Selective Digestive Decontamination: Safety in ICUs
Long-Term Use of Selective Digestive Decontamination in Areas Where Multi-Resistant Bacteria are Endemic. Is it Safe?
Dr Catalina Sánchez-Ramírez ¹, Dr Silvia Hípola-Escalada ², Dr María Adela Hernández-Viera ², Dr Laura Roldán-Furelos ², Dr Alba López-Domínguez ², Dr María de los Ángeles Sosa-Durr ², Dr Laura Lara-Franco ²
PUBLISHED: 28 February 2025
CITATION Sánchez-Ramírez, C., Hípola-Escalada, S., et al., 2025. Long-Term Use of Selective Digestive Decontamination in Areas Where Multi-Resistant Bacteria are Endemic. Is it Safe? Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6188
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6188
ISSN 2375-1924
ABSTRACT
Selective digestive decontamination is a prophylactic strategy to reduce the incidence of infection in intensive care unit (ICU) patients by prevention or eradication of the oropharyngeal and gastrointestinal carrier state of potentially pathogenic microorganisms in the gut flora that precedes the development of most ICU-acquired infections. It is one of the most studied interventions in critically ill patients. A lot of research have shown that selective digestive decontamination prevents severe infections, reduces mortality, has no adverse effects, is cost-effective and its use does not show a significant increase in antimicrobial resistance in areas of low antibiotic resistance. However, the impact of this in areas where multi-resistant Gram-negative bacteria are endemic is less clear and of great interest. Some studies also comment on the risk of developing infections after discharge from the ICU. The aim of this article is to provide a narrative review of the main evidence supporting or not supporting the use of selective digestive decontamination in these settings, whether it is safe to use in and after ICU discharge and in different types of patients.
Keywords
selective digestive decontamination, selective decontamination of the digestive tract, oropharyngeal decontamination, intensive care, antibiotic prophylaxis, prevention, resistance, critical ill patient, infection, mortality.
Introduction
The morbidity and mortality associated with ICU acquired infections is a major problem and it is important to reduce their incidence. A prophylactic strategy known as selective digestive decontamination (SDD) aims to reduce the incidence of infection in the ICU. SDD is a manoeuvre designed to convert carriers of ‘abnormal’ flora to carriers of ‘normal’ flora using non-absorbable enteral antimicrobials. Low-pathogenic micro-organisms such as anaerobes, which generally only cause morbidity, are not affected by SDD by design. There are 15 potentially pathogenic micro-organisms (PPMs) that cause virtually all infections. They can be divided into two groups: ‘normal’, usually present in previously healthy patients and ‘abnormal’, usually present in patients with chronic or acute underlying pathology. “Normal” PPMs include Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, Escherichia coli, Staphylococcus aureus and Candida albicans. The group of “abnormal” PPMs causing ICU-acquired infections would include aerobic Gram-negative bacteria (Klebsiella, Enterobacter, Citrobacter, Proteus, Morganella, Acinetobacter, Serratia and Pseudomonas spp.) and methicillin-resistant Staphylococcus aureus (MRSA).
According to the carrier status of the patient, infections in the ICU are classified. Surveillance specimens together with diagnostic specimens help to distinguish between 3 types of infection: endogenous primary, endogenous secondary and exogenous infections. Primary endogenous infections are caused by PPMs that the patients carried in their digestive tract when they were admitted to ICU. Secondary endogenous infections are those caused by PPMs that the patients did not have in their digestive tract when they were admitted to the ICU, but acquired in their digestive tract during their stay in the ICU. Exogenous infections can occur during ICU admission and are caused by “abnormal” PPMs that were not previously present in the oropharynx and rectum surveillance samples.

PPMs: Potentially Pathogenic Microorganisms
Selective digestive decontamination is a prophylactic technique for the control of the three types of nosocomial infections caused by the 15 PPMs, based on four principles:
- Enteral antimicrobials: usually colistin, tobramycin and nystatin or amphotericin B. To treat secondary endogenous or exogenous infections.
- Four days of intravenous antimicrobials after ICU admission, usually cefotaxime. For primary endogenous or exogenous infections.
- Hygiene measures to prevent cross-contamination between patients or from the environment to the patient.
- Surveillance samples once or twice weekly.
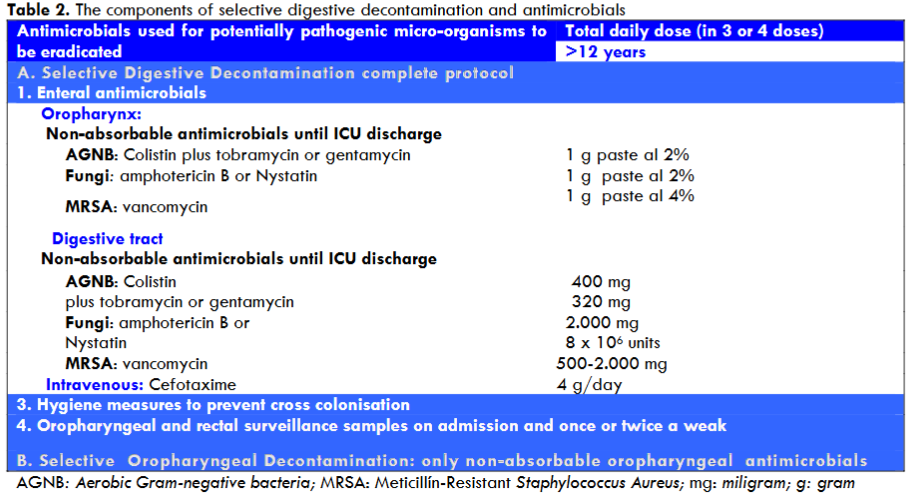
Enteral administration of non-absorbable colistin and tobramycin is used to eradicate ‘abnormal’ aerobic Gram-negative bacteria by decontaminating the gastrointestinal tract and/or suppository. We also administer non-absorbable amphotericin B or nystatin to eradicate fungi. In MRSA-endemic units, topical vancomycin is used. Because of their underlying illness, the ICU patient can’t get rid of non-anaerobic pathogenic flora because their intestinal overgrowth weakens their immune system. The basis for the enteral administration of colistin and tobramycin is the restoration of systemic immunity and the prevention or eradication of “abnormal” non-anaerobic pathogenic flora in the oropharynx and gastrointestinal tract, effectively controlling aspiration and translocation of these microorganisms to the lower respiratory tract and blood.
