SARS-CoV-2 Antibodies in Human Milk During Pregnancy
SARS-CoV-2 IgA and IgG in Human Milk of Women Infected During Pregnancy
Sudha Rani Narasimhan¹², MD; Priya Jegatheesan¹², MD; Jiraporn Lueangsakulthai³, PhD; Claudia Flores¹; Angela Huang¹, RNC, MPH; Christina Anderson¹², MD; Sonya Misra¹², MD, MPH; Rupalee Patel¹, PNP, DNP; David Charles Dallas³, PhD; Dongli Song¹², MD, PhD
- Department of Pediatrics, Division of Neonatology, Santa Clara Valley Medical Center, San Jose, CA, USA
- Department of Pediatrics, Stanford University School of Medicine, Stanford, CA, USA
- College of Health, Oregon State University, Corvallis, CA
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Narasimhan, SR., et al., 2024. SARS-CoV-2 IgG and IgA in Human Milk of Women Infected During Pregnancy. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v1i210.5840
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v1i210.5840
ISSN 2375-1924
ABSTRACT
Background:
SARS-CoV-2 infection is still ongoing worldwide. The passage of SARS-CoV-2 antibodies through human milk provides essential protection to infants. The objective of this study was to determine the presence of immunoglobulins in the human milk at 2 and 6 weeks post-delivery of women who tested positive for the SARS-CoV-2 virus during pregnancy.
Methods:
This prospective observational cohort study was conducted between April 2020 and January 2021. We collected human milk from mothers who tested positive for the SARS-CoV-2 virus during pregnancy. Human milk IgG and IgA antibodies against SARS-CoV-2 S1 RBD protein were measured.
Results:
Eighty-one human milk samples were collected from 57 mothers infected during pregnancy. Human milk collected at 2 weeks post-delivery was positive for SARS-CoV-2 IgG in 82% and IgA in 49% of samples. At 6 weeks, human milk was positive for SARS-CoV-2 IgG in 67% and for IgA in 37% of samples. There was no correlation between the IgG or IgA levels at 2 weeks post-delivery and days from the first positive PCR test to delivery or gestational age at the first positive PCR test. SARS-CoV-2 antibodies were detectable in human milk of mothers who were PCR positive early in gestation. There was no significant difference in human milk IgG or IgA levels when comparing maternal symptom severity. The IgA and IgG levels were lower at 6 weeks compared to 2 weeks in 79% of the paired samples.
Conclusions:
Human milk of mothers infected with the SARS-CoV-2 virus during pregnancy showed the presence of both IgG and IgA against the virus protein. Continued human milk feeding of infants born to mothers infected with SARS-CoV-2 should be encouraged to optimize protective antibodies transferred to the infant.
Introduction
Human milk feeding is the most effective feeding strategy to promote optimal health in newborns. In addition to nutritional components, human milk has many protective properties, including human milk oligosaccharides, bioactive factors, cells, and immunoglobulins.¹ Human milk feeding is associated with improved infant health and immune development when compared to formula fed infants.² Immunoglobulins in human milk provide barrier immunity to the infant, including specificity towards respiratory antigens.³
The COVID-19 pandemic, caused by the SARS-CoV-2 virus, was a major worldwide public health crisis. While the pandemic has ended, infection with this virus is still ongoing. During pregnancy, maternal SARS-CoV-2 antibodies, which are produced after infection or vaccination, are transferred to the fetus through the placenta.⁴ Postnatally, human milk antibodies provide additional protection to the infant. The World Health Organization (WHO) recommends continuation of human milk feeding even if the mother is infected with SARS-CoV-2.⁵ While the transmission of SARS-CoV-2 virus via human milk has been noted to be rare,⁶–⁹ the presence of SARS-CoV-2 antibodies in human milk has been shown in multiple studies, although the number of participants included in these studies are limited to under 200 participants.¹⁰–¹⁴ Studying the native reaction to SARS-CoV-2 infection is now likely confounded by prior COVID infection and/or vaccination, making reports of immunoglobulins in human milk from mothers infected during the early phase of the pandemic important and relevant.
