Spinal Biomechanics and Atopic Dermatitis: A Study
Biomechanical alterations of the spine correlated with the severity of dermatitis and calcitonin gene-related peptide levels
Danial Khorsandi ¹, Mackarena Ccoicca ¹, Alex Ruiz ¹, Miguel Angel Tejero ², Angela Olaru ³⁻⁴, Ignacio Umbert ¹⁻⁴*
¹ Umbert Institute of Dermatology, Corachan Clinic, Plaza Manuel Corachán 4, 08017 Barcelona, Spain.
² Diagnostic Imaging Department, Corachan Clinic, Plaza Manuel Corachán 4, 08017 Barcelona, Spain.
³ Biomedical Vertix, Integrative Medicine and Chiropractic Health Center, Sant Cugat del Valles, Barcelona, Spain.
⁴ Co-senior authors
*Email: [email protected]
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION Khorsandi, D., Ccoicca, M., et al., 2025. Biomechanical alterations of the spine correlated with the severity of dermatitis and calcitonin gene-related peptide levels. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6283
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6283
ISSN 2375-1924
ABSTRACT
Background: Intrinsic atopic dermatitis is a chronic cutaneous inflammatory disease with pruritus and eczematous lesions of skin. A long-lasting cycle of itch-scratch roots results in substantial morbidities and discomfort. Treatment of patients with moderate to severe dermatitis is a challenge.
Objectives: A) To characterize the relationship between spinal biomechanical alterations, the severity of intrinsic atopic dermatitis, and the blood levels of calcitonin gene-related peptide (CGRP) and B) to determine whether chiropractic spinal manipulative therapy can be an effective complementary treatment.
Materials and Methods: In this prospective study, 33 patients with severity index (EASI) score less than 7 were compared with 40 patients with EASI score greater than 7. The severity level of spine biomechanical alterations (spinal biomechanical alterations) was quantified using full spine radiographic descriptions. The expression of CGRP was determined in blood using ELISA tests. All patients were prescribed the same anti-inflammatory topical cream. Of the 73 patients, 51 choose to be also treated by chiropractic and the 22 others were used as control. Data were analyzed before and after the treatment.
Results: A strong correlation was found between overall spinal biomechanical modifications, altered skin status and CGRP levels. The EASI scores were correlated with the different segments of the spine. Although the EASI score of the patients in the control group decreased after 2 weeks of using the anti-inflammatory cream, 3 months later the dermatitis symptoms flared up again and the EASI scores returned to baseline values. In contrast, both the EASI scores and the CGRP levels of treated patients by chiropractic remained low after 3 months.
Conclusions: This study shows that the severity of intrinsic dermatitis is related to that of spinal biomechanical alterations, and that CGRP levels may serve as a valuable pathological marker. Chiropractic proved to be a valuable complementary therapeutic tool.
Keywords: Chiropractic, CGRP, Column, Dermatitis, Dermatology, Spine.
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disease with exclusive clinical manifestations through age, race or ethnicity groups. It is an important public health problem, with a lifetime prevalence of 10-20% in children, and 1-3% in adults. Intensive pruritus and eczematous lesions of skin are the main symptoms. In most patients with AD, a long-lasting cycle of itch-scratch roots results in substantial morbidities and discomfort. Etiologically, AD is multifactorial with genetically, environmental and immunological interacting factors. Similar to many other dermatological diseases, a psychosomatic component might be involved in the development of AD. The clinical phenotype of AD is characterised as the product of several interactions including susceptible genes, environment, defective cutaneous barrier function, and the immunologic diversity of the immune responses. A simple classification divides AD into two groups: extrinsic and intrinsic. The extrinsic group is characterised by high immunoglobulin (Ig) E levels and impaired barriers. The intrinsic group, which comprises 20% of the patients, is defined by normal IgE levels and preserved barriers.
Several studies have shed light on the role of the nervous system in the pathogenesis of AD. The nervous system is critical for disease progression through an irregular expression of neuropeptides in the affected skin. Neurogenic inflammation occurs during cutaneous inflammatory reactions. The nerve endings secrete neuropeptides, such as calcitonin gene-related peptide (CGRP) and substance P, which induce the release of inflammatory mediators (cytokines and neuropeptides) by immune cells increasing inflammation.
Several studies attempted to determine the bone mineral density in AD patients. These studies showed a relationship between AD and a decrease in bone mineral density that could explain the increase in bone fractures of these patients. Based on these findings, a prospective study was designed to explore the relationship between spinal biomechanical alterations and intrinsic AD. Understanding this relationship may help optimise approaches used to control some of the pathological mechanisms in AD and thus contribute to the development of effective treatments.
