Safety of Long-Term Cryopreserved Umbilical Cord MSCs
Long-term cryopreserved umbilical cord derived mesenchymal stem cells: A study on their safety
Kyoko Baba 1,2, Yoshika Sugimoto 1, Kazuno Moriyama 1, Kenichi Kumazawa 1, Takayuki Sugimoto 1, Yasuharu Yamazaki 1, Akira Takeda 1
- Department of Plastic Surgery and Aesthetic Surgery, Kitasato University School of Medicine; 1-15-1 Kitasato, Minami ku, Sagamihara, Kanagawa 252-0374, Japan
- Department of Plastic Surgery, Kitasato University Medical Center; 6-100 Arai, Kitamoto, Saitama 364-8501, Japan
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Baba, K., Sugimoto, Y., et al., 2024. Long-term cryopreserved umbilical cord derived mesenchymal stem cells: A study on their safety. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5944
COPYRIGHT: © 2024 European Society of Medicine. This is an open- access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5944
ISSN 2375-1924
ABSTRACT
Background:
In recent years, mesenchymal stem cells have gained attention in the field of regenerative medicine. While mesenchymal stem cells derived from adipose tissue and bone marrow are well-studied, obtaining sufficient tissue from neonates or infants poses significant challenges. Umbilical cord-derived mesenchymal stem cells are a promising alternative source, as they can be harvested non-invasively and in large quantities with minimal ethical concerns. This study aims to evaluate the safety of long-term cryopreserved umbilical cord-derived mesenchymal stem cells and explore their potential for clinical applications in regenerative medicine.
Methods:
Umbilical cord-derived mesenchymal stem cells were cryopreserved for periods ranging from 4 years and 5 months to 5 years and 9 months. After thawing, the following assessments were performed:
-
Morphological changes: Cell morphology was observed under a light microscope before and after cryopreservation.
-
Multipotency evaluation: Osteogenic and adipogenic differentiation potentials were assessed to confirm MSC characteristics.
-
Chromosomal stability: G-band staining and Spectral Karyotyping were used to identify numerical and structural chromosomal abnormalities.
-
Tumorigenetic potential: Real-time PCR was conducted to measure the expression of telomerase reverse transcriptase, a tumorigenic marker.
Results:
No significant morphological differences were observed before and after cryopreservation. Both osteogenic and adipogenic differentiation potentials were maintained, confirming the multipotency of the cells. Chromosomal abnormalities were found in 4 out of 168 cells using G-banding, but no abnormalities were detected with Spectral Karyotyping. Telomerase reverse transcriptase gene expression was low, indicating a low risk for tumorigenicity.
Conclusion:
Long-term cryopreserved umbilical cord-derived mesenchymal stem cells maintain their safety in terms of morphology and function, showing promise as autologous graft material for regenerative medicine. However, further safety evaluations, particularly concerning chromosomal abnormalities, are essential before clinical application.
Keywords:
Umbilical cord-derived mesenchymal stem cells, Long-term cryopreservation, safety evaluation, regenerative medicine.
1. Introduction
In recent years, regenerative medicine aimed at tissue regeneration has been actively pursued. The usefulness of mesenchymal stem cells (MSCs) as a material for regenerative medicine has been widely recognized. In the field of plastic surgery, research on MSCs derived from adipose tissue and bone marrow has drawn particular attention. However, obtaining sufficient amounts of adipose tissue or bone marrow for neonates and infants presents significant physical challenges. We have identified the umbilical cord (UC) as a promising new source of MSCs. The umbilical cord, which can be collected non-invasively at birth in large quantities, contains umbilical cord-derived mesenchymal stem cells (UC-MSCs). Since the umbilical cord is typically discarded, its use raises minimal ethical concerns. Moreover, UC-MSCs possess high regenerative potential and a favorable immune tolerance profile. Additionally, no evidence has been reported of tumorigenicity when UC-MSCs were transplanted into immunodeficient animals. Based on these factors, we consider the use of UC as a valuable resource for regenerative medicine.
