Subtenon WJ-MSC Therapy for Retinitis Pigmentosa Insights
Subtenon Implantation of Wharton’s Jelly Derived Mesenchymal Stromal Cell for Retinitis Pigmentosa: A 1-2 Year Follow-Up Report
Nor Shahidah Khairullah, FRCPath1, Nor Fariza Ngah, MSOph2, Roslin Azni Abdul Aziz, MSOph2, Daphne Teh, MSOph2, Angelina Tiah, MedSci1
- 123 Century International Life Science Centre, Seri Kembangan, Selangor, Malaysia
- Department of Ophthalmology Hospital Shah Alam, Shah Alam, Selangor, Malaysia
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Khairullah, N.S., et al., 2026. Subtenon Implantation of Wharton’s Jelly Derived Mesenchymal Stromal Cell for Retinitis Pigmentosa: A 2-Year Follow-Up Report. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7196
ISSN 2375-1924
Abstract
Background: Retinitis Pigmentosa (RP) is a group of genetically inherited retinal dystrophies characterized by the progressive degeneration of photoreceptors, leading to irreversible vision loss. With limited therapeutic options currently available, stem cell therapy has emerged as a promising regenerative strategy for retinal repair and vision restoration.
Objective: The study aimed to evaluate the safety, feasibility, and potential therapeutic effects of subtenon implantation of human Wharton’s Jelly-derived Mesenchymal Stem Cells (WJ-MSCs) by inhibiting proinflammatory cytokine expression, thereby suppressing chronic retinal inflammation and preventing apoptosis of photoreceptor cells.
Materials and Methods: We present four case illustrations of patients diagnosed with RP who were treated with WJ-MSCs injected in the deep subtenon space. Each patient received a total of four injection sessions between November 2020 and January 2023. The procedure was well tolerated in all sessions, and patients were monitored closely for clinical outcomes and potential adverse effects.
Result: Patients 1 and 2 underwent a more intensive treatment regimen with 7 subtenon WJ-MSC injection over a 24-month period. This approach aimed to provide sustained therapeutic effects, with both patients reporting stable visual improvements and no adverse events during the follow-up period. Moreover, both patients 3 and 4 received a shorter treatment course with 3 subtenon WJ-MSC injection over 12 months. Despite the reduced number of injection, both patients experienced intermittent episodes of brighter vision, suggesting potential therapeutic benefits. The study found significant improvements in best corrected visual acuity, outer retinal thickness, and full-field electroretinography parameters, supporting the potential benefits of this treatment modality. Our study involving 4 patients with RP demonstrated that subtenon administration with hWJ-MSC treatment was found to be effective and safe, with no serious adverse events or ophthalmic/systemic side effects reported during the follow-up period.
Conclusion: These findings align with previous study, supporting the safety and efficacy of subtenon hWJ-MSC administration in RP patients and suggesting it as a promising therapeutic approach.
Keywords: Retinitis Pigmentosa, Wharton’s Jelly-derived Mesenchymal Stem Cells, Subtenon implantation, Visual impairment
Introduction
Retinitis pigmentosa (RP) is a significant cause of visual impairment in Malaysia, particularly among younger individuals. This hereditary retinal disorder leads to progressive vision loss due to the degeneration of photoreceptor cells in the retina. A study at the University of Malaya Medical Centre found that RP accounted for 10.04% of cases of low vision and blindness, ranking it among the top four causes in their urban patient population. Similarly, research from Miri Hospital in Sarawak reported RP as the third leading cause of irreversible visual impairment, affecting 9.7% of patients. Nationally, RP is a major contributor to childhood blindness. A study of blind school students indicated that 37.7% of inherited childhood blindness cases were due to RP, often linked to consanguinity marriages.
The inheritance patterns of RP include autosomal dominant, autosomal recessive, and X-linked forms. Malaysia’s diverse ethnic composition, including Malay, Chinese, Indian, and indigenous groups, contributes to a rich genetic landscape. This diversity may influence the prevalence and types of genetic mutations associated with RP in different populations. For instance, certain mutations may be more common in specific ethnic groups, affecting the clinical presentation and progression of the disease. Understanding the specific genetic mutations prevalent in Malaysian populations is crucial for accurate diagnosis, genetic counselling, and the development of targeted therapies.
In response to the challenges posed by RP, Malaysia has seen advancements in medical research and treatment options. Stem cell therapy has emerged as a potential treatment for RP, offering hope for patients with limited options. We, 23 Century International Life Science Centre are exploring the use of stem cells to repair damaged retinal tissue, aiming to halt or even reverse the progression of the disease.