Materials and Methods:
PATIENTS:
All study participants were patients Barcelona, Spain. Patients were aged between 20 and 70 years of both sexes (48 females and 25 males) with confirmed atopic dermatitis (American Academy of Dermatology Consensus Criteria) with moderate-to-severe disease activity and affected body surface area 10% or higher at both screening and baseline, with documented history (within 1 year) of inadequate response to topical medications. In any of the patients no significant high levels of IgE were observed. Patients with diagnosis of dermatitis along with other dermatological conditions or other systemic diseases were excluded from the study. The studies were reviewed on 2021/02/23 and approved by the ethics committee, Hospital group Quiron Salud-Catalunya with protocol number INCV-001. All patients gave written informed consent to participate in this study.
CLINICAL EVALUATION:
For the clinical evaluation the following areas: scalp, forehead, eyes, ears, face, chin, neck, trunk, arms, and hands, legs, genital area, and feet were photographed at each visit. The revision of the photographs was used to quantitatively assessed sing “The Eczema Area and Severity Index” (EASI) system.
FULL-SPINE X-RAY VIEW (SPINOGRAPHIC TELEMETRY):
To calculate the severity score of spinal biomechanical alterations full spine x-rays (scoliogram) were taken for each patient. X-ray analysis was performed by a team of MD radiologists and in double blind they quantified the scoliogram according to literature-based criteria in different areas of the spine: cervical, thoracic, lumbar and sacrum. In order to allow statistical analysis a numerical score was assigned to each radiological finding as described in Table S1.
| Parameters Evaluated | Score |
|---|---|
| Preserved atlanto-odontoid relationship | 0 |
| Atlanto-odontoid relationship with asymmetry (rotational subluxation) Right> Left or Left > Right | 1 |
| Preserved bilateral atlanto-axial joint | 0 |
| Mechanical changes of the articular surfaces of the bilateral atlanto-axial joint | 1 |
| Degenerative changes of the articular surfaces of the bilateral atlanto-axial joint | 2 for incipient changes, 3 for moderate changes, and 4 for severe changes. |
| Preserved uncoapophyseal and interapophyseal joints | 0 |
| Generalised mechanical changes of the uncoapophysial and interapophyseal joints of the cervical spine | 1 |
| Generalised degenerative changes of the uncoapophysial and interapophyseal joints of the cervical spine | 2 for incipient changes, 3 for moderate changes, and 4 for severe changes. |
| C7-T1 spinal transition anomaly | 0 |
| Unilateral/bilateral mega transverse processes of C7 with articulated/fused ossicles | 1 |
| Unilateral/bilateral mega transverse processes of C7 with cervical ribs | 2 |
| Preserved closure | 0 |
| Closing defect | 1 |
STUDY DESIGN:
Seventy-three patients (48 females and 25 males) suffering from AD and with no significant high IgE levels participated in this study. Due to the difficulty in recruiting patients without spinal biomechanical alterations, in the first part of the study patients were divided into two groups according to their dermatitis severity measured using the EASI score (see above): patients with EASI score equal to or lower than 7 units (n=33) and patients with EASI score higher than 7 units (n=40). The male/female and age ratios were similar in both groups. Following diagnosis, patients were treated with a topical compound anti-inflammatory cream, 3 times a day for 15 days. The composition of the cream was as follows: beeler C.S.P. 100 g, pentoxifylline 3%, gentamicin 0.1%, triancenalon acetonide 0.1%, despantenol 5%, aloe vera 15%, vitamin E 5%, nicotinamide 5%, glycerin 15%, melatonin 1%, and indomethacin 3%.
In the second part of the study, 51 patients voluntarily chose to be treated through chiropractic spinal manipulative therapy (SMT), a current treatment for biomechanical problems of the spine. The remaining 22 patients were used as control group.
The following data were analyzed in all patients: a. dermatological examination once a month with quantification of the EASI score; b. radiographic description and quantification of the spinal biomechanical alterations (SBA) severity; and c. once a month measurement of plasma CGRP levels.
DETERMINATION OF CALCITONIN GENE-RELATED PEPTIDE:
Blood calcitonin gene-related peptide (CGRP) levels were determined in duplicate using a commercial Elisa kit, EAI kit (K-015 09, Phoenix Pharmaceuticals).
CHIROPRACTIC SPINAL MANIPULATION TREATMENT:
Patients that chose to be treated by chiropractic spinal manipulation treatment (SMT) underwent 12 sessions of chiropractic treatment as follows: twice a week for 3 weeks, once a week for 4 weeks, and twice a month for one month. The duration of the chiropractic treatment was 3 months. The chiropractors aimed to detect and treat with specific manual techniques, called “adjustments”, also known as SMT, any alteration in the normal dynamic, anatomical or physiological joint relationships of adjacent structures in the spine to restore proper spinal biomechanics and physiological balance. Two types of techniques were used: high-velocity, low amplitude (HVLA)-diversified technique and activator technique.
