Traumatic Central Serous Chorioretinopathy Case Report
Presumed Traumatic Central Serous Chorioretinopathy: A Case Report
Meghan Gorniak, MS 1; Anahita Dadali, MBBS iBSc (Hons) 2; Sweta Kavali, MD 3; Niloofar Piri, MD 1
- SSMHealth Medical Group, Department of Ophthalmology, Saint Louis University, School of Medicine, St. Louis, MO
- St. George’s, University of London, London, England
- Ophthalmology Consultants, St. Louis, MO
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION Gorniak, M., Dadali, A., et al., 2025. Presumed Traumatic Central Serous Chorioretinopathy: A Case Report. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6829
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6829
ISSN 2375-1924
ABSTRACT
Background: Central serous chorioretinopathy describes central neurosensory detachment of the retina due to the accumulation of subretinal fluid. Both pathophysiology and proposed etiology/risk factors of the condition have been reported. However, an association between blunt trauma to the eye and central serous chorioretinopathy has not been established.
Objective: To report a unique case of central serous chorioretinopathy development following trauma to the affected eye.
Method: Case report. Diagnosis was confirmed with a comprehensive ocular exam and imaging.
Results: A 42-year-old Caucasian male presented with sudden blurry vision in the left eye one day after being hit with a Nerf gun dart. He denied any risk factors of central serous chorioretinopathy including increased stress, corticosteroid use, or underlying autoimmune disease. Visual acuity was 20/80 in the left eye. Posterior segment examination revealed circumscribed subretinal fluid 4-disc diameter in size involving the fovea. Findings were supported by spectral domain optical coherence tomography showing large neurosensory detachment and fundus autofluorescence demonstrated well defined hyperautofluorescence corresponding to the detached area. Fluorescein angiography revealed an expansile dot pattern, confirming the diagnosis of central serous chorioretinopathy. Patient returned five weeks later with improved vision and significantly improved subretinal fluid with conservative management.
Conclusion: This case report of central serous chorioretinopathy development with a presumed traumatic etiology is only the fourth reported case with no available cohort studies. Thus far, conservative management remains the primary treatment method; yet treatment courses following trauma have been largely understudied to limited case numbers. Additionally, further research is necessary to better distinguish individual risk factors compared to trauma-related risk factors.
Keywords
central serous chorioretinopathy, trauma, case report, ocular examination, imaging
Introduction
Central serous retinopathy, also known as central serous chorioretinopathy (CSCR), is a medical condition caused by accumulation of subretinal fluid (SRF) that causes localized detachment of the neurosensory retina and/or retinal pigment epithelium (RPE) in the posterior pole. Central serous chorioretinopathy is the fourth most common retinopathy behind age-related macular degeneration (ARMD), diabetic retinopathy, and branch retinal vein occlusion. It most commonly occurs unilaterally, causing blurred vision and can be associated with additional symptoms such as micropsia, metamorphopsia, hyperopic shift, central scotoma, reduced contrast sensitivity, and decreased color saturation. Central serous chorioretinopathy typically manifests between 28 and 68 years of age, with males six times more likely to be affected than females. Vision loss in CSCR occurs due to foveal attenuation, chronic macular edema, and damage to the foveal photoreceptor layer. While vision loss mechanisms have been established, the exact pathophysiology remains unknown. Initially, RPE dysfunction was thought to be the primary cause. However, recent studies have shown this dysfunction is likely a sequela due to irregular vasodilation of choroidal veins, choroidal lobular ischemia, or choroidal capillary endothelial dysfunction with choroidal dysfunction the primary cause. Gass’ theory that choroidal hyperpermeability drives the development of CSCR via increased hydrostatic pressure has continued to gain support with indocyanine green angiography reinforcing this proposed mechanism. Since this development in pathophysiology has been established, the pachychoroid-driven process has been introduced.