In general, safety considerations are paramount for transplantation materials. Autologous tissues minimize many of the risks and ethical concerns associated with allogenic or xenogenic materials, such as the risk of viral infection or the ethical implications of tissue donation. For this reason, autologous tissue continues to be regarded as a superior transplantation material in clinical practice. To utilize UC-MSCs as autologous tissue, it is necessary to preserve them until the time of use. Cryopreservation has been reported to affect the quality of cells and tissues. To the best of our knowledge, the longest cryopreservation period reported for the safety of UC-MSCs is 12 months. However, considering the need for clinical applications, longer cryopreservation periods are required. Therefore, this study investigates the clinical viability of UC-MSCs by evaluating their safety after long-term cryopreservation, aiming to establish a reliable source of autologous graft material for future regenerative medicine applications. Specifically, we evaluated chromosomal numerical and structural abnormalities (Karyotype analysis), as well as the expression of tumorigenicity-related markers such as telomerase reverse transcriptase (hTERT) in UC-MSCs after prolonged cryopreservation. This report presents a highly original study conducted to explore the clinical application of our innovative therapeutic strategy, which involves utilizing cryopreserved-UC-MSCs (cUC-MSCs) as autologous graft material.
2. Materials and methods
The present study, approved by the ethics committee at our university (approval number B-07-13), was conducted after the acquisition of written informed consent from pregnant women. Tissues, namely UC, which are usually discarded in clinical practice, were collected as study materials under the conditions in which the safety of neonates and parturient women was ensured after the expulsion of the placenta. The collected samples of UC were numbered in order not to allow individual identification.
2.1. PRETREATMENT OF UMBILICAL CORD-DERIVED MESENCHYMAL STEM CELLS
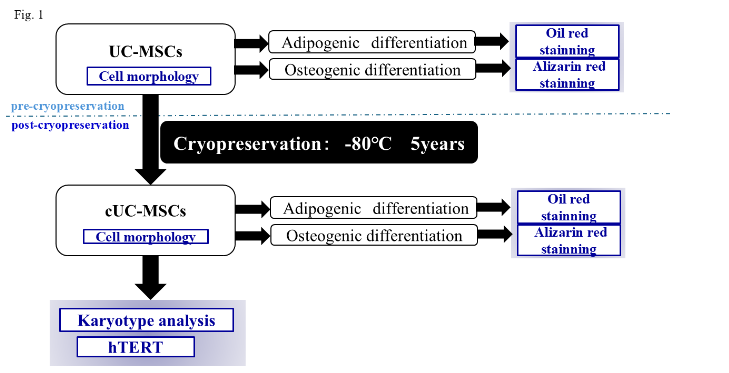
The UC samples were collected from neonates who were born at weeks 37e41 of gestation without any problems during delivery, to mothers whom obstetricians had not found any maternal or fetal complications during pregnancy. UC was cut into about 10-cm sections and was washed with phosphate buffered saline (PBS; Wako Pure Chemical Industries Co., Ltd., Osaka, Japan) until no attached blood remained. UC was treated as per a previously reported method. Briefly, the Wharton jelly (WJ) was chopped into approximately 5-mm sections. Subsequently, the sections were cultured in Dulbecco’s modified Eagle’s medium (DMEM) with the addition of 10% fetal bovine serum (FBS) (SigmaeAldrich MO, USA), antibiotics (100 U/ml penicillin and 100 g/ml streptomycin), in a 25-cm2 flask (Sumilon Sumitomo, Tokyo, Japan) at 37℃ and with 5% CO2. Media were replaced for the first time at 1 week after culture initiation and every 3 days thereafter. The WJ sections were removed at approximately 2 weeks after culture initiation when adhesive spindle-shaped cells emerged. Culture continued until the time when cells became subconfluent. Subsequently, the cells were detached and collected before suspension in a solution for cell cryopreservation (LaboBanker2 ™ TOSC, Tokyo, Japan) and storage at -80℃ until the time of use. UC-MSCs were cryopreserved for approximately 4 years 5 months to 5 years 9 months, and then slowly thawed at room temperature. Cells that became subconfluent in culture after thawing were used for each experiment. The cell morphology of UC-MSCs was observed before and after cryopreservation.