The objective of this study was to determine the levels of IgG and IgA in human milk and compare their changes at 2 and 6 weeks post-delivery of unvaccinated women who tested positive for the SARS-CoV-2 virus during pregnancy.
Methods
SETTING AND STUDY POPULATION
This prospective observational cohort study was conducted between April 2020 and January 2021 at a public safety-net health system with about 4,500 deliveries per year. Approval was obtained from our Institutional Review Board (Reference #20-021). During the study period, all women admitted for delivery underwent universal screening for SARS-CoV-2 by a nasopharyngeal PCR on admission for delivery or within three days prior to admission for elective deliveries. Also, mothers who experienced symptoms concerning for COVID-19 or had close contact with a confirmed SARS-CoV-2 positive person were tested during pregnancy.⁴ Pregnant women who had a confirmed positive SARS-CoV-2 PCR test during the current pregnancy, either prior to admission or at admission, were eligible for the study. This study included 57 women who tested positive for SARS-CoV-2 during pregnancy (2 in the first trimester, 6 in the second trimester, and 49 in the third trimester/at delivery) and were providing human milk for their infants. The timing of maternal SARS-CoV-2 infection was determined by the date of the first positive SARS-CoV-2 PCR test. The Society for Maternal-Fetal Medicine guidelines were used to establish the severity of SARS-CoV-2 symptoms (mild, moderate, severe).¹⁵
HUMAN MILK SAMPLE PREPARATION
Human milk samples were collected at 2 and 6 weeks post-delivery and were kept at -80°C until assayed. Twenty-three human milk samples collected prior to the COVID-19 pandemic were used as negative controls. Human milk samples were thawed at 4°C and centrifuged at 4,250 × g for 10 minutes at 4°C. The infranate was collected by pipette from below the upper fat layer. Multiple aliquots of 400 µL of the supernatant from each sample were stored at -80°C until use for each assay.
MEASUREMENT OF SARS-CoV-2-S1 RBD PROTEIN SPECIFIC ANTIBODIES IN HUMAN MILK
Antibodies (IgG and IgA) against SARS-CoV-2-S1 receptor binding domain (RBD) protein were measured using enzyme linked immunosorbent assays (SARS-CoV-2-S1 RBD protein human IgG and IgA ELISA Kits, IEQ-CoV-S1 RBD-IgG, IEQ-CoV-S1 RBD-IgA, RayBiotech, GA, USA). These ELISA kits were used for analysis of serum samples and were adapted to be used with human milk, similar to previous human milk studies.⁹,¹³,¹⁴,¹⁶,¹⁷ The absorbance was measured with a spectrophotometer (SpectraMax M2, Molecular Devices). Results were calculated using calibration curves as directed by the manufacturer’s specifications. Positive cut-off values for IgG (134 U/ml) and IgA (16262 U/ml) were 2 standard deviations from the average antibody concentration of the 23 control human milk samples collected prior to the pandemic.
DATA ANALYSIS
The maternal and infant demographics, including maternal age, gravida, para, infant gestational age, birth weight, sex, and maternal morbidities (hypertension, diabetes, and obesity), are summarized as percentages, mean and standard deviations, or median and interquartile range (IQR) as indicated. Human milk immunoglobulin levels were compared between maternal symptom groups using Kruskal-Wallis equality of populations rank test for non-parametric variables. Correlation between the timing of maternal infection (time from positive PCR to delivery and gestational age at the time of positive PCR) and human milk IgG and IgA levels at 2 weeks post-delivery were determined using Spearman’s rank correlation coefficient. Human milk IgG and IgA levels in 2 and 6 week paired samples were compared using Wilcoxon signed-rank test. Data analysis was performed using Stata 14.2 (Stata Corp, College Station, TX).