This paper presents a continuation of the initial clinical experience jointly reported by 23 Century Medical Group and Hospital Shah Alam, which demonstrated early functional improvements and safety following subtenon administration of UC-MSCs in RP patients. Building upon these preliminary outcomes, the current study aims to further evaluate the long-term efficacy, safety, and vision-related quality of life improvements in a larger patient cohort. By expanding the clinical dataset and refining the treatment protocol, this study seeks to establish subtenon UC-MSC therapy as a viable adjunct in the management of RP, offering new hope for a condition long considered untreatable. The trial, conducted at Hospital Shah Alam between November 2020 and January 2023.
The Ministry of Health (MOH) Malaysia has recognized the potential of mesenchymal stem cells in treating RP and other degenerative retinal diseases. In 2022, the ministry issued guidelines to ensure the safe and ethical use of stem cell therapies in clinical settings. While stem cell therapy for RP is still in the research and clinical trial stages, the progress made in Malaysia offers hope for patients with limited treatment options. Continued research and adherence to regulatory standards will be crucial in determining the long-term efficacy and safety of these therapies.
In summary, Malaysia is at the forefront of exploring stem cell therapy as a treatment for RP. With ongoing clinical trials and regulatory support, there is optimism that this approach may provide new avenues for managing and potentially reversing the effects of this debilitating condition.
Materials & Methods
This clinical study involved eight eyes from four patients who attended the Ophthalmology Clinic at Shah Alam Hospital between November 2020 and January 2023. The study was conducted in accordance with the tenets of the Declaration of Helsinki. All patients were fully informed regarding the study’s purpose, procedures, and course. Written informed consent was obtained from each subject prior to the initiation of any study related assessments or procedures.
The diagnosis of retinitis pigmentosa (RP) was established based on clinical history, ophthalmological examination findings, visual field (VF) testing, and optical coherence tomography (OCT) results. The inclusion criteria were as follows: (1) a clinical diagnosis of retinitis pigmentosa (RP) confirmed by clinical history, fundus appearance, visual field (VF) testing, optical coherence tomography (OCT), and electroretinography (ERG); (2) age greater than 18 years; (3) ability to perform a reliable VF evaluation; and (4) availability of at least one year of follow-up data.
Patients were excluded if they had a history of ocular surgery other than cataract extraction; ocular media opacities that compromised image quality or interfered with test results; coexisting ocular conditions such as retinal pathology unrelated to RP, glaucoma, uveitis, strabismus, or nystagmus; or systemic diseases, including diabetes, neurological disorders, or hypertension, that could affect the study outcomes.
All surgical procedures and ophthalmic assessments were performed by a single experienced vitreoretinal surgeon (AO). Baseline ophthalmic evaluations included best-corrected visual acuity (BCVA), applanation tonometry, slit-lamp biomicroscopy, color fundus photography, OCT, and VF assessment. OCT was conducted using the Optovue system (Optovue Inc., USA) with a standardized scanning protocol. Visual field testing was performed using the Humphrey Field Analyzer (Threshold 30-2, HFA II 750; Carl Zeiss Meditec AG, Germany). Electroretinography (ERG) recordings were obtained using the ERG-Vision monitor (Monpack 3, Metrovision, France), following the standards set by the International Society for Clinical Electrophysiology of Vision (ISCEV). All tests were conducted with the same instruments and by the same trained technician to ensure consistency.
Preparation of Umbilical Cord-Derived Mesenchymal Cell (MSC)
The WJ-MSCs used in this study were isolated from Wharton’s Jelly of the umbilical cord, collected from a single donor with informed maternal consent. The umbilical cord, initially immersed in a cord preservative solution, was washed with 0.9% sodium chloride injection and soaked in 70% alcohol for disinfection. After a final rinse with 0.9% sodium chloride, the cord was measured by clamping and stretching both ends with hemostats, then cut into 2 to 3 cm segments.
The tissue was subsequently weighed, cut, washed, and centrifuged prior to the culturing process in complete culture medium. The cultured tissues were incubated in a CO2 incubator maintained at 37°C, 5.0% CO2, and 95% relative humidity. All cell preparation and cultivation procedures were conducted by Beike 23 Century International Stem Cell Laboratory (Beike 23C), a facility accredited by the Ministry of Health (MOH) for cGMP and cGTP compliance.
Culture-expanded cells were cryopreserved at passage 4 (P4) using standard cryopreservation protocols until use. The cryopreserved cells were characterized via flow cytometry in accordance with the International Society for Cell & Gene Therapy (ISCT) criteria for mesenchymal stem cells. Positive expression of surface markers CD90, CD73, and CD105 was required to be 95%. CD29 was included as an additional marker to verify the identity of the MSC culture, as part of Beike 23C’s characterization protocol. Concurrently, negative expression (<2%) of CD45, CD34, CD79a, CD14, and HLA-DR was confirmed.
