Simvastatin for Decompensated Cirrhosis: Efficacy & Safety
Treatment of Decompensated Cirrhosis Patients with Simvastatin: Why, Safety, Efficacy, Futility, and Learning
Alberto E. Muñoz1, M. Fernanda Veltens2
- Sección de Hepatología, Hospital de Gastroenterología Dr. Carlos Bonorino Udaondo, Facultad de Medicina, Universidad de Buenos Aires.
OPEN ACCESS
PUBLISHED: 31 October 2025
CITATION: MUÑOZ, Alberto E.; VELTENS, M. Fernanda. Treatment of Decompensated Cirrhosis Patients with Simvastatin: Why, Safety, Efficacy, Futility, and Learning. Medical Research Archives, [S.l.], v. 13, n. 10, oct. 2025. Available at: <https://esmed.org/MRA/mra/article/view/6958>. Date accessed: 08 nov. 2025. doi: https://doi.org/10.18103/mra.v13i10.6958.
DOI: https://doi.org/10.18103/mra.v13i10.6958
ISSN 2375-1924
Abstract
The article reviews the clinical experimental trials of simvastatin in cirrhosis. It will attempt to engage the reader in this interesting hepatic subject through the following four steps. Firstly, the paper explains the rationale for statins use, ranging from portal hypertension to decompensated cirrhosis. Secondly, the article summarizes the drug safety results (the relationship between liver/muscle injuries and simvastatin dosage/cirrhosis severity), and the efficacy outcomes (survival/quality of life improvements, and reduction of hospitalizations due to cirrhosis complications) in patients with the decompensated state. Thirdly, the paper analyzes the negative results of treating severe degrees of cirrhosis –Child-Pugh class C and acute-on-chronic liver failure, with this medication. Lastly, this article aims to summarize the lessons learned from the few clinical trials in simvastatin therapy on the decompensated stage. The paper will encourage readers to consider simvastatin as a potential treatment option for cirrhosis in the future.
Keywords: Liver cirrhosis; Simvastatin; Nitric oxide; Inflammation; Fibrosis; Quality of life; Hospitalization
Abbreviations in Order of Appearance:
- RCTs: randomized clinical trials
- PH: portal hypertension
- NO: nitric oxide
- eNOS: endothelial nitric oxide synthase
- SVT: simvastatin
- NSBB: non-selective β-blockers
- RFX: Rifaximin
- CK: creatine kinase
- CTP: Child-Pugh
- CYP3A4: cytochrome P450 3A4
- MELD score: Model for End-Stage Liver Diseases score
- AST: aspartate aminotransferase
- ALT: alanine aminotransferase
- HRQoL: health-related quality of life
- EST: at the end of the simvastatin safety trial
- WBC: white blood cells
- HDL-cholesterol: high-density lipoprotein cholesterol
- ACLF: acute-on-chronic liver failure
Introduction
According to the worldwide 2020 data estimations, there are 112 million cases of compensated cirrhosis and 10.6 million cases of decompensated cirrhosis, the latter after one or more complications occur. To be noted, in 2023, decompensated cirrhosis was the 11th leading cause of annual worldwide deaths (2 million, 4% of all deaths). The current approach is aimed at preventing or treating each complication of cirrhosis. Randomized clinical trials (RCTs) have demonstrated the efficacy of this strategy; however, it has had little impact on the natural history in this stage. The only procedure that can modify the natural history of decompensated cirrhosis is etiological treatment aimed at eliminating the cause of the disease, such as direct-acting antivirals for the hepatitis C virus or prolonged alcohol abstinence. Thus, in up to one-third of cases, disease progression slowed or even reversed, from a decompensated to a compensated state. The unique definitive treatment for decompensated cirrhosis is liver transplantation, but not all patients may qualify as suitable candidates or have access to it due to organ shortages. Moreover, the long-term prognosis did not improve due to the complications of immunosuppression, such as infections, cancer, and renal failure, and long-term graft injury and/or failure. On the other hand, the discovery and clinical development of a new drug is a long process (1-2 decades), and it is expensive. Furthermore, patients with cirrhosis have exclusion criteria for key clinical trials, which limit their treatment options. For these reasons, a new therapeutic approach has emerged for decompensated cirrhosis: a disease-modifying treatment using drugs such as statins. These act on key and/or multiple pathophysiological mechanisms of decompensated cirrhosis— including portal hypertension, bacterial translocation, circulatory dysfunction, inflammation, oxidative stress, and immune dysfunction—regardless of the complications present, halting or slowing their progression, or even inducing their “recompensation”. Taking into account all the above, this article will discuss the current outcomes of simvastatin use in decompensated cirrhotic patients.