Enteral antibiotics have been effective in the prevention of secondary endogenous infections. On the other hand, selective oropharyngeal decontamination (SOD) with enteral antibiotics does not help with either endogenous or exogenous primary infections. Routine hygiene measures are essential to reduce hand contamination and subsequent transmission from external sources. Finally, the SDD protocol includes surveillance sampling on admission and once or twice a week. Knowledge of carrier status allows the effectiveness of this protocol to be assessed. The SDD is designed to reduce endogenous infections by preventing carrier status. Nevertheless, reducing the frequency of carrier status reduces colonisation pressure and therefore also minimises the likelihood of exogenous infection through patient-to-patient cross-transmission from the hands of healthcare professionals.
The aim of this non-systematic review is firstly to provide an overview of the information that is available on the use of SDD and clinical, antimicrobial resistance and safety in ICUs with low and also moderate to high levels of antimicrobial resistance. A literature search of EMBASE/MEDLINE/PubMed was performed, including articles published between 1984 and 2024.
Clinical effects of selective digestive decontamination
Selective digestive decontamination has been used for more than 40 years. At least 73 randomised trials (RCTs) and 16 meta-analyses in critically ill patients have shown that SDD prevents severe infections, reduces mortality and is cost-effective.
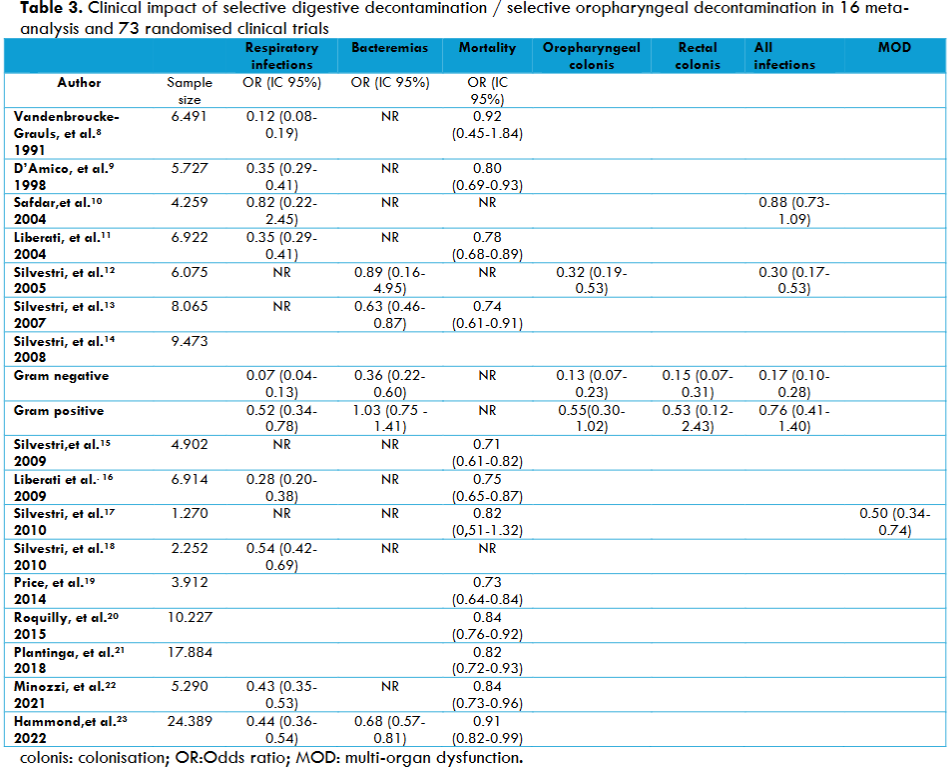
Since 1983, SDD has been proposed as a measure for the prevention of infection in ICU patients, the majority of whom have respiratory failure, are on mechanical ventilation, have reversible morbidity or are admitted after major surgery, coma or shock. A systematic review and meta-analysis of 32 studies involving patients on mechanical ventilation showed that the use of SDD was associated with a lower risk of ventilator-associated pneumonia (VAP) (Relative risk (RR), 0.44 [95% confidence interval (CI), 0.36-0.54]), as did many other meta-analyses. A reduced risk of ICU-acquired bacteraemia (RR, 0.68 [95% CI, 0.57-0.81]) was also found by Hammond et al. In the group of non-catheter-associated bacteraemia caused by Gram-negative bacteria, SDD showed a significant reduction, as in several studies. The hypothesis that this type of infection is predominantly exogenous is supported by the lack of effect of SDD on intravascular catheter-associated bacteraemia a hypothesis supported by the reduced incidence of catheter-associated bacteraemia with purely hygienic measures.
A recent multicentre study in ICUs demonstrated an association between SDD and nosocomial infections in patients on continuous renal replacement therapy (RRT). SDD was associated with a statistically significant decrease in RRT-associated nosocomial infections odds ratio (OR): 0.10, 95% confidence interval [CI]: (0.04-0.26) and the incidence density ratio (IDR): 0.156, 95% CI:(0.048-0.506) of multi-resistant bacteria (MRB). Furthermore SDD is now the standard of care in the Netherlands and is used occasionally in intensive care units in other countries. In De Smet et al. study the SDD group had a lower incidence of ICU-acquired bacteraemia with Enterobacteriaceae than those who received the SOD group.
In critically ill patients, SDD has previously been associated with a low rate of candidaemia. Between 1994 and 2013, only 51 cases of candidaemia were observed among 12.491 ICU patients receiving SDD. In a recent study, the rate of ICU-acquired candidaemia was lower in the SDD group than in the SOD group or in the standard care group. Both in the matched cohort analysis of patients who received mechanical ventilation for at least 48 hours and in the overall population, the rate of ICU-acquired candidaemia was lower in patients who received SDD (0.8% versus 0.3%; p = 0.012 and 0.7% versus 0.3%; p = 0.006, respectively). Patients with ICU-acquired candidaemia had also a higher mortality rate (48.4% versus. 29.8%; p< 0.001).
The SDD cohort had significantly lower rates (p < 0.001) of VAP (1.9 versus 9.3 events per 1000 ventilation days) and MRB infection (0.57 versus 2.28 events per 1000 ICU days) and a non-significant reduction in secondary bacteraemia (0.6 versus 1.41 events per 1000 ICU days) compared with the non-SDD cohort, as well as secondary infections in COVID patients. Van der Meer et al. found that VAP occurred in 10% of patients, although 96% spent more than 5 days in the ICU. Buil et al. showed that their observation indicated that SDD is effective in decolonising yeast from COVID-19 patients in the ICU and may help to reduce candidaemia in this patient group. The same result was found in the study by Ruiz-Santana et al.