2.2. ASSESSMENT OF THE OSTEOBLASTIC- AND ADIPOGENIC-DIFFERENTIATION
The multidifferentiation potential of UC-MSCs and cUC-MSCs before and after cryopreservation was evaluated to confirm their status as MSCs. According to the method reported by Baba et al. (2012), differentiation into osteoblasts and adipocytes was induced to assess their osteoblastic and adipogenic potential. Subconfluent primary cultured cells were divided into two portions, with one portion induced for differentiation into osteoblasts by changing the medium to the NH OsteoDiff Medium (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany), while the other portion was induced for differentiation into adipose cells by changing the medium to NH AdipoDiff Medium (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). Osteoblast-differentiated cells were stained with alizarin red, and adipocyte-differentiated cells were stained with oil red, following standard protocols before being observed under a stereoscopic microscope.
2.3. EVALUATION OF THE SAFETY OF CRYOPRESERVED UMBILICAL CORD-DERIVED MESENCHYMAL STEM CELLS
To evaluate the safety of cUC-MSCs, we focused on two key aspects: chromosomal stability and tumorigenic potential. Chromosomal abnormalities were evaluated using G-band staining and Spectral Karyotyping (SKY) methods. Tumorigenic potential was assessed by measuring the expression of human telomerase reverse transcriptase (hTERT), a marker commonly associated with tumorigenicity.
2.3.1. Karyotype analysis
This evaluation was conducted by the Clinical Laboratory Department of the hospital affiliated with the author’s institution. Chromosomal specimens were prepared from 56 cells per donor, from three donors (a total of 168 cells), and both numerical and structural chromosomal abnormalities were assessed using G-banding. Additionally, chromosomal analysis of cUC-MSCs was performed by SRL, Inc. (Tokyo, Japan) using the SKY method.
2.3.2. Expression of telomerase reverse transcriptase by reverse transcriptase-polymerase chain reaction
Real-time reverse transcriptase-polymerase chain reaction (RT-PCR) was performed to assess the expression of the telomerase reverse transcriptase (hTERT) gene after reverse transcription. Ten specimens were used to determine the expression of the markers. In accordance with published procedures, total RNA was extracted using the RNeasy Mini Kit (Qiagen, Valencia, CA), and cDNA was synthesized. The PCR primer sequences for hTERT were designed using the Primer3 software based on the GeneBank sequence. For quantitative evaluation, iQ™ SYBR® Green Supermix was used. hTERT, along with bone marrow-derived mesenchymal stem cells transduced with HPV E6 and E7, was used as a positive control.
Primer Sequences: Telomerase reverse transcriptase (hTERT)
- Forward: cggtgtgcaccaacatctac
- Reverse: gggttcttccaaacttgctg
TATA box binding protein (TBP)
- Forward: ttcggagagttctgggattg
- Reverse: ctcatgattaccgcagcaaa
3. Results
3.1. Cell morphology
No clear differences in cell morphology were noted before and after cryopreservation, as observed under a light microscope.