Results
A total of 81 human milk samples were collected from 57 mothers, of which 51 were collected at 2 weeks and 30 were collected at 6 weeks, with 24 paired samples at 2 and 6 weeks. The maternal and infant characteristics are summarized in Table 1.
Table 1: Maternal and Infant Characteristics
Maternal demographics and outcomes (n=57)
- Maternal age, years, median (IQR): 27 (22–33)
- Gravida, median (IQR): 3 (1–4)
- Para, median (IQR): 1 (0–2)
- Hispanic, n (%): 53 (93)
Race, n (%)
- White: 55 (96)
- Black: 1 (2)
- Asian: 1 (2)
- C-section delivery, n (%): 14 (25)
Maternal SARS-CoV-2 infection severity, n (%)
- Asymptomatic: 27 (47)
- Mild to moderately symptomatic: 26 (46)
- Severely symptomatic: 4 (7)
- Maternal diabetes, n (%): 13 (23)
- Maternal hypertension, n (%): 10 (18)
- Maternal obesity, n (%): 14 (25)
Infant demographics and outcomes (n=59)
- Infant birth weight, grams, median (IQR): 3370 (2985–3665)
- Gestational age, weeks, median (IQR): 39.1 (38.1–39.7)
- Male, n (%): 24 (41)
- Any human milk feeding in hospital, n (%): 59 (100)
- Exclusive human milk feeding in hospital, n (%): 38 (64)
In this cohort, 47% of the infected mothers were asymptomatic, 43% had mild to moderate symptoms, and only 7% developed severe disease. Human milk collected at 2 weeks post-delivery was positive for SARS-CoV-2 IgG in 82% and IgA in 49% of samples.
At 6 weeks, human milk was positive for SARS-CoV-2 IgG in 67% and IgA in 37% of samples. The concentrations of human milk antibodies are presented in Table 2.
Table 2: Human Milk Antibody Concentration
Human Milk Antibodies
IgG
- 2 weeks: n=51, % positive = 82, concentration = 383 (186–889) U/ml
- 6 weeks: n=30, % positive = 67, concentration = 207 (71–669) U/ml
IgA
- 2 weeks: n=51, % positive = 49, concentration = 14978 (6138–35903) U/ml
- 6 weeks: n=30, % positive = 37, concentration = 6676 (2775–32929) U/ml
There was no correlation between the days from the first positive PCR test to delivery and human milk IgG or IgA levels at 2 weeks post-delivery (Figure 1A, Figure 1B).
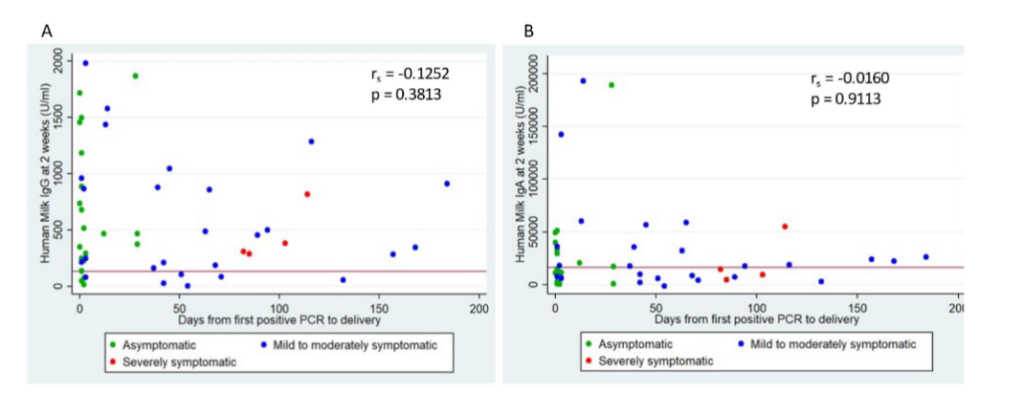
Figure 1: Human Milk Antibody Levels and Days from First Positive PCR to Delivery.
