Therapeutic Implications of Interferon tau in Medicine
Therapeutic implications of interferon tau in medicine
- Center for Research on Infectious Diseases. National Institute of Public Health. Av. Universidad 655, Cuernavaca, Morelos. México, 62100
- Faculty of Medicine, Autonomous University of the State of Mexico. Instituto Literario 100. Toluca Estado de México. CP: 50000
OPEN ACCESS
PUBLISHED:30 November 2024
CITATION:Torres, E.I.H., Peralta, L.D., et al., 2024. Therapeutic implications of interferon tau in medicine. Medical Research Archives, [online] 12(11).
https://doi.org/10.18103/mra.v12.i11.6101
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12.i11.6101
ISSN 2375-1924
Abstract
Interferon tau has been described as a cytokine associated with the pregnancy recognition signal in ruminants. It is structurally and functionally related to type I interferons and binds to the same receptor, but unlike other interferons, it is not inducible by viruses. Interferon tau is less toxic in vitro and in vivo than interferon alpha and Interferon beta even at high concentrations and displays high species cross-reactivity. This review describes the properties of Interferon tau and its applications in health. We emphasize the antiviral, antiproliferative and anti-inflammatory properties of interferon tau, as well as its effects in mitigating diabetes, obesity, autoimmune diseases, and anti-cancer. Therefore, interferon tau has great potential as a promising therapeutic agent with low cytotoxicity particularly for the treatment of infections and chronic diseases, cancer, and autoimmune disorders with application in health.
Keywords
Interferon tau, antiviral properties, inflammation, autoimmune diseases, therapeutic implications
Introduction
Interferon tau (IFN-τ), a type I IFN first discovered for its role in the animal reproductive cycle, is produced by the trophectoderm of ruminants and is associated with the pregnancy recognition signal¹. IFN-τ belongs to the type I interferon family, binds to type 1 interferon receptor (IFNAR),¹ and affects the activation of the JAK/STAT pathway, resulting in the induction of interferon-stimulated genes (ISGs). IFN-τ shares 50% and 70% homology with IFN-α and IFN-ω, respectively but it differs from other type interferons (IFNs) in that it is not inducible by viral infection, unlike IFN-α and IFN-β². In addition, IFN-τ has demonstrated antiviral properties against human and animal viral both in vitro and in vivo, similar to type I IFNs³. It also has broad cross-species reactivity and exhibits 30-fold less cytotoxicity, even at high concentrations both in vitro and in living animals⁴. Likewise, IFN-τ has antiproliferative and immunomodulatory activities ascribed to the other type I IFNs. Furthermore, IFN-τ has been described as having therapeutic implications in experimental encephalomyelitis (EAE), and multiple sclerosis (MS)⁵, and it has implications on modulating the intestinal microbiota, which suggests that the IFN-τ has potential application in the treatment of diseases, such as: cancer, viral infections, MS, intestinal microbiota, etc. Therefore, this review highlights the therapeutic properties and benefits of IFN-τ that hold the potential for health.
Structure and function of IFN-τ
IFN-τ like type I interferon, consists of 172 amino acids in length with two disulfide bridges (1-99, 29-139) and an amino-terminal proline. Its molecular weight ranges between 19 and 24 kDa depending on its glycosylation state. The structure includes five alpha-helices (A–E) separated by loop regions, with a short helix in the middle of the loop connecting helices C and D and a long loop between helices A and B⁶⁷. The carboxyl-terminal region is linked to the receptor binding site, while the biologically active site is located at the amino-terminal⁸. IFN-τ binds to the IFNAR, which consists of two subunits IFNAR1 and IFNAR2⁹, and in the endometrial luminal epithelium of ruminants this binding triggers the activation of both the canonical and non-canonical signaling pathway. These biological events result in the activation of the Janus Kinase-signal transducer and activator of transcription (JAK/STAT) pathway and the interferon regulator factor (IRF) leading to the induction of classical ISGs, in the canonical pathway. On the other hand, in noncanonical pathway involves mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3k) cascades¹⁰¹¹. Additionally, in murine epithelial cells transformed with human papillomavirus (BMK-16/myc), IFN-τ activates both canonical and non-canonical pathways inducing MHC class I, MX and IP10, and is associated with antiviral, antiproliferative and immunoregulatory effects¹².
