Tissue Engineering for Complex Wound Healing Solutions
Tissue-Engineered Approaches for Complex Wounds: A Case Series Using Esterified Hyaluronic Acid Matrix
Robin Evans1, William Starr1, Shreyash Pradhan1, Javier Romero1, Graal Diaz1
- Anacapa Surgical Associates Ventura County Health Care Agency 300 Hillmont Ave., Ventura, CA, USA, 93003
- Graduate Medical Division Community Memorial Healthcare System 147 N. Brent St., Ventura, CA, 93003
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION: Evans, R., Starr, W., Prahdan, S., et al. Tissue-Engineered Approaches for Complex Wounds: A Case Series Using Esterified Hyaluronic Acid Matrix. Medical Research Archives, [online] 13(8).
https://doi.org/10.18103/mra.v13i8.6614
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i8.6614
ISSN 2375-1924
Abstract
Tissue specific injuries are difficult to heal and costly to treat. Current solutions are often insufficient to heal complex wounds. Tissue engineering for acute wounds is a new approach. Tissue engineering focuses on regenerating tissue based on the use of biologic scaffolds and cell signaling. Normal wound healing is a dynamic, multiple-phase process that requires coordinated interactions between inflammatory factors, growth factors, angiogenesis, and collagen formation. New technologies use a tissue engineering approach to ensure a smooth transition between the phases of wound healing. If an acute wound fails to progress through normal healing, the phases can become chronic.
Keywords
Tissue engineering, complex wounds, hyaluronic acid, wound healing, scaffolds.
Introduction
Complex soft tissue injuries are difficult to heal and costly to treat. Current solutions are often insufficient to heal complex wounds. Tissue engineering for acute wounds is a new approach¹. Tissue engineering focuses on regenerating tissue based on the use of biologic scaffolds and cell signaling. Normal wound healing is a dynamic, multiple-phase process that requires coordinated interactions between inflammatory factors, growth factors, angiogenesis, and collagen formation². New technologies use a tissue engineering approach to ensure a smooth transition between the phases of wound healing. If an acute wound fails to progress through normal wound healing phases, it can become a chronic wound³.
For example, negative wound pressure therapy has gained popularity because of its effects on angiogenesis⁴. Through inducing angiogenesis, the time to healing is improved, and chronic wounds have been shown to develop healthy granulation tissue⁵. However, negative wound pressure dressings require specialized care, and significant community support structures unavailable to many patients. As well, stimulating angiogenesis is only one component of successful wound healing. As such, a new tissue engineering approach is required to treat a wider variety of wounds at all stages.
Hyaluronic Acid (HA) has become a recent focus of interest. HA is a critical component in healthy skin⁶. HA is a non-sulfated glycosaminoglycan (GAG) found in the extracellular matrix of the skin and soft tissue. It is comprised of repeating polymeric disaccharides of D-glucuronic acid and N-acetyl-D-glucosamine linked by a glucuronidic β (1–3) bond⁷. Depending on the HA size, salt concentration, pH, and associated cations, HA has a different physiologic effect⁸,⁹.
HA is a key component in successful wound healing¹⁰, as it modifies cell signaling. HA is involved in several different roles. Immediately after injury, hemostasis needs to occur. To accomplish this, high-molecular-weight hyaluronic acid (HMW-HA) is released from platelets. HA recruits neutrophils to perform phagocytosis of debris and dead tissue and release tumor necrosis factor-alpha (TNF-α), IL-1β, and IL-8¹¹. HA has also been shown to interact with the toll-like receptors (TLR2 and TLR4) on lymphocytes and macrophages¹²,¹³. Fibronectin and HA help to guide fibroblasts to the wound¹⁴ and support the formation of type III collagen and a new ECM¹⁵. HA has also been noted to have a role in scarless, fetal wound healing¹⁶.
Uniquely, HA also has excellent properties as a scaffold. Esterified HA has been developed to utilize this characteristic¹³,¹⁷. Esterification allows for improved hydrophobicity and reduces HA degradation¹⁸. The electrospun membrane of esterified HA is covered with silicone sheeting. Currently marketed as Hyalomatrix by Medline, this esterified hyaluronic acid matrix was initially used to treat burns¹⁹. The objective of the current study is to review our current experience to determine future research directions.
