Targeting Female Germline Stem Cells in Ovarian Health
Targeting Female germline stem cells -an attractive modality in ovarian insufficiency / postponed fertility / therapy of infertility following treated malignant tumors -A narrative review
Dr Kulvinder Kochar Kaur, M.D.¹, DR Gautam Allahbadia M.D.², DR Mandeep Singh M.D.DM.³
- (Obstt& Gynae, specialist reproductive endocrinology & Infertility specialist). Scientific Director Dr Kulvinder Kaur Centre For Human Reproduction
https://orcid.org/0000-0003-1473-3419 - (Obstt& Gynae), D.N.B, Scientific Director; Ex-Rotunda-A Centre for Human Reproduction
- (Std) (Neurology), Consultant Neurologist; Swami Satyanand Hospital
OPEN ACCESS
PUBLISHED: 31 March 2026
CITATION: Kaur, K.K., 2026. Targeting Female germline stem cells -an attractive modality in ovarian insufficiency / postponed fertility / therapy of infertility following treated malignant tumors -A narrative review. Medical Research Archives, [online] 14(3).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
In the arena of reproductive medicine, postponement of ovarian aging and perpetuating fertility in cancer patients have long been believed to be main botherations in addition to continued efforts have been put in. Female germline stem cells (FGSCs) have been illustrated to cause healing of aging or injured ovarian structures as well as result in restoration of i) ovarian reproductive, along with ii) endocrine working. With their i) limitless proliferation in addition to ii) guided differentiation into oocytes, iii) FGSCs yield innovative enthusiasm to patients with i) ovarian insufficiency, ii) malignant tumors, and iii) others who require fertility preservation. Previously we had reviewed exhaustively etiopathogenesis of premature ovarian insufficiency (POI)/diminished ovarian reserve (DOR) and the manner manipulating signaling pathways helped in associated generation of early/primordial follicles enhanced ART outcomes in POI&DOR patients. Further we thoroughly reviewed part of stem cells inclusive of very small embryonic-like stem cells (VSELs) in oncofertility/ part of silent information regulator 1 (Sirt1) signalling in improvement of escalating oocyte quality in women presenting with advanced maternal age. In this review, we debut the part of FGSCs in i) ovarian fertility preservation ii) regenerative healing, highlighting the controlling pathways of FGSCs in resulting in restoration of ovarian working. We detail the distinct benefits of FGSCs in infertility therapy, inclusive of i) fertility preservation, ii) animal gene editing, and, iii) regenerative medicine. This objective of our article is yielding innovative research to gather understanding regarding advancement of the clinical translation of FGSCs by evaluating them from plethora of viewpoints, for instance i) origin, ii) controlling, along with iii) implementation.
Keywords: Female germline stem cells (FGSCs); ovarian insufficiency; malignant tumors; postponement of ovarian aging
1. Introduction
Ovarian aging delineates age-associated reduction in ovarian working as well as infertility concern that takes place secondary to ovarian diseases have assumed meaningful botherations in the field of worldwide reproductive health. Primary ovarian insufficiency (POI) possesses the properties of a diminished follicle count in addition to a reduction in oocyte quality, resulting in an irreversible diminishing in ovarian working, along with estrogen quantities in women under 40. In recent years, the incidence of POI has been escalating meaningfully, with a worldwide prevalence of 3.7%. In China, the prevalence has escalated from 2.5% twenty years back to 15%, with ovarian-associated factors working in the form of contributors of 15% to 25% of cases. POI apart from causing amenorrhea as well as infertility, however is further intricately associated with systemic health issues for instance i) osteoporosis, ii) cardiovascular diseases, in addition to iii) cognitive impairments.
The mechanistic modes via which ovarian diseases influence fertility are complicated. For instance, ovarian cysts greater than 5 cm as well as cystectomy have been illustrated to meaningfully affect ovarian reserve six months subsequent to surgery. Chemotherapeutic substances, particularly alkylating compounds, possess the capacity of directly causing injury to follicular structures, resulting in irreversible ovarian injury. i) Radiotherapy, ii) bone marrow transplantation, in addition to iii) autoimmune conditions (such as lupus) stimulates minimally one million POI cases worldwide each year. In recent times, variable arbitration approaches have been posited in reference to tackling such challenges. Successful employment of ovarian tissue cryopreservation in combination with in vitro activation (IVA) has been made in patients with POI who possessed just occasional primordial follicles (PFs) along with healthy pregnancies have been documented. GnRH-a might attenuate chemotherapy stimulated-damage to certain magnitude. Additionally, lifestyle interpositions (for instance antioxidant diet, BMI regulation) as well as early fertility preservation (for instance egg freezing) have been illustrated to postpone reduction in ovarian PFs. Nonetheless, there is existence of requirement of the manner tackling of the dysfunctional ovarian reproductive in addition to endocrine working remodeling has to be attained. Thereby avoidance of along with treatment approaches regarding i) ovarian aging as well as ii) chemotherapy-associated infertility buttressing is required.
2. Corroboration regarding presence of female germline cells in the ovary
2.1 DEBATE IN REFERENCE TO FEMALE GERMLINE CELLS
The query in reference to if there is presence of female germline cells (FGSCs) in mammals as well as their differentiation takes place into working oocytes has been debatable right from the nineteenth century. Substantial studies have illustrated that the ovarian follicle pool is estimated at the time of the perinatal period, in addition to no self-renewing stem cells or newly produced oocytes are existent in the postnatal mouse ovary. The canonical posit that the mammalian ovarian follicle pool is fixed at birth predominated the field for practically four decades. Nevertheless, subsequent to advent of FGSCs, accrual of corroboration has questioned the fixed follicle pool theory that had been pursued throughout last century in reproductive medicine.
To start with Johnson et al. pointed to the existence of proliferative germ cells in the postnatal mammalian ovary in 2004, as well as assisted in follicular in addition to oocyte regeneration. Subsequent to performance of quantification of atretic along with non-atretic follicles in C57BL/6 mice, they found a swift augmentation of follicular atresia subsequent to postnatal day 30, with just continuation of the one-third of the seminal follicle pool by day 42. Dependent on the atresia rate, they anticipated full elimination of the follicle pool amongst weeks. Nonetheless, the full quantity of atretic follicles surpassed the exactitude of elimination, pointing to continuation of the oocyte regeneration. Additionally, transplantation of wild-kind ovaries into transgenic female mice universally, expressing green fluorescent protein (GFP) documented existence of GFP-positive germ cells infiltrating host ovaries as well as new follicles. Such observations defied the canonical belief that the ovary possesses just 1%~33% immature follicles at any stage, resulting in the pathfinder oocyte regeneration hypothesis. Their following work pointed that bone marrow (BM) expresses germline markers in addition to causes rectification of oocyte generation in sterilized mice through transplantation. Donor-obtained oocytes, corroborated by morphology, along with molecular markers, point BM in the form of a plausible germ cell facility maintaining oogenesis in adulthood. The rhetorical of those not in agreement with Johnson’s hypothesis, was that their relative assessment methodologies for atretic as well as non-atretic follicles possess restrictions. The guideline in reference to isolating atretic follicles were believed to be subjective in addition to strain-particular, along with existence of plausible artifacts at the time of tissue fixation might have overdetermined atresia rates. Utilization of single-cell RNA sequencing (scRNA-seq as well as fluorescence-activated cell sorting (FACS) in case of recent studies possessed failure in reference to estimating FGSCs in adult ovarian cortex samples. Nevertheless, Woods and Tilly, in 2023 were critical of such observations, emphasizing main lacunae in Wagner et al.’s workflow. Their observations regarding the removal of cells with gene expression profiles analogous to FGSCs, remarkable cell damage/demise at the time of sample preparation, as well as the requirement for high cell viability in advanced techniques like scRNA-seq. Thus, the lack of FGSCs in such studies continues to be nonconclusive, along with escalating confirmation continues to embrace the existence of FGSCs.
The debate encompassing FGSCs basically revolves on the advocating faction’s corroboration from mouse models, where DEAD-box helicase 4 (DDX4), -positive cells generate oocytes, compared to converse debates that human ovaries possess absence of active germline stem cell niches. This opposes the germaneness for generalizing rodent observations to humans. Nevertheless, with FGSCs escalatingly identified, along with cultured over variable species in addition to side by side persistent continuous advancements would eventually aid in attainment of greater exhaustive resolution of such controversial arena.