3.2. THE OSTEOBLASTIC- AND ADIPOGENIC-DIFFERENTIATION POTENTIAL OF CELLS IN VITRO
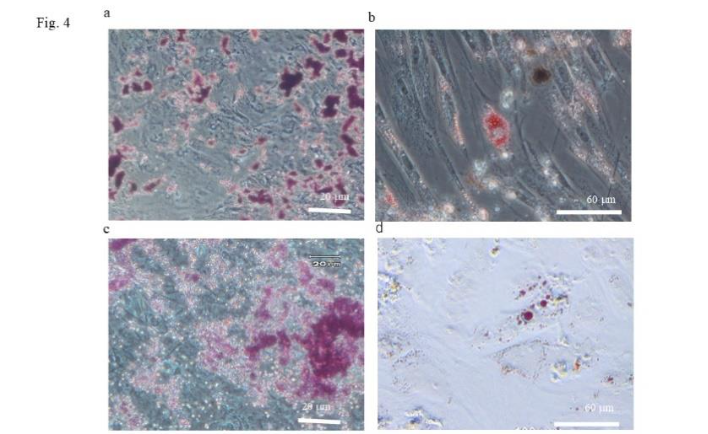
The UC-MSC and cUC-MSC samples that had been induced to differentiate into osteoblasts were stained diffusely throughout the extracellular matrix with alizarin red, confirming the presence of calcium deposits. UC-MSCs and cUC-MSCs that had been induced to differentiate into adipocytes exhibited intracellular lipid droplets that were stained with oil red. These findings confirm the osteogenic and adipogenic differentiation potential of both UC-MSCs and cUC-MSCs, demonstrating their multipotency.
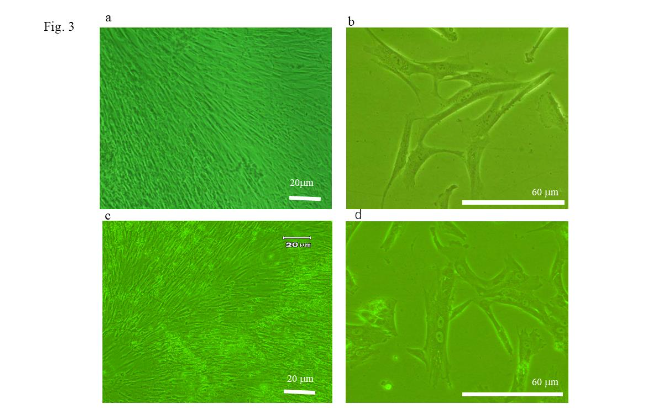 Figure 4. Assessment of the osteoblastic- and adipogenic-differentiation potential of umbilical cord-derived mesenchymal stem cells (UC-MSCs) and cryopreserved-UC-MSCs (cUC-MSCs). (a) Staining with alizarin red. The samples of UC-MSCs, which had been induced to differentiation into osteoblasts, were stained extracellularly and diffusely with alizarin red. (b) Staining with oil red. The samples of UC-MSCs, which had been induced to differentiation into adipose cells, showed intracellular lipid droplets that were stained with oil red (arrow). (c) Staining with alizarin red. The samples of cUC-MSCs, which had been induced to differentiation into osteoblasts, were stained extracellularly and diffusely with alizarin red. (d) Staining with oil red. The samples of cUC-MSCs, which had been induced to differentiation into adipose cells, showed intracellular lipid droplets that were stained with oil red (arrow).
Figure 4. Assessment of the osteoblastic- and adipogenic-differentiation potential of umbilical cord-derived mesenchymal stem cells (UC-MSCs) and cryopreserved-UC-MSCs (cUC-MSCs). (a) Staining with alizarin red. The samples of UC-MSCs, which had been induced to differentiation into osteoblasts, were stained extracellularly and diffusely with alizarin red. (b) Staining with oil red. The samples of UC-MSCs, which had been induced to differentiation into adipose cells, showed intracellular lipid droplets that were stained with oil red (arrow). (c) Staining with alizarin red. The samples of cUC-MSCs, which had been induced to differentiation into osteoblasts, were stained extracellularly and diffusely with alizarin red. (d) Staining with oil red. The samples of cUC-MSCs, which had been induced to differentiation into adipose cells, showed intracellular lipid droplets that were stained with oil red (arrow).