The pachychoroid disease spectrum includes clinical conditions such as CSCR, pachychoroid neovasculopathy (PNV), and polypoidal choroidal vasculopathy (PCV). These diseases involve (1) pachychoroid or thickening of the choroid, (2) pachyvessels or dilation of the veins in the Haller layer and lastly, (3) thinning of inner choroidal layers. The etiology of pachychoroid-driven processes begins with choroidal vessel dilation, leading to increased permeability in the choroid, eventually resulting in serous retinal detachment. Among these factors are vortex venous congestion and compression, mineralocorticoid receptor activation, complement pathway dysregulation, inflammatory processes and/or oxidative stress. Venous congestion plays a significant role in the etiology of pachyvessel and pachychoroid disease. Vortex veins, especially the dominant vortex vein, drain the macula; thus, obstruction of vortex vein outflow results in elevated retrograde venous pressure through the proximal vortex veins, subsequently dilating Haller vessels and leading to SRF build-up. Moreover, malformation of choroidal arteriovenous anastomoses can bypass the choriocapillaries and direct arterial filling, resulting in venous overload that may aid in CSCR progression.
The clinical etiology of CSCR has been studied extensively. Most significantly, patients undergoing corticosteroid treatment for systemic inflammatory diseases and those receiving exogenous glucocorticoid therapy have the greatest associations with CSCR development. Excluding idiopathic distinction, CSCR has also been associated with the following: type A personality, Helicobacter pylori infection, methylenedioxymethamphetamine (MDMA), sildenafil, topiramate, MEK/ERK pathway inhibitors, and obstructive sleep apnea. Nevertheless, association between blunt trauma to the eye and CSCR has not yet been identified.
Case Report
A 42-year-old Caucasian male presented to the ophthalmology clinic for blurry vision after being shot in the left eye with a foam-plastic dart five days prior. He noted central blurry vision in the left eye associated with a single black floater and flashes of light. A Spectral Domain optical coherence tomography (SD-OCT) was obtained, showing neurosensory detachment within the posterior pole in the left eye along with small irregular pigment epithelial detachment (PED) (Figure 1). He was referred to a retina specialist for further evaluation.
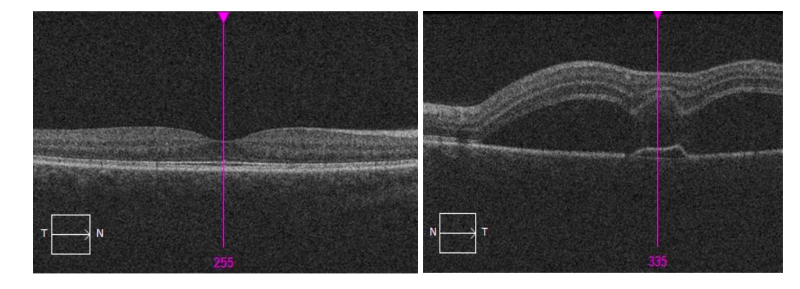
His ocular history was remarkable for vitreous syneresis of both eyes and amblyopia of the left eye. He denied tobacco use with no significant past medical history, surgical history, family history, or previous medication use. The patient denied associated CSCR risk factors such as corticosteroid use, prolonged stress levels, and testosterone use.
The patient’s visual acuity was 20/25-1 in the right eye and 20/80 in the left eye by Snellen Chart without correction. There was no improvement in visual acuity by pinhole in the left eye. Patient’s intraocular pressure (IOP) was measured as 12 mm Hg by Goldmann applanation bilaterally. No relative afferent pupillary defect was noted. Slit lamp examination was within normal limits in both eyes. Posterior examination revealed vitreous syneresis bilaterally. In the right eye, the macula showed good foveal reflex with no hemorrhages, exudates, or pigmentary changes. Left eye showed circumscribed subretinal fluid in the superotemporal macula extending to the fovea. The peripheral exam was within normal limits bilaterally.
Fundus photography showed circumscribed subretinal fluid in the superotemporal macula of the left eye. Fundus autofluorescence demonstrated a well-defined area of hyperautofluorescence corresponding to the location of neurosensory detachment (Figure 2). Photos of the right eye were unremarkable. Spectral Domain optical coherence tomography of the left eye confirmed circumscribed subretinal fluid and small PED; the right eye was within normal limits. Maculopathy secondary to traumatic CSCR was considered the most likely diagnosis after considerations were taken to exclude other causes.
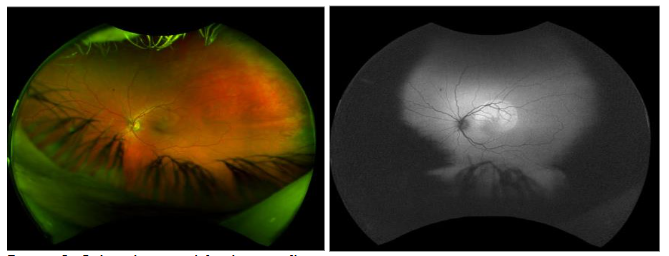
Following initial investigations, the patient was managed conservatively with continued monitoring for possible spontaneous resolution of subretinal fluid. Other treatment options were discussed with the patient in case symptoms persisted, or choroidal neovascularization was seen in the following appointment.
