Terlipressin for Hepatorenal Syndrome: Infusion vs. Bolus
Use of terlipressin in the treatment of hepatorenal syndrome: Intravenous infusion versus intravenous boluses; An Open Label, Pilot, Randomized Controlled Study
Pratap Sagar Tiwari1, Manoj Aryal2, Shital Bhandary3, Sudhamshu KC4
- Department of Hepatology, Nobel Medical College, Biratnagar, Nepal
- Department of Nephrology, National Academy of Medical Sciences, Bir Hospital, Nepal
- Public Health & Medical Education, Patan Academy of Health Sciences, Nepal
- Department of Hepatology, National Academy of Medical Sciences, Bir Hospital, Nepal
OPEN ACCESS
PUBLISHED 31 December 2025
CITATION Tiwari, PS., Aryal, M., et al., 2025. Use of terlipressin in the treatment of hepatorenal syndrome: Intravenous infusion versus intravenous boluses; An Open Label, Pilot, Randomized Controlled Study. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7147
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.7147
ISSN 2375-1924
ABSTRACT
Introduction: The purpose of this study was to evaluate the role of terlipressin in the management of hepatorenal syndrome, focusing on the efficacy of its infusion and bolus regimens.
Objectives: To determine the efficacy and safety of terlipressin intravenous infusion and bolus regimens in treating hepatorenal syndrome.
Materials & Methods: The study employed an open-label, randomized controlled trial design. A total of 56 patients with hepatorenal syndrome were randomly assigned to two groups. The infusion regimen of terlipressin was administered to the TERLI-I group, while the bolus regimen was given to the TERLI-B group. The drug response and its adverse effects were analyzed.
Result: Among the 56 patients, 83.9% responded to the treatment. In the TERLI-I group, 71.4% had a complete response, 14.2% had a partial response, and 14.2% were non-responders. In the TERLI-B group, 60.7% had a complete response, 21.4% had a partial response, and 17.8% were non-responders. Overall, the response to treatment (partial plus complete response) was observed in 85.7% of the TERLI-I group and 82.1% of the TERLI-B group. No significant treatment-related adverse events were observed.
Conclusion: Terlipressin remains the standard of care for the management of patients with hepatorenal syndrome. Both administration regimens were equally effective, with no significant adverse effects.
Keywords: Cirrhosis, Hepatorenal syndrome, Terlipressin.
INTRODUCTION
Hepatorenal syndrome (HRS) is a potentially reversible, volume-non-responsive, functional renal impairment that can occur in patients with advanced chronic liver disease, i.e., cirrhosis with ascites. HRS is classified into types 1 and 2. Type 1 HRS is characterized by rapid progressive renal failure, usually accompanied by multiorgan failure. Type 2 HRS manifests as a slowly progressive functional renal failure associated with refractory ascites. The 2-week mortality rate is as high as 80% in untreated Type 1 HRS patients, with only 10% of patients surviving for 3 months. The prognosis of Type 2 HRS patients is slightly better, with a median survival of 6 months. Overall, HRS carries a dismal prognosis, but in recent years, there has been a trend toward a slight improvement in prognosis with the advent of vasoconstrictors.
The peripheral arterial vasodilation theory is the most widely accepted explanation for the pathophysiology of HRS. The role of vasoconstrictors has been studied as a medical treatment or as a bridging therapy until definitive treatment with liver transplantation can be performed. Among the vasopressin analogs, terlipressin is the most widely studied drug. Terlipressin improves renal perfusion and glomerular filtration in patients with HRS by inducing vasoconstriction of the splanchnic circulation. Several studies have shown that terlipressin is an effective drug in the medical treatment of HRS. Thus, it is recommended as the first-line drug in combination with plasma expansion with albumin by the Acute Dialysis Quality Initiative (ADQI) work group. However, terlipressin is an expensive drug; its high cost makes it less practical to use when used for prolonged durations. Alternative agents such as noradrenaline are widely available and relatively inexpensive, but there are fewer studies supporting their efficacy. Moreover, most studies have used terlipressin in a bolus regimen and have reported adverse events in up to 22%. However, a randomized study by Angeli P showed that terlipressin, when given by continuous intravenous infusion rather than intravenous bolus, required a lower dosage and yet showed equal efficacy and is better tolerated. However, the intravenous terlipressin regimen in HRS requires further validation. Thus, this study aimed to compare the efficacy of terlipressin when used as a continuous intravenous infusion versus intravenous boluses for treating HRS.