DATA ANALYSIS:
To compare non-parametric variables U-Mann Whitney test was performed for group comparison. In addition, non-parametric linear Gaussian test was performed to find correlations between the levels of variables. To study correlations, only the most severe patients with EASI>7 group were used. All statistics analyses were conducted using GraphPad Prism version 10, GraphPad Software, San Diego, California, USA. The results are shown as the mean ± SD.
Results:
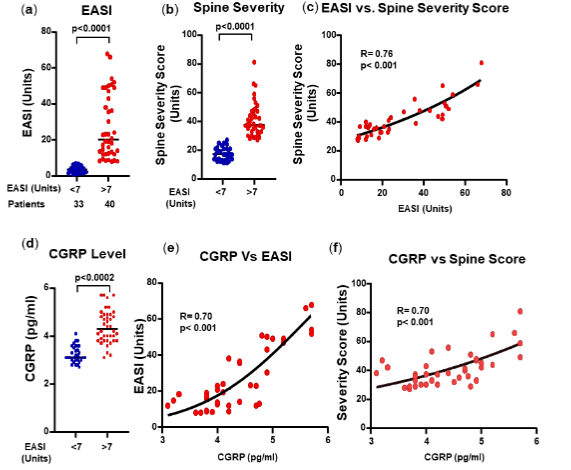
THE SEVERITY OF DEGENERATIVE CHANGES INDUCED BY SBA CORRELATED WITH THE SEVERITY OF DERMATITIS AND THE CGRP LEVELS:
The scores assigned to the different parts of the spine of a patient were then summed, thereby calculating a total spine SBA severity score for that patient. None of the 73 patients participating in this study was free of radiographically evidenced spinal biomechanical alterations and, they were divided into two groups according to the severity of the dermatitis, based on the EASI score. As expected, difference in EASI scores between the statistically significant. Difference in the total spine severity score of the two groups was also statistically significant. A good correlation (R= 0.76) between the severity of AD and the total spine severity score was observed. Interestingly, CGRP levels were also higher in the EASI>7 group and they correlated with EASI scores (R= 0.70) and the spine severity scores (R= 0.70).
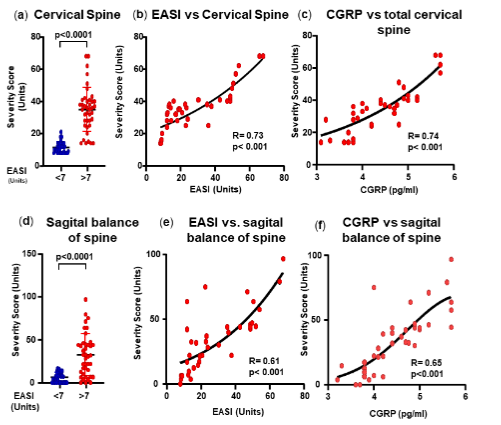
DEGENERATIVE CHANGES IN THE CERVICAL SPINE:
The study then determined which areas of the spine were responsible for the correlation between SBA severity and the dermatological anomalies. For this purpose, the spinal severity score was calculated starting from the upper (C1-Atlas) to the lower spinal section (Sacral section) separately. This calculation aimed at establishing which specific area of the spine (i.e., cervical, thoracic, lumbar, and sacro-coccygeal, including sagittal balance) could be associated with dermatitis. When comparing the severity score of the cervical spine (C1 to C7), a significant difference was observed between group. A good correlation was observed between the spine severity and the EASI (R= 0.73) or the CGRP levels (R= 0.74). The different segments of the cervical spine were analysed in more detail in Supplementary Figures.
THORACIC SPINE SEVERITY SCORE:
These scores were calculated by reviewing the lateral X-ray images was a statistically significant difference in thoracic spine total severity score between patients with and those with EASI>7. While there was a reduced correlation between the severity of spinal degeneration and that of dermatitis, there was a good correlation with the CGRP levels.
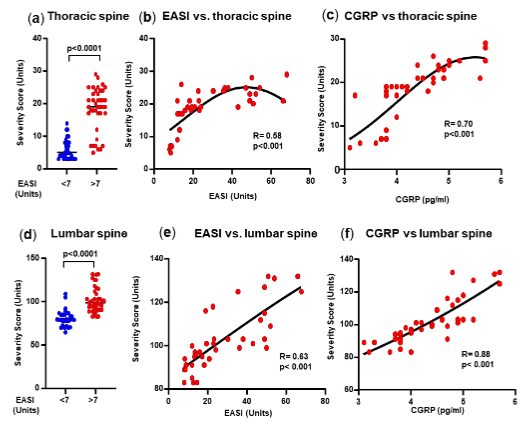
LUMBAR SPINE SEGMENTS SEVERITY SCORE:
The lumbar spine segments are located at the bottom of the spine, between the thoracic and the sacral segments. It consists of five separate vertebrae that are the largest vertebrae in the human spine. The lumbar segments help the spine to support its structure. Concerning the lumbar spine, there was a statistically significant difference between the group and the EASI>7 group. Moreover, the correlation between the EASI score and the lumbar spine severity score was R=0.63, which was close to significance. However, there was an excellent correlation (R= 0.88) between the spine severity scores and the CGRP levels.