Red horizontal line represents positive cut-off values, IgG (134 U/ml) and IgA (16262 U/ml);
rₛ represents Spearman’s correlation coefficient.
A: Scatter plot of human milk IgG levels at 2 weeks post-delivery and days from first positive PCR to delivery.
B: Scatter plot of human milk IgA levels at 2 weeks post-delivery and days from first positive PCR to delivery.
There was no correlation between the gestational age of the first positive PCR test and human milk IgG or IgA levels at 2 weeks post-delivery (Figure 2A, Figure 2B).
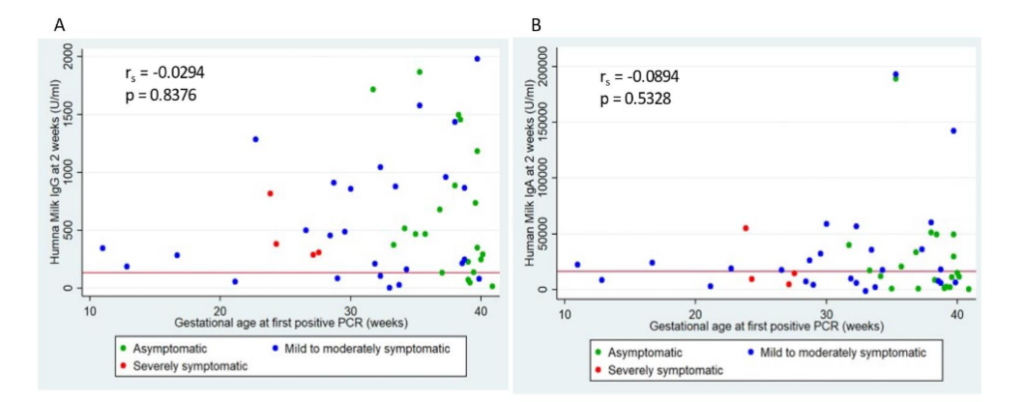
Figure 2: Human Milk Antibody Levels and Gestational Age at First Positive PCR.
Red horizontal line represents positive cut-off values, IgG (134 U/ml) and IgA (16262 U/ml); rₛ represents Spearman’s correlation coefficient.
A: Scatter plot of human milk IgG levels at 2 weeks post-delivery and gestational age at first positive PCR.
B: Scatter plot of human milk IgA levels at 2 weeks post-delivery and gestational age at first positive PCR.
There was no difference in human milk IgG or IgA levels when comparing maternal symptom severity (Figure 3).
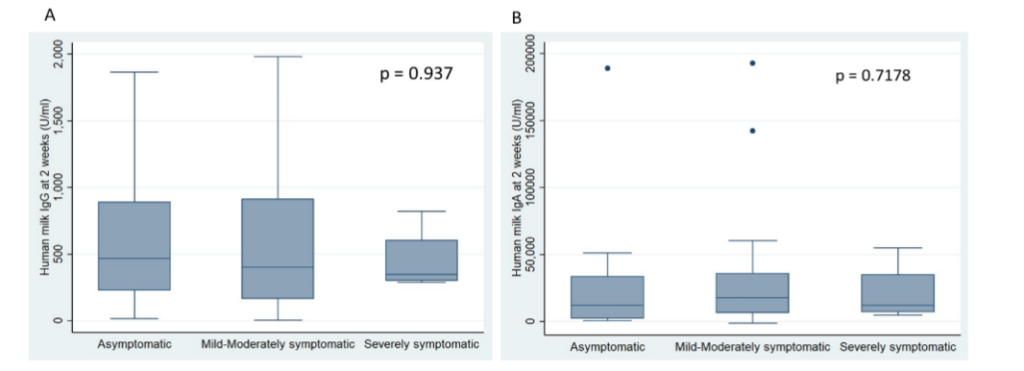
Figure 3: Human Milk Antibody Levels and Maternal Symptom Severity.