METHODOLOGY
This was a hospital-based, open-label, pilot, randomized controlled study of the Hepatology unit, Nobel Medical College, from September 2023 to March 2024.
Patients: All consecutive patients with decompensated chronic liver disease (CLD) with acute kidney injury (AKI) admitted to the Hepatology unit, Nobel Medical College, were included in the study. Chronic liver disease was diagnosed on clinical grounds, including laboratory tests, endoscopic evidence, sonographic findings, and liver histology, if available. The study included patients diagnosed with HRS. The diagnosis of HRS was determined using the criteria proposed by the International Ascites Club (IAC) as shown in Table 1.
| Diagnostic criteria for HRS |
|---|
| 1. CLD with Ascites |
| 2. Diagnosis of AKI according to the IAC AKI Criteria |
| 3. No improvement of serum creatinine concentration (decrease to ≤1.5 mg/dl) after at least 2 days of diuretic withdrawal and volume expansion with albumin at 20 g/day |
| 4. Absence of shock |
| 5. No current or recent treatment with nephrotoxic drugs |
| 6. Absence of parenchymal kidney disease as indicated by proteinuria with a protein concentration >500 mg/day, microhematuria with >50 erythrocytes per high-power field, and/or abnormal renal findings on ultrasonography. |
Definition of AKI: Serum creatinine (SCr) concentration increase ≥0.3 mg/dl in ≤ 48 hours, from the baseline. Baseline SCr concentration: Stable SCr concentration for ≤ 3 months. If not available, a stable SCr concentration closest to the current one. If there is no previous SCr concentration measurement, use the admission SCr concentration.
The exclusion criteria included:
- a. Improvement in renal function after albumin infusion for 2 days
- b. History of coronary artery disease, ventricular arrhythmia, obliterative arterial disease of the limbs, or other severe extrahepatic disease.
- c. Septic shock
- d. Contraindications to the use of terlipressin
- e. CLD with herbal-induced liver injury
- f. Acute on chronic liver failure (ACLF)
The study was an open-label, randomized controlled trial, approved by the institutional ethics committee (IRC/NMCTH/492/2021). Written informed consent was obtained before inclusion in the study.
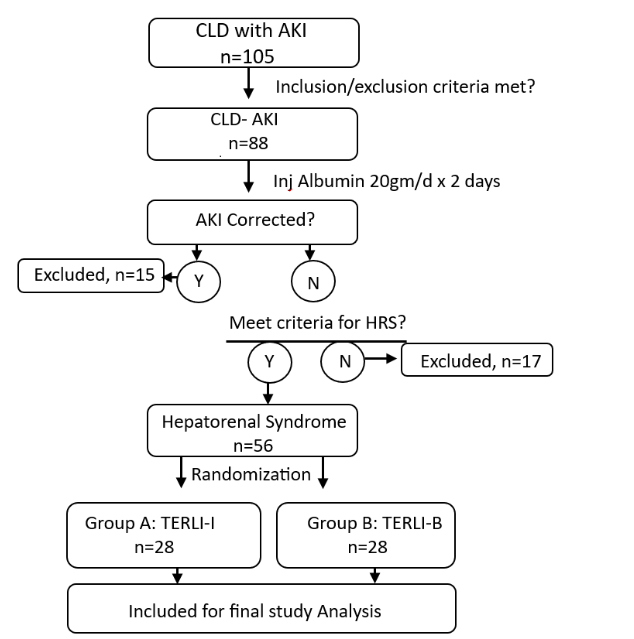
Treatment Protocols: All consecutive patients diagnosed with CLD fulfilling the AKI criteria received initial resuscitation and supportive care as needed. Diuretics and beta blockers were stopped. Before randomization into the two groups, each patient received a first-line treatment for 48 h consisting of an intravenous albumin infusion (20 g). After 48 h, the patient’s AKI status was reassessed. If creatinine did not decrease, they were included in the randomization group. Patients were randomized into two treatment groups, A and B, using the coin flip method. Patients in Group A received an infusion regimen of terlipressin (TERLI-I), whereas those in Group B received a bolus regimen (TERLI-B). Diagram 1 explains the flow chart, below.