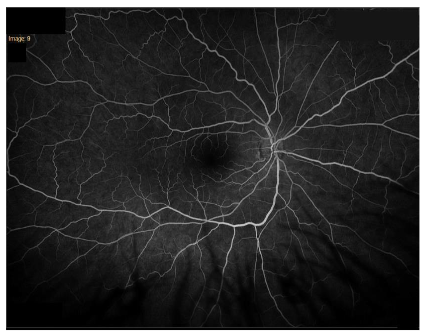
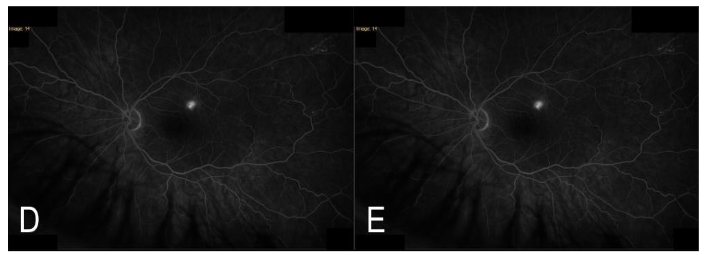
Five weeks later, the patient returned to the clinic with a visual acuity of 20/80-1 in the left eye that improved to 20/40-1 with pinhole. Intraocular pressure was 12 mm Hg in the right eye and 13 mm Hg in the left. Posterior segment exam demonstrated blunted foveal reflex of the left eye; right eye continued to appear within normal limits. Fundus fluorescein angiography was performed and showed a normal pattern in the right eye (Figure 3). Fluorescein angiography demonstrated a classic expansile dot pattern in the left superior macula consistent with CSCR (Figure 4).
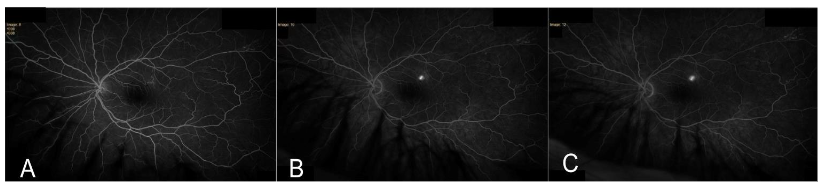
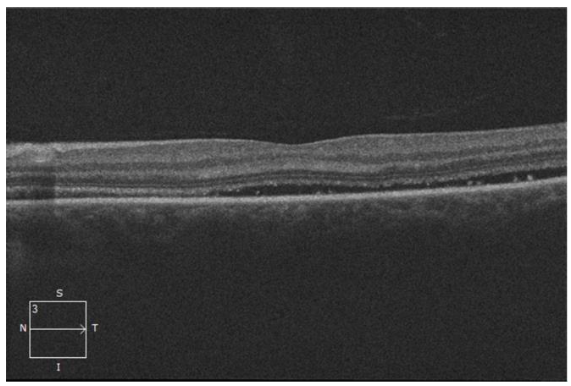
Repeat SD-OCT of the left eye at 5-week follow up showed decreased SRF from prior scan, suggesting improvement from the initial visit (Figure 5).
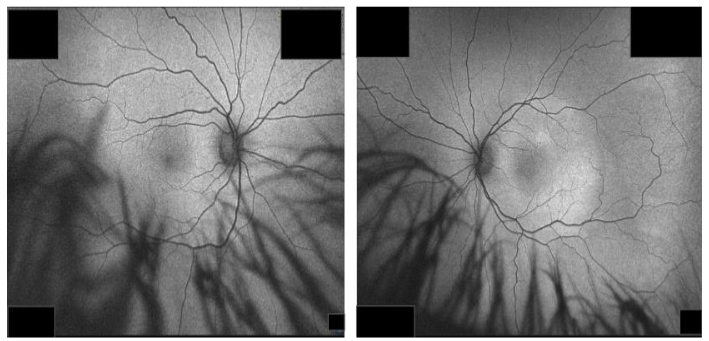
Similarly, repeat autofluorescence imaging showed decreased hyperautofluorescence with diffuse distribution, indicating improvement (Figure 6).