Methods:
This was a single-center, prospective case-series conducted at a Level II trauma center over 16 months (February 2021 to September 2022). Eligible participants were consecutive adults aged 18 years or older treated with eHAM for wounds during the study window; exclusions included use of eHAM outside the study indications, incomplete medical records preventing key data extraction, or loss to follow-up before healing could be ascertained. Intervention details followed the manufacturer’s protocol and local practice, with documentation on whether eHAM served as the primary modality or an adjunct, frequency of dressing changes, device settings, and any adjunctive therapies (debridement, antibiotics, negative-pressure therapy, grafting) with timing relative to eHAM. Clinician training adhered to institutional guidelines, and deviations from the protocol were recorded. Data were abstracted.
from the electronic medical record and wound-care logs by trained staff using a predefined form and data dictionary. Quality assurance included double data entry for critical fields and regular source-note verification with adjudication of discrepancies. Variables collected encompassed demographics and wound characteristics (location, mechanism, injury date, size in cm2, depth, prior treatments, skin grafting dates and type, dates of eHAM applications and re-applications). Outcomes included the primary time to wound healing (defined as complete epithelialization with no drainage) and secondary outcomes such as percent wound size reduction, need for grafting, complications, and days to milestones. Follow-up extended from the first eHAM application to healing or last contact. Wound size and depth were measured using standardized methods; clinicians determined healing. Statistical analyses used SAS 9.4. Descriptive statistics described variables; time-to-event analyses employed Kaplan-Meier estimates with median time to healing and 95% CIs, and bivariate tests for subgroup comparisons. The study received IRB approval, with no external funding.
Figure 1. Participants by gender
Results:
In this cohort of 12 consecutive adults with full-thickness wounds treated with the esterified hyaluronic acid matrix (eHAM), the tables summarize wound-size changes, injury location, timing, and subsequent interventions. Table 1 shows that initial wound sizes ranged widely (4 to 2000 cm2). After the first eHAM application, 11 of 12 wounds decreased in size, while two wounds showed no change (initial 360 cm2 remaining 360 cm2; initial 16 cm2 remaining 16 cm2). The individual delta sizes ranged from a small reduction of 0 to a substantial reduction of 425 cm2, with percent decreases after the first eHAM spanning 0% to 77.8%. The mean decrease across all wounds after the first eHAM was 56.99 cm2, corresponding to an average 30.5% reduction in wound area (mean initial 244.6 cm2; mean post-eHAM 187.6 cm2).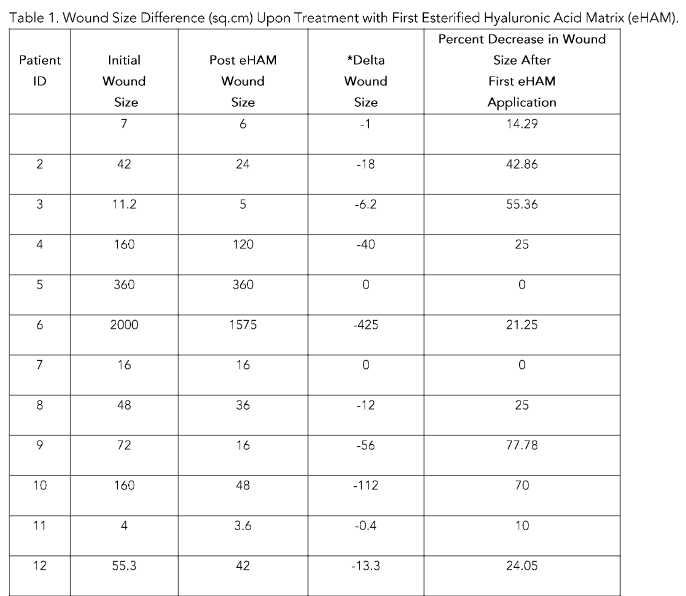
 Summary of Wound Sizes by Area of Injury.