For TERLI-I, terlipressin (starting at 2 mg) was dissolved in 50 mL of 5% dextrose solution and infused over 24 h using a pump. For TERLI-B, the standard practice recommendation was used: terlipressin (starting at 0.5 mg) given as an intravenous bolus every 6 h. In both groups, terlipressin was continued until a complete response occurred or discontinued if any adverse events occurred. The treatment response (defined below) was assessed every 3rd day. For partial responders or non-responders, the dose was doubled and reassessed on the 3rd day. The maximum dose of terlipressin was 12 mg/day, and the maximum treatment duration was 15 days.
Both groups received daily intravenous albumin (20 g/day) along with terlipressin. Blood samples were taken before the initiation of therapy and at days 1, 3, 5, 7, and 15 of treatment to measure standard liver and renal function tests, and blood pressure and weight were recorded. In patients with tense ascites, paracentesis was performed.
Stages of Acute kidney injury:
- Stage 1: SCr concentration increase ≥0.3 mg/dL up to 2 times the baseline level.
- Stage 2: SCr concentration >2-3 times the baseline level.
- Stage 3: SCr concentration >3 times the baseline level or SCr concentration ≥4.0 mg/dL or initiation of renal replacement therapy.
Progression: Progression of AKI to a higher stage or the need for renal replacement therapy.
Regression: Regression of AKI to a lower stage.
Definitions of Response:
- NO RESPONSE: No regression of AKI despite complete treatment protocol
- PARTIAL RESPONSE: Regression of the AKI stage with a decrease in SCr concentration, but to a value ≥0.3 mg/dl above the baseline.
- COMPLETE RESPONSE (REVERSAL OF HRS): decrease in the SCr concentration to <0.3 mg/dl from the baseline.
End Points
PRIMARY END POINT: Complete response (i.e., reversal of HRS).
Statistical Analyses: The sample size was 56, which is consistent with the pilot trial sample sizes reported by Julious and Kieser and Wassmer. A proforma was used to collect data from the patients enrolled in the study. The data from the proforma were entered into Microsoft Excel. After coding, the data were then exported to the Statistical Package for Social Sciences (SPSS) version 25 software for analysis. A p-value < 0.05 was considered significant. Descriptive analysis consisted of presenting the continuous data in mean and standard deviation, while frequency and percentages were reported for categorical data. The results were analyzed at baseline, day 3, day 5, the end of treatment, and day 30 of the study. We enrolled 56 patients, 28 in each treatment group. The characteristics of the responders and non-responders were analyzed regardless of the treatment regimen. Univariate and multivariate analyses were performed to determine the baseline predictors of response.
RESULTS
One hundred and five patients presented with a diagnosis of CLD and AKI. Fifty-six patients met the criteria of hepatorenal syndrome and were thus included in the study after randomization. Twenty-eight patients were included in each group: Group A (TERLI-I) and Group B (TERLI-B). There were no significant differences between the two groups in the clinical and laboratory data at the time of randomization.