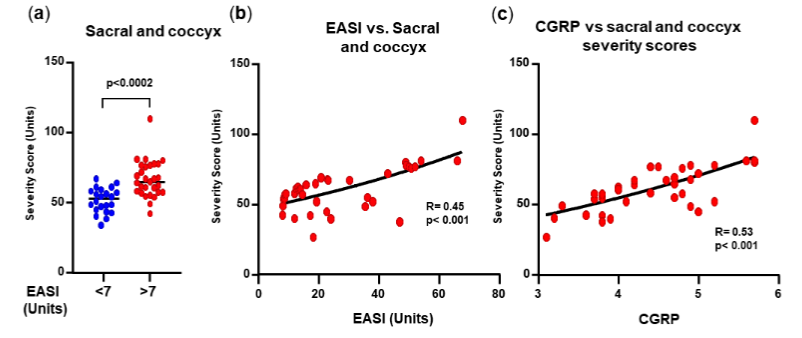
SACRAL AND COCCYX SCORE AREA:
The sacrum is a large and flat bone located below the last lumbar vertebra (L5) and the coccyx is located under the sacrum. The sacrum consists of 5 vertebrae (S1-S5) while the coccyx is made up by 3 to 5 small bones. Both help in supporting the human weight and are essential for walking, standing, and sitting. There was a difference between the two EASI groups. The correlation between the severity score and EASI level was significant, as well with the CGRP values.
THE AGE OF THE PATIENTS CORRELATED WITH THE SEVERITY OF DERMATITIS:
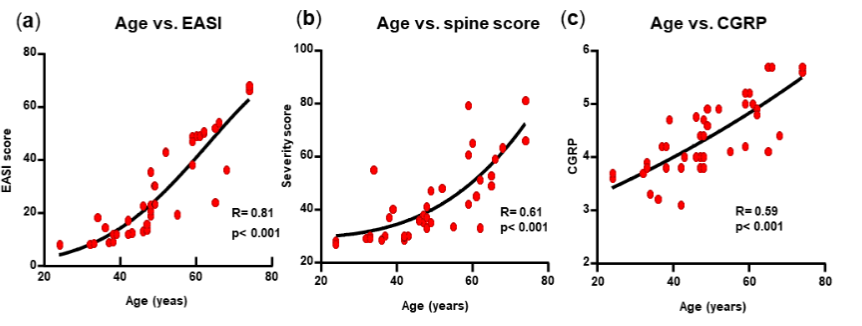
Aging causes important cutaneous modifications. With age the stratum corneum and epidermis becomes thicker, the papillary dermis contains less collagen than the reticular dermis and is more fragmented and clustered, epidermal rete edges and the dermal/epidermal junction dermal papillae become flat due to the retraction of villi. In addition, in the stratum basale, keratinocytes proliferate less and fibroblasts have a lower ability to migrate, due to the fragmented extracellular matrix. Aging in patients with AD has been reported to exhibit unique clinical phenotypes and immunologic endotypes. Hence, the effect of age in relation to the importance of dermatological lesions was evaluated in all patients. An excellent correlation was found between the age of the patients and the EASI score (R= 0.81), thus suggesting that age is associated with more severe clinical presentation of AD. Age was also associated with spinal damage severity score, but with a limited value of R= 0.61, and with the GCRP levels (R= 0.59).