3.3. THE SAFETY OF CRYOPRESERVED UMBILICAL CORD-DERIVED MESENCHYMAL STEM CELLS
3.3.1. Karyotype analysis
Numerical and structural chromosomal abnormalities in cUC-MSCs were observed in 4 out of 168 cells from one donor, specifically involving chromosome 2. The SKY method did not reveal any abnormalities.

3.3.2. Expression of telomerase reverse transcriptase by reverse transcriptase-polymerase chain reaction
The expression of the hTERT gene in cUC-MSCs was significantly lower compared to the positive control.
| Sample | hTERT Expression |
|---|---|
| cUC-MSCs | Significantly lower compared to the positive control |
| Control | Positive control |
4. Discussion
Safety of cryopreserved umbilical cord-derived mesenchymal stem cells. For the clinical application of UC-MSCs obtained from UC at birth, long-term cryopreservation is required when used as autologous tissue. Even when used as allogeneic tissue, long-term cryopreservation is necessary to ensure stable supply for clinical use. Conventionally, cryopreservation involves gradual cooling to -80°C, followed by storage at -196°C using liquid nitrogen. It is widely recognized that cryopreservation can affect cell quality. Mullers et al. reported that cryopreservation causes physical damage to cells, leading to changes in cell viability and potentially compromising clinical benefits. Therefore, verifying the safety and viability of cUC-MSCs stored under appropriate conditions is crucial and beneficial for their clinical application as either autologous or allogeneic tissue. In this study, since we aim for clinical use as autologous tissue, we conducted the research under unique conditions: a long-term cryopreservation period of 5 years and storage at -80°C, a temperature commonly used in general medical facilities. Although different cryopreservation solutions were used, Zhang et al. reported that cryopreservation for one year at -196°C did not affect the cell morphology, surface marker expression, cell viability, proliferative capacity, or chromosomal stability of cUC-MSCs. Additionally, BÁRCIA et al. demonstrated that cryopreservation did not affect the surface marker expression of UC-MSCs, though the storage period was unspecified. Comparing previous studies with this study regarding the effects of cryopreservation on cUC-MSCs is challenging due to limited data on long-term cryopreservation of MSCs and differences in conditions. Previous studies did not actively suggest that cryopreservation compromises the safety of cUC-MSCs. In this study, the negative hTERT gene expression did not suggest tumorigenesis, and no significant differences in cell morphology were observed. However, we did observe abnormalities in chromosome 2 in 4 out of 168 cells from one donor using G-banding. Considering that the UC used in this study was sourced from healthy neonates with no complications during pregnancy or delivery, the effects of experimental procedures (e.g., culture or cryopreservation) were considered. Zhang et al. reported no chromosomal abnormalities in 3 cells. It is unclear whether the observed differences are due to freezing temperature, cryopreservation period, culture conditions, sample size, or other experimental factors. Nevertheless, the chromosomal abnormalities observed in this study do not directly suggest tumorigenesis, and the study suggests that UC-MSCs are amenable to cryopreservation. Therefore, in terms of safety, the potential for utilizing cUC-MSCs as autologous tissue in regenerative medicine is suggested. However, ensuring cell safety is essential for clinical application. This study suggests that safety testing, such as chromosomal analysis before transplantation, is desirable when considering clinical application.
5. Conclusion
In conclusion, this study suggests that cUC-MSCs have significant potential as a safe source of autologous tissue for regenerative medicine, particularly following long-term cryopreservation.
Conflict of interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Funding
This research was funded by a grant-in-aid for scientific research (C)20K09871 from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Acknowledgments
The authors are grateful to Yumiko Sone, for his valuable discussions about the manuscript.