Why use statins in cirrhosis patients?
Portal hypertension (PH) is directly or indirectly responsible for major clinical cirrhosis complications —ascites, variceal bleeding, and hepatic encephalopathy—and it is the leading cause of death in these patients. PH occurs due to the cirrhotic process itself, which causes mechanical disruption of the liver’s vascular architecture. However, Roberto Groszmann et al. observed, for the first time, that the increase of hepatic vascular resistance is the first pathophysiological phenomenon that causes PH. This mechanism accounts for approximately 30% of the increase in portal pressure. Therefore, modulation of intrahepatic vascular resistance has become a key therapeutic target of PH. Studies in experimental cirrhosis in Groszmann’s Hepatic Hemodynamic Laboratory in New Haven suggested increased hepatic resistance because endothelial nitric oxide (NO) release was impaired in liver microvasculature. In addition, the deficient availability of NO may explain the inability of the intrahepatic vasculature to relax in response to an acute increase in portal blood flow (e.g., after meals). This impaired endothelium-dependent relaxation through NO production —endothelial dysfunction— worsened by postprandial hyperglycemia and hypertriglyceridemia. In cirrhosis, studies have reported either unaltered or decreased hepatic protein levels of endothelial nitric oxide synthase (eNOS); however, decreased hepatic eNOS activity has been uniformly reported. The complex post-translational modifications of eNOS would be responsible for this reduction. Therefore, for several years now, upregulating eNOS activity in the cirrhotic liver has been proposed as a new strategy to correct the increased hepatic vascular tone in these patients. Based on previous findings and experimental cardiology trials, the group led by Jaime Bosch in Barcelona hypothesized that simvastatin (SVT) could act as a hepatic NO donor. In 2004 and 2009, two clinical trials conducted by this group in patients with cirrhosis and PH provided the clinical rationale for using statins in Hepatology. In the first study, acute SVT administration significantly increased NO levels in hepatic venous blood and decreased hepatic sinusoidal resistance, while systemic NO levels and hemodynamics remained unchanged. The second trial was a proof-of-concept RCT demonstrating that SVT lowers portal pressure in patients with cirrhosis and PH, with an excellent safety profile. Furthermore, SVT reduced portal pressure in both patients on non-selective β-blockers (NSBB) and those not on NSBB, suggesting an additive effect when combined with these drugs. Finally, SVT improves quantitative liver function, as evaluated by indocyanine green clearance.
Safety of simvastatin in decompensated cirrhosis patients
These results encouraged the evaluation of using statins in cirrhotic patients, with endpoints other than PH. To date, many observational studies have demonstrated the benefits of statins in patients with cirrhosis. However, a systematic review and meta-analysis of these studies revealed that their quality of evidence was low, as most were retrospective. Therefore, hereinafter, this article will rely on the experimental studies published to date to discuss the currently available clinical evidence on the safety, efficacy, and futility of SVT in patients with cirrhosis. Statins-related adverse events in the general population are frequent, relatively mild, and transient. The most commonly reported are diarrhea, abdominal pain, meteorism, constipation, and headache; however, others cause special concern, such as muscle damage and liver injury. Regarding muscle damage, the BLEPS trial (BLEeding Prevention with Simvastatin) showed that there were only 2 patients who developed rhabdomyolysis after receiving SVT 40 mg/day, both were Child-Pugh (CTP) class C, compared to no cases in CTP class A or B. SVT is primarily