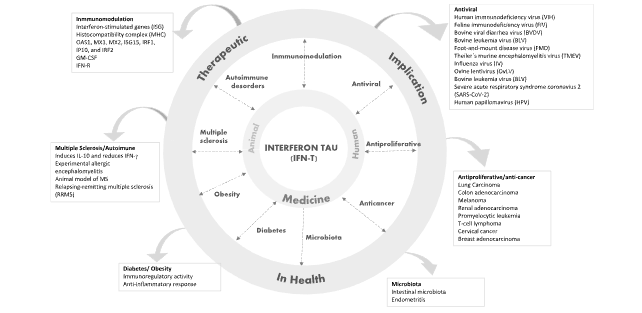
Figure 1: Therapeutic implications of interferon tau in medicine
Antiviral effect of IFN-τ
Unlike type I interferons (IFN-α/β) which are secreted by cells in response to viral infection, IFN-τ is not inducible by viral infection, but still maintains its antiviral properties. IFNs are the effector cytokines of the innate immune response and are expressed at picomolar concentrations by the cells¹³ within a few hours after viral infection to exert their effector actions¹⁴ and maybe to limit host toxicity. IFN-τ is expressed between 12 and 26 days of pregnancy during the elongation period in bovines¹, and can exceed 100 µg per day without causing toxic effects²·¹⁶. Additionally IFN-τ exhibits high species cross-reactivity making it an attractive candidate (with 30 times less cytotoxic effect) to replace IFN-α/β in clinical applications. The antiviral effect of IFN-τ is linked to the activation or increased expression of ISGs such as Mx GTPase 1 (Mx-1), IRF1, ISG15, 2’,5’-oligodeadenylate synthetase (OAS).
It has demonstrated antiviral activity inhibiting virus (HIV) in monocyte-derived macrophages more effectively than human IFN-α without cytotoxicity¹⁵. Similar, IFN-τ shown significant dose-dependent inhibition of reverse transcriptase activity in feline immunodeficiency virus (FIV)¹⁶. IFN-τ has also shown both in vitro and in vivo antiviral effects against bovine viral diarrhea virus (BVDV)¹⁷ and bovine leukemia virus (BLV)¹⁸. Also, IFN-τ inhibited replication of foot-and-mount disease virus (FMD)¹⁹, Theiler’s murine encephalomyelitis virus (TMEV)²⁰, influenza virus (IV)²¹, ovine lentivirus (OvLV)²². Recently, in an in vitro assay, IFN-τ inhibited the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is 1,000X more potent than antiviral drugs and IFN-λ²³, and the oral treatment in a preclinical animal study showed a potent agent against SARS-CoV-2 virus. Additionally, to DNA virus in human keratinocytes transformed with human papillomavirus (HPV-16), type 16, the treatment with IFN-τ decreases intracellular E6 and E7 oncogenes expression²⁴. Likewise, in human (SiHa) and murine (BMK-16/myc) epithelial cells transformed with HPV-16, IFN-τ induces repression of viral E6 and E7 oncogenes, in a time and dose-dependent manner, and with effects similar to those of IFN-α²⁵. These results show the antiviral properties of IFN-τ provide a promising alternative treatment option for viral infections, with implications for both animal and human health.
Immunomodulation functions of IFN-τ
The immunoregulatory properties of the IFN-τ have been primarily established during pregnancy, and over the last three decades, it has emerged as a key molecule in the regulation of immune response. IFN-τ exerts both paracrine and endocrine actions to regulate the innate immune response to prevent conceptus rejection, nevertheless, this mechanism suggests its versatility and efficacy in modulating immune responses¹·²·²⁷. The paracrine action of IFN-τ is via the regulation of expression of interferon-stimulated genes (ISG) after binding to the IFNAR¹²·²⁸. The immunoregulatory genes activated include major histocompatibility complex (MHC)₂, OAS1, MX1, MX2, ISG15, IRF1, IP10, and IRF2 which contribute to uterine receptivity to implantation and prevention of immune rejection of conceptus²⁸·²⁹.