Author contributions
Conceptualization: Kyoko Baba
Methodology: Kyoko Baba
Validation: Yoshika Sugimoto, Kazuno Moriyama, Kenichi Kumazawa, Takayuki Sugimoto
Investigation: Yoshika Sugimoto
Resources: Kyoko Baba
Data curation: Kyoko Baba
Writing—original draft preparation: Kyoko Baba
Writing—review and editing: Akira Takeda
Supervision: Yasuharu Yamazaki
Project administration: Akira Takeda
Funding acquisition: Kyoko Baba
All authors have read and agreed to the published version of the manuscript.
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Kitasato University (B-07-13). Informed consent was obtained from all subjects involved in the study.
References
- Behnia H, Khojasteh A, Soleimani M, Tehranchi A, Atashi A. Repair of alveolar cleft defect with mesenchymal stem cells and platelet derived growth factors: A preliminary report. J Craniomaxillofac Surg. 2012;40(2):7.
- Plachokova AS, van den Dolder J, van den Beucken JJ, Jansen JA. Bone regenerative properties of rat, goat and human platelet-rich plasma. Int J Oral Maxillofac Surg. 2009;38(8):861-869.
- Lee JY, Nam H, Park YJ, et al. The effects of platelet-rich plasma derived from human umbilical cord blood on the osteogenic differentiation of human dental stem cells. In Vitro Cell Dev Biol Anim. 2011;47(2):157-164.
- Qi Y, Niu L, Zhao T, et al. Combining mesenchymal stem cell sheets with platelet-rich plasma gel/calcium phosphate particles: A novel strategy to promote bone regeneration. Stem Cell Res Ther. 2015;6:256.
- Sarugaser R, Lickorish D, Baksh D, Hosseini MM, Davies JE. Human umbilical cord perivascular (HUCPV) cells: A source of mesenchymal progenitors. Stem Cells. 2005;23:220-229.
- Pivoriūnas A, Bernotiene E, Unguryte A, et al. Isolation and differentiation of mesenchymal stem-like cells from human umbilical cord vein endothelium and subendothelium. Biologija. 2006;2:99-103.
- Kern S, Eichler H, Stoeve J, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood or adipose tissue. Stem Cells. 2006;24(5):1294-1301.
- Chen M, Peng J, Xie Q, et al. Mesenchymal stem cells alleviate moderate-to-severe psoriasis by reducing the production of type I interferon (IFN-I) by plasmacytoid dendritic cells (pDCs). Stem Cells Int. 2019;2019:6961052.
- Lin Y, Wang H, Jiang C, et al. Effects of different concentrations of human umbilical cord mesenchymal stem cells to ameliorate psoriasis-like skin lesions in BALB/c mice. Ann Transl Med. 2022;10(2):86.
- Wang Z, Hu Y, Wang X, et al. Comparative analysis of the therapeutic effects of fresh and cryopreserved human umbilical cord-derived mesenchymal stem cells in the treatment of psoriasis. Stem Cell Rev Rep. 2023;19:1922-1936.
- Ahn H, Lee SY, Jung WJ, Pi J, Lee KH. Psoriasis treatment using minimally manipulated umbilical cord-derived mesenchymal stem cells: A case report. World J Clin Cases. 2021;9:6798-6803.
- Ohgushi H, Kitamura S, Kotobuki N, et al. Clinical application of marrow mesenchymal stem cells for hard tissue repair. Yonsei Med J. 2004;45:61-67.
- Ishiguro M, Yamazaki Y, Baba K, et al. Assessment of the osteogenic potential of maxilla-derived mesenchymal stromal cells and the utilization of serum-free medium for culture thereof. Kitasato Med J. 2014;44:84-94.
- Aoyagi K, Yamazaki Y, Matsuo A, et al. Significance of CD271 in bone marrow mesenchymal stem cells: Changes by cryopreservation. J Craniofac Surg. 2010;21(3):666-678.
- Zhang M, Zhao Y, Wang L, et al. Study of the biological characteristics of human umbilical cord mesenchymal stem cells after long-time cryopreservation. Cell Tissue Bank. 2022;23:739-752.