Summary of Wound Sizes by Area of Injury.Table 2 groups wounds by area of injury. Head and neck wounds (n=4) had a mean initial size of 16.05 cm2, a mean delta of –6.4 cm2, and a mean 30.6% reduction, with healing averaging 56 days. Upper extremity wounds (n=1) measured 55.3 cm2 initially, decreasing by 13.3 cm2 (24.05% decrease) and healing in 25 days. The trunk wound (n=1) began at 2000 cm2, decreased by 425 cm2 (21.25%), and healed in 133 days. Lower extremity wounds (n=6) started at a mean 136 cm2, with a mean delta of –36.67 cm2 and a 32.98% reduction, healing over about 74.5 days.
Table 2. Table of Summaries by Area of Injury.
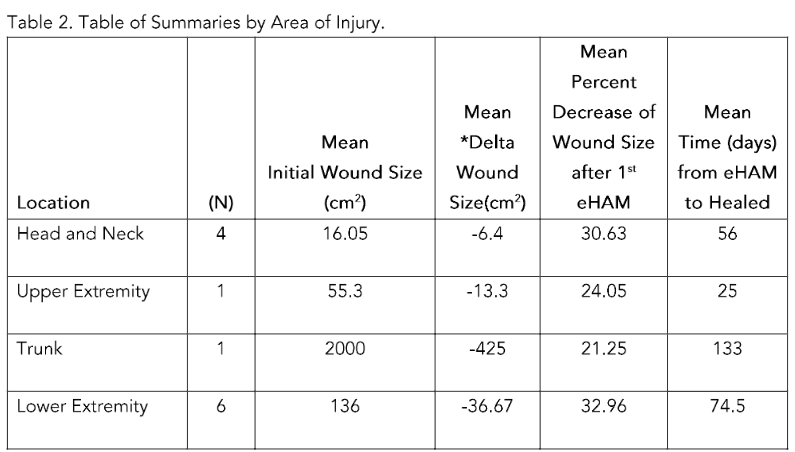
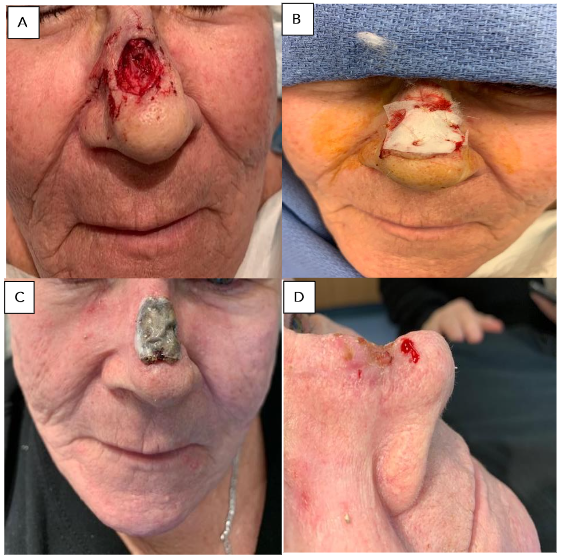
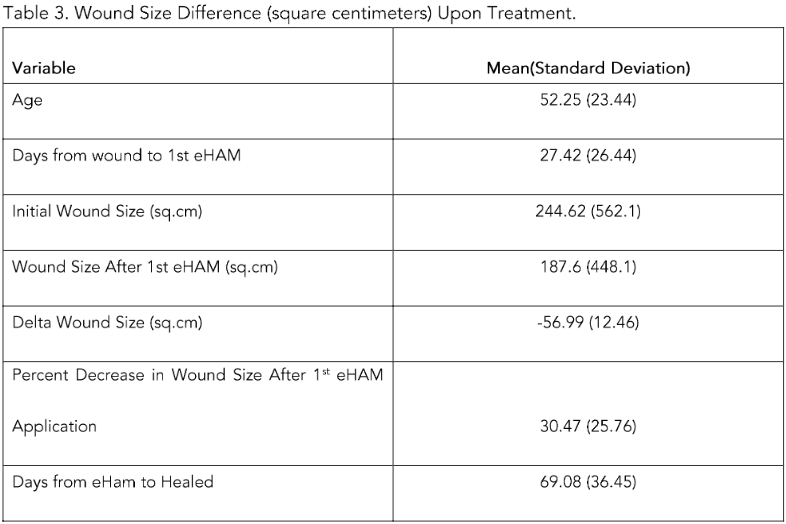
Table 3 provides the overall picture: mean age was 52.25 years (SD 23.44). Time from wound onset to first eHAM averaged 27.4 days (SD 26.4). Initial wounds averaged 244.6 cm2 (SD 562.1), with post-first-eHAM wounds averaging 187.6 cm2 (SD 448.1) and a mean delta of −56.99 cm2 (SD 12.46). The mean percent decrease after the first eHAM was 30.47% (SD 25.76), and the time from eHAM to healed averaged 69.1 days (SD 36.5).