metabolized by hepatic cytochrome P450 3A4 (CYP3A4), known as the first-pass effect. In this regard, an inverse correlation was observed between the severity of cirrhosis, as assessed by the CTP score, and CYP3A4 activity. Thus, the reduced first-pass effect of SVT in CTP class C would lead to higher SVT plasma concentrations and increased risk of adverse events. For these reasons, the authors suggest that a higher proportion of patients with severe hepatic impairment (2.8%) developed muscle damage compared with the general population (0.009-0.1%). In the LIVERHOPE-SAFETY phase II RCT, patients with decompensated cirrhosis (CTP class B or C) were randomly assigned to receive for 12 weeks either: SVT 40 mg/day plus rifaximin 1,200 mg/day (SVT40 + RFX), SVT 20 mg/day plus rifaximin 1,200 mg/day (SVT20 + RFX), or placebo of both drugs. The SVT40 + RFX group showed a significant increase in serum creatine kinase (CK) levels at the end of treatment compared with the placebo; CK levels in the SVT20 + RFX and placebo groups were similar. Three patients (19%) in the SVT40 + RFX group developed muscle toxicity compatible with rhabdomyolysis. Notably, muscle damage occurred in only 8% of CTP class B patients compared to 50% of CTP class C patients. On the other hand, our group conducted a prospective, open-label, uncontrolled phase IIa trial evaluating SVT safety in 30 patients with decompensated cirrhosis (CTP class A [n = 6], CTP class B [n = 22], and CTP class C [n = 2]) receiving 40 mg/day for one year. Muscle injury occurred in 36.7% of patients and was significantly associated with a baseline Model for End-Stage Liver Disease (MELD) score > 12 and a baseline CTP class C. Myalgia caused a transient reduction of SVT to 10 mg/day in 23.4% of patients, and myonecrosis resulted in temporary discontinuation of SVT in another 13.3% of patients. Muscle injury was the only clinically significant adverse event because it required SVT dose modification. Taken together, the results of the three trials showed that muscle injury was associated with 1) the 40 mg/day SVT dose, and 2) advanced cirrhosis severity (CTP class C and MELD score > 12). Liver injury is the other concerning side effect related to statin use. The most common pattern of liver injury is hepatocellular; rarely, a mixed pattern with prolonged symptomatic cholestasis. In the LIVERHOPE-SAFETY trial, based on data safety monitoring board recommendations, 10 patients in the SVT40 + RFX group were withdrawn early because of serious grade 3 hepatic adverse events. In addition, the SVT40 + RFX group showed significantly higher aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels compared with the SVT20 + RFX and placebo groups. At week 12, there were no significant differences in AST and ALT levels between the SVT20 + RFX and placebo groups. It is also noteworthy that the number of patients who discontinued treatment due to side effects was significantly higher in the SVT40 + RFX group (56%) compared with the other two groups (14%). In conclusion, in patients with decompensated cirrhosis, SVT40 + RFX was associated with a significant increase in liver injury, leading to treatment discontinuation compared to SVT20 + RFX or placebo groups. On the contrary, in the safety study that we conducted, no patient developed a liver injury. Unlike the LIVERHOPE-SAFETY trial, when comparing values at the end of the trial versus at baseline, serum AST slightly decreased, serum ALT markedly reduced (32 ± 16 versus 39 ± 20 IU/L, respectively), and serum alkaline phosphatase significantly reduced (119 ± 48 versus 147 ± 67 IU/L, respectively). In brief, the discrepancy in the results of both trials can be attributed to the greater cirrhosis severity in the LIVERHOPE-SAFETY trial compared to our study. However, both safety trials agreed in recommending that in patients with decompensated cirrhosis, the SVT dose should be up to 20 mg/day.