| Features | TERLI-I Group A: n=28 | TERLI-B Group B: n=28 | P value |
|---|---|---|---|
| Age (years) | 51.00±6.76 | 51.71±9.07 | 0.740 (NS) |
| Sex (m/f) | 22/6 | 19/9 | 0.820 (NS) |
| Etiology, Alcohol/not Alcohol | 25/3 | 23/5 | 0.705 (NS) |
| MAP, mm Hg | 84.14±11.30 | 82.54±13.47 | 0.630 (NS) |
| White blood cell, /ul | 7776.75±4606.42 | 9028.57±4577.96 | 0.312 (NS) |
| Serum Na, mmol/L | 125.68±3.47 | 126.79±3.08 | 0.212 (NS) |
| Serum Creatinine, mg/dl | 2.61±0.92 | 2.95±1.25 | 0.252 (NS) |
| Serum total bilirubin, mg/dl | 1.26±1.16 | 1.65±1.42 | 0.263 (NS) |
| AST, U/L | 93.18±44.11 | 74.21±53.64 | 0.154 (NS) |
| ALT, U/L | 69.29±34.29 | 61.18±39.19 | 0.413 (NS) |
| Albumin, g/L | 2.49±0.34 | 2.30±0.45 | 0.080 (NS) |
| INR | 1.66±0.22 | 1.63±0.34 | 0.612 (NS) |
| CTP score | 9.46±1.04 | 9.68±1.34 | 0.505 (NS) |
| MELD score | 21.43±4.83 | 22.36±5.34 | 0.501 (NS) |
| MELD-Na score | 27.54±3.51 | 27.79±4.20 | 0.810 (NS) |
Abbreviations: MAP, mean arterial pressure; AST, Aspartate transaminase; ALT, Alanine transaminase; INR, international normalized ratio; CTP, Child-Turcotte-Pugh; MELD, Model for End-Stage Liver Disease; MELD-Na, MELD including sodium; NS, not significant; TERLI-BOL, terlipressin by intravenous boluses; TERLI-INF, terlipressin by intravenous infusion.
Response to treatment
Of the 56 patients, 47 (83.9%) responded to treatment. In the TERLI-I group, 20 patients (71.4%) had a complete response, four patients (14.2%) had a partial response, and four patients (14.2%) were non-responders. In the TERLI-B group, 17 patients (60.7%) had a complete response, six patients (21.4%) had a partial response, and five patients (17.8%) were non-responders. Overall, the response to treatment (partial plus complete response) was seen in 85.7% of the TERLI-I group and 82.1% of the TERLI-B group. Note: For partial responders or non-responders, the dosing of terlipressin was doubled and further reassessed on the next 3rd day.
Among the responders, the mean daily dose of terlipressin was 2.43±0.63 mg and 2.35±0.57 mg in Groups A and B, respectively, and the duration of treatment was 6.14±1.48 and 5.79±1.37 days, but no significant difference was observed regarding the dose and length of treatment. Similarly, no significant differences were observed in the mean increase in mean arterial pressure or in the mean reduction in serum creatinine between the two groups.
| Features | TERLI-I Group A: n=28 | TERLI-B Group B: n=28 | P value |
|---|---|---|---|
| Duration of treatment (days) | 6.14±1.48 | 5.79±1.37 | 0.354 (NS) |
| Mean daily dose of terlipressin (mg) | 2.43±0.63 | 2.35±0.57 | 0.610 (NS) |
| SCr, at end of treatment (mg/dl) | 1.63±1.08 | 1.93±1.39 | 0.365 (NS) |
| Delta MAP, mm Hg | 89.07±11.75 | 85.46±13.78 | 0.297 (NS) |
| Delta CP-score | 0.214±0.63 | 0.429±0.63 | 0.210 (NS) |
| Delta-MELD | 5.18±4.20 | 5.46±3.90 | 0.793 (NS) |
| Delta Meld-Na | 3.93±3.43 | 4.14±3.23 | 0.811 (NS) |
| Delta MAP Day 3 of treatment versus baseline | -2.5±3.40 | -2.82±6.03 | 0.807 (NS) |
| End of treatment versus baseline | -4.93±4.03 | -2.79±6.41 | 0.140 (NS) |
Abbreviations: CP: Child Pugh score; MAP: mean arterial pressure; MELD: Model for End-Stage Liver Disease; MELD-Na: MELD including sodium
Adverse Events
Infection was present in 16 (28.5%) patients in the form of spontaneous bacterial peritonitis (SBP) and urinary tract infection. All patients received intravenous cefotaxime (2 g) three times a day. A total of 3 patients (10.7%) in the TERLI-I group and 5 patients (17.8%) in the TERLI-B group developed diarrhea, although it was not statistically significant. No other treatment-related adverse events were observed.