2.2 PRESENCE OF FEMALE GERMLINE CELLS
Bukovsky et al., in 2008, identified a minimal quantity of adult ovarian surface tissue as well as cultured it in vitro for 5~6 days, finding the generation of follicle-like structures which expelled polar bodies in addition to possess the properties of secondary follicles. Nonetheless, they identified, ovarian surface epithelial cells or ovarian stem cells (OSE), not particular FGSCs. The manner detailed in their study, the identified OSE might differentiate into different cell kinds inclusive of i) mesenchymal, ii) epithelial, iii) granulosa, iv) neural kind cells, along with v) oocytes. The initial identification as well as purification of FGSCs, got performed by Zou et al., with success in 2009, demonstrating unique germline stem cell characteristics. Following that Lu, White, along with Zhang autonomously identified germline stem cells from ovaries of different species with the utilization of variable approaches in reference to stem cells working. Tackling cynicism, recent observations illustrate that idealized single-cell RNA sequencing (scRNA-seq) workflows possess the capacity of isolation of occasional germline cells in adult ovarian cortical tissues which harmonize ovarian stem cells (OSCs) however vary from other cell kinds, inclusive of i) oocytes in addition to ii) perivascular cells (PVCs). Further evaluation displayed pivotal molecular corroboration of such germline cells beginning the initial phase of meiosis amongst mature ovarian tissues. Up till now, researchers have consistently isolated ovarian germline stem cells (FGSCs) in the ovaries of i) rats, ii) pigs, iii) sheep, in addition to iv) humans, pointing to their broader existence across mammals. Present botherations exist in exhaustively evaluating the properties of cell populations amongst adult mammalian ovaries as well as defining situations which might embrace in vitro in addition to in vivo neooogenesis.
2.3 PROPERTIES OF FEMALE GERMLINE STEM CELLS
FGSCs apart from perpetuating the follicular pool, however further illustrated canonical properties of adult stem cells. Pivotal properties of FGSCs are illustrated as follows in Figure 1.
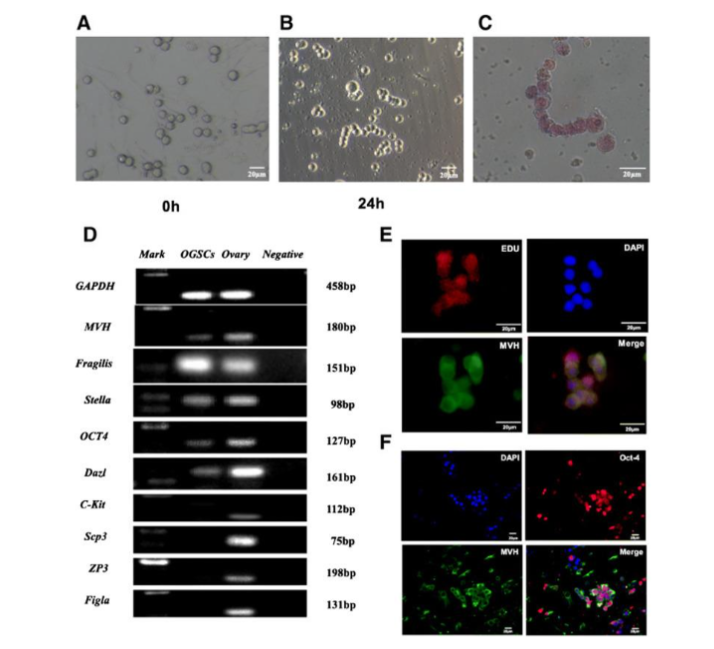
(1) Morphology as well as Growth designs: FGSCs are analogous spermatogonial stem cells (SSCs), displaying a spherical shape, grape-like clusters, large in addition to bright spherical nuclei, along with distinct boundaries amongst nuclei in addition to cytoplasm.
(2) Germline-Specific Markers: FGSCs express germ cell-specific markers for instance Mouse Vasa Homologue (MVH), Dazl, octamer-binding transcription-4 (OCT-4), as well as Fragilis, coordinating with the molecular profile of SSCs.
(3) Proliferative Capacity: Dual immunofluorescence staining for MVH in addition to B5-Bromo-2′-deoxyuridine (BrDU) corroborated that FGSCs possess the retention of mitotic activity whereas conserving identity of germline.
(4) Cell Cycle controlling: FGSCs express cell cycle associated transcription factors (for instance c-MYC, EGR-1) alongside high telomerase activity, along with TERT in addition to alkaline phosphatase (AP).
(5) Probability of Differentiation: subsequent to estrogen supplementation, FGSCs differentiate into oocytes in vitro, along with form fertile offspring subsequent to transplantation into mouse ovaries.
3. Controlling of female germline stem cells
Sustenance of ovarian reserve in addition to working homeostasis via self-renewal as well as directed differentiation gets attained by FGSCs. FGSCs facilitate follicular regeneration by activating signaling pathways for instance Notch along with Hedgehog (Hh), whereas liberating antioxidant factors (for instance i) superoxide dismutase (SOD), ii) glutathione peroxidase (GPx) to circumvent oxidative stress (OS)-stimulated injury to oocytes. FGSCs control ovarian working via plethora of pathways, with crucial mechanistic modes delineated hereafter.
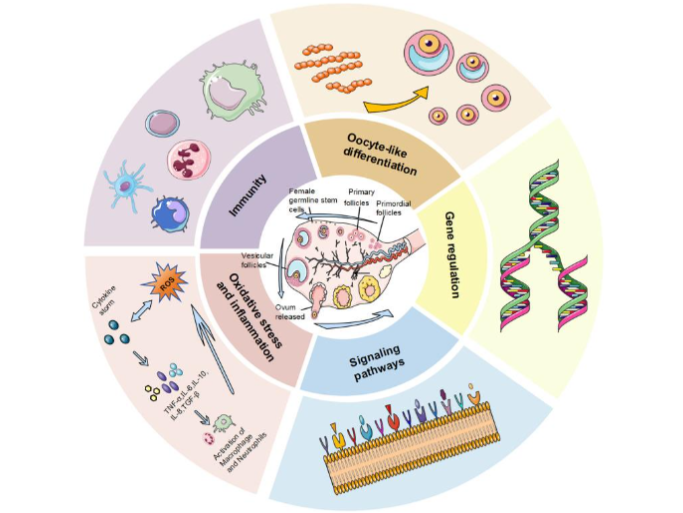
(1) Immune Cell modulated: Immune cells as well as their liberated factors take part in addition to facilitate the proliferation, along with differentiation of ovarian germline stem cells (OGSCs), thereby causes restoration of ovarian endocrine working.
(2) Direct Differentiation: FGSCs directly differentiate into primary oocytes, that further generates into mature follicles, resulting in renewal of the follicular pool.
(3) Diminishing of Stress & Inflammation: Diminishing oxidative stress (OS)- as well as inflammation causes restoration of ovarian working in addition to FGSC actions, facilitating FGSC survival.
(4) Gene controlling: Subsequent to estrogen (E2) triggering, binding of ERα takes place with the Stra8 gene promoter (that is activated by retinoic acid) to stimulate Stra8 transcription, eventually stimulating oocyte generation.
(5) Controlling of signaling pathways: Modulating signaling pathways stimulates an autophagic protection conferring reactions in FGSCs, attenuating cellular senescence.
3.1 SIGNALING PATHWAYS
Plethora of signaling pathways control FGSCs, therefore leading to improvement of ovarian working.