Efficacy of simvastatin in decompensated cirrhosis patients
Only two trials evaluated the survival of patients with decompensated cirrhosis following SVT treatment for one year or more. The BLEPS trial assessed the SVT effects in patients after variceal bleeding by adding it to standard prophylaxis, NSBB, and variceal ligation. Patients were stratified by (CTP class A/B or C) and then randomly assigned to receive SVT (n = 69) or placebo (n = 78) for up to 2 years. The primary endpoint was a composite of rebleeding or death. During a median follow-up of approximately one year, a similar proportion in the placebo group (39%) and the SVT group (32%) reached the primary endpoint. Nonetheless, when only death was evaluated, mortality was 22% in the placebo group compared to 9% in the SVT group (HR = 0.39; P = 0.030). In a pre-planned subgroup analysis, SVT significantly reduced mortality in CTP class A/B patients, but not in CTP class C patients (HR = 0.16; P = 0.006), resulting in an 84% reduction in mortality in the former subgroup. There were no differences in the rate of cirrhosis complications between the two groups. Overall, the BLEPS trial demonstrated that adding SVT to the standard of care in patients who recover from an acute variceal bleeding episode improves survival in CTP class A or B, without reducing cirrhosis complications. Taking into account all outcomes of this trial, the authors concluded that in CTP class C patients, SVT would not be safe nor effective, because it would cause muscle injury and not improve survival, respectively. Our group evaluated the survival of patients with decompensated cirrhosis and cardiovascular risk factors in a retrospective, matched study of cases (agreed to add SVT to standard treatment) and series (did not agree to add SVT). Nine patients were included in each group and were matched 1:1 by age, gender, etiology of cirrhosis, CTP, and MELD scores. The median survival in the case group was 107 months, whereas in the series group, it was 20 months (HR = 0.14; P < 0.001). It is also worth noting the 86% reduction in mortality. Two findings underpinned this outcome. Firstly, further complications were reduced: during the follow-up, the mean interval between cirrhosis complications in the case group was significantly longer (33.6 ± 19.9 months) than in the series group (9.4 ± 8.2 months). Secondly, in the series group, there was a significant increase in cirrhosis severity at the end of the study compared with baseline (evaluated through CTP and MELD scores); remaining unchanged in the case group. Thus, this study showed that the addition of SVT to the standard therapy in patients with decompensated cirrhosis and cardiovascular risk factors was clinically relevant compared to standard treatment, as this intervention significantly improved survival. Taken together, both the BLEPS trial and our study showed an 85% reduction in mortality. However, considering that death was a secondary endpoint in the BLEPS trial, and our study design was retrospective and non-randomized, RCTs with mortality as a primary endpoint should confirm this outcome. An editorial with a suggestive title, “Statins in Cirrhosis: The Magic Pill?” makes relevant reflections about the efficacy of SVT in patients with decompensated cirrhosis in the BLEPS trial. The authors point out that SVT increased survival when added to the standard treatment for variceal bleeding, but does not prevent further cirrhosis complications. They speculate that SVT might improve some unknown liver function or mechanism. In this sense, the safety trial led by our group also demonstrated that SVT administration reduced the severity of cirrhosis (assessed by CTP class) in patients with decompensated cirrhosis. This outcome led us to make a secondary analysis of this trial. The primary endpoint was the cirrhosis severity, and the secondary endpoints were the health-related quality of life (HRQoL) and the hospitalizations for cirrhosis complications. At baseline, 93% of patients were CTP class A or B, which means mild-to-moderate cirrhosis severity. The study demonstrated that cirrhosis severity decreased significantly when comparing baseline with the end of the safety trial (EST), only across the CTP score (7.3 ± 1.3 versus 6.7 ± 1.7) and CTP class. In this regard, the major outcome was that 12 patients improved from CTP class B to CTP class A, and only three patients worsened from CTP class A to CTP class B. This result suggests that SVT reduces cirrhosis severity in patients with relatively preserved liver function at baseline, such as CTP class A or CTP class B. On the contrary, increased cirrhosis severity would hinder the improvement of compromised liver function in CTP class C patients. On the other hand, due to changes in cirrhosis severity and differences in clinical outcomes, fifteen patients completed the trial as CTP class A, while another fifteen completed it as CTP class B/C. This result occurred in a context of chronic, low-grade inflammation at baseline, characterized by white blood cell (WBC) counts greater than 5.0×109/L. In addition, when evaluating at baseline CTP class A versus CTP class B/C patients at EST, the first group showed: 1) higher albumin (> 3.0 g/dL), and 2) higher high-density lipoprotein cholesterol (HDL-cholesterol) (> 38 mg/dL). When analyzing EST versus baseline, only in the CTP class A at EST, there was a significant reduction in WBC, neutrophil, and monocyte counts, as well as C-reactive protein levels. Likewise, these patients showed a significant increase in serum albumin and a recovery in HRQoL. Moreover, admissions for cirrhosis complications significantly decreased in CTP class A (9%) compared to CTP class B/C at EST (69%). Taken together, SVT would reduce cirrhosis severity only in CTP class A/B patients with chronic low-grade inflammation and a suitable anti-inflammatory protein and lipid milieu at baseline (possibly by reducing systemic chronic inflammation). Furthermore, through this anti-inflammatory effect, only the CTP class A at EST would improve HRQoL and lessen admissions due to cirrhosis complications. Finally, a recent trial reported that in patients with suboptimal response to NSBB, the administration of carvedilol and SVT significantly reduced the CTP score at the end of the trial compared to baseline, an improvement not seen in the group that received carvedilol plus placebo.