Beyond its role in reproduction, the immunoregulatory properties of IFN-τ demonstrate a versatile immunoregulatory profile in various contexts, including other cell lines, organisms, and diseases. In peripheral blood leukocytes (PBMC) treated with IFN-τ, there is an increase of OAS1, MX2 and ISG15, evidencing the response to the PBMC to the IFN-τ in components of the circulating immune systems³⁰. Additionally, IFN-τ stimulates granulocyte-macrophage colony-stimulating factor (GM-CSF) gene expression in lymphocytes, endometrial stromal cells³¹, and in vitro culture of ovine embryos³². GM-CSF is an important cytokine that induces the differentiation and proliferation of granulocytes and macrophages derived from hematopoietic progenitor cells and has a potential
Therapeutic target in several autoimmune and inflammatory disorders (rheumatoid arthritis, multiple sclerosis, etc.)³³. Furthermore, the IFN-τ has clinical implications as various cell lines and tissues are susceptible to interferon, which may eventually induce immunoregulatory genes in favor of controlling diseases.
In addition, IFN-τ has been shown to suppress IL-1β release in human macrophages, alleviating obesity-associated inflammation in murine models by reducing proinflammatory cytokines³⁴. Furthermore, IFN-τ can mitigate lipopolysaccharide (LPS)-induced inflammation in endometrial epithelial cells and activate non-classical pathways such as PI3K/AKT, underscoring its ability to modulate inflammatory responses³⁵. In models of autoimmune diabetes, IFN-τ prevented the development of cyclophosphamide-induced and exacerbated anti-islet cytotoxic activity, suggesting its therapeutic potential in autoimmune conditions³⁵. Moreover, in autoimmune diseases of the central nervous system, such as multiple sclerosis, IFN-τ has shown the ability to reduce the stimulation of allogeneic lymphocytic responses, mitigating inflammation and neuronal damage.⁵ Finally, in infectious diseases, IFN-τ inhibits Staphylococcus aureus-induced inflammation in mice with pneumonia and has been associated with the suppression of NF-κB activation as well as the suppression of TNF-α, IL-1β and IL-6³⁶. Therefore; IFN-τ may have the potential as a therapeutic agent against infectious diseases, inflammation, and autoimmune diseases.
Anticancer properties
IFN-τ has emerged as a cytokine with remarkable antiproliferative potential, due to its high species cross-reactivity; its anticellular activity has been demonstrated in various cellular and animal models, including human and murine cells³⁷. Its ability to inhibit cell multiplication has been observed in a variety of tumor cell lines, including amnion epithelium, large cell carcinoma of the lung, colon adenocarcinoma, malignant melanoma, renal adenocarcinoma, promyelocytic leukemia, T-cell lymphoma, cervical cancer cells and breast adenocarcinoma⁵⁸. The antiproliferative ability is localized in the carboxyl-terminal region of IFN-τ protein. Replacement of Ile143 with Thr in helix E reduces receptor binding affinity and completely abolishes antiproliferative activity³⁸. Moreover, the antiproliferative effects of IFN-τ are associated with the up-regulation of IFN-stimulated genes (ISGs) such as IRF1, ISG15, OAS and Mx-1, as well as increased expression of major histocompatibility complex (MHC) class I and beta-2-microglobulin¹²·³⁷.
A crucial aspect of the mechanism of action of IFN-τ is its ability to induce apoptosis in tumor cells. IFN-τ activates signaling pathways that lead to the activation of caspases, including caspases 3, 7, 8 and 11, which are essential in the apoptotic cascade and pyroptosis³⁸·³⁹. This process ensures the elimination of cells with uncontrolled proliferation, contributing to the reduction of tumor mass. In addition, IFN-τ can arrest the cell cycle by negatively regulating cyclins and cyclin-dependent kinases (CDKs)⁴⁰. This cell cycle arrest prevents tumor cells from progressing through subsequent phases, limiting their ability to increase excessively.