CHIROPRACTIC SPINAL MANIPULATIVE THERAPY AS COMPLEMENTARY THERAPEUTIC TOOL:
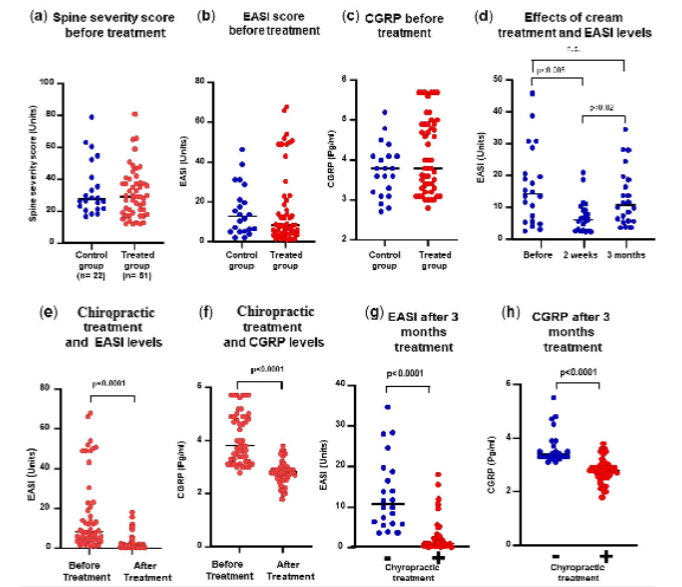
All 73 patients were treated with a topical anti-inflammatory cream 3 times a day for 15 days as soon as dermatitis was diagnosed. Patients chose to be treated by the chiropractic spinal manipulative therapy (SMT). The 22 patients who chose not to undergo chiropractic treatment were used as control group. The chiropractic SMT consisted of 12 sessions during 3 months distributed as follows: twice a week for 3 weeks, once a week for 4 weeks, and twice a month for one month. Before chiropractic SMT, the spine severity scores, the EASI scores and the CGRP levels of the treatment and control groups were similar. In the control group treated only with the cream, the EASI score decreased significantly after 2 weeks. However, after 3 months, the EASI score increased again. No significant differences were found between the EASI score before cream treatment and after 3 months. After 3 months of chiropractic SMT, the EASI scores and the CGRP levels decreased significantly. When we compared the effect of chiropractic treatment in relation to the group untreated, both the levels of EASI and CGRP levels were significantly reduced.
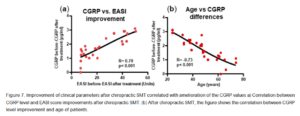
Moreover, the EASI scores before and after treatment correlated with the CGRP levels before and after treatment, suggesting a close relationship between both parameters. Finally, the decrease of CGRP levels before and after treatment correlated negatively with the age of patients. This may be related to the different spinal biomechanical alterations associated with aging.
Discussion:
The results obtained in this study showed a strong correlation between the altered biomechanics of spine and the extent of dermatological lesions of intrinsic AD. Regarding the different spinal segments, the EASI scores of patients suffering from AD were well correlated, particularly with SBAs in the cervical spine as well as in the sagittal balance of the spine. In the upper cervical spine (C1-C2 and C3-C7 segments), a good correlation was found between SBAs and EASI scores. Remarkably, in this study patients with manifestation of dermatitis on the arms and hands showed more severe SBAs in the C3-C7 segment where the nervous fibres directly affect the epithelial cell in the chest, shoulders, arms, and hands. Of note, both measurements are related areas of high spinal mobility and are probably caused by modern habits, such as the use of smartphones.
Regarding the thoracic column, no correlations were found between the EASI scores and the radiographic alterations in the T1 to T9 column area. However, alterations in the T10 vertebrae correlated with the EASI scores, as well as with the CGRP levels. This may be related to the different biomechanical behaviour with greater mobility of this area in relation to the rest of the thoracic spine.
The lumbar spine is one of the most injured areas because it bears the most pressure when holding and pushing, hence resulting in more damage and injury. In this study, a correlation was found between the lumbar severity scores and the EASI scores and the CGRP levels. The spinal nerves of the sacrum and coccyx can directly affect epithelial cells in the sacral area, legs, feet, anus, and genital parts, resulting in dermatitis of the vagina (vaginitis), penis and scrotum. Neurogenic inflammation caused by altered biomechanics of the sacrum has also been reported to correlate with inflammatory diseases in the genital area.
However, concerning the sacral and coccyx area, the results of this study did not indicate a good correlation between the EASI scores and the severity of spinal biomechanical alterations. Interestingly, this study found a strong correlation between the cutaneous disease involvement, the SBA and the CGRP levels. Peptide CGRP is released by sensory neurons and is one of the main components involved in neurogenic inflammation regulating pruritus in AD. By using degenerated intervertebral discs from patients with low back pain and healthy, painless ones from human organ donors, cultured ex vivo, researchers observed a greater release of tumor necrosis factor (TNF)-α, interleukin-1β, nerve growth factor (NGF) and brain derived neurotrophic factor (BDNF), both associated with inflammation and chronic low back pain. Furthermore, factors released by degenerating intervertebral discs such as NGF increased neurite growth and CGRP expression.
These interactions may be triggered by the activation of Toll-like receptors by endogenous alarmins such as fragmented extracellular matrix proteins found in degenerating discs or cartilage that produce NGF. The role of CGRP depends on its localisation. In the dorsal horn, skin-mediated central pruritus causes neuroinflammation, neurogenic vasodilation, and modulation of cutaneous and immune cell function. To amplify neuroinflammation, both immune cells, and keratinocytes release CGRP.