Futility of simvastatin in high cirrhosis severity and in acute-on-chronic liver failure patients
In a cirrhotic rat model, administering SVT before the development of acute-on-chronic liver failure (ACLF) induced by lipopolysaccharide injection, it was noted: 1) amelioration of hepatic sinusoid dysfunction, 2) conferred a hepatic and systemic anti-inflammatory effect, 3) decreased hepatic oxidative stress, 4) improved hepatic NO production, and 5) enhanced survival. These experimental SVT outcomes encouraged its investigation in ACLF –a syndrome characterized by a severe systemic inflammatory response, single or multiple organ failure, a 90-day mortality exceeding 50%, and no established treatment. Shortly thereafter, the Veterans Health Administration published two retrospective cohort trials on statins and ACLF. In the first one, of 84,963 patients with cirrhosis, 8,558 were hospitalized for ACLF, 3,290 received statins, and 5,268 did not. Statins exposure reduced the risk of developing ACLF by 38%. Higher statin doses and longer treatment duration intensified the protective effect of these drugs against new ACLF events. In the second trial, 11,731 patients were admitted for ACLF, 3,017 took statins, and 8,714 did not. Prior statin use was associated with an 18% reduction in ACLF-related mortality at 28 days, and a 24% reduction at 90 days. In brief, the results of these studies showed that statins would be effective in patients with ACLF. However, both studies claimed that there is a need for prospective studies to validate these outcomes, as well as to evaluate the safety of statins in patients with severely decompensated cirrhosis and ACLF. Recently, a parallel group, placebo-controlled trial was conducted in 14 European university hospitals in decompensated cirrhosis. Patients received either SVT 20 mg/day and RFX 1,200 mg/day or a placebo of both drugs for 12 months. The study included only CTP class B/C patients. The primary endpoint was ACLF incidence, and the secondary endpoints were transplant or death and a composite of complications of cirrhosis. Among the 237 participants randomized, 194 were CTP class B, and 43 were CTP class C. Results showed that there were no differences between the treatment vs. placebo groups in ACLF development (17.9% vs. 14.2%), transplant or death (18.8% vs. 24.2%), or further decompensations (42.7% vs. 45.8%), respectively. In addition, serious adverse events (37.6% vs. 41.7%) and fatal adverse events (14.5% vs. 14.2%) were similar between the groups. Only in the SVT + RFX group, three patients (2.6%) developed rhabdomyolysis. In summary, this RCT did not confirm that adding SVT + RFX to standard treatment for severe liver disease, such as CTP class C, would reduce the occurrence of ACLF or improve survival. In line with the outcomes of this trial, in the context of sepsis (which is also associated with acute high-grade inflammatory response and high life-threatening organ failure and mortality), a systematic review and meta-analysis of six RCTs involving 1,720 patients showed that statins did not improve survival compared to placebo.
Learning about simvastatin treatment in patients with decompensated cirrhosis
Based on the points discussed above, this article aims to summarize the lessons learned from the few published clinical trials on the treatment of decompensated cirrhosis with SVT.
- In decompensated cirrhosis patients, SVT 40 mg/day and/or CTP class C were associated with concerning side effects, especially muscle damage, and to a lesser extent, liver injury.
- Muscle damage and liver injury (the latter in one trial) were clinically significant adverse events because they led to SVT dose reduction or SVT discontinuation.
- Based on 1 and 2, in decompensated cirrhosis patients, the SVT dose would be up to 20 mg/day, and should be administered only to CTP class A/B and MELD score < 12 patients.