Quality control analyses including sterility testing, mycoplasma detection, and bacterial endotoxin testing were conducted prior to product release. The average cell viability exceeded 90%, and each patient received 10 to 11 million viable cells suspended in 1.5 mL of electrolyte solution per eye.
Injection of WJ-MSC
A total of 1.5mL of Wharton’s Jelly-derived mesenchymal stromal cell (WJ-MSC) suspension was administered into the subtenon space of each eye. The procedure was performed under local anaesthesia using proparacaine hydrochloride eye drops (Alcaine®, Alcon, USA) under sterile conditions. A 25G curved subtenon cannula (BD Visitec, UK) was used to deliver the suspension into either the superotemporal or inferonasal region to ensure effective distribution.
Post-operatively, patients were prescribed Guttae Maxitrol® (neomycin and dexamethasone) eye drops to be applied four times daily for one week. Oral Ibuprofen 200 mg was administered three times daily for one week, and amoxicillin-clavulanate 500 mg was given four times daily for three days as prophylactic therapy.
Results
Clinical Outcomes of Subtenon MSC Administration in Four Patients
Patients 1 and 2
Both patients received a total of seven mesenchymal stem cell (MSC) transplantations, administered every 4-6 weeks, with follow-up from November 2020 to November 2022. Both patients reported no deterioration in vision since the first subtenon implantation in November 2020. Optical coherence tomography (OCT) images consistently demonstrated the maintenance of a hyperreflective region, with no notable increase in intensity. The previously reported improvements in visual function remained stable throughout the two-year follow-up, with no evidence of regression.
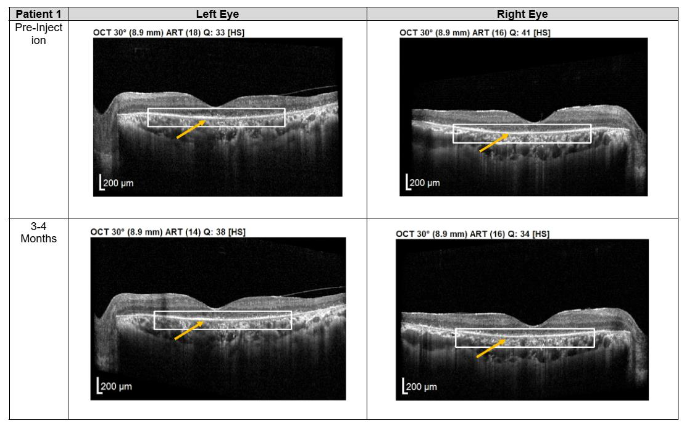
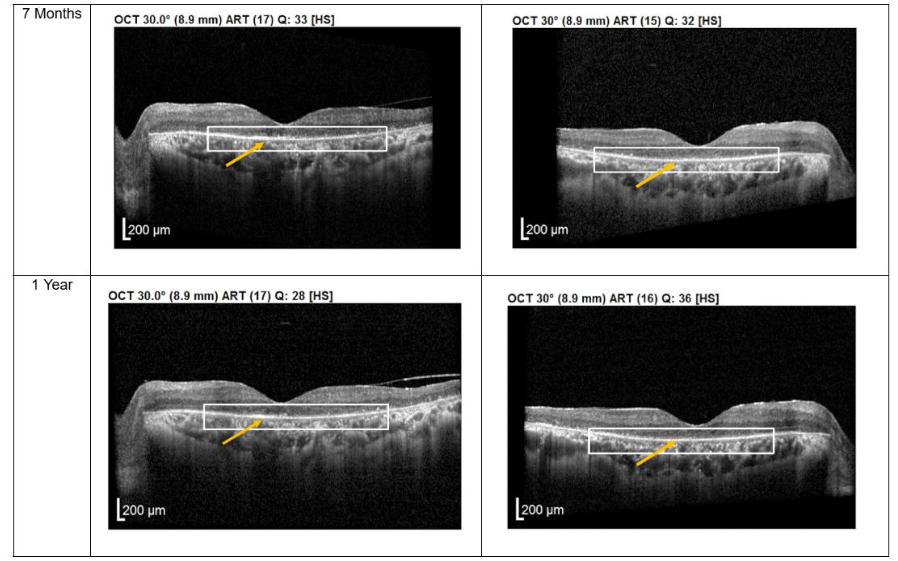
Figure 1. Optical coherence tomography (OCT) images for both eyes of Patient 1 from pre-injection up to 1 year follow-up. Orange arrow demonstrates the presence of hyperreflective material at the interdigitation area of the photoreceptors at the macula and extramacular region. These were observed and maintained until the last follow-up examination.