A retrospective before and after cohort study between January 2017 and June 2023 found that 21 (77.8%) and 21 (46.7%) burn patients in the non-SDD and SDD groups, respectively, developed nosocomial infections (p = 0.009). The number of hospital-acquired candidaemia episodes was 2.52 (1.21-3.82) and 1.13 (0.54-1.73), respectively (p = 0.029). SDD was linked to a reduced incidence of bacterial nosocomial infections and a reduction in the number of nosocomial infections per patient.
In addition, Massart et al. showed that there were 35/143 deaths in the standard care group versus 22/151 in the post-implementation group (p = 0.046) and this difference remained in a multivariable Cox model (HR = 0.58; 95CI [0.34-0.95] p = 0.048). SDD appears to be associated with improved outcomes in critically ill immunocompromised patients.
Against SDD, Hurley JC believes that there are various other paradoxical discrepancies between the results of RCTs on SDD and the results of studies of interventions to prevent infections in ICUs other than SDD, as summarised in Cochrane reviews. The median event rates in the control groups within RCTs of SDD are generally high, whereas the median event rates in the intervention groups are not unusually low. The results for bacteraemia in the meta-analysis also highlight this discrepancy. The rates of ICU-acquired bacteraemia are generally high in the control groups of the SDD RCTs, whereas the rates of bacteraemia in the intervention groups are not unusually low. In contrast, the bacteraemia rates in the control and intervention groups of the SDD CRTs are all close to a contemporary literature benchmark derived from French ICUs of 7%.
In favour of SDD, the Spanish Zero VAP programme has a good rate of infection. As in the Netherlands, Spain and France have national recommendations for the use of SDD to prevent ICU-acquired infections such as ventilator-associated pneumonia. The Whale JJ et al. also recommended use of SDD is one of the few settings in critical care where large RCTs and systematic reviews demonstrate a clear benefit on major outcomes, including mortality and nosocomial infections.
Mortality
A number of meta-analyses have shown a significant reduction in mortality. Two recent studies have provided evidence of a reduction in mortality with SDD. A Cochrane review found that patients treated with SDD had a significant reduction in mortality compared with those receiving placebo or no treatment (RR: 0.84 [95% CI: 0.73-0.96]). This corresponds to a mortality rate of 0.05% in patients treated with SDD compared to 0.1% in patients receiving placebo or no treatment. This means that for every 26 patients treated with SDD, one death was prevented.
The meta-analysis by Hammond HE and colleagues showed that in patients with mechanical ventilation the posterior probability of reduced in-hospital mortality associated with SDD was 99.3%, in a systematic review of 32 RCTs with 24.389 participants. The summary risk ratio was 0.91, indicating a statistically significant reduction in mortality compared with standard care. In another study, the pooled estimated risk ratio (RR) for mortality for SDD compared with standard care was 0.91 (95% credible interval [CrI], 0.82-0.99; I2 = 33.9%; moderate certainty), with a 99.3% posterior probability that SDD reduced hospital mortality. The beneficial association of SDD was clear in trials with an intravenous agent (RR, 0.84 [95% CrI, 0.74-0.94]). It was not clear in trials without an intravenous agent (RR, 1.01 [95% CrI, 0.91-1.11]).
The SuDDICU investigators concluded that the use of SDD did not significantly reduce in-hospital mortality compared with standard care (27 versus 29.1%; OR: 0.91 [95% CI: 0.82-1.02]; p = 0.12). This finding reflected a 1.7% reduction in mortality (95% CI: -4.8 – 1.3%) in patients who received SDD. The 2-percentage point reduction in mortality observed in the SuDDICU trial corresponds to a number needed to treat of 50 to avoid 1 death, which is a clinically relevant effect size. In the post-hoc analysis of the SuDDICU randomised trial in critically ill, mechanically ventilated patients with acute brain injury, SDD significantly reduced in-hospital mortality compared to standard care without SDD. In patients with acute brain injury, in-hospital mortality rates were 32.3% and 38.0% in the SDD and standard care groups, respectively. The unadjusted OR for SDD was 0.76 (95% CI: 0.63- 0.92; p = 0.004). Wittekamp BH et al. showed a significant reduction in mortality in a randomised trial. Luque-Paz et al. suggested that SDD may be associated with a reduction in bacterial VAP and improved 28-day survival in COVID-19 patients.
Conversely, Hurley JC considers that the basis for the difference in mortality between control and intervention groups in SDD trials is still unclear, Several contradictory results are consistent with a spillover effect that would confound the inference of benefit from RCTs. Moreover, this spillover would be a herd effect. Although there has been repeated opposition to SDD, those who use SDD in the ICU, who began using it many years ago, continue to do so because clinical experience tells them that it is a good and safe intervention for patients. Those who oppose it may never have used it and in the meantime a potentially effective intervention is being withheld from patients while others with much less clinical evidence are used. Few things in medicine significantly reduce mortality in critically ill patients.
Selective digestive decontamination and antimicrobial resistance
Expert concern about the emergence of antimicrobial resistance is the main reason why SDD is not used, despite numerous RCTs and meta-analyses showing a reduction in healthcare-associated infections. Most RCTs were conducted in areas with low antimicrobial resistance for multidrug-resistant Gram-negative bacteria. Some studies were conducted in countries with moderate to high levels of resistance, such as Spain, Portugal, Belgium or France.
Ecological studies suggest that SDD increases antimicrobial resistance. An increase in antibiotic resistance, particularly aminoglycoside resistance genes, in the anaerobic flora during SDD has been shown in a study using metagenomic techniques. Van Seane et al. reported significant inactivation of colistin by faeces. In addition, aminoglycoside resistance increases the likelihood of acquiring colistin resistance. Indeed, several studies have described outbreaks of colistin- and aminoglycoside-resistant Enterobacteriaceae during SDD. Therefore, it is necessary to monitor resistance to both antibiotics during the use of SDD. Buelow E et al. found that the gut microbiota of SDD-treated ICU patients was significantly different from that of healthy subjects. Negative effects on the resistome were limited to selection for four resistance genes. The data suggest that the risks associated with ICU admission and SDD on selection for antibiotic resistance were limited, although it was not possible to separate the effects of SDD from confounding variables in the patient cohort. Nonetheless, we found evidence that recolonisation of the gut with antibiotic-resistant bacteria may occur after discharge from the ICU and discontinuation of SDD.