- SVT decreases cirrhosis severity, possibly through a reduction of the systemic chronic inflammation, only in patients who at baseline: I) are classified as CTP class A/B, and II) have a record of a suitable protein and lipid anti-inflammatory environment.
- SVT would be clinically beneficial in patients who have achieved a CTP class A, by improving HRQoL and reducing hospitalizations due to cirrhosis complications.
- The addition of SVT to standard treatment in CTP class A/B patients would increase survival.
- In CTP B (probably without a basal anti-inflammatory milieu) and C patients, the efficacy of SVT would decrease significantly.
Conclusions
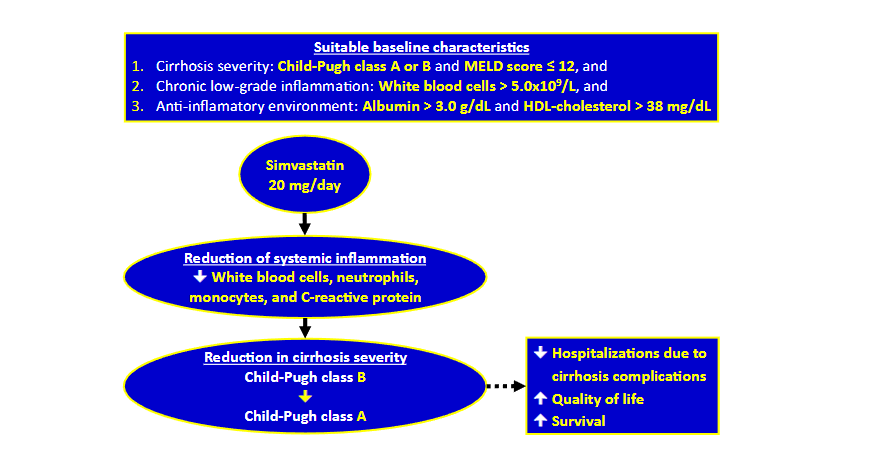
Based on lessons learned SVT 20 mg/day should only be administered to patients who at baseline showed: 1) mild to moderate cirrhosis severity (CTP class A/B and MELD score ≤ 12), 2) chronic low-grade inflammation (WBC > 5.0 x 109/L), and 3) an adequate anti-inflammatory milieu (albumin > 3.0 g/dL and HDL-cholesterol > 38 mg/dL), which would result in fewer hospitalizations for cirrhosis complications, improved quality of life, and increased survival. A summary of the conclusions is shown in Figure 1. Finally, given the limited availability of treatments targeting key pathophysiological mechanisms of decompensated cirrhosis, future RCTs should demonstrate that an inexpensive drug like SVT could become a valuable tool in its treatment.
Financial Support: No funding was received for writing this manuscript.
Conflict of Interest: The authors declare no conflicts of interest.
Dedication: In honor of Jaime Bosch, who, among his many brilliant ideas, was the first one to think that simvastatin could be useful in the treatment of patients with cirrhosis.
References
1. GBD 2017 Cirrhosis Collaborators. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(3):245-266.
2. Devarbhavi H, Asrani SK, Arab JP, et al. Global burden of liver disease: 2023 update. J Hepatol. 2023;79(2):516-537.
3. Caraceni P, Abraldes JG, Gines P, et al. The search for disease-modifying agents in decompensated cirrhosis: From drug repurposing to drug discovery. J Hepatol. 2021;75 Suppl 1:S118-S134.
4. Verna EC, Morelli G, Terrault NA, et al. DAA therapy and long-term hepatic function in advanced/decompensated cirrhosis: Real-world experience from HCV-TARGET cohort. J Hepatol. 2020;73(3):540-548.
5. Adam R, Karam V, Cailliez V, et al. 2018 Annual Report of the European Liver Transplant Registry (ELTR) – 50-year evolution of liver transplantation. Transpl Int. 2018;(12):1293-1317.
6. Jadlowiec CC, Taner T. Liver transplantation: Current status and challenges. World J Gastroenterol. 2016;22(18):4438-45.