Among the five patients who developed diarrhea in the Terli-B group, one patient achieved a complete response on Day 3; thus, terlipressin was stopped. In the other 4 patients, terlipressin was continued for another 24 h. Among the 4 patients, 1 had a partial response, and 3 showed no response and were thus discontinued. In the Terli-I group, three patients had diarrhea; however, a complete response was observed in 1 patient on Day 3. Terlipressin was continued to observe the tolerance, and 2 patients tolerated well until Day 7; however, 1 patient had a partial response, and the third was a non-responder and thus stopped.
Predictors of Response
Baseline mean arterial pressure, total bilirubin, serum albumin, and CP, MELD & MELD-Na scores were significantly associated with the response.
| Univariate Analysis | Responders | Non-responders | P value |
|---|---|---|---|
| Treatment: bolus/infusion | 23/28, 24/28 | 5/28, 4/24 | 1.00 |
| Infection (yes/no) | 13/16, 34/40 | 3/16, 6/40 | 0.705 |
| Baseline MAP, mm Hg | 86.09±11.51 | 69.00±3.08 | <0.001 |
| Baseline white blood cell count | 7518.06±3400.62 | 13022.22±7070.32 | 0.001 |
| Baseline serum creatinine, mg/dl | 2.623±0.91 | 3.58±1.63 | 0.016 |
| Baseline total serum bilirubin, mg/dl | 1.034±0.47 | 3.67±1.96 | <0.001 |
| Baseline international normalized ratio | 1.60±0.28 | 1.89±0.20 | 0.004 |
| Baseline albumin, g/L | 2.48±0.37 | 1.98±0.32 | <0.001 |
| Baseline serum Na, mmol/L | 126.38±3.28 | 125.44±3.47 | 0.439 |
| Baseline CP-Score | 9.24±0.88 | 11.44±0.73 | <0.001 |
| Baseline MELD | 20.66±4.27 | 28.33±4.27 | <0.001 |
| Baseline MELD-Na | 26.79±3.39 | 32.22±2.73 | <0.001 |
Multivariate Analysis
OR 95% CI p
Baseline serum creatinine, mg/dl 0.163 0.031 – 0.865 0.033
DISCUSSION
Renal dysfunction is not uncommon in chronic liver disease. Almost 50% of CLD patients with ascites develop AKI during their illness. In the study by Garcia et al., 19% of hospitalized patients with CLD had AKI, and among those, 17% had HRS. Hepatorenal syndrome has a grave prognosis. Gines and Arroyo have previously reported a 2-week mortality rate as high as 80% in untreated T1 HRS patients, with only 10% of patients surviving for 3 months. In recent years, however, there has been a trend toward a slight improvement in HRS prognosis. For example, in a multicenter study by Salerno et al., the 3-month survival was 20% and 40% for T1 and T2 HRS, respectively.
Based on all studies to date, terlipressin has been the cornerstone therapy for HRS. Terlipressin causes splanchnic vasoconstriction, diverting the blood to the systemic circulation, lowering the sympathetic nervous system and Renin-Angiotensin-Aldosterone System activation, decreasing the production of arginine vasopressin, and eventually leading to improved renal perfusion.
Terlipressin has been studied in several randomized controlled trials (RCT) in patients with HRS comparing its bolus doses plus albumin versus either albumin alone or placebo with or without albumin. The studies showed that terlipressin was able to significantly improve renal function in 24-80% of patients. Overall, terlipressin is considered effective in 40-50% of cases. In a meta-analysis of terlipressin trials, there was an overall 29% reduction in mortality.
Terlipressin is used for HRS in many parts of the world and is included in the Clinical Practice Guidelines in Europe. However, it was not approved in the US until September 2022 owing to its adverse effects. Later, following the results of 3 major studies OT-0401, REVERSE, and CONFIRM, the drug became the first FDA-approved medication for the treatment of HRS, but with a box warning due to the safety issues over respiratory failure.