The box represents the IQR from 25th to 75th percentile (IQR). The marker within the box is the median and the “whiskers” reach the 1.5 times IQR.
A: Box plots of human milk IgG levels for each maternal symptom severity.
B: Box plots of human milk IgA levels for each maternal symptom severity.
Of the 24 paired samples at 2 and 6 weeks, 54% were positive for IgG and 25% were positive for IgA at both time points. No samples that were negative at 2 weeks became positive at 6 weeks. In 19 out of the 24 (79%) paired samples, the IgG level was lower at 6 weeks compared to 2 weeks (Figure 4A). In 19 out of the 24 (79%) paired samples, the IgA level was lower at 6 weeks compared to 2 weeks (Figure 4B).
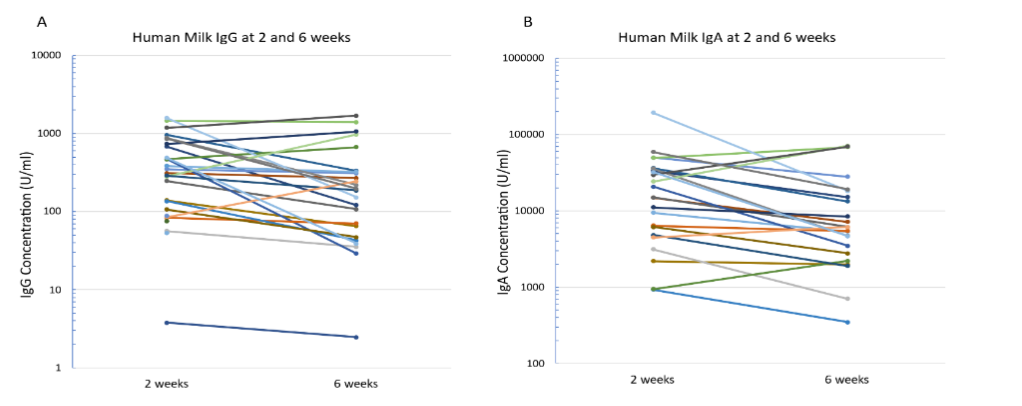
Figure 4: Paired Antibody Levels.
Levels of IgG and IgA are shown in logarithmic scale. Each line represents a paired sample.
A: Paired human milk IgG levels at 2 and 6 weeks.
B: Paired human milk IgA levels at 2 and 6 weeks.
Discussion
This study showed a strong presence of IgG and IgA immunoglobulins against the virus RBD protein in the human milk of mothers infected with the SARS-CoV-2 virus during pregnancy. There was no correlation between human milk SARS-CoV-2 IgG and IgA levels and the timing of maternal SARS-CoV-2 PCR positivity or maternal symptom severity.
In this cohort, 82% of human milk at 2 weeks were positive for SARS-CoV-2 IgG antibodies, whereas 49% were positive for IgA antibodies. The relative concentrations of IgA antibodies were higher than IgG given that human milk immunoglobulins are predominantly IgA. Our findings are consistent with three previous studies that analyzed antibodies in the milk of mothers with SARS-CoV-2 during pregnancy. Fox et al. analyzed 15 human milk samples and showed that 80% of milk samples contained IgA, and 67% of them were also positive for IgG and/or IgM.¹³ Pace et al., reported that 80% and 76% of 37 milk samples had IgG and IgA, respectively.¹³ Szczygiel et al. detected IgG antibodies in 85% and IgA in 86% of 72 milk samples.¹⁶ We found no association between the antibody response and the severity of maternal illness. Even though half of our patients were asymptomatic, they had a robust antibody presence in human milk, which is similar to a previous report.¹⁶
Our study included women who had infections at different gestational ages with the majority of cases in the third trimester. Notably, SARS-CoV-2 antibodies remain detectable in the 2 week post-delivery milk samples of mothers who were PCR positive more than 6 months prior to delivery. There have been case reports showing the presence of SARS-CoV-2-specific IgA in human milk up to 6–8 months after infection.¹⁸ Although we did not evaluate neutralizing activity, previous studies have reported that SARS-CoV-2 antibodies in human milk have neutralizing capacity.⁹,¹⁴,¹⁹ Together, these studies indicate that human milk remains protective for infants even when the infection was early in gestation.