At this time, diagnosis and treatment options were rediscussed with the patient, and he was reassured that symptoms generally continue to improve without treatment. He was advised to return in one month; however, the patient was lost to follow up afterwards.
Discussion
Eye injuries contribute significantly to sight-threatening emergencies in ophthalmology. Blunt trauma to the eye is most common in the male gender and advanced age with outcomes of injury dependent on mechanism of injury and patient demographics. Blunt injury to the eye has been associated with many retinal complications such as commotio retinae, retinal tear, retinal detachment, retinal hemorrhage, and traumatic proliferative retinopathy. However, a clear association between ocular trauma and CSCR has not yet been identified. This case reflects presumed CSCR secondary to blunt trauma given temporal association. Our patient did not exhibit known risk factors for development of CSCR. To the best of our knowledge, apart from the patient case discussed in this report, only three cases of traumatic CSCR have been reported in the literature.
Psychosocial stressors have a major impact on the onset of CSCR, especially in patients with poor coping mechanisms. In a study by Gelber et al., an acute stress reaction preceded the onset of CSCR in patients by a week on average, whilst minor stressors preceded the onset of recurrent symptoms by only a few hours. Moreover, resolution of said stressor led to improved visual acuity within weeks, yet it remains unclear whether there is a causal relationship between removal or resolution of stressors and reduced risk of progression to chronic CSCR. Evidence suggests pharmacological treatments which reduce the sympathetic response to stress (e.g. beta-blockers) potentially have a role in preventing recurrence of symptoms. This patient did not report any additional stressors, making this association a less likely explanation for his development of CSCR.
Apart from psychosocial factors, personality traits may also play a role in CSCR with type A personality types at highest risk. This etiology has been proposed to be related to elevated levels of catecholamines and corticosteroids in patients with type A personality traits compared to those with type B personalities. Personality was not fully assessed in this study and remains a potential confounding variable to this presentation. In CSCR, elevated levels of catecholamines, especially adrenaline, correlate significantly to central macular thickness. Additionally, patients with CSCR have been reported to have elevated serum cortisol levels and urinary cortisol excretion rates. In approximately 5% of cases of endogenous hypercortisolism, the presenting symptom reported is CSCR. Cushing’s syndrome, whether endogenous or exogenous etiology, has been associated with increased risk of CSCR. Similarly, exogenous corticosteroid intake, irrespective of route of administration, has been associated with increased risk of developing this condition. Among patients with solid organ transplant, those having undergone renal transplant appear to be at a particularly higher risk of developing CSCR following long-term corticosteroid use possibly due to several risk factors including but not limited to underlying renal pathology, hypertension, small vessel disease and previous history of hemodialysis leading to choroidal changes. Even though high serum glucocorticoid levels are unambiguously associated with the development of CSCR, the pathophysiology remains unclear. A review by Liegl and Ulbig mentioned several other risk factors for CSCR such as pregnancy, alcohol consumption, untreated hypertension, bone marrow or organ transplantation, and infection of the respiratory tract. Though cases are limited, we propose a rare association between traumatic injury to the eye and CSCR development.
Previous literature has introduced CSCR as a self-limiting condition; however, recent literature has introduced a wide spectrum of presentations with various courses of disease chronically. The development of CSCR tends to occur in male patients 20-50 years of age with type A personality, but presentations have been seen amongst older individuals, females, and varying racial backgrounds. This patient’s fluorescein angiography testing revealed an expansile dot or ink blot appearance, a finding seen in 93% of CSCR cases, and OCT findings were consistent with a typical presentation. The patient case presented here exhibited no previous history of CSCR. In two out of the three previous reported cases of traumatic CSCR, there was no previous history of the condition in the affected eye or the contralateral eye; this is in contrast to the third case, where the patient experienced idiopathic CSCR six years prior to CSCR development in his left eye following a traumatic blow by a hard plastic object. The authors in the latter case noted that their patient possibly had an underlying predisposition to developing CSCR. Regardless of past ocular history, all aforementioned cases reported spontaneous resolution and required no further treatment.