Recently, van Doorn-Schepens et al. demonstrated that the SDD group clustered into two subgroups. One subgroup showed a decrease in Proteobacteria. The other subgroup showed a shift in Proteobacteria species. This study demonstrates that SDD not only reduces the colonisation of the gastrointestinal tract with potentially pathogenic Gram-negative microorganisms but also decreases the abundance of normal colonisers of our gastrointestinal system and results in a change in the overall composition of the microbiota. Similarly, in a 16-year ecological study in Spain, Lloréns-Villar Y et al. found no relevant changes in the overall susceptibility rate after the implementation of SDD; susceptibility rates were not lower than in Spanish ICUs without SDD. Buitinck S et al. in a study of more than 21 years of SDD found that the incidence rates of resistant microbes at the ICU level did not increase significantly over time, but the background resistance rates increased. An overall ecological effect of prolonged use of SDD by counting resistant microorganisms in the ICU was not demonstrated in a country with relatively low rates of resistant microorganisms.
In the enteral solution and paste of SDD, topical colistin and tobramycin are usually used. In a French intensive care unit, SDD was compared with chlorhexidine body wash plus intranasal mupirocin and placebo in a 2 × 2 factorial design. The proportion of patients developing colistin-resistant Gram-negative bacteria infections ranged from 11% in patients who received a double placebo to 2% in patients who received both SDD and chlorhexidine body washing/mupirocin (p = 0.005). A similar (although not significant) effect was found for tobramycin-resistant Gram-negative bacteria infections, from 17% to 9% in patients treated with double placebo and SDD plus chlorhexidine/mupirocin, respectively. Sánchez-Ramírez C et al. found a significant reduction (p < 0.001) in MRB infections after 4 years of SDD use (RR: 0.31; 95% CI: 0.23-0.41), which was correlated with low rates of colistin- and tobramycin-resistant colonisation.
A retrospective study by Halaby et al. has been published that supports the view that SDD favors the emergence of resistance. They analysed the impact of SDD over a five-year period in a Dutch ICU. Logistic regression analysis showed a significant association between SDD and tobramycin resistance and also that colistin resistance emerged in Extended spectrum Beta-lactamase after the introduction of SDD, with a significant increase in bacteraemia due to colistin-resistant organisms. It was therefore recommended that SDD should not be used in outbreaks where resistant bacteria are prevalent. Similarly, Lübbert et al. in their study using SDD for 7 days with colistin and gentamicin in 14 consecutive patients with Klebsiella pneumoniae Carbapenemase found an increase in resistance of 19% to colistin and 45% to gentamicin compared with no resistance in the control group. In contrast, an almost simultaneous analysis of a single-centre Dutch study by Oostdijk et al. was published, in which the acquisition rates of colistin-resistant Gram-negative bacteria in the respiratory tract were low and comparable with and without SDD and topical colistin use.
In a letter to the editor, Zanstra et al. considered the study by Halaby et al. to be an inappropriate with a low level of evidence. The reasons given were: 1) it did not provide data on the type of patients included; 2) it did not use appropriate epidemiological estimators such as prevalence, cumulative incidence, etc; 3) Imported and ICU-acquired pathogens were not distinguished; 4) it did not define whether pathogens occurred in the SDD or non-SDD period; 5) it did not detail the periodicity of sampling, taking diagnostic samples without taking surveillance samples. They also pointed out that resistance in SDD is rare.
A meta-analysis by Daneman et al. of the effect of SDD on resistance in the ICU included 64 studies using SDD and SOD, of which 47 were RCTs and 35 provided resistance data. No association was found between use and emergence of resistance. No differences were found in Gram-negative bacteria resistance to aminoglycosides and quinolones, but In patients with SDD, a reduction in colistin-resistant gram-negative bacteria (OR 0.58, 95% CI: 0-46-0.72) and third-generation cephalosporins was observed.
There is only 1 cluster randomised trial, the RGNOSIS trial, for settings with a higher prevalence of MRB. Wittekamp BH el al. applied SDD daily chlorhexidine 2% body wash and a programme to improve hand hygiene were used as standard for a short period. Each ICU was randomised to 3 separate 6-month intervention periods with either clorhexidine 2% mouthwash, SOD or SDD, all applied 4 times daily, after a baseline period of 6 to 14 months. Adjusted SDD ratios for 28-day mortality were 1.07 (95%CI: 0.86-1.32), 1.05 (95%CI: 0.85-1.29) and 1.03 (95%CI: 0.80-1.32) for clorhexidine, SOD and SDD, respectively, compared with baseline. Among patients receiving mechanical ventilation in ICUs with moderate to high antibiotic resistance prevalence, the use of clorhexidine mouthwash, SOD or SDD was not associated with a reduction in ICU-acquired bloodstream infections caused by MRB compared with standard care. The completed SDD protocol needs to be evaluated according to surveillance sample cultures, sometimes it is necessary to adjust and switch to another aminoglycoside such as gentamycin or use paromomycin if there is an MRB that is resistant to colistin.
Selective digestive decontamination and multi-resistant bacteria infections in COVID-19 patients
A study by Ruiz-Santana S et al. on COVID-19 patient found that patients in the SDD cohort had significantly lower rates (p < 0.001) of VAP and MRB infections (0.57 versus 2.28 events per 1000 ICU days) and a nonsignificant reduction in secondary bacteraemia among patients in the non-SDD cohort. Infections caused by multi-resistant pathogens occurred in 5 patients in the SDD cohort and 21 patients in the non-SDD cohort (p = 0.006). Similarly, a study in France by Ejzenberg M et al. showed that the use of SDD reduced the occurrence of VAP (OR 0.536; CI: 0.338-0.851; p = 0.017) in a multivariate analysis adjusted for confounders. Their pre-post observational study suggests that the use of SDD in a structured VAP prevention protocol appears to reduce the incidence of VAP in COVID-19 patients without changing the incidence of MRB.