Importantly, no severe ophthalmic or systemic complications were observed during this period, supporting the safety of subtenon MSC therapy over a prolonged follow-up. No serious adverse effects were reported by either patient.
Patients 3 and 4
Patients 3 and 4 each received three subtenon MSC administrations at 4-6 weeks intervals, followed by follow-up visits every 3-4 months from December 2022 to November 2023. OCT imaging revealed a pronounced layer of photoreceptors accompanied by hyperreflective material at the interdigitation zone of the photoreceptor cells (PRCs), both at the macula and extramacular regions. These structural changes correlated with subjective reports from both patients of intermittent brighter vision, which persisted through the final follow-up.
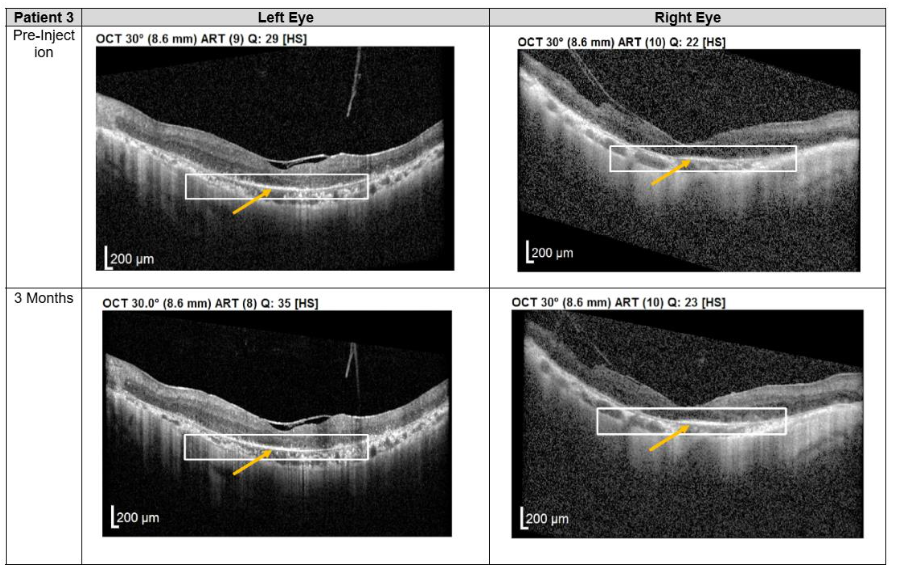
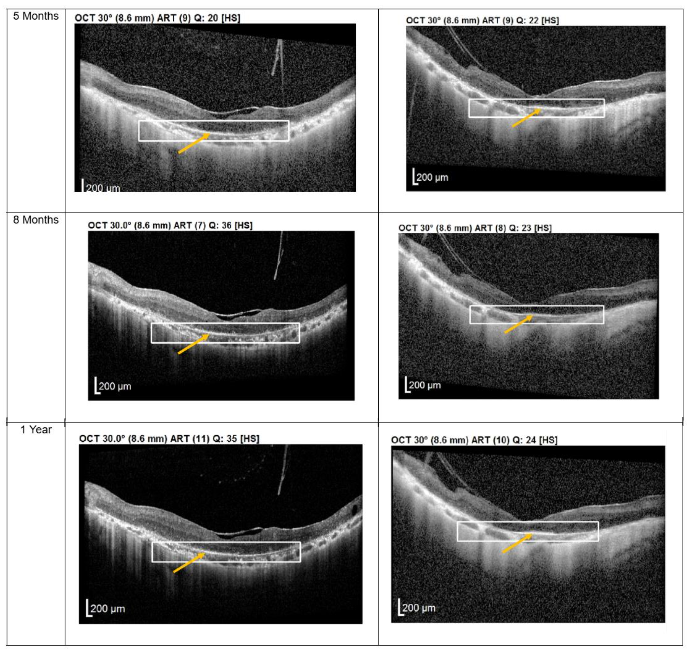
Figure 3. Optical coherence tomography (OCT) images for both eyes of Patient 3 from pre-injection up to 1 year follow-up. Orange arrow demonstrates the presence of hyperreflective material at the interdigitation area of the photoreceptors at the macula and extramacular region. These were observed and maintained until the last follow-up examination.