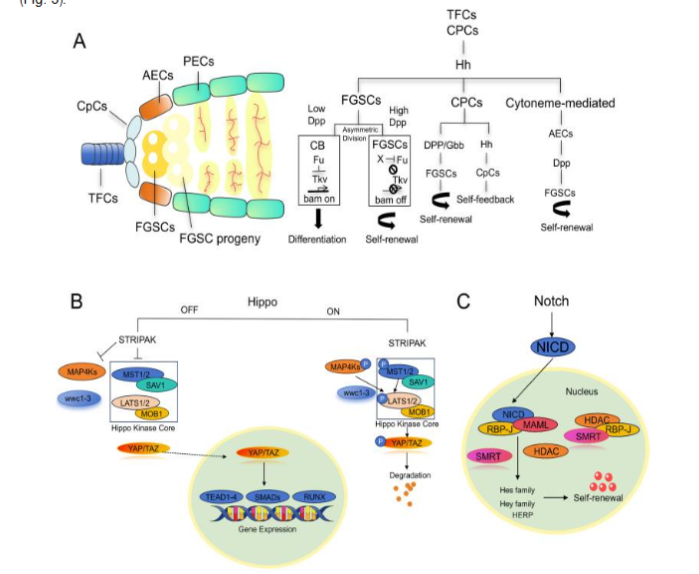
(3.1A) The Hedgehog (Hh) pathway directly serves on FGSCs by activating downstream target genes to suppress differentiation as well as maintenance of self-renewal. Hh further indirectly controls FGSCs via micromilieu stepwise patterns. For instance, CpCs (cap cells) activate Hh signaling to stimulate expression of Bone morphogenetic proteins (BMP) family members Dpp in addition to Gbb in anterior epithelial cells (AECs), whereas directly facilitating Dpp transcription in CpCs themselves. Escalated BMP (Dpp) signaling impedes the differentiation factor Bam, further barricading FGSCs differentiation. FGSCs differentiation, is dynamically orchestrated at the time of beginning of differentiation, the Hh pathway’s positive governer Fused (Fu) is upregulated. Fu facilitates FGSCs conversion into mature germ cells by modulating ubiquitin-based breakdown of the BMP receptor Tkv, therefore ameliorating BMP-modulated differentiation repression.
(3.1B) The Notch pathway possesses pivotal parts in i) cell proliferation, ii) differentiation, as well as iii) fate estimation, meaningfully affecting iv) both somatic in addition to v) germline stem cells. Notch controls sustenance of i) FGSCs stemness, along with ii) differentiation through iii) intercellular connection. In Drosophila, ovarian stem cell niche development a) depends on Notch signaling. b) Germline stem cells (GSCs) possess placement in somatic niches where Notch is crucial: c) activating Delta in germ cells or Notch in somatic cells escalates niche cell numbers, triggering extra GSC generation. d) On the other hand, GSCs possessing absence of working Notch ligands (Delta/Serrate) illustrated failure to activate transforming growth factor beta (TGF-β) signaling, resulting in differentiation as well as niche exit. e) There is existence of association of diminished Notch signaling i) working with ovarian aging, ii) owing to diminished Notch activity elimination of FGSCs in addition to iii) amplified primordial follicle atresia.
(3.1C) The Hippo pathway pivotally controls ovarian working, specifically FGSCs. Via kinase stepwise patterns, Hippo regulates i) cell proliferation, ii) apoptosis, along with iii) organ size. Hippo activation phosphorylates downstream coactivators the Yes-associated protein (YAP) along with transcriptional co-activator with post synaptic density protein, drosophila disc large tumor suppressor [PDZ] and zonula occludens-1-binding motif (TAZ), holding them in the cytoplasm for breakdown, therefore hampering proliferation as well as facilitating apoptosis. On the other hand, Hippo hampering aids in translocation of dephosphorylated YAP/TAZ into the nucleus, leading to binding of DNA binding proteins TEA domain (TEAD) family transcription factors to activate genes guiding proliferation as well as stem cell replenishment. In ovaries, Hippo signaling is imperative for i) FGSCs homeostasis. ii) Decontrolled Hippo signaling is associated with ovarian aging in addition to iii) disease, iv) yielding an understanding into pathology along with v) therapeutic approaches. In mouse ovarian cortex, co-expression of Hippo constituents Large Tumor Suppressor Kinase 2 (LATS2), mammalian Sterile 20-like kinase 1 (MST1) as well as germline markers MVH, OCT4 takes place. Their quantities in addition to colocalization reduction takes place with age, whereas YAP1 is prevalent in 2-month-old mice however lacking in 20-month-old mice, pointing to Hippo’s part in FGSCs dynamics at the time of physiological, along with pathological ovarian aging.
3.2 STEM CELL NICHE
Schofield’s theory in 1978 regarding stem cell niche hypothesized that niches are constituted of i) extracellular matrix (ECM), ii) niche cells, iii) granulocytes, iv) blood vessels, v) immune cells, as well as vi) liberated factors. Escalated confirmation implicates FGSCs niche impairment in the form of a pivotal guide of a) ovarian failure, which probably possesses greater influence in contrast to c) FGSCs aging by itself. Nevertheless, inflammatory micromilieu disturb niche wholeness. Macrophages, with their multifaceted nature in addition to cytokine-liberating flexibility, might result in stabilization of the niches by removal of senescent red blood cells along with necrotic tissue through interleukin-10 (IL-10), as well as Tumor necrosis factor alpha (TNFα).
3.3 OXIDATIVE STRESS (OS) ALONG WITH CHRONIC INFLAMMATION
i) Oxidative stress (OS) as well as ii) chronic inflammation amplify stem cell aging. Follicular hypoxia directs ovarian aging by i) reducing intracellular pH in addition to ii) oocyte metabolism, iii) destabilizing meiotic spindles. Hypoxia activates oxygen sensor hypoxia inducible factor 1 α (HIF 1 α), escalating i) glycolysis, ii) angiogenesis, iii) leukocyte migration, iv) inflammation, along with v) follicular wall rupture. It further stimulates i) ROS overgeneration, ii) accelerating oxidative ovarian injury. ROS in addition to inflammation generate a vicious cycle: ROS activates i) nucleotide-binding domain, leucine-rich-repeat containing family, pyrin domain-containing (NLRP3) inflammasome as well as ii) nuclear factor κB (NF-κB), upregulating iii) IL-1β, iv) IL-6, in addition to v) TNF-α, that also augment a) OS, along with b) aging. Antioxidant arbitration for instance a) resveratrol (RES) causes restoration of ovarian working as well as viability of FGSCs in POI models by diminishing OS in addition to inflammation. b) Chitosan oligosaccharides result in improvement of ovarian micromilieu along with induce FGSCs proliferation through immune-correlated factors. c) Spermidine (SPD) possesses protection conferring FGSCs actions from H2O2-stimulated senescence by stimulating autophagy through phosphatidyl inositol 3-kinase (PI3K) / protein kinase B (AKT). Such approaches might postpone reproductive aging as well as conserve fertility.
3.4 OTHERS
Polycystic Ovary Syndrome (PCOS), a common endocrine disorder, is associated with i) hyperandrogenism, ii) glucose intolerance, as well as iii) cystic ovaries. Metformin, a first-line antidiabetic agent, meaningfully escalates i) proliferative FGSCs in addition to upregulates ii) proliferating cell nuclear antigen (PCNA) iii) cyclin D2, iv) phosphorylated mammalian target of rapamycin (p-mTOR), along with v) phosphorylated 5 AMP-activated protein kinase (p-AMPK) in PCOS mice, leading to restoration of FGSCs working through AMPK/mTOR signaling. D) Soy isoflavones activate Akt signaling by upregulating C-type lectin domain family 11 member A (Clec11a), promoting FGSCs survival as well as proliferation. Such observations, along with transplantation in addition to genetic lineage-tracing outcomes, corroborate capability of FGSCs in creating healthy oocytes, embryos, along with offspring, buttressing their plausibility of tackling female fertility as well as ovarian conditions.
4. Clinical employment along with translational prospects of female germline stem cells
4.1 FERTILITY PRESERVATION
FGSCs offer innovative approaches for fertility preservation along with the postponement of menopause. FGSCs possess the capacity of either getting transplanted into ovaries regarding beginning of the oogenesis in reference to creating fertile offspring or cultured in vitro as well as differentiated into oocytes subsequent to injection into human ovarian cortical tissue, with following xenotransplantation into immunodeficient adult female mice. Working oocytes further possess the capacity of getting obtained in vitro from SSCs.
White et al. isolated FGSCs from adult human ovaries in addition to transplanted them into mouse ovarian cortices, finding immature oocytes, along with granulosa cell generation. Satirapod’s along with his coworkers corroborated that in mouse FGSCs express estrogen receptor-α (ERα), that crosstalks with retinoic acid-induced Stra8 at the time of oogenesis for guiding Stra8 expression. Xiong et al. illustrated restoration of fertility that resulted subsequent to chemotherapy- in case of infertile mice by transplantation in vitro-cultured FGSCs into their ovaries. Wu et al. illustrated in 2017 that FGSCs start differentiation into early oocytes subsequent to their arrival to the ovarian cortex, with transplanted FGSCs (F-TFs) leading to restoration of ovarian working in addition to creating offspring. Taken together, such observations corroborate the presence of proliferative germ cells in postnatal mammalian ovaries, implicated sustaining oocyte as well as follicle development.