Terlipressin can be administered both as intravenous boluses. The continuous infusion regime was shown to be associated with a significantly lower incidence of adverse effects such as pain in the abdomen, persistent diarrhea, peripheral ischemia, cardiovascular abnormalities, and circulatory overload. These results could be due to the short-term effect (3–4 h) of terlipressin on the portal pressure.
Traditionally, terlipressin is administered as a slow IV bolus injection in the hospital setting, although the 2021 American Association for the Study of Liver Diseases (AASLD) guideline has recommended administering it either as an IV bolus or continuous IV infusion. There are fewer studies of increased interest in using terlipressin as a 24-h infusion. Furthermore, there are no adequate data on the stability of terlipressin in infusor devices suitable for 24 h continuous infusion. In the only study by Bui et al., it was reported that terlipressin was physically and chemically stable when used by the infusion method and all reconstituted infusor concentrations retained above 90% of the original concentration over the test conditions. In our study, we used a syringe pump as an infusor device to give terlipressin by continuous infusion.
LIMITATIONS OF THE STUDY
As this study was a pilot project, the number of patients was small. The upcoming study, with a larger patient enrollment, will reveal the clearer efficacy and adverse effects of the drug.
CONCLUSION
Terlipressin remains the standard of care for the management of patients with hepatorenal syndrome. Both administration regimens demonstrated equal efficacy, with no significant adverse effects. However, the cautious use of terlipressin is recommended in patients with evidence of volume overload.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: Authors want to extend sincere gratitude to the resident doctors of the Department of Medicine at the Nobel Medical College for their support.
Authors List and ORCID ID of each author:
Pratap Sagar Tiwari: https://orcid.org/0000-0003-4084-5879
Manoj Aryal: https://orcid.org/0009-0002-1323-3615
Shital Bhandary: https://orcid.org/0000-0002-7483-3043
Sudhamshu KC: https://orcid.org/0000-0001-5493-9521
REFERENCES
- Arroyo V, Ginès P, Gerbes AL, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. Hepatology. 1996;23(1):164-176. doi:10.1002/hep.510230122
- Ginès A, Escorsell A, Ginès P, et al. Incidence, predictive factors, and prognosis of the hepatorenal syndrome in cirrhosis with ascites. Gastroenterology. 1993;105(1):229-236. doi:10.1016/0016-5085(93)90031-7
- Schrier RW, Arroyo V, Bernardi M, Epstein M, Henriksen JH, Rodés J. Peripheral arterial vasodilation hypothesis: A proposal for the initiation of renal sodium and water retention in cirrhosis. Hepatology. 1988;8(5):1151-1157. doi:10.1002/hep.1840080532
- Saner FH, Canbay A, Gerken G, Broelsch CE. Pharmacology, clinical efficacy and safety of terlipressin in esophageal varices bleeding, septic shock and hepatorenal syndrome. Expert Rev Gastroenterol Hepatol. 2007;1(2):207-217. doi:10.1586/17474124.1.2.207
- Sagi SV, Mittal S, Kasturi KS, Sood GK. Terlipressin therapy for reversal of type 1 hepatorenal syndrome: A meta-analysis of randomized controlled trials. J Gastroenterol Hepatol. 2010;25(5):880-885. doi:10.1111/j.1440-1746.2009.06132.x
- Nadim MK, Kellum JA, Forni L, et al. Acute kidney injury in patients with cirrhosis: Acute Disease Quality Initiative (ADQI) and International Club of Ascites (ICA) joint multidisciplinary consensus meeting. J Hepatol. 2024;81(1):163-183. doi:10.1016/j.jhep.2024.03.031