Of note, the CGRP levels before and after chiropractic treatment decreased with the age of the patients. This may be explained because despite its recognised benefit in elderly people, physical activity is much reduced. This reduced mobility may also border on the correlation between chronic dermatitis and low bone mineral density associated with a high prevalence of osteopenia of the spine and hip.
The treatment of AD is based on reducing itch symptoms. As a primary anti-inflammatory drug, topical corticosteroids are the first-line therapy for acute flares of eczema. In this study, treatment with topical anti-inflammatory compounds reduced to normal values the EASI scores and the CGRP levels of all patients. However, evidence suggests a limited effect for topical and systemic targeted neural therapies. Given our clinical observation of the presence of spinal biomechanical alterations in all patients with AD, we decided to treat the patients with chiropractic SMT for 3 months. In accord with a review study about the usefulness of CAM (Complementary Alternative Medicine) in treating AD, our results showed that chiropractic can be valid a supportive treatment.
Conclusions:
This study was undertaken because there are emerging evidences suggesting a role for neuro-immune interactions in dermatological diseases. However, at the present, no guideline-based method is available for treating dermatitis and the pathogenesis of intrinsic atopic dermatitis remains unknown. This comprehensive study of the spine reveals for the first time several items: a) There is a strong correlation between the severity of spine biomechanical alterations and the intensity of atopic dermatitis (EASI score). b) The levels of calcitonin gene-related peptide (CGRP) correlated with both the severity of spinal biomechanical alterations and with the EASI score. c) The treatment with anti-inflammatory drugs reduced the EASI score and the CGRP levels, but both increased again after 3 months. However, these parameters remained low after chiropractic spinal manipulation therapy. The implication of this study is that spinal biomechanical alterations (SBA) are one of the main causal factors of the pathophysiology of atopic dermatitis and neurogenic inflammation may be the bridge linking spine biomechanical alterations and the intensity of atopic dermatitis since both are correlated with CGRP levels. Finally, complementary alternative therapies such as chiropractic may be beneficial for patients with atopic dermatitis.
Conflict of Interest:
Authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding Statement:
None.
Acknowledgements:
We would like to thank the patients for their time and support in this study.
Data availability statement:
The data and the code that support the findings of this study are available on reasonable request from the corresponding author.
Disclosure Summary:
The authors have nothing to disclose.
Ethical Approval:
The study was reviewed on 2021/02/23 and approved by the ethics committee research with medications (CEIm), Hospital group Quiron Salud-Catalunya with protocol number INCV-001. All patients gave written informed consent to participate in this study.
Author contribution:
Danial Khorsandi: Data curation, Formal analysis, Methodology, Investigation, Resources, Validation, Visualization, Writing Original Draft. Mackarena Ccoicca: Data curation, Investigation, Resources. Alex Ruiz: Formal analysis. Miguel Angel Tejero: Data curation, Investigation, Resources. Angela Olaru: Conceptualization, Project administration, Supervision, Validation, Writing, Review & Editing. Ignacio Umbert: Conceptualization, Investigation, Data curation, Methodology, Project administration, Supervision, Validation, Visualization, Writing, Review & Editing.
Consent for publication:
All the co-authors have consented to the publication of the study results.
References:
1. Leung DY, Bieber T. Atopic dermatitis. Lancet 2003; 361: 151-60.
2. Langan SM, Irvine AD, Weidinger S. Atopic dermatitis. Lancet 2020; 396: 345-60.
3. Kader HA, Azeem M, Jwayed SA et al. Current Insights into Immunology and Novel Therapeutics of Atopic Dermatitis. Cells 2021; 10.
4. Roguedas AM, Machet L, Fontes V et al. [Atopic dermatitis: which are the diagnostic criteria used in medical literature?]. Ann. Dermatol. Venereol. 2004; 131: 161-4.
5. Tokura Y, Hayano S. Subtypes of atopic dermatitis: From phenotype to endotype. Allergol Int 2022; 71: 14-24.
6. Misery L. Atopic dermatitis and the nervous system. Clin Rev Allergy Immunol 2011; 41: 259-66.
7. Fan J, Mishra SK. The emerging role of neuroimmune interactions in atopic dermatitis and itch. FEBS J 2022; 289: 2723-35.
8. Zhang Y, Zhang H, Jiang B et al. Current views on neuropeptides in atopic dermatitis. Exp. Dermatol. 2021; 30: 1588-97.
9. Marek-Jozefowicz L, Nedoszytko B, Grochocka M et al. Molecular Mechanisms of Neurogenic Inflammation of the Skin. Int. J. Mol. Sci. 2023; 24.