At the 12-month follow-up, no adverse events were reported. As with the first two patients, no serious ophthalmic or systemic complications were observed, further supporting the safety profile of subtenon MSC administration.
| Clinical Outcome | Patients 1 & 2 | Patients 3 & 4 |
|---|---|---|
| Number of MSC Injections | 7 injections (every 4-6 weeks) | 3 injections (every 4-6 weeks) |
| Follow-up Period | November 2020 – November 2022 (2 years) | December 2022 – November 2023 (1 year) |
| Vision Outcome | No deterioration in vision reported | Intermittent brighter vision reported, persistent through follow-up |
| OCT Findings | Maintenance of hyperreflective region, no intensity increase | Pronounced layer of photoreceptors, hyperreflective material at PRC interdigitation zone at both macula and extramacular regions |
| Visual Function | Improvements in visual function remained stable, no regression | Improvement in brightness, corresponding with OCT findings |
| Safety Profile | No severe ophthalmic or systemic complications | No severe ophthalmic or systemic complications |
| Adverse Events | None reported | None reported |
| OCT Structural Changes | Stable, no progression | Positive structural changes in photoreceptor layer |
| Follow-up Frequency | Every 4-6 weeks for MSC injections; then biannual follow-ups | Every 3-4 months post 3 MSC injections |
| Overall Conclusion | Stable visual function and safety over two years | Subjective improvement in vision, structural changes observed |
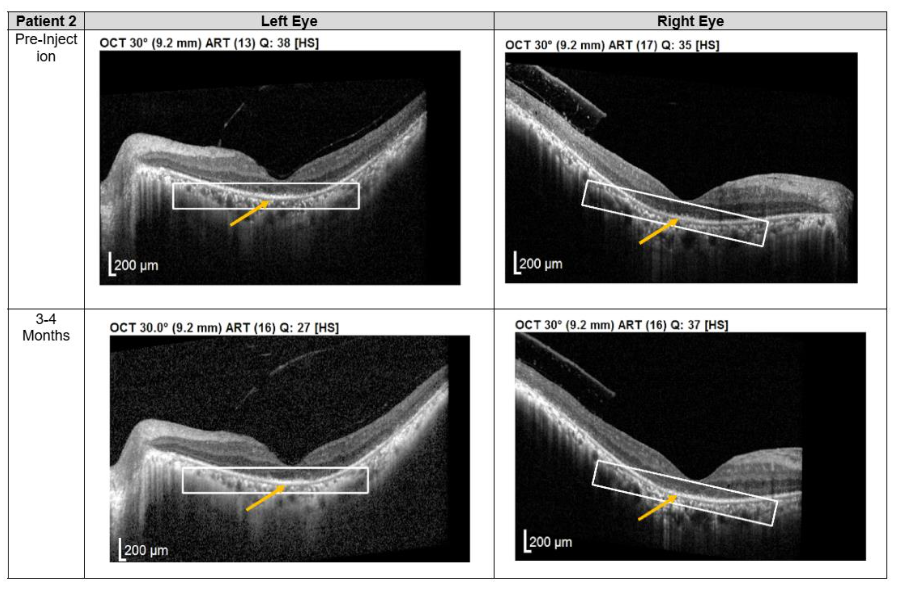
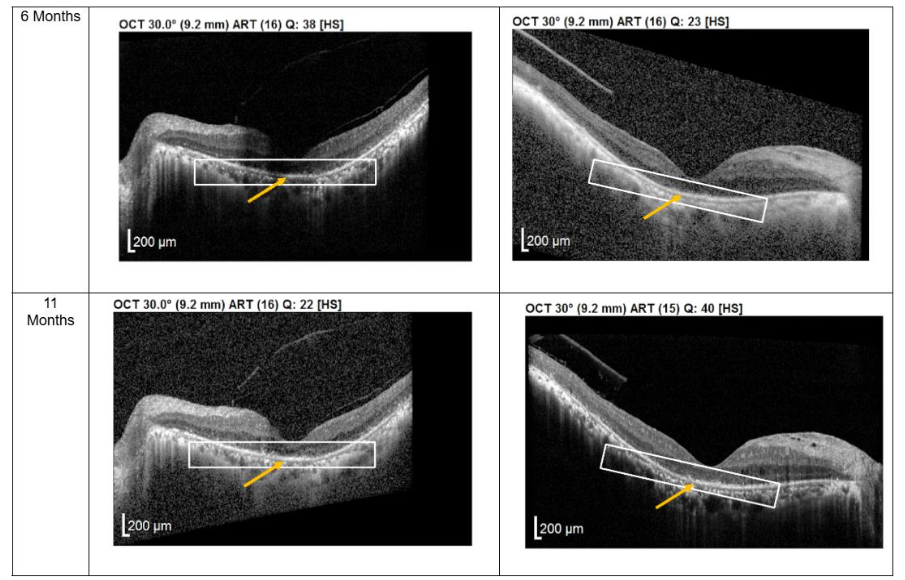
Figure 2. Optical coherence tomography (OCT) images for both eyes of Patient 2 from pre-injection up to 11 month follow-up. Orange arrow demonstrates the presence of hyperreflective material at the interdigitation area of the photoreceptors at the macula and extramacular region. These were observed and maintained until the last follow-up examination.