Bukovsky and Presl posited in 1979 that immune system-ovary crosstalk takes place in controlling reproduction in addition to reproductive conditions. Studies that followed embrace this: athymic (nude) female mice illustrated diminished gonadotropin quantities reversible through neonatal thymosin treatment. In case of humans, i) ovarian surface epithelial cells (OSCs) serve in the form of ii) bipotent stem cells iii) producing germ along with iv) granulosa cells. v) Immune controlling takes part in physiological neo-oogenesis as well as vi) follicular replenishment at the time of a) fetal in addition to b) reproductive stages, with i) FGSCs proliferation, along with ii) differentiation manipulated by iii) immune cells as well as their iv) liberated factors.
4.2 ORGANOIDS ALONG WITH IN VITRO MODELS
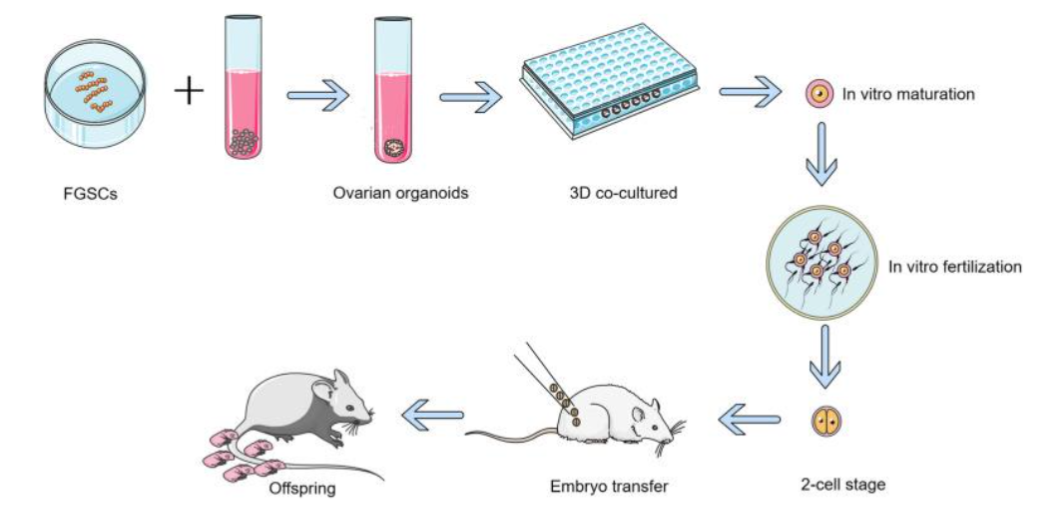
Li X et al. in 2021 generated ovarian organoids from mouse FGSCs by utilization of a 3D culture system. Such organoids, analogous to normal ovaries, possessed follicles along with liberated hormones. Single-cell sequencing isolated six cell populations, inclusive of i) germ, ii) granulosa, as well as iii) theca cells, with unique gene expression profiles. Follicles from organoids matured in vitro in addition to generated normal offspring through fertilization. One additional group utilized total-organ decellularization to generate 3D bioscaffolds simulating the natural ovarian micromilieu, perpetuating native microarchitecture, along with biochemical tips. Purification of the FGSCs through MACS complied to such scaffolds amongst 24 h, i) that colonized the extracellular matrix (ECM), as well as ii) developed cluster-like structures. In 2023 Luo et al. formed semiconductor polymer dot (Pdot)- dependent siRNA nanocomposites for targeted gene knockdown in FGSCs. Pdots illustrated i) greater fluorescence, ii) making real-time following up of cellular uptake, iii) intracellular trading, in addition to iv) exocytosis. v) Noticeably, Pdots- illustrated least cytotoxicity, along with differentiation disturbance, evading lysosomes for efficacious extracellular liberation, that enables them to be optimal nanocarriers. Further penetration of Pdot-siRNA was brought about in reference to the FGSC-obtained 3D ovarian organoids, efficaciously downregulating target genes. Therefore, bioengineered ovarian prosthetics (see Figure 4) utilization of FGSCs delineates an innovative approach regarding rectification of fertility with safety, specifically in cancer survivors.
4.3 STEM CELL ALONG WITH GENE THERAPY
FGSCs are escalatingly getting acknowledged for their plausibility regarding therapy of infertility, perpetuating fertility, aiding in animal gene editing, as well as resulting in advancements in regenerative medicine. Ovarian impairment or insufficiency, resulting from i) genetic abnormalities, ii) autoimmunity, or iii) damage, as well as chemotherapy/radiation-induced ovarian failure, constitute main etiological factors of infertility. Present modalities (oocyte/embryo cryopreservation, ovarian tissue freezing) possess the risk of transportation of the malignant cells back into cancer survivors. Stem cell therapies, are inclusive of i) embryonic stem cells (ESCs), ii) mesenchymal stem cells (MSCs), in addition to iii) induced pluripotent stem cells (iPSCs), are incepting in the form of attractive methodologies for alternatives modes of treatments. FGSCs, possess the capabilities of self-replenishment along with differentiation, offer distinct plausibility regarding therapy of the ovarian diseases.
5. Drawbacks of ovarian germline stem cell research
5.1 TECHNICAL HINDRANCES OF FEMALE GERMLINE STEM CELLS
The isolation as well as identification of ovarian germline stem cells (FGSCs) continues to be bothesome. FGSCs possess absence of considerably particular surface markers, in addition to current identification modalities usually aids in contamination of samples with other ovarian cells, jeopardizing purity. In the clinical scenario identification of FGSCs needs ovarian tissue extraction an event restrained by restricted accessibility of sample, facilities, along with invasive risks. In humans, FGSCs are yielded from ovarian cortical tissue within follicular aspirates. Nevertheless, lesser ovarian tissue obtained in aspirates results in noncommensurate success rates in generating cell lines. FGSCs illustrated lesser effectiveness in differentiating into mature oocytes in vitro as well as usually frequently go through senescence or elimination of plausibility of differentiation at the time of expansion leading to, meaningful omission of stemness subsequent to passaging. Canonical 2-dimensional (2D) culture systems do not possess the capacity of sufficiently mimicking the ovarian micromilieu in vitro. Contrasting with mesenchymal stem cells (MSCs), long-term culture protocols for FGSCs continues to be nonidealized. Whereas Matrigel-dependent 3D culture escalates i) cell viability as well as ii) sustenance of stemness, iii) vital frameworks inclusive of iv) Matrigel content, v) embedding modalities (e.g., coculture with feeder cells), in addition to vi) batch-to-batch consistency need continued idealization.
5.2 CLINICAL SAFETY OF FEMALE GERMLINE STEM CELLS THERAPY
There is absence of the existence of long-term outcomes presently in reference to corroboration of safety of FGSC owing to it might stimulate tumor generation along with possesses the capability of epigenetic memory retention from source cells, which makes it imperative to take into account the plausible clinical risks. Whereas transplanted cells need to be incorporated into the i) host ovarian stroma, ii) fibrotic or iii) inflammatory micromilieu might impede FGSC engraftment. Furthermore, allogeneic FGSCs possess the capacity of stimulating host, immune attacks. Germline gene editing in human FGSCs needs considerable precaution owing to inheritance by the offspring. The complicated nature of the ovarian micromilieu, combination with stem cell treatments continues to be in clinical trial/research phases, which leads to meaningful interpersonal discrepancies in reactions to therapy within subjects. The results of post-transplantation are capable of getting impacted by age of the patient as well as the etiological factors that lie beneath.