- Martín–Llahí M, Pépin M, Guevara M, et al. Terlipressin and Albumin vs Albumin in Patients With Cirrhosis and Hepatorenal Syndrome: A Randomized Study. Gastroenterology. 2008;134(5):1352-1359. doi:10.1053/j.gastro.2008.02.024
- Sanyal AJ, Boyer T, Garcia–Tsao G, et al. A Randomized, Prospective, Double-Blind, Placebo-Controlled Trial of Terlipressin for Type 1 Hepatorenal Syndrome. Gastroenterology. 2008;134(5):1360-1368. doi:10.1053/j.gastro.2008.02.014
- Angeli P, Gines P, Wong F, et al. Diagnosis and management of acute kidney injury in patients with cirrhosis: revised consensus recommendations of the International Club of Ascites. Gut. 2015;64(4):531-537. doi:10.1136/gutjnl-2014-308874
- Julious SA. Sample size of 12 per group rule of thumb for a pilot study. Pharm Stat. 2005;4(4):287-291. doi:10.1002/pst.185
- Kieser M, Wassmer G. On the Use of the Upper Confidence Limit for the Variance from a Pilot Sample for Sample Size Determination. Biometrical J. 1996;38(8):941-949. doi:10.1002/bimj.4710380806
- Garcia-Tsao G, Parikh CR, Viola A. Acute kidney injury in cirrhosis. Hepatology. 2008;48(6):2064-2077. doi:10.1002/hep.22605
- Montoliu S, Ballesté B, Planas R, et al. Incidence and Prognosis of Different Types of Functional Renal Failure in Cirrhotic Patients With Ascites. Clinical Gastroenterology and Hepatology. 2010;8(7):616-622. doi:10.1016/j.cgh.2010.03.029
- Salerno F, Cazzaniga M, Merli M, et al. Diagnosis, treatment and survival of patients with hepatorenal syndrome: A survey on daily medical practice. J Hepatol. 2011;55(6):1241-1248. doi:10.1016/j.jhep.2011.03.012
- Belcher JM, Parada XV, Simonetto DA, et al. Terlipressin and the Treatment of Hepatorenal Syndrome: How the CONFIRM Trial Moves the Story Forward. American Journal of Kidney Diseases. 2022;79(5):737-745. doi:10.1053/j.ajkd.2021.08.016
- Papaluca T, Gow P. Terlipressin: Current and emerging indications in chronic liver disease. J Gastroenterol Hepatol. 2018;33(3):591-598. doi:10.1111/jgh.14009
- Solà E, Guevara M, Ginès P. Current treatment strategies for hepatorenal syndrome. Clin Liver Dis (Hoboken). 2013;2(3):136-139. doi:10.1002/cld.209
- Mattos A, Mattos A, Ribeiro R. Terlipressin Versus Noradrenaline In The Treatment Of Hepatorenal Syndrome – Systematic Review With Meta-Analysis And Full Economic Evaluation. Eur J Gastroenterol Hepatol. 2016;28(3):345-351. doi:10.1016/j.jval.2015.09.522
- Wong F, Pantea L, Sniderman K. Midodrine, octreotide, albumin, and TIPS in selected patients with cirrhosis and type 1 hepatorenal syndrome. Hepatology. 2004;40(1):55-64. doi:10.1002/hep.20262
- Dundar HZ. Management of hepatorenal syndrome. World J Nephrol. 2015;4(2):277. doi:10.5527/wjn.v4.i2.277
- Neri S, Pulvirenti D, Malaguarnera M, et al. Terlipressin and Albumin in Patients with Cirrhosis and Type I Hepatorenal Syndrome. Dig Dis Sci. 2008;53(3):830-835. doi:10.1007/s10620-007-9919-9
- Boyer TD, Sanyal AJ, Wong F, Frederick R, Lake JR, Jamil K. Terlipressin plus albumin is more effective than albumin alone in Improving renal function in patients with cirrhosis and hepatorenal syndrome type 1. Gastroenterology. 2016;150(7):1579-1589. doi:10.1053/j.gastro.2016.02.026
- Solanki P, Chawla A, Garg R, Gupta R, Jain M, Sarin SK. Beneficial effects of terlipressin in hepatorenal syndrome: a prospective, randomized placebo-controlled clinical trial. J Gastroenterol Hepatol. 2003;18(2):152-156. doi:10.1046/j.1440-1746.2003.02934