In epithelial cells transformed with human papillomavirus (HPV), such as SiHa and BMK-16/myc cells, IFN-τ exhibits antiproliferative effects with a decreased suppression of cell division comparable to that observed with other type I IFNs. IFN-τ also suppresses the expression of E6 and E7 oncogenes and induces apoptosis²⁵·⁴¹. These results suggest that IFN-τ may represent a less cytotoxic and promising alternative to traditional type I interferons for therapy against cancers associated with viral infections.
In a tumor model of immunocompetent HPV 16-positive mice, the treatment with IFN-τ showed significant antitumor activity, reducing tumor growth in a dose- and time-dependent manner and increasing mice survival²⁵. This result indicates that IFN-τ may be a good candidate for immunotherapy against cervical cancer.
In a murine breast cancer model (4T1) IFN-τ induced a 40–70% inhibition of cell proliferation, promoted caspases activation and consequently induced apoptosis. Additionally, IFN-τ inhibited tumor growth in a triple-negative breast cancer (TNBC) murine tumor model, which was associated with increased expression of granulocyte and macrophage colony-stimulating factor (GM-CSF)⁴². Therefore, IFN-τ may be a promising immunotherapeutic cytokine for the treatment of TNBC.
IFN-τ represents a breakthrough in anticancer therapies, standing out for its ability to induce apoptosis, inhibit cell proliferation and regulate the immune response, with a favorable toxicity profile. However, to maximize its therapeutic potential, it is essential to continue detailed research to explore its mechanisms of action and optimize delivery strategies.
Modulation of microbiota
Pleiotropic effects of IFN-τ have been identified including its anti-inflammatory activity and its influence on the modulation of the intestinal microbiota. Supplementation with IFN-τ in mice affects the diversity and composition of the intestinal microbiota, decreasing the percentage of Firmicutes in the jejunum and ileum while increasing Bacteroidetes. However, Firmicutes are enhanced in the colon and feces. These events are associated with decreased expression of IL-17 in both normal mice and those infected with intestinal pathogens. The reduced expression of its cytokine may be related to its role in blocking the recruitment of monocytes and neutrophils to sites of inflammation, which are involved in the pathogenesis of various diseases. Moreover, IFN-τ supplementation increases the feed and liquid intake in mice although it has modest effects on body weight⁴³.
Furthermore, in a mouse model of endometritis induced by Escherichia coli, IFN-τ alleviated endometrial damage and improved the barrier function of endometrial tissues. The treatment with IFN-τ also altered gut microbiota and metabolic pathways. A reduction in the abundance of Enterorhabdus in the gut microbiota of mice was linked to an anti-inflammatory mechanism mediated by IFN-τ⁴⁴. These findings demonstrate the potential of IFN-τ modulating the microbiota in the context of bacterial infections highlighting its anti-inflammatory effects, and therapeutic potential in infectious diseases.
Diabetes, Obesity and Multiple sclerosis autoimmune disorders
The involvement of type I IFN in diabetes control has been proposed in preclinical models, however, their clinical use has been limited due to their toxicity. As a result, other interferons with lower toxicity, such as IFN-τ have been suggested. In non-obese diabetic (NOD) mice, treatment with IFN-τ reduced the development of spontaneous diabetes and was associated with the immunoregulatory activity of splenic cells (CD25+CD4+ T cells). It is proposed that IFN-τ could be a better therapeutic option than IFN-α for the clinical treatment of diabetes, showing promising results with lower toxicity⁴⁵.
Additionally, IFN-τ has been shown to mitigate obesity-associated insulin resistance in a diet-induced obesity mouse model and this effect was associated with macrophage activation and an anti-inflammatory response. Therefore, IFN-τ represents a novel bio-therapeutic cytokine for treating obesity-associated syndrome and type 2 diabetes⁴⁶.