5.3 MODALITIES OF RESOLUTIONS OF RESEARCH DRAWBACKS OF FEMALE GERMLINE STEM CELLS
Idealization of the culture as well as differentiation systems for FGSCs possess the capacity of getting attained by the utilization of combination of natural stimulators (for instance Cistanche polysaccharides) to target the TGF-β pathway, therefore escalating differentiation orchestration. Efficacious hampering of EED actions leads to improvement of culture effectiveness in addition to cell procured. Serum-free along with feeder-free FGSC culture systems possess the capacity of getting generated by the utilization of Matrigel idealization. Regarding tackling of the botherations in reference to clinical safety, plethora of imaging modalities (for instance MRI-fluorescence dual labeling) aids in real-time monitoring of organization of transplanted cell in vivo for early determination of aberrant proliferation. Categorization of patients as per causative factors diminishes multifaceted nature of therapies results. Long-term keeping track of animal transplantation for greater than 6 months requires implementation of mesenchymal stem cells (MSCs) safety protocols evaluation, regarding monitoring of teratoma generation along with intergenerational to reproductive actions. Establishing ovarian cell (granulosa/embracing cells)-FGSC co-culture organoids simulates physiological follicular micromilieu, causing advancing differentiation mechanistic modes research. Furthermore, 3D bioscaffolds possess the capability of reproducibility of natural ovarian niches possess the capacity of getting engineered. Repopulation of purified FGSCs of decellularized ovarian scaffolds takes place subsequent to triggering that might result in restoration of working. Such modalities delineate attractive technologies regarding assessment of ovarian reproductive in addition to endocrine working glutathione peroxidase (GPx). Further earlier we had reviewed parthenogenetic embryonic stem cells, induced pluripotent stem cells, very small embryonic-like stem cells (VSELs) in Oncofertility, targeting SIRT Signaling Pathway in reference to escalating oocyte quality in women presenting with advanced maternal age.
6. Role of Proanthocyanidins
Bhartiya D & Sharma D isolated two kinds of stem cells in ovary surface epithelial (OSE) of mammals: i) very small embryonic-like stem cells (VSELs) as well as ii) ovarian germline stem cells (OGSCs). Such cells possess the capacity of going through long-term in vitro expansion in addition to eventually differentiating into an oocyte-like structure, leading to resulting in the development of fertile pups. OGSCs are robustly associated with the FGSCs that Wu et al. are presently evaluating. Sharma D observed no disappearance of OGSCs subsequent to chemotherapy or aging, as well as outcomes illustrated that some quantity of VSELs survived in ovarian tissue subsequent to chemoablation in addition to possessed the capacity of retention of differentiating into oocyte-like structures. In future empirical modulations, usable cells from OSE would further get identified for finding in reference to the manner protection gets conferred against age correlated reduction or by external factor in further studies.
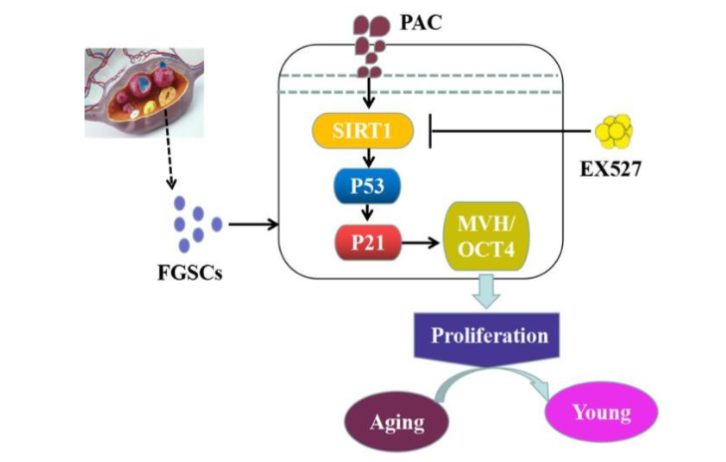
Proanthocyanidins (PACs), delineate natural antioxidants broadly acknowledged regarding their i) anti-aging as well as ii) anti-inflammatory characteristics, in addition to are frequently utilized in variable avenues for instance i) food, ii) medicine iii) environmental protection, iv) cosmetics, along with v) agriculture. PACs portray water-soluble phenolic substances which canonically are present in i) fruits, ii) vegetables, iii) nuts as well as iv) plants. Their antioxidant capability is 40 fold larger than that of vitamin C or E. Owing to their natural antioxidant characteristics, PACs have been pointed in the form of a plausible protection conferring against oxidative stress (OS) injury to separate organs in addition to tissues, inclusive of the ovaries. Research has illustrated that PACs possess the capacity of efficaciously ameliorating pathological alteration in granulosa cells by inhibiting autophagy, along with apoptosis, that eventually account for postponement of ovarian aging. Nevertheless, studies evaluating the actions of PACs on FGSCs continues to be not revealed. A natural reduction of ovarian follicle pool takes place with ageing of women. Nevertheless, FGSCs possess a distinct capability of differentiating into oocytes as well as indelibly self-replenish, yielding an efficacious vi arena of postponement of ovarian aging by renewal of the primordial follicle pool. Thereby, activating FGSCs is vital in reshaping in addition to safeguarding ovarian working. Wu et al. conducted the evaluation of the biological actions of proanthocyanidins (PACs), that delineate natural antioxidants that illustrate anti-aging as well as anti-inflammatory characteristics, which is advantageous for both male in addition to female reproduction. Their in vivo, along with in vitro experiments illustrate that PACs facilitate FGSCs proliferation whereas postponing ovarian aging. Their observations were that PACs escalated the quantity of i) primordial follicles, ii) primary follicles, iii) corpus luteum whereas diminishing iv) cystic follicles, as well as v) escalated estradiol (E2) quantities along with anti-mullerian hormone (AMH) quantities in mice. Furthermore, PACs meaningfully buttressed FGSCs proliferation time- in addition to in a dose-based fashion by upregulating mRNA, along with protein expressions for FGSCs- iii) particular markers for instance i) MVH as well as ii) OCT4 whereas downregulating p53/p21 through activation of iv) silent information regulator 1 (Sirt1) signaling pathway. The actions of PACs on FGCSs were observed to be hindered by the Sirt1 hampering agent EX527. Thereby Conclusion drawn by them were that PACs cause postponement of premature ovarian insufficiency (POI) via controlling the Sirt1-p53-p21 signaling pathway implicating FGSCs.
7. Conclusions
Perpetuating fertility continues to be a crucial concentration in medicine, enabling women to retain reproductive health. Stem cells in adult ovaries hold meaningful repercussions for reproductive medicine. Present fertility preservation modalities are inclusive of i) embryo cryopreservation, ii) oocyte cryopreservation, along with iii) ovarian tissue cryopreservation. Whereas embryo as well as oocyte freezing are well generated, ovarian tissue cryopreservation is still needing surgical elimination, freezing, in addition to followed by transplantation of thawed tissue continues to be in its early budding clinical stages.
Advancements in cancer therapy have resulted in improvement in survival rates, yet surgery, chemotherapy, along with radiotherapy usually results in robust injuries in reference to ovarian working. Adolescent or reproductive-age female patients achieving long-term survival subsequent to cancer remission or cure pose reduced or fertility omission. Internationally, ovarian tissue cryopreservation as well as transplantation (OTCT) is acknowledged in the form of the exclusive strategy regarding perpetuating ovarian working in addition to fertility in children, along with women who do not possess the capacity of postponement of cancer therapies. OTCT further represents the maximum efficacious as well as attractive strategy regarding fertility preservation. Decades subsequently, thawed autologous ovarian tissue possess the capacity of getting transplanted back, in reference to restoration of ovarian working in addition to fertility. Nevertheless, present studies have illustrated that double injures from cryopreservation along with ischemia results in robust elimination of ovarian reserve in addition to mitochondrial energy metabolism impairment in oocytes. In reference to tackling the concern, the plausible employment of FGSCs in this field yields extensive attractiveness in reference to reproductive medicine for scientific investigators. On one side, FGSCs are capable of differentiating directionally into working oocytes; Conversely, they might be co-cultured with other cells or biomaterials, plausibly making the formation of ovarian organoids. The ovary possesses the capability of enzymatically getting detached into i) identified cells or ii) primordial follicles. Subsequent to reagglomeration as well as transplantation into recipient animals, such cells regenerate working follicles possessing the capability of ovulation. At the time of ovarian transplantation as well as tissue healing, i) germ in addition to ii) granulosa cells might redistribute iii) amongst donor, along with iv) host primordial follicles. Noticeably, a) somatic in addition to b) germ cells identified from mouse along with c) rat ovaries possess the capacity of getting recombined in vitro in the context of reconstruction of follicles, which get subsequently transplanted into mice which had severe combined immunodeficient (SCID). Therefore generating innovative contrived ovarian models co-cultured with diverse ovarian cells to study working restoration technologies poses substantial vital botherations. Whereas germline stem cells like FGSCs are believed to be valuable regarding their plausibility in restoration of fertility, their restricted availability as well as absence of complete insights in oocyte transformation mechanistic modes work in the form of hurdles regarding therapeutic implementation. Furthermore, the tumorigenicity of stem cell treatments continues to be a botheration. Correspondingly, with spermatogonial stem cells (SSCs) in reference to male infertility therapies, FGSCs possess the retention of risk of malignant conversion. Collectively, FGSCs yield innovative approaches in reference to treatment of ovarian conditions in addition to advancement of scientific investigators work on transgenic animal or breeding of elite livestock. Despite challenges, the scientific community along with public continues to be enthusiastic regarding their plausibility to reform management of ovarian disease as well as infertility treatments.