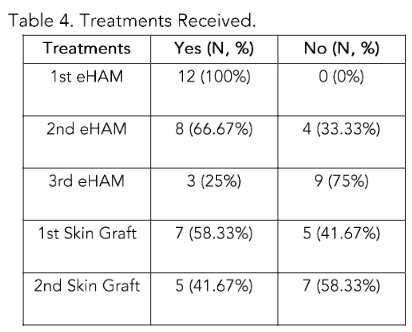
Table 4 describes treatments: all patients received a first eHAM, eight (66.7%) received a second, and three (25%) a third. First skin grafts were performed in 7 patients (58.3%), with a second graft in 5 patients (41.7%).
Discussion:
The development of eHAM. Tissue engineering is an emerging interdisciplinary field that applies principles of bioengineering and life sciences to develop substitutes that guide tissue regeneration. Langer and Vacanti²⁰ categorized three components that govern the outcome of tissue-engineered constructs: scaffolding, instructive environment, and responsive cells. Tissues are engineered in one of two ways: fabricated matrices or the use of cultured human epithelial cells. In the 1980s, the replacement of skin with cells in collagen-glycosaminoglycan scaffolds to facilitate tissue formation was gaining traction. Burke et al.²¹ successfully used porous collagen-chondroitin fibrillar dermis to physiologically promote wound healing in burn patients. Most of the early foundations of tissue engineering relied on the use of human cell cultures.
The development of cultured human epithelial autografts (CEA)²² was a major advance in tissue engineering. However, cultured autografts were found to be fragile and expensive²³. The dermal-epidermal junction requires a biocompatible scaffold to help these cells grow effectively. Hyaluronic acid was a biocompatible molecule and potential scaffold for CEA. Initially, the ethyl and n-propyl esters of hyaluronic were used to create a polymer film and show the biocompatibility of the approach²⁴. Zacchi et al.²⁵ cultured fibroblasts and keratinocytes on a non-woven scaffold of a benzylester of hyaluronic acid to re-create the epidermal-dermal junction seen in normal skin. Galassi et al.²⁶ used this approach to show an increase in the reconstruction of autologous ECM in both acute and chronic wounds.
Biodegradable temporizing matrices (BTM) have gained popularity in the treatment of acute burn wounds. Early burn debridement has been shown to improve outcomes²⁷–³¹. However, the extent of debridement can be limited by the availability of skin graft donor sites, especially in large burns. BTM such as eHAM provides alternatives for early coverage. Gravante et al. demonstrated eHAM use in deep partial thickness³² and full thickness¹⁸ burn patients. Some of these patients went on to heal without skin grafting. A silicone sheet bilayer in these patients theoretically improves fluid loss and granulation tissue formation monitoring. Osti and Enzo³³ found an improvement in wound pH after eHAM application, suggesting improvement in burn healing through alteration of cell signalling. Specifically, it appears that hyaluronic acid plays a role via specific HA receptors, inflammation, cellular migration, and angiogenesis³⁴.