7. Morgan S, Grootendorst P, Lexchin J, et al. The cost of drug development: a systematic review. Health Policy. 2011;100(1):4-17.
8. Lai AG, Chang WH, Parisinos CA, et al. An informatics consult approach for generating clinical evidence for treatment decisions. BMC Med Inform Decis Mak. 2021;21(1):281.
9. Bosch J. Vascular deterioration in cirrhosis: the big picture. J Clin Gastroenterol. 2007;41 Suppl 3:S247-53.
10. Bhathal PS, Grossman HJ. Reduction of the increased portal vascular resistance of the isolated perfused cirrhotic rat liver by vasodilators. J Hepatol. 1985;1(4):325-37.
11. Marteau P, Ballet F, Chazouilleres O, et al. Effect of vasodilators on hepatic microcirculation in cirrhosis: a study in the isolated perfused rat liver. Hepatology. 1989;9(6):820-3.
12. Bosch J, Abraldes JG, Groszmann R. Current management of portal hypertension. J Hepatol. 2003;38 Suppl 1:S54-68.
13. Gupta TK, Toruner M, Chung MK, et al. Endothelial dysfunction and decreased production of nitric oxide in the intrahepatic microcirculation of cirrhotic rats. Hepatology. 1998;28(4):926-31.
14. Rockey DC, Chung JJ. Reduced nitric oxide production by endothelial cells in cirrhotic rat liver: endothelial dysfunction in portal hypertension. Gastroenterology. 1998;114(2):344-51.
15. Shah V, Toruner M, Haddad F, et al. Impaired endothelial nitric oxide synthase activity associated with enhanced caveolin binding in experimental cirrhosis in the rat. Gastroenterology. 1999;117(5):1222-8.
16. Shah V, Cao S, Hendrickson H, et al. Regulation of hepatic eNOS by caveolin and calmodulin after bile duct ligation in rats. Am J Physiol Gastrointest Liver Physiol. 2001;280(6):G1209-16.
17. Bellis L, Berzigotti A, Abraldes JG, et al. Low doses of isosorbide mononitrate attenuate the postprandial increase in portal pressure in patients with cirrhosis. Hepatology. 2003;37(2):378-84.
18. Ceriello A, Taboga C, Tonutti L et al. Evidence for an independent and cumulative effect of postprandial hypertriglyceridemia and hyperglycemia on endothelial dysfunction and oxidative stress generation: effects of short- and long-term simvastatin treatment. Circulation. 2002;106(10):1211-8.
19. Van de Casteele M, Omasta A, Janssens S, et al. In vivo gene transfer of endothelial nitric oxide synthase decreases portal pressure in anaesthetised carbon tetrachloride cirrhotic rats. Gut. 2002;51(3):440-5.
20. Sarela AI, Mihaimeed FM, Batten JJ, et al. Hepatic and splanchnic nitric oxide activity in patients with cirrhosis. Gut. 1999;44(5):749-53.
21. Shah V. Cellular and molecular basis of portal hypertension. Clin Liver Dis. 2001 Aug;5(3):629-44.
22. Wiest R, Groszmann RJ. The paradox of nitric oxide in cirrhosis and portal hypertension: too much, not enough. Hepatology. 2002;35(2):478-91.
23. Liao JK. Inhibition of Gi proteins by low density lipoprotein attenuates bradykinin-stimulated release of endothelial-derived nitric oxide. J Biol Chem. 1994;269(17):12987-92.
24. Plenz GA, Hofnagel O, Robenek H. Differential modulation of caveolin-1 expression in cells of the vasculature by statins. Circulation. 2004;109(2):e7-8.
25. Dimmeler S, Fleming I, Fisslthaler B, et al. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature. 1999;399 (6736):601-5.
26. Laufs U, Liao JK. Post-transcriptional regulation of endothelial nitric oxide synthase mRNA stability by Rho GTPase. J Biol Chem. 1998;273(37):24266-71.
27. Zafra C, Abraldes JG, Turnes J, et al. Simvastatin enhances hepatic nitric oxide production and decreases the hepatic vascular tone in patients with cirrhosis. Gastroenterology. 2004;126(3):749-55.