References
- Touraine P, Chabbert-Buffet N, Plu-Bureau G, et al. Premature ovarian insufficiency. Nat Rev Dis Primers. 2024;10(1):63. doi:10.1038/s41572-024-00547-5
- Le DC, Ngo MHT, Kuo YC, et al. Secretome from estrogen-responding human placenta-derived mesenchymal stem cells rescues ovarian function and circadian rhythm in mice with cyclophosphamide-induced primary ovarian insufficiency. J Biomed Sci. 2024;31(1):95. doi:10.1186/s12929-024-01085-8
- Federici S, Rossetti R, Moleri S, et al. Primary ovarian insufficiency: update on clinical and genetic findings. Front Endocrinol (Lausanne). 2024;15:1464803. doi:10.3389/fendo.2024.1464803
- Hong W, Wang B, Zhu Y, et al. Female germline stem cells: aging and anti-aging. J Ovarian Res. 2022;15(1):79. doi:10.1186/s13048-022-01011-2
- Jiang M, Gao Y, Hou H, Guo J, Li W, Qin T, et al. Bone mineral density in patients with primary ovarian insufficiency: a systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2024;295:219-227. doi:10.1016/j.ejogrb.2024.02.013
- Theodorou A, Karagiannakis DS, Stefanaki K, et al. Female-specific risk factors for cardiovascular disease: an update. Hormones (Athens). 2024;23(4):637-653. doi:10.1007/s42000-024-00576-5
- Bareghamyan H, Chopikyan A, Petrosyan M, et al. Influence of ovarian cysts on ovarian reserve and fertility: a case-control study. Int J Gynaecol Obstet. 2024;165(2):424-430. doi:10.1002/ijgo.15284
- Shandley LM, Spencer JB, Kipling LM, et al. The risk of infertility after surgery for benign ovarian cysts. J Womens Health (Larchmt). 2023;32(5):574-582. doi:10.1089/jwh.2022.0385
- Pu X, Zhang L, Zhang P, et al. Human UC-MSC-derived exosomes facilitate ovarian renovation in rats with chemotherapy-induced premature ovarian insufficiency. Front Endocrinol (Lausanne). 2023;14:1205901. doi:10.3389/fendo.2023.1205901
- Kulvinder Kochar Kaur K, Allahbadia GN, Singh M. The mechanism by which chemotherapy with alkylating agents causes follicular activation: is there any further mode for the loss of primordial follicles? J Gynecol. 2023;6(3). doi:10.23880/oajg-16000222
- McGlacken-Byrne SM, Conway GS. Premature ovarian insufficiency. Best Pract Res Clin Obstet Gynaecol. 2022;81:98-110. doi:10.1016/j.bpobgyn.2021.09.011
- Lee HN, Chang EM. Primordial follicle activation as a new treatment for primary ovarian insufficiency. Clin Exp Reprod Med. 2019;46(2):43-49. doi:10.5533/cerm.2019.46.2.43
- Kulvinder Kochar Kaur K, Allahbadia GN, Singh M. Utilization of manipulation of signaling pathways accompanying generation of early/primordial follicles for enhancement of ART outcomes in POI and DOR patients: updated comprehensive review of in vitro activation with clinical experiences. J Med Adv Clin Case Rep. 2022;1:1-13.
- Blumenfeld Z, Evron A. Preserving fertility when choosing chemotherapy regimens: the role of gonadotropin-releasing hormone agonists. Expert Opin Pharmacother. 2015;16(7):1009-1020. doi:10.1517/14656566.2015.1031654
- Mendez M, Fabregues F, Ferreri J, et al. Biomechanical characteristics of the ovarian cortex in POI patients and functional outcomes after drug-free in vitro activation. J Assist Reprod Genet. 2022;39(8):1759-1767. doi:10.1007/s10815-022-02483-5
- Mintziori G, Veneti S, Kolibianakis EM, et al. Egg freezing and late motherhood. Maturitas. 2019;125:1-4. doi:10.1016/j.maturitas.2019.03.017
- Shelling A, Nasef NA. The role of lifestyle and dietary factors in the development of premature ovarian insufficiency. Antioxidants (Basel). 2023;12(8):1601. doi:10.3390/antiox12081601
- Borum K. Oogenesis in the mouse. Exp Cell Res. 1961;24(3):495-507. doi:10.1016/0014-4827(61)90449-9
- Green SH, Zuckerman S. Further observations on oocyte numbers in mature rhesus monkeys (Macaca mulatta). J Endocrinol. 1954;10(3):284-290. doi:10.1677/joe.0.0100284
- Horan CJ, Williams SA. Oocyte stem cells: fact or fantasy? Reproduction. 2017;154(1):R23-R35. doi:10.1530/REP-17-0008
- Peters H, Levy E, Crone M. Deoxyribonucleic acid synthesis in oocytes of mouse embryos. Nature. 1962;195(4844):915-916. doi:10.1038/195915a0
- Johnson J, Canning J, Kaneko T, et al. Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature. 2004;428(6979):145-150. doi:10.1038/nature02316
- Johnson J, Bagley J, Skaznik-Wikiel M, et al. Oocyte generation in adult mammalian ovaries by putative germ cells in bone marrow and peripheral blood. Cell. 2005;122(2):303-315. doi:10.1016/j.cell.2005.06.031
- Gosden RG. Germline stem cells in the postnatal ovary: is the ovary more like a testis? Hum Reprod Update. 2004;10(3):193-195. doi:10.1093/humupd/dmh023
- Faddy MJ, Gosden RG, Edwards RG. Ovarian follicle dynamics in mice: a comparative study of three inbred strains and an F1 hybrid. J Endocrinol. 1983;96(1):23-33. doi:10.1677/joe.0.0960023
- Wagner M, Yoshihara M, Douagi I, et al. Single-cell analysis of human ovarian cortex identifies distinct cell populations but no oogonial stem cells. Nat Commun. 2020;11(1):1147. doi:10.1038/s41467-020-14936-3
- Woods DC, Tilly JL. Revisiting claims of the continued absence of functional germline stem cells in adult ovaries. Stem Cells. 2023;41(2):200-204. doi:10.1093/stmcls/sxac083
- Bukovsky A, Gupta SK, Virant-Klun I, et al. Study of origin of germ cells and formation of new primary follicles in adult human and rat ovaries. In: Germline Stem Cells. Springer; 2008:233-265. doi:10.1007/978-1-60327-214-8_16
- Zou K, Yuan Z, Yang Z, et al. Production of offspring from a germline stem cell line derived from neonatal ovaries. Nat Cell Biol. 2009;11(5):631-636. doi:10.1038/ncb1869
- Lu Z, Wu M, Zhang J, et al. Improvement in isolation and identification of mouse oogonial stem cells. Stem Cells Int. 2016;2016:2749461. doi:10.1155/2016/2749461
- White YAR, Woods DC, Takai Y, et al. Oocyte formation by mitotically active germ cells purified from ovaries of reproductive-age women. Nat Med. 2012;18(3):413-421. doi:10.1038/nm.2669
- Zhang C, Wu J. Production of offspring from a germline stem cell line derived from prepubertal ovaries of germline reporter mice. Mol Hum Reprod. 2016;22(7):457-464. doi:10.1093/molehr/gaw030
- Alberico H, Fleischmann Z, Bobbitt T, et al. Workflow optimization for identification of female germline or oogonial stem cells in human ovarian cortex using single-cell RNA sequence analysis. Stem Cells. 2022;40(5):523-536. doi:10.1093/stmcls/sxac015
- Li X, Yao X, Mu C, et al. Serum- and feeder-free culture of juvenile monkey female germline stem cells and testosterone regulation of their self-renewal. Stem Cell Rev Rep. 2022;18(1):336-345. doi:10.1007/s12015-021-10278-9
- Martin JJ, Woods DC, Tilly JL. Implications and current limitations of oogenesis from female germline or oogonial stem cells in adult mammalian ovaries. Cells. 2019;8(2):93. doi:10.3390/cells8020093
- Meng L, Zhang Y, Hua Y, et al. Identification of oogonial stem cells in chicken ovary. Cell Prolif. 2023;56(3):e13371. doi:10.1111/cpr.13371