Multiple sclerosis (MS) is considered an inflammatory disease of the central nervous system (CNS). A type I IFN therapy has been approved for the treatment of multiple sclerosis (MS), but its clinical use is limited by toxicity. As an alternative, interferons like IFN-τ which have lower toxicity, are being evaluated⁴⁷. IFN-τ is substantially less toxic even at high concentrations. In an animal model of MS, IFN-τ has been shown to be highly effective⁵, regardless of whether it is administered intraperitoneally, orally, or systemically, it does not lose its activity. Furthermore, IFN-τ induces the production of IL-10 and reduces IFN-γ levels in both rodents and humans, demonstrating efficacy against experimental allergic encephalomyelitis.
(EAE), an animal model of MS⁴⁸. The toxicity of IFN-τ has been evaluated in human T-cells, confirming its remarkably non-toxic nature even at high concentrations alongside its immunomodulatory properties⁴⁹. In this context, phase I and II clinical trials in humans are being conducted for the treatment of relapsing-remitting multiple sclerosis (RRMS)⁵⁰, further confirming IFN-τ as a potentially less toxic therapeutic option for immune-mediated disorders including MS.
Conclusions
In recent years, research on IFN-τ has generated significant scientific evidence of its therapeutic potential. Due to its cross-reactivity between species and very low toxicity, IFN-τ has been applied in the treatment of infectious diseases, chronic diseases and cancer. It has demonstrated immunoregulatory, antiviral, antiproliferative, anticancer and anti-inflammatory effects, as well as involvement in diabetes, obesity, and certain neurological diseases. Many of these properties have been validated in in vitro systems and animal models, suggesting that the therapeutic and clinical implications could be highly relevant for treating animals and more specifically human diseases. Although few clinical studies have investigated the use of IFN-τ in humans, it is essential to expand clinical trials targeting diseases of health importance involving infectious diseases, chronic diseases, and cancer. Additionally, IFN-τ could be an emergency-use molecule for emerging and re-emerging healthcare and public health diseases. Therefore, the future of IFN-τ is promising for its therapeutic use in humans.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding Statement
ScholarChip CONAHCYT: 1022338 (EIH), 798196 (LDP), FOINS CONACYT: 2013-215484.
Acknowledgements
Authors would like to thank Consejo Nacional de Humanidades, Ciencia y Tecnologías (CONAHCYT).
References
1. Kowalczyk A, Czerniawska-Piątkowska E, Wrzecińska M. The Importance of Interferon-Tau in the Diagnosis of Pregnancy. Biomed Res Int. 2021; 2021:9915814. doi:10.1155/2021/9915814
2. Subramaniam PS, Khan SA, Pontzer CH, Johnson HM. Differential recognition of the type I interferon receptor by interferons tau and alpha is responsible for their disparate cytotoxicities. Proc Natl Acad Sci U S A. 1995;92(26):12270-12274. doi:10.1073/ pnas.92.26.12270
3. Chon TW, Bixler S. Interferon-tau: current applications and potential in antiviral therapy. J Interferon Cytokine Res. 2010;30(7):477-485. doi:10.1089/jir.2009.0089
4. Alexenko AP, Ealy AD, Roberts RM. The cross-species antiviral activities of different IFN-tau subtypes on bovine, murine, and human cells: contradictory evidence for therapeutic potential. J Interferon Cytokine Res. 1999;19(12):1335-1341. doi:10.1089/107999099312795
5. Khan OA, Jiang H, Subramaniam PS, Johnson HM, Dhib-Jalbut SS. Immunomodulating functions of recombinant ovine interferon tau: potential for therapy in multiple sclerosis and autoimmune disorders. Mult Scler. 1998;4(2):63-69. doi:10.1177 /135245859800400204.
6. Bazer FW, Ying W, Wang X, et al. The many faces of interferon tau. Amino Acids. 2015;47 (3):449-460. doi:10.1007/s00726-014-1905-x.
7. Senda T, Saitoh SI, Mitsui Y, Li J, Roberts RM. A three-dimensional model of interferon-tau. J Interferon Cytokine Res. 1995;15(12):1053-1060. doi:10.1089/jir.1995.15.1053.