Changes in human milk antibody levels have been characterized after infection or vaccination during lactation. We compared changes of antibody levels in paired 2 and 6 weeks samples from mothers who had SARS-CoV-2 during pregnancy. In the majority of the paired samples, we observed a decrease in IgG and IgA levels over time. Consistent with our observation, Bauerl et al. followed 12 mothers with paired longitudinal samples and found a generalized decrease in IgA levels after infection.²⁰ Wackman et al. showed a decrease in human milk antibody levels at 6 weeks compared to birth in women infected during pregnancy.²¹ Jakuszko et al. showed the duration of SARS-CoV-2-specific IgG in human milk was longer than that of IgA after vaccination.²²
However, Young et al. followed 47 lactating women after infection up to 90 days and found an upward trend of IgG and IgA response in 36% of participants and no change in IgG and IgA response in 38%.¹⁷ The differences of the antibody changes overtime among these studies likely reflect the differences of the timing of infection/vaccination and milk sample collection.
COVID-19 vaccination during pregnancy or lactation is highly protective to the mothers and their infants.¹⁷,²³ Differential human milk antibody responses to the various vaccines have been reported, with mRNA vaccines producing a more robust antibody response, making mRNA vaccines highly sought after.²⁴ Worldwide, COVID-19 vaccination is limited in many regions, especially in low-income countries.³⁰ However, even in countries where vaccines are available to everyone, universal vaccination is challenging. According to the recent CDC report, 18% of all adult females, 23% of pregnant females, and 23% of breastfeeding females are unvaccinated for COVID-19 in the US.³¹ Thus, infection-induced SARS-CoV-2 specific antibodies in human milk provide important protection to many infants, especially in regions without ready access to vaccination. Although several previous studies have shown a strong presence of SARS-CoV-2 antibody in human milk after natural infection during pregnancy, the total number of samples analyzed was less than 200.¹⁶,²⁰,²¹ We were able to study the native reaction to infection during pregnancy in 57 mothers, adding strength to the literature. Another strength of this study is that paired samples were used to describe time-dependent variation in the human milk IgG and IgA antibodies. One limitation of our study is that we cannot determine the precise timing of infection of asymptomatic mothers who screened positive for SARS-CoV-2 PCR at the time of delivery, resulting in uncertainty of infection duration in these cases.
Conclusions
Our study confirms the strong presence of SARS-CoV-2-specific IgG and IgA in the human milk of mothers who have had SARS-CoV-2 infection during pregnancy. Continued human milk feeding by mothers infected with SARS-CoV-2 should be encouraged to provide protection to the infant.
Conflicts of Interest Statement:
The authors have no conflicts of interest to declare.
Funding Statement:
This work was supported by the Valley Health Foundation (formerly the Valley Medical Center Foundation).
Acknowledgements:
We thank the mothers, newborns, and their families who participated in the study, outpatient pediatric clinics, and BRIDGE home follow up program. We thank First 5 of Santa Clara County for their support.