Currently, there is no treatment specifically recommended for CSCR and observational therapy for the first three months remains the mainstay of treatment in the absence of foveal leakage, choroidal neovascularization or other underlying inflammatory conditions requiring systemic therapy. Other treatments such as verteporfin photodynamic monotherapy are used in cases when there is no spontaneous resolution, or if there is occupational need for faster visual recovery. In cases of secondary complications like choroidal neovascularization, intravitreal anti-vascular epithelial growth factor (anti-VEGF) agents are indicated. Our patient exhibited improvement at his 5 week follow-up appointment and was subsequently lost to follow up. Given spontaneous improvement in 5 weeks, it is highly likely his CSCR will completely resolve.
Conclusion
Blunt trauma to the eye can pose several sight-threatening emergencies and thorough investigations, including comprehensive ophthalmic examination and imaging modalities, are required to rule out other conditions before the diagnosis of CSCR can be confirmed. Previous literature has established important modifiable and non-modifiable risk factors for development of CSCR, and three previous case reports have introduced an association between blunt trauma and development of CSCR. Due to the limited number of cases in previous literature, development secondary to trauma has not been researched adequately. While CSCR typically resolves spontaneously, the full treatment course of traumatic CSCR remains unknown; nevertheless, trauma-related stress has a probable role. To better understand the etiology and course of traumatic CSCR, larger populations of patients need to be followed over the course of their disease to help distinguish individual risk factors from trauma-related risk factors. Moreover, the effect of systemic disease and the possible association of previous CSCR with development of traumatic CSCR should be further explored.
Conflicts of Interest:
The authors have no conflicts of interest to declare.
Acknowledgements:
Thank you to the technicians at Ophthalmology Consultants for completing necessary testing (SD-OCT, FAF, FA, fundus photos) during appointments.
References
- Zhang, X., Lim, C. et al (2023). Central serous chorioretinopathy: updates in the pathogenesis, diagnosis and therapeutic strategies, BMC, 10: pp. 33.
- Wang, M., Munch, I. et al (2008) Central serous chorioretinopathy, Acta Ophthalmologica, 86: pp. 126-145.
- Liew, G., Quin, G., et al (2013) Central serous chorioretinopathy: a review of epidemiology and pathophysiology, Clinical and Experimental Ophthalmology, 41: pp. 201-214.
- Gäckle, H., Lang G., et al (1998) Central serous chorioretinopathy. Clinical, fluorescein angiography and demographic aspects, Ophthalmole, 95: pp. 529-533.
- Spaide, R., Campeas, L., et al (1996) Central Serous Chorioretinopathy in younger and older adults, Ophthalmology, 103: pp. 2070-2080.
- Giovannini, A., Scassellati-Sforzolini, B., et al (1997) Choroidal Findings in the Course of Idiopathic Serous Pigment Epithelium Detachment by Indocyanine Green Videoangiography, Retina, 17: pp. 286-296.
- Cheung, C., Lee, W., Koizumi, H., et al (2019) Pachychoroid disease. Eye (London), 33: pp. 14–33.
- Zarnegar, A., Ong, J., Matsyaraja, T., et al (2023) Pathomechanisms in central serous chorioretinopathy: A recent update, International Journal of Retinal and Vitreous, 9: pp. 3.
- Gass, J. (1967) Pathogenesis of disciform detachment of the neuroepithelium. American Journal of Ophthalmology, 63: pp.1–139.
- Wang, Z., Xin, Z. and Yang, J. (2021) Choriocapillaris ischemia at the leakage point of patients with acute central serous chorioretinopathy. Front Med (Lausanne), 8: pp. 675876.
- Lim, J., Kim, M. and Shin, M. (2010) Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina, 30: pp. 1465–1471.
- Prünte, C. and Flammer, J. (1996) Choroidal capillary and venous congestion in central serous chorioretinopathy. American Journal of Ophthalmology, 121: pp. 26–34.
- Spaide, R., Gemmy Cheung, C., Matsumoto, H., et al (2021) Venous overload choroidopathy: A hypothetical framework for central serous chorioretinopathy and allied disorders. Prog Retin Eye Res, 86: pp. 100973.
- Lejoyeux, R., Benillouche, J., Ong, J., et al (2022) Choriocapillaris: fundamentals and advancements. Prog Retin Eye Res, 87: pp. 100997.