Antimicrobial resistance after ICU discharge
The incidence of infections was monitored for 14 days after ICU discharge in two university hospitals by De Smet et al. There was no difference in the incidence of hospital-acquired infections after ICU discharge, which were 11.2, 12.9 and 8.3 per 1000 risk days for patients receiving SDD (n = 296), SOD (n = 286) and standard care (n = 289), respectively. Of patients who died in hospital after ICU discharge (n = 58), 8 (14%) developed a nosocomial infection after ICU discharge: 3 of 21 after SDD, 3 of 15 after SOD and 2 of 22 after standard care. They concluded that they rejected the hypothesis that an increased rate of infection after ICU discharge would affect the clinical outcome and mortality of patients who had received SDD or SOD in the ICU, although there was a trend towards more infections, particularly superficial surgical wound infections, in these patients after ICU discharge. De Jonge E et al. found that intestinal carriage at ICU discharge and the rate of MRB acquisition after ICU discharge were lower after SDD than after SOD. The prevalence of intestinal MRB carriage 10 days after ICU discharge was similar in both groups, suggesting rapid intestinal MRB clearance after ICU discharge.
Effects of long-term use of Selective digestive decontamination/Selective oropharyngeal decontamination
Several studies have shown that there is no increase in resistance with long-term use of SDD. This is described in 2 longitudinal studies, one French and the other German. The French study was a 6-year retrospective case-control study, also in an ICU of a tertiary hospital. The German study was observational and prospective over 5 years in a third level surgical ICU. In the Spanish study, Ochoa-Ardila et al. showed no increase in resistance over 5 years in an ICU with SDD. They included 1.588 patients with SDD in their 5-year study. The incidence of MRB remained stable: 18.91 per 1000 bed days. The incidence of resistant enterobacteria remained stable: Pseudomonas aeruginosa resistance to tobramycin, amikacin and ciprofloxacin was significantly reduced; there was an increase in P. aeruginosa resistant to ceftazidime and imipenem, linked to an increase in imipenem consumption; the incidence of other non-fermenting bacilli and MRSA was nearly zero. They concluded that prolonged use of SDD was not associated with increased acquisition of resistant flora.
In another Dutch longitudinal study involving 38 ICUs with or without SDD and SOD use over 4 years, they found a statistically significant increase in tobramycin-resistant Enterobacteriaceae resistance in ICUs without SDD or SOD, and no such trend in ICUs using SDD or SOD but found a decrease in Gram-negative bacteria resistance to the antibiotics studied, such as colistin, tobramycin, ceftazidime and cefotaxime or ceftriaxone. Wittekamp BH et al. in a post hoc analysis of 2 consecutive 7-year multicentre cluster RCTs in 5 ICUs showed that resistance to tobramycin in respiratory and rectal specimens decreased significantly and resistance to colistin in respiratory specimens did not change with long-term use of SDD or SOD compared to standard treatment. Long-term use did not increase the prevalence of colistin- and tobramycin-resistant Gram-negative bacteria of SDD or SOD compared with standard treatment. Tobramycin resistance did not increase during a median of 7 years of SDD or SOD treatment. Similarly, Sanchez-Ramirez C et al. described low rates of colistin and tobramycin resistance in a 4-year prospective study in a high-resistance ICU. In another 16-year Spanish study mentioned above, no significant changes in the overall susceptibility rate were observed after the introduction of the SDD; susceptibility rates were not less than in Spanish ICUs without SDD. Another 21-year longitudinal study using SDD observed no significant increase in the incidence rates of resistant microorganisms over time, but the baseline resistance rates measured at admission for cephalosporins, polymyxin B/colistin, and ciprofloxacin showed increases of 7.9%, 3.5%, and 8.0%, respectively. Rodríguez-Gascón A et al. in a 4-year retrospective ecological study found a significant reduction in Escherichia coli resistance to amoxicillin/clavulanic acid in ICUs using the SDD protocol and resistance of Enterococcus faecalis to high-concentration gentamycin and high-concentration streptomycin. A significant increase in the resistance of coagulase-negative staphylococcus to linezolid was also observed in the non-SDD ICU. Overall, the level of resistance in the SDD ICU was lower or of the same order of magnitude as in the ICU without SDD and as reported in the Spanish national registry.
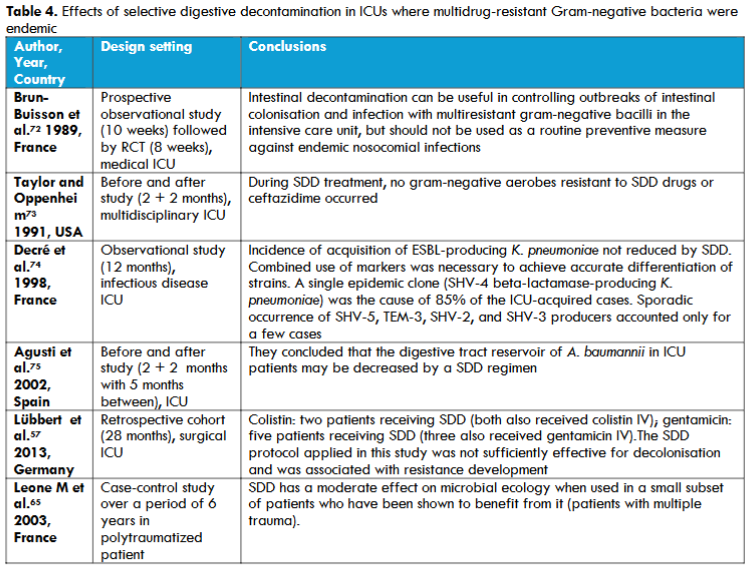
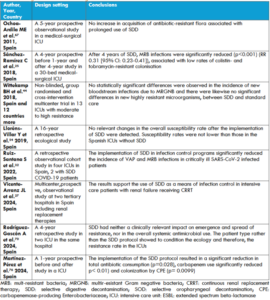
Table 4. Effects of selective digestive decontamination in ICUs where multidrug-resistant Gram-negative bacteria were endemic
Adverse effects of Selective digestive decontamination
The administration of SDD is a safe procedure. Oesophageal or jejunal obstruction has been described as a complication of oral administration of paste and enteral solution. This complication can be avoided with good oral hygiene control. The most recent Cochrane review concluded that no evidence was available on the adverse effects of SDD (gastrointestinal disorders or allergic reactions) due to insufficient reporting and poor data.
Conclusions
The use of SDD is one of the few fields in the ICU where large RCTs and systematic reviews demonstrate a clear benefit on major mortality, nosocomial infections and morbidity, with no apparent adverse effects. In settings with a low prevalence of antimicrobial resistance, SDD is consistently associated with reduced antimicrobial resistance and improved patient outcomes. For settings with a higher prevalence of multidrug-resistant pathogens, there is only 1 cluster-randomised trial and this showed no evidence of better patient outcomes. But it had several problems: it did not use the full SDD protocol with intravenous antibiotics and it only used it for six months, which is a short time. Some doctors believe that large-scale trials are still needed to determine the effectiveness of SDD in specific patient populations and settings with a high prevalence of multidrug-resistant pathogens, but are they really necessary? Several ICUs that started using SDD years ago continue to do so because their resistance monitoring data and clinical effects show that it is.