- Saber M, Tavakol P, Esfandiari F. Isolation of female germline stem cells from mouse and human ovaries by differential adhesion. Int J Cell Biol. 2022;2022:5224659. doi:10.1155/2022/5224659
- Huang Y, Ye H. Female germline stem cells: recent advances, opportunities, and challenges to overcome. Cell Regen. 2025;14:34. doi:10.1186/s13619-025-00256-8
- Xie W, Wang H, Wu J. Similar morphological and molecular signatures shared by female and male germline stem cells. Sci Rep. 2014;4:5580. doi:10.1038/srep05580
- Huang Y, Ye H, Zhu F, et al. The role of chito-oligosaccharide in regulating ovarian germ stem cells function and restoring ovarian function in chemotherapy mice. Reprod Biol Endocrinol. 2021;19(1):14. doi:10.1186/s12958-021-00699-z
- Wang J, Du H, Ma L, et al. MitoQ protects ovarian organoids against oxidative stress during oogenesis and folliculogenesis in vitro. Int J Mol Sci. 2023;24(2):924. doi:10.3390/ijms24020924
- Wang J, Fang J, Feng M, et al. Inhibition of EED activity enhances cell survival of female germline stem cells and improves oocyte production during oogenesis in vitro. Open Biol. 2023;13(1):220211. doi:10.1098/rsob.220211
- Zheng K, Hong W, Ye H, et al. Chito-oligosaccharides and macrophages have synergistic effects on improving ovarian stem cell function by regulating inflammatory factors. J Ovarian Res. 2023;16(1):76. doi:10.1186/s13048-023-01143-z
- Wang H, Jiang M, Bi H, et al. Conversion of female germline stem cells from neonatal and prepubertal mice into pluripotent stem cells. J Mol Cell Biol. 2014;6(2):164-171. doi:10.1093/jmcb/mju004
- Li S, Wang M, Chen Y, et al. Role of the hedgehog signaling pathway in regulating the behavior of germline stem cells. Stem Cells Int. 2017;2017:5714608. doi:10.1155/2017/5714608
- Lu W, Casanueva MO, Mahowald AP, et al. Niche-associated activation of Rac promotes the asymmetric division of Drosophila female germline stem cells. PLoS Biol. 2012;10(7):e1001357. doi:10.1371/journal.pbio.1001357
- Rojas-Rios P, Guerrero I, Gonzalez-Reyes A. Cytoneme-mediated delivery of hedgehog regulates the expression of bone morphogenetic proteins to maintain germline stem cells in Drosophila. PLoS Biol. 2012;10(4):e1001298. doi:10.1371/journal.pbio.1001298
- Inaba M, Yamashita YM, Buszczak M. Keeping stem cells under control: new insights into the mechanisms that limit niche-stem cell signaling within the reproductive system. Mol Reprod Dev. 2016;83(8):675-683. doi:10.1002/mrd.22682
- Xia L, Jia S, Huang S, et al. The fused/Smurf complex controls the fate of Drosophila germline stem cells by generating a gradient BMP response. Cell. 2010;143(6):978-990. doi:10.1016/j.cell.2010.11.022
- Xia L, Zheng X, Zheng W, et al. The niche-dependent feedback loop generates a BMP activity gradient to determine the germline stem cell fate. Curr Biol. 2012;22(6):515-521. doi:10.1016/j.cub.2012.01.056
- Hsu HJ, Bahader M, Lai CM. Molecular control of the female germline stem cell niche size in Drosophila. Cell Mol Life Sci. 2019;76(21):4309-4317. doi:10.1007/s00018-019-03223-0
- Ward EJ, Shcherbata HR, Reynolds SH, et al. Stem cells signal to the niche through the Notch pathway in the Drosophila ovary. Curr Biol. 2006;16(23):2352-2358. doi:10.1016/j.cub.2006.10.022
- Pan Z, Sun M, Li J, et al. Expression of markers related to ovarian germline stem cells in mouse ovarian surface epithelium and correlation with the Notch signaling pathway. Cell Physiol Biochem. 2015;37(6):2311-2322. doi:10.1159/000438586
- Song X, Call GB, Kirilly D, et al. Notch signaling controls germline stem cell niche formation in the Drosophila ovary. Development. 2007;134(6):1071-1080. doi:10.1242/dev.003392
- Fu M, Hu Y, Lan T, et al. The Hippo signalling pathway and its implications in human health and diseases. Signal Transduct Target Ther. 2024;9(1):5. doi:10.1038/s41392-023-01682-3
- Kwon Y, Vinayagam A, Sun X, et al. The Hippo signaling pathway interactome. Science. 2013;342(6159):737-740. doi:10.1126/science.1243971
- Yin F, Yu J, Zheng Y, et al. Spatial organization of Hippo signaling at the plasma membrane mediated by the tumor suppressor Merlin/NF2. Cell. 2013;154(6):1342-1355. doi:10.1016/j.cell.2013.08.025
- Pocaterra A, Romani P, Dupont S. YAP/TAZ functions and their regulation at a glance. J Cell Sci. 2020;133(2):jcs230425. doi:10.1242/jcs.230425
- Clark KL, George JW, Przygrodzka E, et al. Hippo signaling in the ovary: emerging roles in development, fertility, and disease. Endocr Rev. 2022;43(6):1074-1096. doi:10.1210/endrev/bnac013
- Li J, Zhou F, Zheng T, et al. Ovarian germline stem cells and the Hippo signaling pathway association with physiological and pathological ovarian aging in mice. Cell Physiol Biochem. 2015; 36(5):1712-1724. doi:10.1159/000430144
- Schofield R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells. 1978;4(1-2):7-25.
- Dooley D, Vidal P, Hendrix S. Immunopharmacological intervention for successful neural stem cell therapy: new perspectives in CNS neurogenesis and repair. Pharmacol Ther. 2014;141(1):21-31. doi:10.1016/j.pharmthera.2013.08.001
- Casanova-Acebes M, A-Gonzalez N, Weiss LA, et al. Innate immune cells as homeostatic regulators of the hematopoietic niche. Int J Hematol. 2014;99(6):685-694. doi:10.1007/s12185-014-1561-7
- Molinari E, Bar H, Pyle AM, et al. Transcriptome analysis of human cumulus cells reveals hypoxia as the main determinant of follicular senescence. Mol Hum Reprod. 2016;22(8):866-876. doi:10.1093/molehr/gaw038
- Kim J, Bagchi IC, Bagchi MK. Signaling by hypoxia-inducible factors is critical for ovulation in mice. Endocrinology. 2009;150(7):3392-3400. doi:10.1210/en.2008-0948
- McGarry T, Biniecka M, Veale DJ, et al. Hypoxia, oxidative stress and inflammation. Free Radic Biol Med. 2018;125:15-24. doi:10.1016/j.freeradbiomed.2018.03.042
- Atalay Ekiner S, Gęgotek A, Skrzydlewska E. Inflammasome activity regulation by PUFA metabolites. Front Immunol. 2024;15:1452749. doi:10.3389/fimmu.2024.1452749
- Tucker PS, Scanlan AT, Dalbo VJ. Chronic kidney disease influences multiple systems: describing the relationship between oxidative stress, inflammation, kidney damage, and concomitant disease. Oxid Med Cell Longev. 2015;2015:806358. doi:10.1155/2015/806358
- Zhang Y, Hu X, Zou LQ. Flavonoids as therapeutic agents for epilepsy: unveiling anti-inflammatory and antioxidant pathways for novel treatments. Front Pharmacol. 2024;15:1457284. doi:10.3389/fphar.2024.1457284
- Breuss JM, Atanasov AG, Uhrin P. Resveratrol and its effects on the vascular system. Int J Mol Sci. 2019;20(7):1523. doi:10.3390/ijms20071523
- Jiang Y, Zhang Z, Cha L, et al. Resveratrol plays a protective role against premature ovarian failure and promotes female germline stem cell survival. Int J Mol Sci. 2019;20(14):3605. doi:10.3390/ijms20143605
- Nadile M, Retsidou MI, Gioti K, et al. Resveratrol against cervical cancer: evidence from in vitro and in vivo studies. Nutrients. 2022;14(24):5273. doi:10.3390/nu14245273
- Pyo IS, Yun S, Yoon YE, et al. Mechanisms of aging and the preventive effects of resveratrol on age-related diseases. Molecules. 2020;25(20):4649. doi:10.3390/molecules25204649