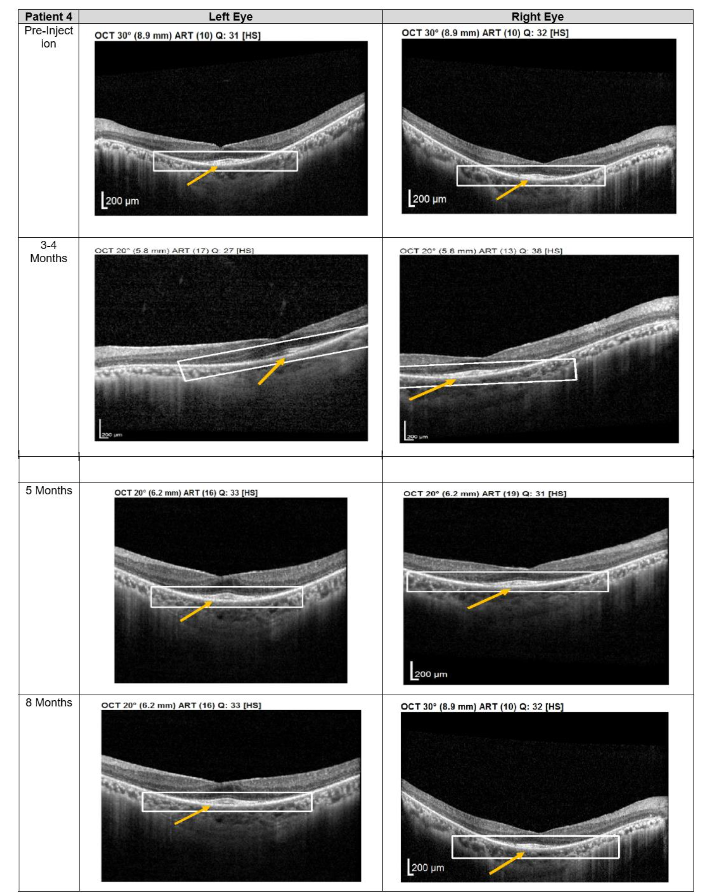
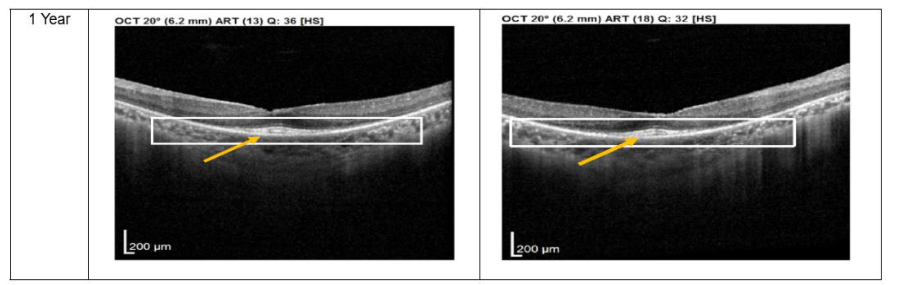 Figure 4. Optical coherence tomography (OCT) images for both eyes of Patient 4 from pre-injection up to 1 year follow-up. Orange arrow demonstrates the presence of hyperreflective material at the interdigitation area of the photoreceptors at the macula and extramacular region. These were observed and maintained until the last follow-up examination.
Figure 4. Optical coherence tomography (OCT) images for both eyes of Patient 4 from pre-injection up to 1 year follow-up. Orange arrow demonstrates the presence of hyperreflective material at the interdigitation area of the photoreceptors at the macula and extramacular region. These were observed and maintained until the last follow-up examination.Discussion
Retinitis pigmentosa (RP) is a progressive inherited retinal neurodegenerative disorder characterized by photoreceptor apoptosis, chronic neuroinflammation, and secondary inner retinal remodeling, ultimately leading to irreversible vision loss. Despite advances in genetic diagnostics, therapeutic options capable of altering disease progression remain limited. In this context, mesenchymal stromal cell (MSC)-based therapy has gained increasing attention as a disease-modifying strategy aimed at preserving residual retinal structure and function rather than replacing lost photoreceptors.