Conflicts of interest Statement
No conflicts of interest.
Funding statement
None.
We are grateful to Sergio Ruiz Santana, Professor Emeritus of Medicine at the University of Las Palmas de Gran Canaria, for his great dedication and scientific contribution over the years, which allowed us to initiate and consolidate the routine use of SDD in our ICU. Also, to Pedro Saavedra Santana, Professor of Mathematics and Informatics at the University of Las Palmas de Gran Canaria and statistical advisor in our SDD studies, without whose collaboration this review would not have been possible.
References
2. Van Saene HKF, Damjanovic V, Murray AE, de la Cal MA. How to classify infections in intensive care units the carrier state, a criterion whose time has come? J Hosp Infect. 1996; 33:1-12.
3. Cerdá E, Abella A, de la Cal MA, et al. Enteral vancomycin controls methicillin-resistant Staphylococcus aureus endemicity in an intensive care burn unit: A 9- year prospective study. Ann Surg. 2007; 245:397-407.
4. Silvestri L, van Saene HK, Milanese M, et al. Prevention of MRSA pneumonia by oral vancomycin decontamination: a randomised trial. Eur Respir J. 2004; 23:921-6.
5. Taylor N, van Saene HKF, Abella A, Silvestri L, Vucic M, Peric M. Selective digestive decontamination. Why don’t we apply the evidence in the clinical practice? Med Intensiva. 2007; 31:.3.
6. Tejerina-Álvarez EE, de la Cal López MÁ. Selective decontamination of the digestive tract: concept and application. Med Intensiva (Engl Ed). 2023; 47(10):603-615.
7. Álvarez Lerma F, Sánchez García M, Lorente L, et al. Guidelines for the prevention of ventilator associated pneumonia and their implementation. The Spanish ‘‘Zero-VAP’’ bundle. Med Intensiva. 2014; 38:226-36,
8. Vandenbroucke-Grauls CM, Vandenbroucke JP. Effect of selective decontamination of the digestive tract on respiratory tract infections and mortality in the intensive care unit. Lancet. 1991; 338:859-62.
9. D’Amico R, Pifferi S, Leonetti C, Torri V, Tinazzi A, Liberati A. Effectiveness of antibiotic prophylaxis in critically ill adult patients: systematic review of randomised controlled trials. Br Med. 1998; 316:1275-85.
10. Safdar N, Said A, Lucey MR. The role of selective decontamination for reducing infection in patients undergoing liver transplantation: a systematic review and meta-analysis. Liver Transpl. 2004; 10:817-27.
11. Liberati A, D’Amico R, Pifferi S, Torri V, Brazzi L. Antibiotic prophylaxis to reduce respiratory tract infections and mortality in adults receiving intensive care. Cochrane Database Syst Rev 2004; CD000022.
12. Silvestri L, van Saene HK, Milanese M, Gregori D. Impact of selective decontamination of the digestive tract on fungal carriage and infection: systematic review of randomised controlled trials. Intensive Care Med. 2005; 31:898-910.
13. Silvestri L, van Saene HK, Milanese M, Gregori D, Gullo A. Selective decontamination of the digestive tract reduces bloodstream infections and mortality in critically ill patients. Systematic review of randomised, controlled trials. J Hosp Infect. 2007; 65:187-203.
14. Silvestri L, van Saene HK, Casarin A, Berlot G, Gullo A. Impact of selective decontamination of the digestive tract on carriage and infection due to Gram-negative and Gram-positive bacteria: a systematic review of randomised controlled trials. Anaesth Intensive Care. 2008; 36:324-38.
15. Silvestri L, van Saene HK, Weir I, Gullo A. Survival benefit of the full selective digestive decontamination regimen. J Crit Care. 2009;24: 474.e7-14.
16. Liberati A, D’Amico R, Pifferi S, Torri V, Brazzi L, Parmenlli E. Antibiotic prophylaxis to reduce respiratory tract infections and mortality in adults receiving intensive care. Cochrane Database Syst Rev. 009 Oct 7; CD000022.
17. Silvestri L, van Saene HK, Zandstra DF, Marshall JC, Gregori D, Gullo A. Impact of selective decontamination of the digestive tract on multiple organ dysfunction syndrome: systematic review of randomised controlled trials. Crit Care Med. 2010; 38:1370-6.
18. Silvestri L, Milanese M, Taylor N, Piacente N, Zandstra DS, van Saane. Selective digestive decontamination reduces ventilator-associated tracheobronchitis. Respir Med. 2010; 104:1953-5.
19. Price R, MacLennan G, Glen J. SuDDICU Collaboration. Selective digestive or oropharyngeal decontamination and topical oropharyngeal chlorhexidine for prevention of death in general intensive care: Systematic review and network meta-analysis. BMJ. 2014; 348:g2197.
20. Roquilly A, Marret E, Abraham E, Asehnovu K. Pneumonia prevention to decrease mortality in the ICU. A systematic review and metaanalysis. Clin Infect Dis. 2015; 60:64-75.
21. Plantinga NL, de Smet AMGA, Oostdijk EAN, et al. Selective digestive and oropharyngeal decontamination in medical and surgical ICU patients: Individual patient data meta-analysis. Clin Microbiol Infect. 2018; 24:505-13.
22. Minozzi S, Pifferi S, Brazzi L, Pecoraro V, Montrucchio G, D’Amico R. Topical antibiotic prophylaxis to reduce respiratory tract infections and mortality in adults receiving mechanical ventilation. Cochrane Database Syst Rev. 2021:CD000022.
23. Hammond NE, Myburgh J, Seppelt I, et al. Association Between Selective Decontamination of the Digestive Tract and In-Hospital Mortality in Intensive Care Unit Patients Receiving Mechanical Ventilation: A Systematic Review and Meta-analysis. 2022; 328:1922-34.
24. Stoutenbeek CP, van Saene HK, Miranda DR, Zandstra DF. The effect of selective decontamination of the digestive tract on colonisation and infection rate in multiple trauma patients. Intensive Care Med. 1984; 10: 185-192.