8. Pontzer CH, Ott TL, Bazer FW, Johnson HM. Structure/function studies with interferon tau: evidence for multiple active sites. J Interferon Res. 1994;14(3):133-141. doi:10.1089/jir.1994.14.133.
9. Brooks K, Spencer TE. Biological roles of interferon tau (IFNT) and type I IFN receptors in elongation of the ovine conceptus. Biol Reprod. 2015;92(2):47. doi:10.1095/biolreprod.114.124156.
10. Platanias LC. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat Rev Immunol. 2005;5(5):375-386. doi:10.1038/nri1604.
11. Stewart MD, Johnson GA, Vyhlidal CA, et al. Interferon-tau activates multiple signal transducer and activator of transcription proteins and has complex effects on interferon-responsive gene transcription in ovine endometrial epithelial cells [published correction appears in Endocrinology. 2010 May;151(5):2400. Stewart, D M [corrected to Stewart, M D]. Endocrinology. 2001;142(1):98-107. doi:10.1210/endo.142.1.7891.
12. Fierros-Zárate G, Olvera C, Salazar-Guerrero G, et al. Bovine Interferon-Tau Activates Type I interferon-Associated Janus-signal Transducer in HPV16-positive Tumor Cell. J Cancer. 2020;11(16) :4754-4761. doi:10.7150/jca.33527.
13. Ivashkiv LB, Donlin LT. Regulation of type I interferon responses. Nat Rev Immunol. 2014;14 (1):36-49. doi:10.1038/nri3581.
14. Mihaescu G, Chifiriuc MC, Filip R, et al. Role of interferons in the antiviral battle: from virus-host crosstalk to prophylactic and therapeutic potential in SARS-CoV-2 infection. Front Immunol. 2024;14: 1273604. doi:10.3389/fimmu.2023.1273604.
15. Maneglier B, Rogez-Kreuz C, Dereuddre-Bosquet N, et al. Anti-HIV effects of IFN-tau in human macrophages: role of cellular antiviral factors and interleukin-6. Pathol Biol. 2008; (7-8):492-503. doi: 10.1016/j.patbio.2008.06.002.
16. Pontzer CH, Yamamoto JK, Bazer FW, Ott TL, Johnson HM. Potent anti-feline immunodeficiency virus and anti-human immunodeficiency virus effect of IFN-tau. J Immunol. 1997;158(9):4351-4357.
17. Kohara J, Nishikura Y, Konnai S, Tajima M, Onuma M. Effects of interferon-tau on cattle persistently infected with bovine viral diarrhea virus. Jpn J Vet Res. 2012;60(2-3):63-70.
18. Kohara J, Yokomizo Y. In vitro and in vivo effects of recombinant bovine interferon-tau on bovine leukemia virus. J Vet Med Sci. 2007;69(1): 15-19. doi:10.1292/jvms.69.15.
19. Usharani J, Park SY, Cho EJ, et al. Antiviral activity of ovine interferon tau 4 against foot-and-mouth disease virus. Antiviral Res. 2017; 143:134-141. doi: 10.1016/j.antiviral.2017.01.018.
20. Tennakoon DK, Smith R, Stewart MD, Spencer TE, Nayak M, Welsh CJ. Ovine IFN-tau modulates the expression of MHC antigens on murine cerebrovascular endothelial cells and inhibits replication of Theiler’s virus. J Interferon Cytokine Res. 2001;21(10):785-792. doi:10.1089/107999001 753238015.
21. Martín V, Pascual E, Avia M, et al. A Recombinant Adenovirus Expressing Ovine Interferon Tau Prevents Influenza Virus-Induced Lethality in Mice. J Virol. 2016;90(7):3783-3788. doi:10.1128/JVI.03258-15.
22. Juste RA, Ott TL, Kwang J, Bazer FW, de La Concha-Bermejillo A. Effects of recombinant ovine interferon-tau on ovine lentivirus replication and progression of disease. J Gen Virol. 2000;81(Pt 2):525-532. doi:10.1099/0022-1317-81-2-525.
23. Wanjin Tang, Xiaojun Liu, Fuller W Bazer, et al. Interferon Tau Is a Potent Oral Agent against SARS CoV-2 Infection. SM J Infect Dis 2023. 6:12.