Mesenchymal stromal cells exert their therapeutic effects predominantly through paracrine mechanisms rather than direct cellular replacement. Although MSCs have demonstrated limited differentiation capacity toward retinal-like lineages in experimental settings, current evidence indicates that their primary benefit arises from the secretion of neurotrophic, anti-apoptotic, antioxidative, and immunomodulatory factors. These bioactive molecules including brain-derived neurotrophic factor, ciliary neurotrophic factor, glial cell line derived neurotrophic factor, hepatocyte growth factor, and vascular endothelial growth factor support photoreceptor survival, stabilize synaptic connectivity, and attenuate secondary neuronal degeneration.
Wharton’s Jelly derived MSCs (WJ-MSCs) offer distinct biological advantages over adult tissue derived MSCs, including higher proliferative capacity, lower immunogenicity, and a more potent anti-inflammatory secretome. WJ-MSCs have been shown to downregulate proinflammatory cytokines such as tumor necrosis factor α, interleukin (IL)-1β, and IL-6, while upregulating anti-inflammatory mediators including IL-10 and transforming growth factor β. This immunomodulatory effect is particularly relevant in RP, where persistent microglial activation and chronic inflammation continue to drive photoreceptor loss even after the initial genetic insult.
The subtenon route of MSC administration represents a minimally invasive delivery method that provides sustained trophic support to the posterior segment while avoiding the risks associated with intravitreal or subretinal injection. Prior clinical and experimental studies have demonstrated that MSCs delivered via the subtenon or peribulbar space can exert prolonged paracrine effects without direct intraocular migration, thereby reducing the risk of intraocular inflammation, retinal detachment, or endophthalmitis. The absence of serious ocular or systemic adverse events in the present case series, including patients followed for up to 24 months, further supports the favorable safety profile of subtenon WJ-MSC administration.
Structural changes observed on optical coherence tomography (OCT) provide additional insight into the biological effects of treatment. All patients demonstrated persistent hyperreflective material localized at the interdigitation zone of the photoreceptors. Rather than representing fibrotic change, this OCT finding may reflect photoreceptor outer segment remodeling, improved mitochondrial activity, or enhanced metabolic integrity of surviving photoreceptors. Similar hyperreflective changes have been reported in studies evaluating MSC-mediated neuroprotection and are often associated with functional stabilization rather than disease progression. The stability of these OCT findings over extended follow-up in patients 1 and 2 suggests a sustained therapeutic effect rather than a transient inflammatory response.
Functionally, patients receiving a greater number of subtenon injections over a longer treatment duration demonstrated stable visual function without further deterioration, whereas those receiving fewer injections reported intermittent subjective improvements in brightness perception. This observation aligns with emerging evidence that repeated MSC administrations may be necessary in chronic neurodegenerative diseases to maintain a favorable microenvironment, as transplanted MSCs have limited survival and exert time-dependent paracrine activity. The progressive nature of RP likely necessitates periodic reinforcement of neuroprotective signaling to counter ongoing inflammatory and apoptotic processes.
Importantly, the stabilization of visual function observed in this cohort contrasts with the expected natural history of RP, which is typically characterized by continuous functional decline. While the small sample size and non-randomized design preclude definitive efficacy conclusions, the absence of disease progression over 1 to 2 years suggests a potential disease-modifying effect. These findings are consistent with prior clinical reports of MSC-based therapies in RP, which similarly demonstrated safety and modest functional or structural benefits.
Taken together, the present findings support subtenon administration of WJ-MSCs as a safe and biologically plausible adjunctive therapy for RP. Although MSC therapy does not correct the underlying genetic defect, it may delay disease progression by preserving photoreceptor viability, modulating retinal inflammation, and stabilizing retinal architecture. Larger controlled studies with standardized outcome measures and extended follow-up are warranted to further define optimal dosing strategies and long-term efficacy.
Conclusion
This study provides extended longitudinal clinical evidence supporting the safety, feasibility, and biological plausibility of subtenon administration of Wharton’s Jelly derived mesenchymal stromal cells (WJ-MSCs) as a therapeutic approach for retinitis pigmentosa, a rare, progressive, and irreversible inherited retinal disorder with no approved disease-modifying treatment. As a continuation of our previously published clinical findings in the Medical Journal of Malaysia, the present study demonstrates consistent and reproducible clinical outcomes over an extended follow-up period of up to 24 months, with sustained stabilization of visual function and persistent structural retinal changes observed on optical coherence tomography. The concordance between early improvements reported in the initial publication and the longer-term outcomes observed in this cohort supports the durability of the therapeutic effect and reduces the likelihood of transient or placebo-driven responses.