References
1. Cacho NT, Lawrence RM. Innate Immunity and Breast Milk. Front Immunol. 2017;8:584. doi:10.3389/fimmu.2017.00584
2. Oddy WH. The impact of breastmilk on infant and child health. Breastfeed Rev. Nov 2002; 10(3):5-18.
3. Atyeo C, Alter G. The multifaceted roles of breast milk antibodies. Cell. Mar 18 2021;184(6): 1486-1499. doi:10.1016/j.cell.2021.02.031
4. Song D, Prahl M, Gaw SL, et al. Passive and active immunity in infants born to mothers with SARS-CoV-2 infection during pregnancy: Prospective cohort study. medRxiv. May 03 2021; doi:10.1101/2021.05.01.21255871
5. WHO. Clinical management of COVID-19: Interim guidance, 27 May 2020. World Health Organization. Accessed November 7, 2022. https://creativecommons.org/licenses/by-nc-sa/3.0/igo
6. Kumar J, Meena J, Yadav A, Kumar P. SARS-CoV-2 detection in human milk: a systematic review. The journal of maternal-fetal & neonatal medicine : the official journal of the European Association of Perinatal Medicine, the Federation of Asia and Oceania Perinatal Societies, the International Society of Perinatal Obstet. Feb 08 2021:1-8. doi:10.1080/14767058.2021.1882984
7. Centeno-Tablante E, Medina-Rivera M, Finkelstein JL, et al. Transmission of SARS-CoV-2 through breast milk and breastfeeding: a living systematic review. Ann N Y Acad Sci. 01 2021;1484 (1):32-54. doi:10.1111/nyas.14477
8. Krogstad P, Contreras D, Ng H, et al. No Evidence of Infectious SARS-CoV-2 in Human Milk: Analysis of a Cohort of 110 Lactating Women. medRxiv. Apr 07 2021; doi:10.1101/2021.04.05.21254897
9. Pace RM, Williams JE, Järvinen KM, et al. COVID-19 and human milk: SARS-CoV-2, antibodies, and neutralizing capacity. medRxiv. Sep 18 2020; doi:10.1101/2020.09.16.20196071
10. Gao X, Wang S, Zeng W, et al. Clinical and immunologic features among COVID-19-affected mother-infant pairs: antibodies to SARS-CoV-2 detected in breast milk. New Microbes New Infect. Sep 2020;37:100752.
doi:10.1016/j.nmni.2020.100752
11. Dong Y, Chi X, Hai H, et al. Antibodies in the breast milk of a maternal woman with COVID-19. Emerg Microbes Infect. Dec 2020;9(1):1467-1469. doi:10.1080/22221751.2020.1780952
12. Lebrão CW, Cruz MN, Silva MHD, et al. Early Identification of IgA Anti-SARSCoV-2 in Milk of Mother With COVID-19 Infection. J Hum Lact. Nov 2020;36(4):609-613. doi:10.1177/0890334420960433
13. Fox A, Marino J, Amanat F, et al. Robust and Specific Secretory IgA Against SARS-CoV-2 Detected in Human Milk. iScience. Nov 20 2020; 23(11):101735. doi:10.1016/j.isci.2020.101735
14. Pace RM, Williams JE, Järvinen KM, et al. Characterization of SARS-CoV-2 RNA, Antibodies, and Neutralizing Capacity in Milk Produced by Women with COVID-19. mBio. 02 09 2021;12(1) doi:10.1128/mBio.03192-20
15. Halscott TM, MS, Vaught JM, Miller EM, MPH. Management Considerations for Pregnant Patients with Covid-19. Society for Maternal Fetal Medicine High Risk Pregnancy Experts; 2020. Accessed November 7, 2022.
https://www.smfm.org/covidclinical
16. Szczygiol P, Lukianowski B, Koscielska-Kasprzak K, et al. Antibodies in the breastmilk of COVID-19 recovered women. BMC Pregnancy Childbirth. Aug 11 2022;22(1):635. doi:10.1186/s12884-022-04945-z
17. Young BE, Seppo AE, Diaz N, et al. Association of Human Milk Antibody Induction, Persistence, and Neutralizing Capacity With SARS-CoV-2 Infection vs mRNA Vaccination. JAMA Pediatr. 02 01 2022;176(2):159-168.