24. Johnson JA, Hochkeppel HK, Gangemi JD. IFN-tau exhibits potent suppression of human papillomavirus E6/E7 oncoprotein expression. J Interferon Cytokine Res. 1999;19(10):1107-1116. doi:10.1089/107999099313046.
25. Padilla-Quirarte HO, Trejo-Moreno C, Fierros-Zarate G, et al. Interferon-Tau has Antiproliferative effects, Represses the Expression of E6 and E7 Oncogenes, Induces Apoptosis in Cell Lines Transformed with HPV16 and Inhibits Tumor Growth In Vivo. J Cancer. 2016;7(15):2231-2240. doi:10.7150/jca.15502.
26. Rocha CC, da Silveira JC, Forde N, Binelli M, Pugliesi G. Conceptus-modulated innate immune function during early pregnancy in ruminants: a review. Anim Reprod. 2021;18(1): e20200048. 10. doi:10.1590/1984-3143-AR2020-0048.
27. Rashid NA, Lalitkumar S, Lalitkumar PG, Gemzell-Danielsson K. Endometrial receptivity and human embryo implantation. Am J Reprod Immunol. 2011;66 Suppl 1:23-30. doi:10.1111/j.1 600-0897.2011.01048. x.
28. Binelli M, Subramaniam P, Diaz T, et al. Bovine interferon-tau stimulates the Janus kinase-signal transducer and activator of transcription pathway in bovine endometrial epithelial cells. Biol Reprod. 2001;64(2):654-665. doi:10.1095/biolreprod64.2.654.
29. Spencer TE, Ott TL, Bazer FW. Expression of interferon regulatory factors one and two in the ovine endometrium: effects of pregnancy and ovine interferon tau. Biol Reprod. 1998;58(5):1154-1162. doi:10.1095/biolreprod58.5.1154.
30. Casano AB, Barile VL, Menchetti L, et al. Interferon Tau (IFNt) and Interferon-Stimulated Genes (ISGs) Expression in Peripheral Blood Leukocytes and Correlation with Circulating Pregnancy-Associated Glycoproteins (PAGs) during Peri-Implantation and Early Pregnancy in Buffalo Cows. Animals (Basel). 2022;12(22):3068. doi:10.33 90/ani12223068.
31. Emond V, Asselin É, Fortier MA, Murphy BD, Lambert RD. Interferon-Tau Stimulates Granulocyte-Macrophage Colony-Stimulating Factor Gene Expression in Bovine Lymphocytes and Endometrial Stromal Cells1. Biol Reprod. 2000;62(6):1728-1737 . doi:10.1095/biolreprod62.6.1728.
32. Rooke J, Ewen M, McEvoy T, Entrican G, Ashworth C. Effect of inclusion of serum and granulocyte-macrophage colony stimulating factor on secretion of interferon-tau during the in vitro culture of ovine embryos. Reprod Fertil Dev. 2005;17(5):513-521. doi:10.1071/rd05014.
33. Wicks IP, Roberts AW. Targeting GM-CSF in inflammatory diseases. Nat Rev Rheumatol. 2016;12(1):37-48. doi:10.1038/nrrheum.2015.161.
34. Hara K, Shirasuna K, Usui F, et al. Interferon-tau attenuates uptake of nanoparticles and secretion of interleukin-1β in macrophages. PLoS One. 2014;9(12): e113974. doi: 10.1371/journal. pone.0113974.
35. Jiang K, Cai J, Jiang Q, et al. Interferon-tau protects bovine endometrial epithelial cells against inflammatory injury by regulating the PI3K/AKT/β-catenin/FoxO1 signaling axis. J Dairy Sci. 2024; 107(1):555-572. doi:10.3168/jds.2022-22983.
36. Zhao G, Wu H, Jiang K, et al. IFN-τ inhibits S. aureus-induced inflammation by suppressing the activation of NF-κB and MAPKs in RAW 264.7 cells and mice with pneumonia. Int Immunopharmacol. 2016; 35:332-340. doi: 10.1016/j.intimp.2016.02.016.