The clinical use of eHAM for complex wounds. Even complex wounds benefit from eHAM application. Complex wounds include wounds with exposed bone, tendon, or other difficult-to-heal clinical situations such as diabetic foot wounds. These unique wounds pose a challenge to any clinician. Notably, the exposure of tendon or bone can lead to tendon necrosis and osteomyelitis, respectively. Ultimately increased delayed healing, morbidity, and mortality. As well, there are often limited dressing and clinical options. A skin graft does not have the ability to take without a blood supply from its recipient site and a local, regional, or free flap is often required. Although negative wound pressure therapy (NWPT) has been successfully employed in the treatment of complex wounds³⁵, it is painful, expensive, and requires a bulky machine to be attached to the wound at all times.
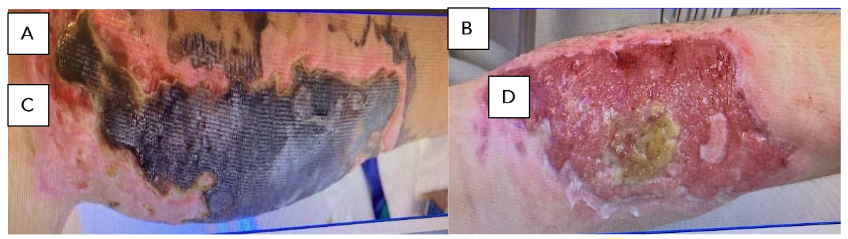
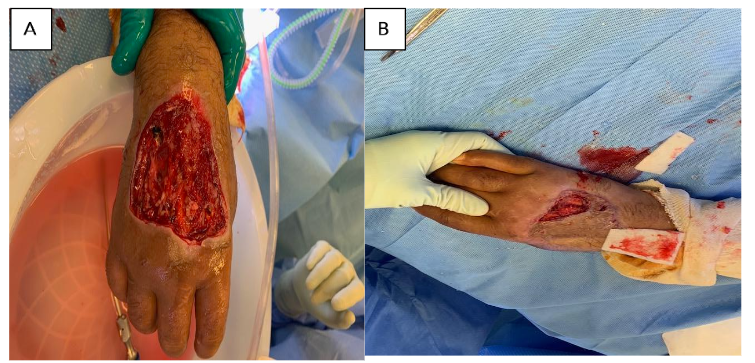
Figure 4. Full thickness skin loss of calf after cast removal for a closed tibial fracture: A. Eschar managed with Silvadene dressing (20 cm × 8 cm); B. Sloughed off eschar; C. Calf after removal of eHAM for 3 weeks (14.5 cm × 6 cm); D. Calf 60 days after eHAM application.
Liu et al. illustrated the effectiveness of eHAM in patients with exposed tendon or bone from trauma, chronic wounds, diabetic foot wounds, and burns³⁶. In this series, eHAM was used for approximately three weeks and followed by coverage with split-thickness skin grafting. However, multiple applications of eHAM have also been used. Vaienti et al. has shown that multiple applications of eHAM improve wound healing outcomes³⁷. This includes healing by secondary intention and preparation for skin grafting.
The primary therapeutic aim of esterified hyaluronic acid matrix (eHAM) in complex wounds is to induce the formation of a neodermis—a dermal-like, organized tissue that serves as an integrative bed for subsequent healing steps. This neodermis was first identified through work by Myers and colleagues, who described a distinct
issue resulting from the interaction of the eHAM scaffold with host tissue. The neodermis forms as a product of HA signaling within the matrix, promoting a cascade of cellular events that culminate in a highly vascularized, structurally organized layer that more closely resembles native dermis than conventional granulation tissue.
Histologically, the neodermis appears more organized and vascular than typical granulation tissue, with Aballey and colleagues reporting that the dermal characteristics within the neodermis resemble those of normal skin dermis. The extended vascular network supports robust perfusion and nutrient delivery, which is particularly important in wounds with exposed structures such as tendons and bones, where a stable, well-vascularized bed is essential for protection and healing. The neodermis thus provides a durable, scaffold-like foundation that accommodates re-epithelialization and enhances graft take, enabling skin grafts to adhere and integrate more reliably and potentially reducing the need for more complex flap coverage in some cases.