- Ren B, Kwah MXY, Liu C, et al. Resveratrol for cancer therapy: challenges and future perspectives. Cancer Lett. 2021;515:63-72. doi:10.1016/j.canlet.2021.05.001
- Zhou DD, Luo M, Huang SY, et al. Effects and mechanisms of resveratrol on aging and age-related diseases. Oxid Med Cell Longev. 2021;2021:9932218. doi:10.1155/2021/9932218
- Huang Y, Ye H, Zhu F, et al. The role of chito-oligosaccharide in regulating ovarian germ stem cells function and restoring ovarian function in chemotherapy mice. Reprod Biol Endocrinol. 2021;19(1):14. doi:10.1186/s12958-021-00699-z
- Li X, Ye H, Su T, et al. Immunity- and reproduction-protective effects of chitosan oligosaccharides in a cyclophosphamide/busulfan-induced premature ovarian failure mouse model. Front Immunol. 2023;14:1185921. doi:10.3389/fimmu.2023.1185921
- Yuan X, Tian G, Pei X, et al. Spermidine induces cytoprotective autophagy of female germline stem cells in vitro and ameliorates aging caused by oxidative stress through upregulated sequestosome-1/p62 expression. Cell Biosci. 2021;11(1):107. doi:10.1186/s13578-021-00614-4
- Wang CH, Wang QQ, Su YS, et al. Metformin improves polycystic ovary syndrome and activates female germline stem cells in mice. Sheng Li Xue Bao. 2022;74(3):370-380. doi:10.13294/j.aps.2021.0090
- Li X, Yao X, Mu C, et al. Serum- and feeder-free culture of juvenile monkey female germline stem cells and testosterone regulation of their self-renewal. Stem Cell Rev Rep. 2022;18(1):336-345. doi:10.1007/s12015-021-10278-9
- Ding X, Liu G, Xu B, et al. Human GV oocytes generated by mitotically active germ cells obtained from follicular aspirates. Sci Rep. 2016;6:28218. doi:10.1038/srep28218
- Cheng H, Shang D, Zhou R. Germline stem cells in human. Signal Transduct Target Ther. 2022;7(1):345. doi:10.1038/s41392-022-01197-3
- Zou K, Yuan Z, Yang Z, et al. Production of offspring from a germline stem cell line derived from neonatal ovaries. Nat Cell Biol. 2009;11(5):631-636. doi:10.1038/ncb1869
- Luo H, Li X, Tian GG, et al. Offspring production of ovarian organoids derived from spermatogonial stem cells by defined factors with chromatin reorganization. J Adv Res. 2021;33:81-98. doi:10.1016/j.jare.2021.03.006
- Satirapod C, Wang N, MacDonald JA, et al. Estrogen regulation of germline stem cell differentiation as a mechanism contributing to female reproductive aging. Aging (Albany NY). 2020;12(8):7313-7333. doi:10.18632/aging.103080
- Xiong J, Lu Z, Wu M, et al. Intraovarian transplantation of female germline stem cells rescues ovarian function in chemotherapy-injured ovaries. PLoS One. 2015;10(10):e0139824. doi:10.1371/journal.pone.0139824
- Wu C, Xu B, Li X, et al. Tracing and characterizing the development of transplanted female germline stem cells in vivo. Mol Ther. 2017;25(6):1408-1419. doi:10.1016/j.ymthe.2017.04.019
- Bukovsky A, Presl J. Ovarian function and the immune system. Med Hypotheses. 1979;5(4):415-436. doi:10.1016/0306-9877(79)90108-7
- Goya RG, Reggiani PC, Vesenbeckh SM, et al. Thymulin gene therapy prevents the reduction in circulating gonadotropins induced by thymulin deficiency in mice. Am J Physiol Endocrinol Metab. 2007;293(1):E182-E187. doi:10.1152/ajpendo.00085.2007
- Bukovsky A, Caudle MR. Immunoregulation of follicular renewal, selection, premature ovarian failure, and menopause in vivo versus neo-oogenesis in vitro, premature ovarian failure and ovarian infertility treatment, and a clinical trial. Reprod Biol Endocrinol. 2012;10(1):97. doi:10.1186/1477-7827-10-97
- Ye H, Li X, Zheng T, et al. The effect of the immune system on ovarian function and features of ovarian germline stem cells. Springerplus. 2016;5(1):990. doi:10.1186/s40064-016-2390-3
- Li X, Zheng M, Xu B, et al. Generation of offspring-producing 3D ovarian organoids derived from female germline stem cells and their application in toxicological detection. Biomaterials. 2021;279:121213. doi:10.1016/j.biomaterials.2021.121213
- Pennarossa G, Ghiringhelli M, Gandolfi F, et al. Creation of a bioengineered ovary: isolation of female germline stem cells for the repopulation of a decellularized ovarian bioscaffold. In: Brevini TAL, Fazeli A, Turksen K, eds. Next Generation Culture Platforms for Reliable In Vitro Models. Springer; 2021:139-149.
- Luo Y, Yin M, Mu C, et al. Engineering female germline stem cells with exocytotic polymer dots. Adv Mater. 2023;35(24):2210458. doi:10.1002/adma.202210458
- Chon SJ, Umair Z, Yoon MS. Premature ovarian insufficiency: past, present, and future. Front Cell Dev Biol. 2021;9:672890. doi:10.3389/fcell.2021.672890
- Ishizuka B. Current understanding of the etiology, symptomatology, and treatment options in premature ovarian insufficiency. Front Endocrinol (Lausanne). 2021;12:626924. doi:10.3389/fendo.2021.626924
- Szeliga A, Calik-Ksepka A, Maciejewska-Jeske M, et al. Autoimmune diseases in patients with premature ovarian insufficiency our current state of knowledge. Int J Mol Sci. 2021;22(5):2594. doi:10.3390/ijms22052594
- Duffy C, Allen S. Medical and psychosocial aspects of fertility after cancer. Cancer J. 2009;15(1):27-33. doi:10.1097/PPO.0b013e3181976602
- Jadoul P, Dolmans MM, Donnez J. Fertility preservation in girls during childhood: is it feasible, efficient and safe and to whom should it be proposed? Hum Reprod Update. 2010;16(6):617-630. doi:10.1093/humupd/dmq010
- Park SJ, Han JY, Kim SW, et al. Current position of oncofertility in adolescent female cancer patients: a comparative review of society guidelines. In Vivo. 2024;38(1):48-57. doi:10.21873/invivo.13409
- Yan L, Wang L, Wu J, et al. Multi-biofunctional graphene oxide-enhanced poly-L-lactic acid composite nanofiber scaffolds for ovarian function recovery of transplanted tissue. NPJ Regen Med. 2022;7(1):52. doi:10.1038/s41536-022-00236-5
- Sadeghi S, Mosaffa N, Huang B, et al. Protective role of stem cells in premature ovarian insufficiency: current status and mechanism of action. Heliyon. 2024;10(1):e23271. doi:10.1016/j.heliyon.2023.e23271
- Wilmut I, Hooper ML, Simons JP. Genetic manipulation of mammals and its application in reproductive biology. J Reprod Fertil. 1991;92(2):245-279. doi:10.1530/jrf.0.0920245
- Zhang Y, Yang Z, Yang Y, et al. Production of transgenic mice by random recombination of targeted genes in female germline stem cells. J Mol Cell Biol. 2011;3(2):132-141. doi:10.1093/jmcb/mjq043
- Pursel VG, Pinkert CA, Miller KF, et al. Genetic engineering of livestock. Science. 1989;244(4910):1281-1288. doi:10.1126/science.2499927
- Brinster RL, Zimmermann JW. Spermatogenesis following male germ-cell transplantation. Proc Natl Acad Sci U S A. 1994;91(24):11298-11302. doi:10.1073/pnas.91.24.11298
- Nagano M, Brinster CJ, Orwig KE, et al. Transgenic mice produced by retroviral transduction of male germ-line stem cells. Proc Natl Acad Sci U S A. 2001;98(23):13090-13095. doi:10.1073/pnas.231473498
- Zhang W, Cheng Y, Zhang S, et al. Application of Matrig