25. Sánchez-Ramírez C, Hípola-Escalada S, Cabrera-Santana M, et al. Long-term use of selective digestive decontamination in an ICU highly endemic for bacterial resistance. Crit. Care. 2018; 22;141.
26. Pronovost P, Needham D, Berenholtz S, et al. An intervention to decrease catheter related bloodstream infections in the ICU. N Engl J Med. 2006; 355:2725-32.
27. Vicente-Arranz JL, Sánchez-Ramírez C, Saavedra P, et al. The Relationship between Selective Digestive Decontamination and Nosocomial Infections in Patients Receiving Continuous Renal Replacement Therapy in ICUs: A Multicenter Study, J Clin Med.2024; 13:1.
28. Francis, JJ, Duncan EM, Prior ME, et al. Comparison of four methods for assessing the importance of attitudinal beliefs: An international Delphi study in intensive care settings. Br. J. Health Psychol. 2014; 19: 274-291.
29. De Smet AM, Kluytmans JA, Cooper BS, et al. Decontamination of the digestive tract and oropharynx in ICU patients. N. Engl. J. Med. 2009; 360, 20-31.
30. Buitinck SH, Zandstra DF, van der Voort PH. ICU-acquired candidemia within SDD: low incidence in a 20-year longitudinal database. Intensive Care Med 2016; 42(6):1094-1095.
31. Reizine F, Massart N, Joussellin V, et al. Association between selective digestive decontamination and decreased rate of acquired candidemia in mechanically ventilated ICU patients: a multicenter nationwide study. Critical Care. 2023; 27:1: 494.
32. Ruiz-Santana S, Mora-Quintero ML, Saavedra P, et al. COVID-19 Secondary Infections in ICU Patients and Prevention Control Measures: A Preliminary Prospective Multicenter Study. Antibiotics (Basel). 2022; 11(8):1016.
33. Van der Meer S.B, Figaroa G, van der Voort PHJ, Nijsten MW. Pillay J. Ventilator-associated pneumonia in critically-ill patients with COVID-19 in a setting of selective decontamination of the digestive tract. Crit. Care. 2021; 25, 445.
34. Buil JB, Schouten JA, Wauters J, van de Hoeven H, Verweij PE. Absence of candidemia in critically ill patients with COVID-19 receiving selective digestive decontamination. Intensive Care Medicine. 2022; 48:5 611-612.
35. Gangneux JP, Dannaoui E, Fekkar A, et al. Fungal infections in mechanically ventilated patients with COVID 19 during the first wave: the French multicentre MYCOVID study. Lancet Respir Med. 2022; 10(2):180-90.
36. Pérez-Torres D, Martín-Luengo AI, Cuenca-Rubio C, et al. Selective decontamination of the digestive tract in a burns unit reduces the incidence of hospital-acquired infections: A retrospective before-and-after cohort study. Med Intensiva 2024; 48:12 677-685.
37. Massart N, Dupin C, Legris E, et al. Prevention of ICU-acquired infection with decontamination regimen in immunocompromised patients: a pre/post observational study. Eur J Clin Microbiol Infect Dis. 2023; 42:10 1163-1172.
38. Hurley JC. Selective digestive decontamination-Con. Intensive Care Med. 2023; 49:982-983.
39. Garrouste-Orgeas M, Timsit JF, Tafflet M, et al. Excess risk of death from intensive care unit-acquired nosocomial bloodstream infections: a reappraisal. Clin Infect Dis. 2006; 42:1118-1126.
40. Leone M, Bouadma L, Bouhemad, et al. Hospital acquired pneumonia in ICU. Anaesth Crit Care Pain Med. 2018; 37:83.
41. De Waele JJ, Leroux Roels I Depuydt P. Selective digestive decontamination Pro. Intensive Care Med. 2023; 49:979-981.
42. Myburgh JA, Seppelt IM, Goodman F, et al. SuDDICU Investigators for the Australian and New Zealand Intensive Care Society Clinical Trials Group. Effect of Selective Decontamination of the Digestive Tract on Hospital Mortality in Critically Ill Patients Receiving Mechanical Ventilation: A Randomised Clinical Trial. Jama 2022; 328, 1911-1921.
43. Young PJ, Devaux A, Li Q, et al. Selective digestive tract decontamination in critically ill adults with acute brain injuries: a post hoc analysis of a randomised clinical trial. Intensive Care Med. 2024; 50:1 56-67.
44. Wittekamp BH, Plantinga NL, Cooper BS, et al. Decontamination strategies and bloodstream infections with antibiotic resistant microorganisms in ventilated patients: A randomised clinical trial. JAMA. 2018; 320:2087-98.
45. Luque Paz D, Tattevin P, Jaubert P, Reizine F, Kouatchet A. Selective digestive decontamination to reduce the high rate of ventilator-associated pneumonia in critical COVID-19. Anaesth Crit Care Pain Med. 2021; 41(1): 100987.
46. Sánchez Ramírez C, Hípola Escalada S, Lara Franco L, et al. Eleven years of implementation of selective digestive decontamination in a mixed ICU: impact on nosocomial multidrug-resistant infection, antibiotic consumption and colistin and tobramycin colonization. Intensive Care Med Exp .2023; 11(1):A0333.
47. Jernberg C, Löfmark S, Edlund C, Jansson JK. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology. 2010; 156:3216-23.
48. Buelow E, Gonzalez TB, Versiuis D, et al. Effects of selective digestive decontamination (SDD) on the gut resistome. J Antimicrob Chemother 2014; 69: 2215-23.
49. Van Saene JJ, van Saene HK, Stoutenbeek CP, Lerk CF. Influence of faeces on the activity of antimicrobial agents used for decontamination of the alimentary canal. Scand J Infect Dis. 1985; 17:295-300.
50. Halaby T, Al Nalemi N, Kluytmans J, van der Palen J, Vandenbroucke-Grauls CM. Emergence of colistin resistance in Enterobacteriaceae after the introduction of selective digestive tract decontamination for the treatment in an intensive care unit. Antimicrobial Agents Chemother. 2013; 57:3224-9.
51. Al Naiemi N, Heddema ER, Bart A, et al. Emergence of multidrug-resistant Gram-negative bacteria during selective decontamination of the digestive tract on an intensive care unit. J Antimicrob Chemother. 2006; 58:853-6.
52. Buelow E, Bello González TDJ, Fuentes S, et al. Comparative gut microbiota and resistome profiling of intensive care patients receiving selective digestive tract decontamination and healthy subjects. Microbiome. 2017; 14;5(1):88.