In essence, eHAM’s value lies not only in temporary wound coverage but in actively guiding tissue remodeling toward a dermal substitute that supports stability, vascularity, and subsequent epidermal regeneration. While the precise extent and kinetics of neodermis formation can be influenced by factors such as infection, perfusion, and patient comorbidities, the concept underscores how HA-based matrices create a dermal analogue that bridges the gap between the wound bed and definitive closure.
Conclusion:
Our experience demonstrates that the electrospun non-woven esterified hyaluronic acid matrix (eHAM) is a valuable tool in the treatment of complex wounds. The nanofibrous scaffold provides increased opportunities for cell adhesion and growth. Matrix revascularization allows for healing by secondary intention or coverage with a skin graft. In our study, many patients were able to heal using eHAM therapy alone. Clinical benefit was demonstrated after the first eHAM application, where wound size was markedly reduced for all patients. Multiple eHAM applications showed improved efficacy in larger wounds. Further investigation will allow us to improve our tissue engineering paradigm to optimize outcomes for patients with complex wounds.
Conflict of Interest:
None.
Funding Statement:
This study did not receive funding. Dr. Evans is a member of the Advisory Board of True Digital Surgery, and the rest of the authors have nothing to disclose.
Acknowledgements:
None.
References
1. Vig, K. et al. Advances in skin regeneration using tissue engineering. International journal of molecular sciences 18, 789 (2017).
2. Sinno, H. & Prakash, S. Complements and the wound healing cascade: an updated review. Plastic surgery international 2013 (2013).
3. Krzyszczyk, P., Schloss, R., Palmer, A. & Berthiaume, F. The role of macrophages in acute and chronic wound healing and interventions to promote pro-wound healing phenotypes. Frontiers in physiology 9, 419 (2018).
4. Glass, G., Murphy, G., Esmaeili, A., Lai, L. M. & Nanchahal, J. Systematic review of molecular mechanism of action of negative‐pressure wound therapy. British Journal of Surgery 101, 1627-1636 (2014).
5. Bassetto, F. et al. Histological evolution of chronic wounds under negative pressure therapy. Journal of plastic, reconstructive & aesthetic surgery 65, 91-99 (2012).
6. Papakonstantinou, E., Roth, M. & Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermato-endocrinology 4, 253-258 (2012).
7. Weissmann, B., Meyer, K., Sampson, P. & Linker, A. Isolation of oligosaccharides enzymatically produced from hyaluronic acid. Journal of Biological Chemistry 208, 417-429 (1954).
8. Fallacara, A., Baldini, E., Manfredini, S. & Vertuani, S. Hyaluronic acid in the third millennium. Polymers 10, 701 (2018).
9. Tavianatou, A. G. et al. Hyaluronan: molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J 286, 2883-2908, doi:10.1111/febs.14777 (2019).
10. Neuman, M. G., Nanau, R. M., Oruña-Sanchez, L. & Coto, G. Hyaluronic acid and wound healing. Journal of Pharmacy & Pharmaceutical Sciences 18, 53-60 (2015).
11. Graca, M. F. P., Miguel, S. P., Cabral, C. S. D. & Correia, I. J. Hyaluronic acid-Based wound dressings: A review. Carbohydr Polym 241, 116364, doi:10.1016/j.carbpol.2020.116364 (2020).
12. Aya, K. L. & Stern, R. Hyaluronan in wound healing: rediscovering a major player. Wound repair and regeneration 22, 579-593 (2014).
13. Chen, W. J. in Hyaluronan 147-156 (Elsevier, 2002).
14. Webber, J., Jenkins, R. H., Meran, S., Phillips, A. & Steadman, R. Modulation of TGFβ1-dependent myofibroblast differentiation by hyaluronan. The American journal of pathology 175, 148-160 (2009).
15. Stern, R., Asari, A. A. & Sugahara, K. N. Hyaluronan fragments: an information-rich system. European journal of cell biology 85, 699-715 (2006).
16. Longaker, M. T. et al. Studies in fetal wound healing. V. A prolonged presence of hyaluronic acid characterizes fetal wound fluid. Annals of surgery 213, 292 (1991).