10. Garg NK, Silverberg JI. Eczema is associated with osteoporosis and fractures in adults: a US population-based study. JACI 2015; 135: 1085-7 e2.
11. Lowe KE, Mansfield KE, Delmestri A et al. Atopic eczema and fracture risk in adults: A population-based cohort study. JACI 2020; 145: 563-71 e8.
12. Bieber T. Atopic dermatitis. NEJM 2008; 358: 1483-94.
13. Leshem YA, Hajar T, Hanifin JM et al. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. BJD 2015; 172: 1353-7.
14. Hanifin JM, Thurston M, Omoto M et al. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp. Dermatol. 2001; 10: 11-8.
15. Silverberg JI, Lei D, Yousaf M et al. What are the best endpoints for Eczema Area and Severity Index and Scoring Atopic Dermatitis in clinical practice? A prospective observational study. BJD 2021; 184: 888-95.
16. Harrop JS, Vaccaro AR, Hurlbert RJ et al. Intrarater and interrater reliability and validity in the assessment of the mechanism of injury and integrity of the posterior ligamentous complex: a novel injury severity scoring system for thoracolumbar injuries. J. Neurosurg. Spine 2006; 4: 118-22.
17. Lee JY, Vaccaro AR, Lim MR et al. Thoracolumbar injury classification and severity score: a new paradigm for the treatment of thoracolumbar spine trauma. J. Orthop. Sci. 2005; 10: 671-5.
18. Kotil K, Alan MS, Bilge T. Medical management of Pott disease in the thoracic and lumbar spine: a prospective clinical study. J. Neurosurg. Spine 2007; 6: 222-8.
19. Kaale BR, Krakenes J, Albrektsen G et al. Whiplash-associated disorders impairment rating: neck disability index score according to severity of MRI findings of ligaments and membranes in the upper cervical spine. J. Neurotrauma 2005; 22: 466-75.
20. Hasegawa K, Kitahara K, Shimoda H et al. Facet joint opening in lumbar degenerative diseases indicating segmental instability. J. Neurosurg. Spine 2010; 12: 687-93.
21. Lotz JC, Haughton V, Boden SD et al. New treatments and imaging strategies in degenerative disease of the intervertebral disks. Radiology 2012; 264: 6-19.
22. Garg A, Kapellusch JM. Applications of biomechanics for prevention of work-related musculoskeletal disorders. Ergonomics 2009; 52: 36-59.
23. Anderson PA, Moore TA, Davis KW et al. Cervical spine injury severity score. Assessment of reliability. J. Bone Joint Surg. Am. 2007; 89: 1057-65.
24. Umbert I, Sans G, Valls J et al. EP2311454 treatment for atopic dermatitis: therapeutic efficiency in patients and anti-inflammatory in vitro. Clin. Dermatol. 2017; 5: 5-11.
25. Trager RJ, Bejarano G, Perfecto RT et al. Chiropractic and Spinal Manipulation: A Review of Research Trends, Evidence Gaps, and Guideline Recommendations. J. Clin. Med. 2024; 13.
26. Young KJ, Leboeuf-Yde C, Gorrell L et al. Mechanisms of manipulation: a systematic review of the literature on immediate anatomical structural or positional changes in response to manually delivered high-velocity, low-amplitude spinal manipulation. Chiropr. Man. Ther. 2024; 32: 28.
27. Rubinstein SM, de Zoete A, van Middelkoop M et al. Benefits and harms of spinal manipulative therapy for the treatment of chronic low back pain: systematic review and meta-analysis of randomised controlled trials. BMJ 2019; 364: l689.
28. Qaseem A, Wilt TJ, McLean RM et al. Noninvasive Treatments for Acute, Subacute, and Chronic Low Back Pain: A Clinical Practice Guideline From the American College of Physicians. Ann. Intern. Med. 2017; 166: 514-30.
29. Haavik H, Kumari N, Holt K et al. The contemporary model of vertebral column joint dysfunction and impact of high-velocity, low-amplitude controlled vertebral thrusts on neuromuscular function. Eur. J. Appl. Physiol. 2021; 121: 2675-720.
30. Bussieres AE, Stewart G, Al-Zoubi F et al. Spinal Manipulative Therapy and Other Conservative Treatments for Low Back Pain: A Guideline From the Canadian Chiropractic Guideline Initiative. J Manipulative Physiol Ther 2018; 41: 265-93.
31. Le Huec JC, Thompson W, Mohsinaly Y et al. Sagittal balance of the spine. Eur. Spine J. 2019; 28: 1889-905.
32. Ling FP, Chevillotte T, Leglise A et al. Which parameters are relevant in sagittal balance analysis of the cervical spine? A literature review. Eur. Spine J. 2018; 27: 8-15.