53. Van Doorn-Schepens MLM, Abis GSA, Oosterling SJ, et al. The effect of selective decontamination on the intestinal microbiota as measured with IS-pro: a taxonomic classification tool applicable for direct evaluation of intestinal microbiota in clinical routine. Eur Journal of Clinical Microb and Infect Diseases. 2022; 41:11 (1337-1345).
54. Lloréns-Villar Y, Tusell F, Canut A, et al. Antibiotic susceptibility trend before and after long-term use of selective digestive decontamination: a 16 year ecological study. Journal of Antimicrobial Chemotherapy. 2019; 74:8 2289-2294.
55. Buitinck S, Jansen R, Rijkenberg S, et al. The ecological effects of selective decontamination of the digestive tract (SDD) on antimicrobial resistance: A 21-year longitudinal single-centre study. Critical Care. 2019; 23:1
56. Camus C, Bellissant E, Sebille V, et al. Prevention of acquired infections in intubated patients with the combination of two decontamination regimens. Crit Care Med. 2005; 33:307-14.
57. Lübbert C, Faucheux S, Becker-Rux D, et al. Rapid emergence of secondary resistance to gentamicin and colistin following selective digestive decontamination in patients with KPC-2- producing Klebsiella pneumoniae: a single-centre experience. Int J. Antimicrob Agents. 2013; 42:565-70.
58. Oostdijk EA, Smits L, De Smet AM, Leverstein-van Hall MA, Kesecioglu J, Bonten MJ. Colistin resistance in gram negative bacteria during prophylactic topical colistin use in intensive care units. Intensive Care Med. 2013; 39:653-60.
59. Zandstra DF, Rommes JH, de la Cal MA, Silvestri L, Taylor N, van Saene HK. Colistin resistance during selective digestive tract decontamination is uncommon. Antimicrob Agents Chemother [Letter] 2014; 58:626.
60. Daneman N, Sarwar S, Fowler A, Cuthbentson BH. Effect of selective decontamination on antimicrobial resistance in intensive care units: a systematic review and meta-analysis. Lancet Infect Dis. 2013; 13:328-41.
61. Sánchez-Ramírez C, Hernández Viera MA, Morales Sirgado RE, et al. Assesment of enteral paromomycin to eradicate colistin and carbapenemase resistant microorganisms in rectal colonisation to prevent ICU-acquired infections. Intensive Care Med Exp. 2019; 7(3): A1067.
62. Ejzenberg M, Wicky PH, Patrier J, et al. Influence of selective digestive decontamination on survival and prevention of ICU acquired infections in Sars-Cov-2 infected patients with mechanical ventilation for acute respiratory failure. J. Annals of Intensive Care. 2023 ;13 Suppl 1.
63. De Smet AM, Hopmans TE, Minderhoud AL, et al. Decontamination of the digestive tract and oropharynx: hospital acquired infections after discharge from the intensive care unit. Intensive Care Med 2009; 35:1609-13.
64. De Jonge E, De Wilde RBP, Juffermans NP, et al. Carriage of antibiotic-resistant Gram-negative bacteria after discontinuation of selective decontamination of the digestive tract (SDD) or selective oropharyngeal decontamination (SOD). Crit Care. 2018; 22:243.
65. Leone M, Albanese J, Antonini F, Nguyen-Michel A, Martin C. Long-term (6-year) effect of selective digestive decontamination on antimicrobial resistance in intensive care, multiple-trauma patients. Crit Care Med 2003; 31:2090-5.
66. Heininger A, Meyer E, Schwab F, Marschal M, Unertl K, Krueger WA. Effects of long-term routine use of selective digestive decontamination on antimicrobial resistance. Intensive Care Med. 2006; 32:1569-76.
67. Ochoa-Ardila ME, García Cañas A, Gómez-Mediavilla K, et al. Long term use of selective decontamination of the digestive tract does not increase antibiotic resistance; a 5-year prospective cohort study. Intensive Care Med. 2011; 37:1458-65.
68. Houben AJ, Oostdijk EA, van der Voort PH, Monen JC, Bonten MJ, van der Bij AK; ISIS-AR Study Group. Selective decontamination of the oropharynx and the digestive tract, and antimicrobial resistance: a 4 year ecological study in 38 intensive care units in the Netherlands. J Antimicrob Chemother. 2014; 69:797-804.
69. Wittekamp BH, Oostdijk EA, de Smet AMG, Bonten MJ. Colistin and tobramycin resistance during long- term use of selective decontamination strategies in the intensive care unit: A post hoc analysis. Crit Care. 2015; 19:113.
70. Rodríguez-Gascón A, Lloréns-Villar Y, Solinís MA, Barrasa H, Canut-Blasco A. Does selective digestive decontamination (SDD) increase antibiotic resistance? Long-term comparison of two intensive care units (with and without SDD) of the same tertiary hospital. Eur J of Clin Microbiol & Infect Dis. 2024, 43: 885-893.
71. Smit MJ, van der Spoel JI, de Smet AM, de Jonge E, Kuiper RA, van Lieshout EJ. accumulation of oral antibiotics as an adverse effect of selective decontamination of the digestive tract; a series of three cases. Intensive Care Med. 2007; 33:2025-6.
72. Brun-Buisson C, Legrand P, Rauss A, et al. Intestinal decontamination for control of nosocomial multiresistant gram-negative bacilli. Study of an outbreak in an intensive care unit. Ann Intern Med. 1989; 110:873-81.
73. Taylor ME, Oppenheim BA. Selective decontamination of the gastrointestinal tract as an infection control measure. J Hosp Infect. 1991; 17:271-8.
74. Decré D, Gachot B, Lucet JC, Arlet G, Bergogne-Berezin E, Regnier B. Clinical and bacteriologic epidemiology of extended-spectrum beta-lactamase-producing strains of Klebsiella pneumoniae in a medical intensive care unit. Clin Infect Dis. 1998; 27:834-44.
75. Agusti C, Pujol M, Argerich MJ, et al. Short-term effect of the application of selective decontamination of the digestive tract on different body site reservoir ICU patients colonized by multi-resistant Acinetobacter baumannii. J Antimicrob Chemother.2002; 49:205-8.
76. Martínez-Pérez M, Fernández-Fernández R, Morón R, et al. Descontaminación digestiva selectiva: un enfoque integral para reducir las infecciones nosocomiales y la resistencia a los antimicrobianos en la UCI.J Clin Med. 2024; 13(21):6482.