- Baba K, Yamazaki Y, Ikemoto S, et al. Osteogenic potential of human umbilical cord-derived mesenchymal stromal cells cultured with umbilical cord blood-derived autoserum. J Craniomaxillofac Surg. 2012;40:768-772.
- Ogata S, Ogihara Y, Nomoto K, et al. Clinical score and transcript abundance patterns identify Kawasaki disease patients who may benefit from the addition of methylprednisolone. Pediatr Res. 2009;19:577-584.
- Baba K, Yamazaki Y, Ishiguro M, et al. Osteogenic potential of human umbilical cord-derived mesenchymal stromal cells cultured with umbilical cord blood-derived fibrin: A preliminary study. J Craniomaxillofac Surg. 2013;41:775-782.
- Badowski M, Muise A, Harris DT. Mixed effects of long-term frozen storage on cord tissue stem cells. Cytotherapy. 2014;16(9):1313-1321.
- Shimazu T, Mori Y, Takahashi A, et al. Serum- and xeno-free cryopreservation of human umbilical cord tissue as mesenchymal stromal cell source. Cytotherapy. 2015;17(5):593-600.
- Müller L, Tunger A, Wobus M, et al. Immunomodulatory properties of mesenchymal stromal cells: An update. Front Cell Dev Biol. 2021;9:637725.
- Bárcia RN, Santos JM, Teixeira M, et al. Umbilical cord tissue-derived mesenchymal stromal cells maintain immunomodulatory and angiogenic potencies after cryopreservation and subsequent thawing. Cytotherapy. 2017;19(3):360-370.
- Marquez-Curtis LA, Elliott JA. Mesenchymal stromal cells derived from various tissues: Biological, clinical and cryopreservation aspects: Update from 2015 review. Cryobiology. 2024;115:104856.
- Mukai T, Tojo A, Nagamura-Inoue T. Mesenchymal stromal cells as a potential therapeutic for neurological disorders. Regen Ther. 2018;9:32-37.
- Skiles ML, Marzan AJ, Brown KS, Shamonki JM. Comparison of umbilical cord tissue-derived mesenchymal stromal cells isolated from cryopreserved material and extracted by explantation and digestion methods utilizing a split manufacturing model. Cytotherapy. 2020;22(6):581-591.
- Guan YT, Xie Y, Li DS, et al. Comparison of biological characteristics of mesenchymal stem cells derived from the human umbilical cord and decidua parietalis. Mol Med Rep. 2019;20(1):633-639.
- Zhao L, Chen S, Yang P, Cao H, Li L. The role of mesenchymal stem cells in hematopoietic stem cell transplantation: Prevention and treatment of graft-versus-host disease. Stem Cell Res Ther. 2019;10(1):1-13.
- Le Thi Bich P, Nguyen Thi H, Dang Ngo Chau H, et al. Allogeneic umbilical cord-derived mesenchymal stem cell transplantation for treating chronic obstructive pulmonary disease: A pilot clinical study. Stem Cell Res Ther. 2020;11(1):1-14.
- Kol A, Wood JA, Carrade Holt DD, et al. Multiple intravenous injections of allogeneic equine mesenchymal stem cells do not induce a systemic inflammatory response but do alter lymphocyte subsets in healthy horses. Stem Cell Res Ther. 2015;6(1):1-9.
- Marquez-Curtis LA, Elliott JA. Mesenchymal stromal cells derived from various tissues: Biological, clinical and cryopreservation aspects: Update from 2015 review. Cryobiology. 2024;115:104856.
- Baba K, Yamazaki Y, Sone Y, et al. An in vitro long-term study of cryopreserved umbilical cord blood-derived platelet-rich plasma containing growth factors—PDGF-BB, TGF-β, and VEGF. J Craniomaxillofac Surg. 2019;47(5):668-675.