28. Abraldes JG, Albillos A, Bañares R, et al. Simvastatin lowers portal pressure in patients with cirrhosis and portal hypertension: a randomized controlled trial. Gastroenterology. 2009;136(5):1651-8.
29. Kamal S, Khan MA, Seth A, et al. Beneficial Effects of statins on the rates of hepatic fibrosis, hepatic decompensation, and mortality in chronic liver disease: A systematic review and meta-analysis. Am J Gastroenterol. 2017;112(10):1495-1505.
30. Boccuzzi SJ, Bocanegra TS, Walker JF, et al. Long-term safety and efficacy profile of simvastatin. Am J Cardiol. 1991;68(11):1127-31.
31. Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet. 2016;388(10059):2532-2561.
32. Abraldes JG, Villanueva C, Aracil C, et al. Addition of simvastatin to standard therapy for the prevention of variceal rebleeding does not reduce rebleeding but increases survival in patients with cirrhosis. Gastroenterology. 2016;150(5):1160-1170.e3.
33. Albarmawi A, Czock D, Gauss A, et al. CYP3A activity in severe liver cirrhosis correlates with Child-Pugh and model for end-stage liver disease (MELD) scores. Br J Clin Pharmacol. 2014;77(1):160-9.
34. Thompson PD, Panza G, Zaleski A, et al. Statin-associated side effects. J Am Coll Cardiol. 2016;67(20):2395-2410.
35. Pose E, Napoleone L, Amin A, et al. Safety of two different doses of simvastatin plus rifaximin in decompensated cirrhosis (LIVERHOPE-SAFETY): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Gastroenterol Hepatol. 2020;(1):31-41.
36. Muñoz AE, Pollarsky F, Marino M, et al. Safety of chronic simvastatin treatment in patients with decompensated cirrhosis: Many adverse events but no liver injury. Dig Dis Sci. 2021;66(9):3199-3208.
37. Russo MW, Scobey M, Bonkovsky HL. Drug-induced liver injury associated with statins. Semin Liver Dis. 2009;29(4):412-22.
38. Muñoz AE, Pollarsky FD, Marino M, et al. Addition of statins to the standard treatment in patients with cirrhosis: Safety and efficacy. World J Gastroenterol. 2021;27(28):4639-4652.
39. Malizia G, D’Amico G. Statins in Cirrhosis: The magic pill? Hepatology. 2016;63(6):2047-9.
40. Muñoz AE, Pollarsky F, Marino M, et al. Baseline severity and Inflammation would influence the effect of simvastatin on clinical outcomes in cirrhosis patients. Dig Dis Sci. 2023;68(8):3442-3450.
41. Alvarado-Tapias E, Brujats A, Puente A, et al. Hemodynamic effects of carvedilol plus simvastatin in cirrhosis with severe portal hypertension and suboptimal response to β-blockers: A double-blind, placebo-controlled, randomized trial. Hepatology. 2025;82(1):140-154.
42. Tripathi DM, Vilaseca M, Lafoz E, et al. Simvastatin prevents progression of acute on chronic liver failure in rats with cirrhosis and portal hypertension. Gastroenterology. 2018;155(5):1564-1577.
43. Arroyo V, Moreau R, Jalan R. Acute-on-chronic liver failure. N Engl J Med. 2020;382(22):2137-2145.
44. Mahmud N, Chapin S, Goldberg DS, et al. Statin exposure is associated with reduced development of acute-on-chronic liver failure in a Veterans Affairs cohort. J Hepatol. 2022;76(5):1100-1108.
45. Chapin S, Kaplan DE, Taddei T, et al. Association between statin exposure and short-term mortality in patients with high-grade acute-on-chronic liver failure. JHEP Rep. 2023;5(6):100740.
46. Pose E, Jimenez C, Zaccherini G, et al. Simvastatin and rifaximin in decompensated cirrhosis: A randomized clinical trial. JAMA. 2025;333(10):864-874.
47. Deshpande A, Pasupuleti V, Rothberg MB. Statin therapy and mortality from sepsis: a meta-analysis of randomized trials. Am J Med. 2015;128(4):410-7.e1.





 http://orcid.org/0000-0002-5458-4692
http://orcid.org/0000-0002-5458-4692