doi:10.1001/jamapediatrics.2021.4897
18. Duncombe CJ, McCulloch DJ, Shuey KD, et al. Dynamics of breast milk antibody titer in the six months following SARS-CoV-2 infection. J Clin Virol. 09 2021;142:104916. doi:10.1016/j.jcv.2021.104916
19. Cabanillas-Bernal O, Cervantes-Luevano K, Flores-Acosta GI, Bernáldez-Sarabia J, Licea-Navarro AF. COVID-19 Neutralizing Antibodies in Breast Milk of Mothers Vaccinated with Three Different Vaccines in Mexico. Vaccines (Basel). Apr 18 2022;10(4)doi:10.3390/vaccines10040629
20. Bauerl C, Randazzo W, Sanchez G, et al. SARS-CoV-2 RNA and antibody detection in breast milk from a prospective multicentre study in Spain. Archives of disease in childhood Fetal and neonatal edition. Mar 2022;107(2):216-221. doi:10.1136/archdischild-2021-322463
21. Wachman EM, Snyder-Cappione J, Devera J, et al. Maternal, Infant, and Breast Milk Antibody Response Following COVID-19 Infection in Early Versus Late Gestation. Pediatr Infect Dis J. Mar 1 2023;42(3):e70-e76. doi:10.1097/INF.0000000000003802
22. Jakuszko K, Kościelska-Kasprzak K, Żabińska M, et al. Immune Response to Vaccination against COVID-19 in Breastfeeding Health Workers. Vaccines (Basel). Jun 17 2021;9(6) doi:10.3390/vaccines9060663
23. Golan Y, Prahl M, Cassidy AG, et al. COVID-19 mRNA Vaccination in Lactation: Assessment of Adverse Events and Vaccine Related Antibodies in Mother-Infant Dyads. Front Immunol. 2021;12:77 7103. doi:10.3389/fimmu.2021.777103
24. Kelly JC, Carter EB, Raghuraman N, et al. Anti-severe acute respiratory syndrome coronavirus 2 antibodies induced in breast milk after Pfizer-BioNTech/BNT162b2 vaccination. Am J Obstet Gynecol. Jul 2021;225(1):101-103. doi:10.1016/j.ajog.2021.03.031
25. Perl SH, Uzan-Yulzari A, Klainer H, et al. SARS-CoV-2-Specific Antibodies in Breast Milk After COVID-19 Vaccination of Breastfeeding Women. JAMA. May 18 2021;325(19):2013-2014. doi:10.1001/jama.2021.5782
26. Rosenberg-Friedman M, Kigel A, Bahar Y, et al. BNT162b2 mRNA vaccine elicited antibody response in blood and milk of breastfeeding women. Nat Commun. Oct 28 2021;12(1):6222. doi:10.1038/s41467-021-26507-1
27. Gray KJ, Bordt EA, Atyeo C, et al. Coronavirus disease 2019 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. Sep 2021;225(3):303 e1-303 e17. doi:10.1016/j.ajog.2021.03.023
28. Lechosa-Muniz C, Paz-Zulueta M, Mendez-Legaza JM, et al. Induction of SARS-CoV-2-Specific IgG and IgA in Serum and Milk with Different SARS-CoV-2 Vaccines in Breastfeeding Women: A Cross-Sectional Study in Northern Spain. Int J Environ Res Public Health. Aug 21 2021;18(16) doi:10.3390/ijerph18168831
29. Selma-Royo M, Bauerl C, Mena-Tudela D, et al. Anti-SARS-CoV-2 IgA and IgG in human milk after vaccination is dependent on vaccine type and previous SARS-CoV-2 exposure: a longitudinal study. Genome Med. Apr 21 2022;14(1):42. doi:10.1186/s13073-022-01043-9
30. Total Covid-19 Vaccine doses administered per 100 people. Our World in Data. Updated August 14, 2024. Accessed September 11, 2024. https://ourworldindata.org/grapher/covid-vaccination-doses-per-capita?tab=map&time=latest
31. Covid-19 Vaccination Coverage and Vaccine Confidence Among Adults. Accessed September 11, 2024. https://www.cdc.gov/vaccines/imz-managers/coverage/covidvaxview/interactive/adults.html