17. Harding, K. (Elsevier BV, 2000).
18. Gravante, G. et al. Hyalomatrix PA in burn care practice: results from a national retrospective survey, 2005 to 2006. Ann Plast Surg 64, 69-79, doi:10.1097/SAP.0b013e31819b3d59 (2010).
19. Longinotti, C. The use of hyaluronic acid based dressings to treat burns: A review. Burns & trauma 2, 2321-3868.142398 (2014).
20. Langer, R., Lanza, R., Langer, R. S. & Vacanti, J. P. Principles of tissue engineering. (Academic Press, 2000).
21. Burke, J. F., Yannas, I. V., Quinby Jr, W. C., Bondoc, C. C. & Jung, W. K. Successful use of a physiologically acceptable artificial skin in the treatment of extensive burn injury. Annals of surgery 194, 413 (1981).
22. Green, H., Kehinde, O. & Thomas, J. Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proceedings of the National Academy of Sciences 76, 5665-5668 (1979).
23. Atiyeh, B. S. & Costagliola, M. Cultured epithelial autograft (CEA) in burn treatment: three decades later. Burns 33, 405-413 (2007).
24. Cortivo, R., Brun, P., Rastrelli, A. & Abatangelo, G. In vitro studies on biocompatibility of hyaluronic acid esters. Biomaterials 12, 727-730 (1991).
25. Zacchi, V. et al. In vitro engineering of human skin‐like tissue. Journal of Biomedical Materials Research: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and the Australian Society for Biomaterials 40, 187-194 (1998).
26. Galassi, G. et al. In vitro reconstructed dermis implanted in human wounds: degradation studies of the HA-based supporting scaffold. Biomaterials 21, 2183-2191 (2000).
27. Janzekovic, Z. A new concept in the early excision and immediate grafting of burns. The Journal of trauma 10, 1103-1108 (1970).
28. Janzekovic, Z. Early surgical treatment of the burned surface. (1972).
29. Burke, J. F., Bondoc, C. C. & Quinby, W. C. Primary burn excision and immediate grafting: a method shortening illness. Journal of Trauma and Acute Care Surgery 14, 389-395 (1974).
30. Herndon, D. N. et al. A comparison of conservative versus early excision. Therapies in severely burned patients. Annals of surgery 209, 547 (1989).
31. Desai, M. H. et al. Early burn wound excision significantly reduces blood loss. Annals of surgery 211, 753 (1990).
32. Gravante, G. et al. The use of Hyalomatrix PA in the treatment of deep partial-thickness burns. Journal of burn care & research 28, 269-274 (2007).
33. Osti, E. Skin ph variations from the acute phase to re-epithelialization in burn patients treated with new materials (burnshield®, semipermeable adhesive film, dermasilk®, and hyalomatrix®). Non-invasive preliminary experimental clinical trial. Annals of burns and fire disasters 21, 73 (2008).
34. Litwiniuk, M., Krejner, A., Speyrer, M. S., Gauto, A. R. & Grzela, T. Hyaluronic acid in inflammation and tissue regeneration. Wounds 28, 78-88 (2016).
35. Park, H., Copeland, C., Henry, S. & Barbul, A. Complex wounds and their management. Surgical Clinics 90, 1181-1194 (2010).
36. Liu, X., Dadireddy, K. & Velamuri, S. R. Esterified Hyaluronic Acid Matrix in Lower Extremity Reconstruction With Exposed Tendon and Bone: A Retrospective Review. Journal of Burn Care & Research 41 (2020).
37. Vaienti, L. et al. Limb trauma: the use of an advanced wound care device in the treatment of full-thickness wounds. Strategies in trauma and limb reconstruction 8, 111-115 (2013).
38. Myers, S. R., Partha, V. N., Soranzo, C., Price, R. D. & Navsaria, H. A. Hyalomatrix: a temporary epidermal barrier, hyaluronan delivery, and neodermis induction system for keratinocyte stem cell therapy. Tissue engineering 13, 2733-2741 (2007).
39. Aballay, A. & Hermans, M. H. Neodermis formation in full thickness wounds using an esterified hyaluronic acid matrix. Journal of Burn Care & Research 40, 585-589