- Pang, C., Shah, V., Sarraf, D., Freund, K. (2014) Ultra-widefield imaging with autofluorescence and indocyanine green angiography in central serous chorioretinopathy. American Journal of Ophthalmology, 158: pp. 362–371.
- Brinks, J., van Dijk E., Meijer, O., et al (2022) Choroidal arteriovenous anastomoses: a hypothesis for the pathogenesis of central serous chorioretinopathy and other pachychoroid disease spectrum abnormalities. Acta Ophthalmology, 100: pp. 946-959.
- Terao, N., Imanaga, N., Wakugawa, S., et al (2021). Short axial length is related to asymmetric vortex veins in central serous chorioretinopathy. Ophthalmology Scientific, 1: pp. 100071.
- Bacci, T., Oh, D., Singer, M., et al (2022) Ultra-widefield indocyanine green angiography reveals patterns of choroidal venous insufficiency influencing pachychoroid disease. Invest Ophthalmol Vis Sci, 63: pp. 17.
- Mori, K., Gehlbach, P., Yoneya, S., Shimizu, K. (2004) Asymmetry of choroidal venous vascular patterns in the human eye. Ophthalmology, 111: pp. 507–512.
- Hiroe, T., Kishi, S. (2018) Dilatation of asymmetric vortex vein in central serous chorioretinopathy. Ophthalmol Retina, 2: pp. 152–161.
- Garg, S., Dada, T., Talwar, D., Biswas, N. (1997) Endogenous cortisol profile in patients with central serous chorioretinopathy, British Journal of Ophthalmology, 81: pp. 962-964.
- Haimovici, R., Koh, S., Gagnon, D., Lehrfeld, T., Wellik, S. (2004) Risk factors for central serous chorioretinopathy: A case–control study. Ophthalmology, 111: pp. 244-249.
- Mohseni, M., Blair, K., Gurnani, B., et al (2024) Blunt Eye Trauma. StatPearls Publishing, Available from: https://www.ncbi.nlm.nih.gov/books/NBK470379/
- Steeples, L., Sharma, V., & Mercieca, K. (2015). Traumatic central serous chorioretinopathy. Indian Journal of Ophthalmology, 63, pp. 536-538.
- Ponce, C. M. P., Mohidat, H. M., & Garcia, C. A. (2012). Central serous chorioretinopathy after blunt trauma. BMJ Case Reports, 2012.
- Jackson, T. E., Sood, V., & Haigh, P. M. (2012). Central serous chorioretinopathy secondary to trauma. Oman Journal of Ophthalmology, 5, pp. 51-52.
- Gelber GS, Schatz H. (1987) Loss of vision due to central serous chorioretinopathy following psychological stress. American Journal of Psychiatry, 144: pp. 46–50.
- Tatham A, Macfarlane A. (2006) The use of propranolol to treat central serous chorioretinopathy: an evaluation by serial OCT, Journal of Ocular Pharmacology and Therapeutics, 22: pp. 145–149.
- Spahn C, Wiek J, Burger T. (2004) Operationalized psycho-dynamic diagnostics (OPD) in patients with central serous chorioretinopathy, Psychotherapie Psychosomatik, medizinische Psychologie, 54: pp. 52–57.
- Jinghua, S., Junfeng, T., Zhitao, W., et al (2003) Effect of catecholamine on central serous chorioretinopathy, Current Medical Science, 23: pp. 313–316.
- Haimovici, R., Rumelt, S., Melby, J., (2003) Endocrine abnormalities in patients with central serous chorioretinopathy, Ophthalmology, 110: pp. 698–703.
- Bouzas, E., Scott, M., Mastorakos, G., et al (1993) Central serous chorioretinopathy in endogenous hypercortisolism. Arch Ophthalmol, 111: pp. 1229–1233.
- Jonas, J., Kamppeter, B. (2005) Intravitreal triamcinolone acetonide and central serous chorioretinopathy. British Journal of Ophthalmology, 89: pp. 386–387.
- Lee, C., Kang, E., Lee, K., et al. (2011) Central serous chorioretinopathy after renal transplantation. Retina, 31: pp. 1896–1903.
- Liegl, R., & Ulbig, M. W. (2014). Central serous chorioretinopathy, Ophthalmologica, 232: pp. 65-76.
- Gemenetzi, M., De Salvo, G., and Lotery, A. (2010) Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye (London), 24: pp. 1743 – 1756.
- Lim, J., