilepsies.
Post-injury epileptogenesis and its experimental model. After any brain insult, acute symptomatic seizures may emerge within the first days post trauma. Head-traumas, cerebrovascular accidents or other brain-injuries cause 20-60% of epilepsies. Most acute symptomatic seizures is followed by a second one in the next two years, however, 25-40% long-term remission rate has been reported. The estimated rate of early post-ischemic stroke-seizures range from 2% to 33%, and the incidence of early seizures and epilepsy after parenchymal brain hemorrhages is 11.8–15.4%.
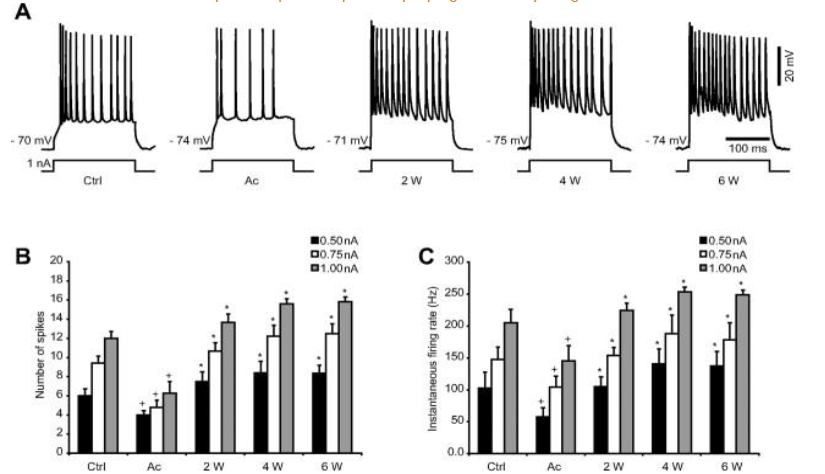
The Steriade-school studied posttraumatic epileptogenesis in the nineties, Timofeev and co-workers have continued this research. The starting point was that isolated neo-cortical areas were hyper-excitable and produced local suppression-burst activity. It was hypothesized, that the bursts had a homeostatic stabilizing function, compensating low neuronal activity after de-afferentation. The results supported the role of homeostatic synaptic. After a large transection of white matter underneath the suprasylvian cortex producing the isolated cortex model, Avramescu and Timofeev studied cats with intra-, and extra-cellular electrodes for 2-, 4 and 6 weeks. The EEG-parameters were compared with those seen before surgery. First, a low neuronal activity of the de-afferented slabs had appeared. Later, this activity increased, reaching the level of paroxysmal high amplitude bursts; finally, in free-moving animals, epilepsy developed. They attributed the increase of activation to a homeostatic compensating process in response to de-afferentation; leading to an uninhibited facilitation (overshot) of homeostatic plasticity. In sleep, they found an increase of the silent down-states (hyperpolarized periods) of the bi-stable slow oscillation.
Recently, an interesting work of Massimini appeared, evidencing a special bistability-like compensating process after stroke-related brain injuries; penetrating into NREM sleep, wakefulness and REM sleep. These studies confirm the importance of slow wave bistability in homeostatic healing processes after brain damages and the potential (inherent) epileptic derailment.
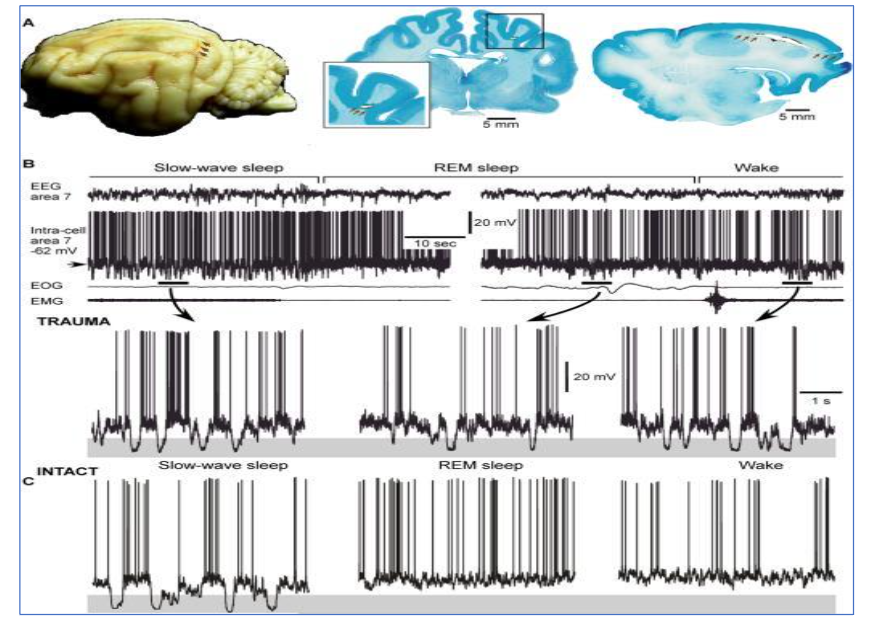
Figure 7: Cat brain depicting the localization of the undercut (arrows), global view – left, frontal section – middle and sagittal section – right. B. Intracellular and field potential recordings during different states of vigilance. Epochs indicated by horizontal bars are expanded below. Note the presence of large amplitude hyperpolarizing potentials, indicated by shadowed area, during NREM, REM sleep and waking (in the highlight). C. Intracellular neuronal recording in the intact cortex. EOG = electro-oculogram, EMG = electromyogram, TRAUMA = injured animal (Modified from Timofeev et al 2010) (108).
Discussion
In this paper, we applied the system-epilepsy concept, for studying epileptogenesis; a long-lasting process whatever the precipitating agent is. This concept is not new. The epileptic derailment of NREM sleep homeostatic plasticity is an inherent hazard. In epilepsies we reviewed, one may recognize the common ways of transformation; normal sleep-EEG patterns serving plasticity turn to epileptic transients. The features of the affected systems and their relation to sleep, determine the types of transformations. Postinjury epilepsy seems to be a good model of epileptic transformation, deserving further studies.
The epileptic derailment in the vigilance-regulating twin-systems (AE linked to sleep promotion and SHE linked to the arousal system) is special, highlighting that the normal functioning of an epilepsy-prone system, may trigger seizures.
The link between DOA and SHE is also worth discussing. The first one is a sleep disorder; the second one is epilepsy, while they share several features, such as sleep-relatedness, symptoms, genetic overlaps, and an arousal-related sleep-dissociation mechanism with autonomic, motor and emotional arousals occurring within a sleeping brain. The regions of activation in DOA-episodes, overlap with the seizure-onset zones of SHE-seizures and co-localize with hubs of the salience network. Since the SN is interconnected with the hypothalamo-hypopituitary-adrenal axis producing the Cannon-Selye type acute stress reaction, DOA and hypermotor seizures may both manifest false alarm-symptoms (without any danger).
In epilepsies we reviewed, IEDs in NREM sleep carry more important roles in epileptogenesis than seizures. In case of SeLECTS, IEDs (CTS) appear to be the functional building-stones signalizing the state of the epileptic process both in progression and in regression. It is unclear whether the paramount role of NREM sleep seen in childhood epilepsies prevails also in epilepsies starting in adulthood; featured by less plasticity.
In contrast to HFO, traditional IEDs’ (spikes’) rate is not a measure of seizure-threshold or clinical severity. Those epilepsies with abundant IEDs and hardly any seizures, challenge the old dogma that epilepsy does not exist without seizures.
There is an implicit agreement that „sleep-related epilepsy” means sleep-related seizures. We feel that also IEDs need to be taken into account in this respect. Recently, the development of fMRI has helped understanding the hidden network and effect of IEDs, including CTS.
A big mass of evidence on IEDs’ influence on the cognitive sphere has come from studies on SeLECTS children. It has been evidenced that SeLECTS may progress to EE-SWAS, where augmented CTS flood NREM sleep in association with increased pathological HFO.
CTS may signalize a time-limited regional delay of cortical development with increased cortical excitability that can progress to epilepsy. Without this epileptic evolution, normal development may restore and CTS disappear, while a further epileptic evolution may interfere with cognitive development.
IEDs interfere with sleep-plastic functions, therefore, the actual cognitive loss originates from the impairment of NREM sleep-related synaptic plasticity.
SeLFE and reading-induced seizures seem to be the products of PN, hosting language functions. PN anchors several known variants of epileptic derailment in the human communication-system, the majority waiting for categorization.
NREM sleep-homeostatic plasticity has a major role in epilepsy. Homeostatic plasticity is a compensatory (healing) process, correcting or recovering cell-loss or functional decrease, however, it goes together with the increase of excitability that may reach an epileptic level. In system-epilepsies we investigated, we found supports to this attractive theoretical model. During epileptogenesis, normal functions partially transform to an epileptic working mode.
Conclusion
The epileptic transformation can be traced in several physiological brain systems. Epileptogenesis is intertwined with sleep. Sleep-homeostatic plasticity repairs any disturbance of NREM sleep, it provides a strong protection and recovery-mechanism for the brain enabling it to develop; but it also carries a risk of epilepsy. If the powerful NREM sleep homeostatic-force derails, sleep EEG patterns may transform to epileptic spikes and pathological ripples.
NREM sleep is a mediator, in which exaggerated homeostatic plasticity may transform physiologic networks to dysfunctional and discharging epileptic systems; up-and-down states of sleep slow oscillations may play a key role. Epilepsy seems to be the most important sleep disorder. The parameters of pathologic HFO signalize epileptogenesis and the epileptic progress. NREM sleep potentiates epileptic phenomena, which in turn, interfere with sleep-functions. This vicious circle working every night, may transform brain-functions, fuel seizures and cognitive impairment, especially in flexible childhood periods and plastic systems of development. MTLE, AE, JME, SHE and SeLFE (including language induced reflex-epilepsies), are hosted by plastic brain systems. Each of them links to sleep homeostatic plasticity. The system-epilepsy concept has led us to a potential new approach of epilepsy taxonomy, which would consider spectral lines, too.
Practical consequence:
Whole-night EEG is not simply an “activation” procedure (like hyperventilation), but a sampling probe revealing the dynamics of the epileptic process, acting unnoticeably every night. Therefore, whole night EEG studies are most important diagnostic tools to be used in each patient with epilepsy.
Suffix
This paper does not systematize all epilepsies according to the system approach. The most important shortage is the scrutiny of epileptogenesis in the visual system. The understanding of the photo-paroxysmal response, the axial phenomenon related to visual system-epilepsy, is still lacking; it is present in a much wider-, even non-epileptic-population, beyond epileptic patients. The biology of photosensitivity would exceed the frames of the present review.
References
- Pitkänen A, Engel J Jr. Past and present definitions of epileptogenesis and its biomarkers. Neurotherapeutics. 2014;11(2):231-41. Doi:10.1007/s13311-014-0257-2.
- Klein P, Dingledine R, Aronica E, et al. Commonalities in epileptogenic processes from different acute brain insults: Do they translate? Epilepsia.2018;59(1):37-66. Doi:10.1111/epi.13965.
- Steriade M. Neuronal substrates of sleep and epilepsy. Cambridge: University Press. 2003.
- Goddard GV, McIntyre D, Leech C. A permanent change in brain function resulting from daily electrical stimulation. Exp. Neurol 1969; 25:295–330.
- Bliss TV, Lomo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J Physiol. 1973;232(2):331-56.
- Wada JA, Sato M. Generalized convulsive seizure state induced by daily stimulation of the amygdala in split-brain cats. Paper presented at the 27th annual meeting of the American Electroencephalographic Society. 1973. Boston.
- Morrell F. Goddard’s kindling phenomenon: A new model of the “mirror focus”. In: H. C. Sabel Ji (Ed.) Chemical modulation of brain New York: Raven 1973.
- Morrell F, deToledo-Morrell L. From mirror focus to secondary epileptogenesis in man: an historical review. Adv Neurol 1999;81:11-23.
- Tononi G, Cirelli C. Sleep and synaptic homeostasis: a hypothesis. Brain Research Bulletin 2003;62(2):143-50.
- Tononi G, Cirelli C. Sleep function and synaptic homeostasis. Sleep Medicine Reviews 2006;10(1):49–62.
- Tononi G, Cirelli C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 2014; 81(1):12-34.
- Gibbs: Gibbs FA, Gibbs EL. Atlas of electroenecephalography 1,2 1950.
- Nobili L, Frauscher B, Eriksson S, et al. Sleep and epilepsy: A snapshot of knowledge and future research lines. J Sleep Res. 2022;31(4):e13622.
- Beenhakker MP, Huguenard JR. Neurons that fire together also conspire together: is normal sleep circuitry hijacked to generate epilepsy? Neuron 2009;62:612–32.
- Halász P. Are absence epilepsy and nocturnal frontal lobe epilepsy system epilepsies of the sleep/wake system? Behav Neurol 2015; 231676.
- Avanzini G, Manganotti P, Meletti S, et al. The system epilepsies: a pathophysiological hypothesis. Epilepsia. 2012; 53: 71-8.
- Wolf P, Yacubian EM, Avanzini G, et al. Juvenile myoclonic epilepsy: A system disorder of the brain. Epilepsy Res 2015;114:2-12.
- Striano P, Striano S. Reading epilepsy and its variants: a model for system epilepsy. Epilepsy Behav. 2011;20(3):591. Doi:10.1016/j.yebeh.2011.01.021.
- Halász P, Kelemen A, Clemens B, et al. The perisylvian epileptic network. A unifying concept. Ideggyogy Sz. 2005; 5920;58(1-2):21-31.
- Szűcs A, Rosdy B, Kelemen A, Horváth A, Halász P. Reflex seizure triggering: Learning about seizure producing systems. Seizure. 2019;69:25-30. Doi:10.1016/j.seizure.2019.03.019.
- Scoville WB, Milner B. Loss of recent memory after bilateral hippocampal lesions. J Neurol Neurosurg Psychiatry 1957;20(1):11–21. Doi:10.1136/jnnp.20.1.11.
- Born J. Rasch B. Gais S. Sleep to remember. Neuroscientist 2006;12(5):410-24.
- Rasch B, Born J. About sleep’s role in memory. Physiol Rev 2013;93(2):681-766. Doi:10.1152/physrev.00032.2012.
- Buzsáki G. Hippocampal Sharp Wave-Ripple: A cognitive Biomarker for Episodic Memory and Planning. Hippocampus. 2015;25–8, 10738.
- Clemens Z, Mölle M, Erőss L, et al. Fine-tuned coupling between human parahippocampal ripples and sleep spindles. Eur J Neuro Sci. 2011;33:511-20.
- Latchoumane CV, Ngo HV, Born J, Shin, HS. Thalamic spindles promote memory formation during sleep through triple phase-locking of cortical, thalamic, and a hippocampal rhythms. Neuron. 2018;95:424–35.
- Staresina BP, Niediek J, Borger V, Surges R, Mormann F. How coupled slow oscillations, spindles and ripples coordinate neuronal processing and communication during human sleep. Nat Neurosci. 2023;26(8):1429-37. Doi:10.1038/s41593-023-01381-w.
- Mölle M, Born J. Slow oscillations orchestrating fast oscillations and memory consolidation. Prog Brain Res. 2011;193:93-110. Doi:10.1016/B978-0-444-53839-0.00007-7.
- Fogel SM, Smith CT. Learning-dependent changes in sleep spindles and Stage 2 sleep. J Sleep Res. 2006;15:250–5.
- Gais S, Molle M, Helms K, Born J. Learning-dependent increases in sleep spindle density. J Neurol Sci. 2002;22:6830–4.
- Schabus M, Gruber G, Parapatics S, et al. Sleep spindles and their significance for declarative memory consolidation. Sleep 2004;27(8):1479-85. Doi:10.1093/sleep/27.7.1479.
- Jackson JH. On a particular variety of epilepsy (“intellectual aura”), one case with symptoms of organic brain disease. Brain. 1888;11:179–207. Doi:10.1093/brain/11.2.179.
- Penfield W, Jasper HH. Epilepsy and the Functional Anatomy of the Human Brain.1954. Little Brown and Company – Boston.
- Bancaud J, Talairach J. Sémiologie clinique des crises du lobe temporal (méthodologie et investigations SEEG de 233 malades). In: Crises épileptiques et épilepsies du lobe temporal, tome II. Gentilly: documentation médicale Labaz, 1991. Boston: 1954 Little, Brown.
- Sano K, Malamud N. Clinical significance of sclerosis of the cornu Ammonis: ictal psychic phenomena. Arch. Neurol. Psychiat (Chic.) 1953;70:40–53.
- Margerison JH, Corsellis JA. Epilepsy and the temporal lobes. A clinical, electroencephalographic and neuropathological study of the brain in epilepsy, with particular reference to the temporal lobes. Brain. 1966;89(3):499-530. Doi:10.1093/brain/89.3.4991007.
- Babb TL, Kupfer WR, Pretorius JK, Crandall PH, Levesque MF. Synaptic reorganization by mossy fibers in human epileptic fascia dentata. Neuroscience. 1991;42:351-63.
- Represa A, Pollard H, Moreau J, Ghilini G, Khrestchatisky M, Ben-Ari Y. Mossy fiber sprouting in epileptic rats is associated with a transient increased expression of alpha-tubulin. Neurosci Lett. 1993;156(1-2):149-52. Doi:10.1016/0304-3940(93)90460-3.
- Tauck DL, Nadler JV. Evidence of functional mossy fiber sprouting in hippocampal formation of kainic acid-treated rats. J Neurosci 1985;5:1016-22.
- Turski WA, Cavalheiro EA, Schwarz M, Czuczwar SJ, Kleinrok Z, Turski L. Limbic seizures produced by pilocarpine in rats: behavioural, electroencephalographic and neuropathological study. Behav Brain Res. 1983;9:315-35.
- Maglóczky Z, Freund TF. Impaired and repaired inhibitory circuits in the epileptic human hippocampus. Trends Neurosci. 2005;28(6):334-40. Doi:0.1016/j.tins.2005 04.002.
- Clemens Z, Janszky J, Szűcs A, Békésy M, Clemens B, Halász P. Interictal epileptic spiking during sleep and wakefulness in mesial temporal lobe epilepsy: a comparative study of scalp and foramen ovale electrodes. Epilepsia 2003;44:186-92.
- Shatskikh TN, Raghavendra M, Zhao Q, Cui Z, Holmes GL. Electrical induction of spikes in the hippocampus impairs recognition capacity and spatial memory in rats. Epilepsy Behav. 2006;9:549-56.
- Kleen J, Scott RC, Holmes GL, et al. Hippocampal interictal epileptiform activity disrupts cognition in humans. Neurology. 20131:18–24.
- Lambert I, Tramoni-Negre E, Lagarde S, et al. Accelerated long-term forgetting in focal epilepsy: Do interictal spikes during sleep matter? Epilepsia. 2021;62(3):563-569. Doi:10.1111/epi.16823.
- Gelinas JN, Khodagholy D, Thesen T, Devinsky O, Buzsáki G. Interictal epileptiform discharges induce hippocampal-cortical coupling in temporal lobe epilepsy. Nat Med. 2016;22: 641-8.
- Frauscher B, Bernasconi N, Caldairou B, von Ellenrieder N, Bernasconi A, Gotman J. Interictal Hippokampal Spiking Influences the Occurrence of Hippokampal Sleep Spindles. Sleep 2015;38(12):1927-33. Doi:10.5665/sleep.5242.
- Gloor P. Generalized cortico-reticular epilepsies. Some considerations on the pathophysiology of generalized bilaterally synchronous spike and wave discharge. Epilepsia. 1968;9(3):249-63. Doi:10.1111/j.1528-1157.1968.tb04624.x.
- Frauscher B, Bartolomei F, Kobayashi K, et al. High-frequency oscillations: The state of clinical research. Epilepsia. 2017;58(8):1316-1329. Doi:10.1111/epi.13829.
- Gulyás AI, Freund TT. Generation of physiological and pathological high frequency oscillations: the role of perisomatic inhibition in sharp-wave ripple and interictal spike generation. Curr Opin Neurobiol. 2015;31:26-32. Doi:10.1016/j.conb.2014.07.020.
- Hermann B, Seidenberg M, Lee EJ, Chan F, Rutecki P. Cognitive phenotypes in temporal lobe epilepsy. J Int Neuropsychol Soc. 2007;13(1):12-20.
- Hermann B, Conant LL, Cook CJ, et al. Network, clinical and sociodemographic features of cognitive phenotypes in temporal lobe epilepsy. Neuroimage Clin 2020;27:102341. Doi:10.1016/j.nicl.2020.
- Steriade M. (2003). Neuronal substrates of sleep and epilepsy. Cambridge University Press, 322-48.
- Steriade M. Synchronized activities of coupled oscillators in the cerebral cortex and thalamus at different levels of vigilance. Cereb Cortex. 1997;7(6):583-604.
- von Krosigk M, Bal T, McCormick DA. Cellular mechanisms of a synchronized oscillation in the thalamus. Science. 1993;261:361–4.
- Steriade M, Contreras D. Relations between cortical and thalamic cellular events during transition from sleep patterns to paroxysmal activity. J Neurosci. 1995;15(1 Pt 2):623-42.
- Crunelli V, Lőrincz ML, McCafferty C, et al. Clinical and experimental insight into pathophysiology, comorbidity and therapy of absence seizures, Brain, 143(8),2020, 2341–68.
- Meeren H, Pijn JP, Van Luijtelaar EL, Coenen AM, Lopes da Silva FH. Cortical focus drives widespread corticothalamic networks during spontaneous absence model of corticoreticular epilepsy. Trans Am Neurol Assoc. 2002;98:203–5.
- Meeren H, van Luijtelaar G, Lopes da Silva F, Coenen A. Evolving concepts on the pathophysiology of absence seizures: the cortical focus theory. Arch Neurol. 2005;62:37.
- Moeller F, Siebner HR, Wolff S, et al. Simultaneous EEG-fMRI in drug-naive children with newly diagnosed absence epilepsy. Epilepsia. 2008;49(9):1510-9. Doi:10.1111/j.1528-1167.2008.01626.x.
- Aung T, Tenney JR, Bagić AI. Contributions of Magnetoencephalography to Understanding Mechanisms of Generalized Epilepsies: Blurring the Boundary Between Focal and Generalized Epilepsies? Front Neurol. 2022;13:831546. Doi:10.3389/fneur.2022.831546.
- Bancaud J, Talairach J, Morel P, et al. “Generalized” epileptic seizures elicited by electrical stimulation of the frontal lobe in man. Electroencephalogr Clin Neurophysiol. 1974; 37(3):275-82. Doi:10.1016/0013-4694.
- Jocić-Jakubi B, Jovanović M, Janković DS, et al. Frontal-onset absences in children: associated with worse outcome? A replication study. Seizure. 2009 May;18(4):275-8. Doi:10.1016/j.seizure.2008.10.013.
- Chaitanya G, Sinha S, Narayanan M, Satishchandra P. Scalp high frequency oscillations (HFOs) in absence epilepsy: An independent component analysis (ICA) based approach. Epilepsy Res. 2015;115:133-40. Doi:10.1016/j.eplepsyres.2015.06.008.
- Manning JP, Richards DA, Leresche N, Crunelli V, Bowery NG. Cortical area-specific block of genetically determined absence seizures by ethosuximid. Neuroscience 2004;123:5–9.
- Aghakhani Y, Bagshaw AP, Bénar CG, Hawco C, Andermann F, Dubeau F, et al.