- Boyer TD, Sanyal AJ, Pappas SC, Wong F, Jamil K. Percentage change in serum creatinine (SCr) is a sensitive indicator of therapeutic response to terlipressin in hepatorenal syndrome type 1 (HRS-1). J Hepatol. 2015;62(2). doi:10.1016/S0168-8278(15)30417-7
- European Association for the Study of the Liver. EASL clinical practice guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69(2):406-460. doi:10.1016/j.jhep.2018.03.024
- Sanyal AJ, Boyer TD, Frederick RT, et al. Reversal of hepatorenal syndrome type 1 with terlipressin plus albumin vs. placebo plus albumin in a pooled analysis of the OT-0401 and REVERSE randomised clinical studies. Aliment Pharmacol Ther. 2017;45(11):1390-1402. doi:10.1111/apt.14052
- Wong F, Pappas SC, Curry MP, et al. Terlipressin plus Albumin for the Treatment of Type 1 Hepatorenal Syndrome. N Engl J Med. 2021;384(9):818-828. doi:10.1056/nejmoa2008290
- Angeli P, Volpin R, Gerunda G, et al. Reversal of Type 1 Hepatorenal Syndrome With the Administration of Midodrine and Octreotide. Hepatology. 1999;29(6):1690-1697. doi:10.1002/hep.510290629
- Escorsell À, Bandi JC, Moitinho E, et al. Time profile of the haemodynamic effects of terlipressin in portal hypertension. J Hepatol. 1997;26(3):621-627. doi:10.1016/s0168-8278(97)80428-x
- Biggins SW, Angeli P, Garcia‐Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74(2):1014-1048. doi:10.1002/hep.31884
- Bui T, Sandar S, Luna G, Beaman J, Sunderland B, Czarniak P. An investigation of reconstituted terlipressin infusion stability for use in hepatorenal syndrome. Scientific Reports. 2020;10(1). doi:10.1038/s41598-020-78044-4
- Cavallin M, Piano S, Romano A, et al. Terlipressin given by continuous intravenous infusion versus intravenous boluses in the treatment of hepatorenal syndrome: A randomized controlled study. Hepatology. 2016;63(3):983-992. doi:10.1002/hep.28396
- Gerbes AL, Huber E, Gülberg V. Terlipressin for hepatorenal syndrome: continuous infusion as an alternative to iv bolus administration. Gastroenterology. 2009;137(3):1179. doi:10.1053/j.gastro.2009.03.064
- Mukhtar A, Salah M, Aboulfetouh F, et al. The use of terlipressin during living donor liver transplantation: Effects on systemic and splanchnic hemodynamics and renal function. Crit Care Med. 2011;39(6):1329-1334. doi:10.1097/ccm.0b013e3182120842
- Ding C, Wu X, Fan X, He C, Li J. Hemodynamic effects of continuous versus bolus infusion of terlipressin for portal hypertension: A randomized comparison. J Gastroenterol Hepatol. 2013;28(7):1242-1246. doi:10.1111/jgh.12195
- Guevara M, Terra C, Nazar A, et al. Albumin for bacterial infections other than spontaneous bacterial peritonitis in cirrhosis. A randomized, controlled study. J Hepatol. 2012;57(4):759-765. doi:10.1016/j.jhep.2012.06.013
- Thévenot T, Bureau C, Oberti F, et al. Effect of albumin in cirrhotic patients with infection other than spontaneous bacterial peritonitis. A randomized trial. J Hepatol. 2015;62(4):822-830. doi:10.1016/j.jhep.2014.11.017
- Fernández J, Angeli P, Trebicka J, et al. Efficacy of Albumin Treatment for Patients with Cirrhosis and Infections Unrelated to Spontaneous Bacterial Peritonitis. Clinical Gastroenterology and Hepatology. 2020;18(4):963-973.e14. doi:10.1016/j.cgh.2019.07.055
- Moore K, Jamil K, Verleger K, et al. Real-world treatment patterns and outcomes using terlipressin in 203 patients with the hepatorenal syndrome. Aliment Pharmacol Ther. 2020;52(2):351-358. doi:10.1111/apt.15836