Importantly, repeated subtenon WJ-MSC administration was well tolerated, with no serious ocular or systemic adverse events observed across both studies, supporting a favorable safety profile suitable for chronic administration in a degenerative condition. Given the progressive nature of retinitis pigmentosa and the absence of effective pharmacologic or biologic interventions capable of altering disease trajectory, the observed stabilization of disease progression represents a clinically meaningful outcome in this patient population.
The therapeutic rationale for WJ-MSCs is supported by established paracrine-mediated mechanisms, including immunomodulation, suppression of chronic retinal inflammation, and neuroprotection of residual photoreceptors, rather than irreversible structural replacement. This mechanism is particularly well suited to rare neurodegenerative retinal diseases, where preservation of remaining function is a primary clinical objective.
Furthermore, the minimally invasive subtenon delivery route offers a practical and scalable administration strategy with a favorable risk profile, enabling repeat dosing to maintain therapeutic benefit in alignment with the time-limited paracrine activity of mesenchymal stromal cells. The observed association between treatment intensity and sustained functional stabilization further supports the biological consistency of this approach.
Taken together, the cumulative safety data, reproducibility of clinical stabilization across sequential studies, mechanistic plausibility, and substantial unmet medical need support further regulatory consideration of subtenon WJ-MSC therapy under an orphan drug development framework for retinitis pigmentosa. Larger, controlled clinical studies are warranted to confirm efficacy, optimize dosing schedules, and define appropriate clinical endpoints in accordance with regulatory requirements.
Acknowledgement:
We would like to thank the Director General of Health Malaysia for allowing the publication of this manuscript and the staff of Department of Ophthalmology of Shah Alam Hospital for their contribution and support to this study. We also thank the Chief Executive Officer of 23 Century International Life Science Centre for her continuous support in this case study.
References:
- Khairullah, S. N., Fariza, N. N., Roslin, A. A. Z., & Angelina, T. (2022). Subtenon implantation of Wharton’s Jelly-derived mesenchymal stromal cells in retinitis pigmentosa. Med J Malaysia, 77(5), 564-568.
- Reddy, S. C., & Tan, B. C. (2001). Causes of childhood blindness in Malaysia: results from a national study of blind school students. International ophthalmology, 24, 53-59.
- Reddy, S. C., Tajunisah, I., Low, K. P., & Karmila, A. B. (2008). Prevalence of eye diseases and visual impairment in urban population a study from University of Malaya Medical Centre. Malaysian family physician: the official journal of the Academy of Family Physicians of Malaysia, 3(1), 25.
- Kevin-Tang, X. H., Tajunisah, I., Lott, P. P. W., & Reddy, S. C. (2024). Prevalence of visual impairment and eye diseases in Malaysia: A cross-sectional prospective study at the University of Malaya Medical Centre. Malaysian family physician: the official journal of the Academy of Family Physicians of Malaysia, 19, 30. https://doi.org/10.51866/oa.549
- https://www.moh.gov.my/Executive summary-mesenchymal stem cell for retinitis pigmentosa.
- Zarbin, M. (2016). Cell-based therapy for degenerative retinal disease. Trends in molecular medicine, 22(2), 115-134.
- Liang, X., Ding, Y., Zhang, Y., Tse, H. F., & Lian, Q. (2014). Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell transplantation, 23(9), 1045-1059.
- Kyurkchiev, D., Bochev, I., Ivanova-Todorova, E., Mourdjeva, M., Oreshkova, T., Belemezova, K., & Kyurkchiev, S. (2014). Secretion of immunoregulatory cytokines by mesenchymal stem cells. World journal of stem cells, 6(5), 552.
- Weiss JN, Levy S, Benes SC. SCOTS study in retinitis pigmentosa. Stem Cells Transl Med. 2015; 4(7):873-885.
- Satarian L, et al. Intravitreal MSCs in advanced RP. J Ophthalmic Vis Res. 2017;12(1):58-64.
- Ding SL, Kumar S, Mok PL. MSC reparative mechanisms in retinal disease. Int J Mol Sci. 2017; 18(7):1406.
- Harrell CR, et al. MSC secretome in retinal degeneration. Stem Cells Int. 2019;2019:1-16.
- Yu B, et al. MSC-derived exosomes in retinal degeneration. Stem Cell Res Ther. 2020;11:526.
- Nazarov PV, et al. Immunomodulatory properties of WJ-MSCs. Cell Mol Life Sci. 2020;77:3491-3511.