37. Chon TW, Bixler S. Interferon-tau: current applications and potential in antiviral therapy. J Interferon Cytokine Res. 2010;30(7):477-485. doi:10.1089/jir.2009.0089.
38. Li J, Roberts RM. Structure-function relationships in the interferon-tau (IFN-tau). Changes in receptor binding and in antiviral and antiproliferative activities resulting from site-directed mutagenesis performed near the carboxyl terminus. J Biol Chem. 1994;269(40):24826-24833.
39. Suzuki T, Sakumoto R, Hayashi KG, et al. Involvement of interferon-tau in the induction of apoptotic, pyroptotic, and autophagic cell death-related signaling pathways in the bovine uterine endometrium during early pregnancy. J Reprod Dev. 2018;64(6):495-502. doi:10.1262/jrd.2018-063.
40. Subramaniam PS, Johnson HM. A role for the cyclin-dependent kinase inhibitor p21 in the G1 cell cycle arrest mediated by the type I interferons. J Interferon Cytokine Res. 1997;17(1):11-15. doi:10.1089/jir.1997.17.11.
41. Johnson JA, Hochkeppel HK, Gangemi JD. IFN-tau exhibits potent suppression of human papillomavirus E6/E7 oncoprotein expression. J Interferon Cytokine Res. 1999;19(10):1107-1116. doi:10.1089/107999099313046.
42. Espin-Rivera AM, Meza-Aparicio FU, Reyna-Flores F, Burguete-Garcia AI, Guzman-Olea E, Bermudez-Morales VH. Interferon-tau (IFN-τ) Has Antiproliferative Effects, Induces Apoptosis, and Inhibits Tumor Growth in a Triple-negative Breast Cancer Murine Tumor Model. In Vivo. 2023;37 (6):2517-2523. doi:10.21873/invivo.13359.
43. Ren W, Chen S, Zhang L, et al. Interferon Tau Affects Mouse Intestinal Microbiota and Expression of IL-17. Mediators Inflamm. 2016; 2016:2839232. doi:10.1155/2016/2839232.
44. Xue G, Zheng Z, Liang X, Zheng Y, Wu H. Uterine Tissue Metabonomics Combined with 16S rRNA Gene Sequencing to Analyze the Changes of Gut Microbiota in Mice with Endometritis and the Intervention Effect of Tau Interferon. Microbiol Spectr. 2023;11(3): e0040923. doi:10.1128/spectrum.00409-23.
45. Sobel DO, Ahvazi B, Amjad F, Mitnaul L, Pontzer C. Interferon-tau inhibits the development of diabetes in NOD mice. Autoimmunity. 2008; 41(7):543-553. doi:10.1080/08916930802194195.
46. Ying W, Kanameni S, Chang CA, et al. Interferon tau alleviates obesity-induced adipose tissue inflammation and insulin resistance by regulating macrophage polarization. PLoS One. 2014;9(6): e98835. doi: 10.1371/journal.pone.0098835.
47. Reder AT, Feng X. How type I interferons work in multiple sclerosis and other diseases: some unexpected mechanisms. J Interferon Cytokine Res. 2014;34(8):589-599. doi:10.1089/jir.2013.0158.
48. NIH 2004 Oral IFN-tau treatment in RRMS patients | www.inknowvation.com. Home | www.inknowvation.com. Accessed on September 21, 2024. https://www.inknowvation.com/sbir/awards/nih-2004-oral-ifn-tau-treatment-rrms-patients
49. Find and participate in clinical trials and research studies happening around the world | TrialScreen. Find and participate in clinical trials and research studies happening around the world | TrialScreen. Accessed on September 21, 2024. https://app.trialscreen.org/trials/phase-2-multiple-sclerosis-safety-efficacy-oral-ifn-tau-patients-relapsing-trial-actrn12606000241538
50. Lorelie V. Oral IFN-tau treatment in RRMS patients. Grantome. March 1, 2004. Accessed September 21, 2024. https://grantome.com/grant/NIH/R43-AI055227-01A1.