33. Blair MJ, Jones JD, Woessner AE et al. Skin Structure-Function Relationships and the Wound Healing Response to Intrinsic Aging. Adv. Wound Care 2020; 9: 127-43.
34. Williamson S, Merritt J, De Benedetto A. Atopic dermatitis in the elderly: a review of clinical and pathophysiological hallmarks. Br. J. Dermatol. 2020; 182: 47-54.
35. Tanei R. Atopic Dermatitis in Older Adults: A Review of Treatment Options. Drugs Aging 2020; 37: 149-60.
36. Wang S, Zhu R, Gu C et al. Distinct clinical features and serum cytokine pattern of elderly atopic dermatitis in China. JEADV 2020; 34: 2346-52.
37. Coskun Benlidayi I, Basaran S. Comparative study of lumbosacral alignment in elderly versus young adults: data on patients with low back pain. ACER 2015; 27: 297-302.
38. Jinkins JR. Acquired degenerative changes of the intervertebral segments at and suprajacent to the lumbosacral junction. A radioanatomic analysis of the nondiskal structures of the spinal column and perispinal soft tissues. Radiol Clin North Am 2001; 39: 73-99.
39. Galbusera F, van Rijsbergen M, Ito K et al. Ageing and degenerative changes of the intervertebral disc and their impact on spinal flexibility. Eur. Spine J. 2014; 23 Suppl 3: S324-32.
40. Stecco C, Pirri C, Fede C et al. Dermatome and fasciatome. Clin. Anat. 2019; 32: 896-902.
41. Park JHM, Kang SYP, Lee SGP et al. The effects of smart phone gaming duration on muscle activation and spinal posture: Pilot study. Physiother. Theory Pract. 2017; 33: 661-9.
42. Rodriguez-Sanz J, Carrasco-Uribarren A, Cabanillas-Barea S et al. Validity and reliability of two Smartphone applications to measure the lower and upper cervical spine range of motion in subjects with chronic cervical pain. J Back Musculoskelet Rehabil 2019; 32: 619-27.
43. Burgos J, Barrios C, Mariscal G et al. Non-uniform Segmental Range of Motion of the Thoracic Spine During Maximal Inspiration and Exhalation in Healthy Subjects. Front. Med. 2021; 8: 699357.
44. Morita D, Yukawa Y, Nakashima H et al. Range of motion of thoracic spine in sagittal plane. Eur. Spine J. 2014; 23: 673-8.
45. Liyew WA. Clinical Presentations of Lumbar Disc Degeneration and Lumbosacral Nerve Lesions. Int. J. Rheumatol. 2020; 2020: 2919625.
46. Brooks BK, Southam SL, Mlady GW et al. Lumbar spine spondylolysis in the adult population: using computed tomography to evaluate the possibility of adult onset lumbar spondylosis as a cause of back pain. Skelet. Radiol. 2010; 39: 669-73.
47. Wesselmann U. Neurogenic inflammation and chronic pelvic pain. World J. Urol. 2001; 19: 180-5.
48. Steinhoff M, Ahmad F, Pandey A et al. Neuroimmune communication regulating pruritus in atopic dermatitis. JACI 2022; 149: 1875-98.
49. Krock E, Rosenzweig DH, Chabot-Dore AJ et al. Painful, degenerating intervertebral discs up-regulate neurite sprouting and CGRP through nociceptive factors. J Cell Mol Med 2014; 18: 1213-25.
50. Krock E, Currie JB, Weber MH et al. Nerve Growth Factor Is Regulated by Toll-Like Receptor 2 in Human Intervertebral Discs. J. Biol. Chem. 2016; 291: 3541-51.
51. Pahor M, Guralnik JM, Ambrosius WT et al. Effect of structured physical activity on prevention of major mobility disability in older adults: the LIFE study randomized clinical trial. JAMA 2014; 311: 2387-96.
52. Keadle SK, McKinnon R, Graubard BI et al. Prevalence and trends in physical activity among older adults in the United States: A comparison across three national surveys. Prev. Med. 2016; 89: 37-43.
53. Haeck IM, Hamdy NA, Timmer-de Mik L et al. Low bone mineral density in adult patients with moderate to severe atopic dermatitis. BJD 2009; 161: 1248-54.
54. Tadicherla S, Ross K, Shenefelt PD et al. Topical corticosteroids in dermatology. JDD 2009; 8: 1093-105.
55. Elmariah SB. Adjunctive Management of Itch in Atopic Dermatitis. Dermatol. Clin. 2017; 35: 373-94.
56. Vieira BL, Lim NR, Lohman ME et al. Complementary and Alternative Medicine for Atopic Dermatitis: An Evidence-Based Review. Am. J. Clin. Dermatol. 2016; 17: 557-81.




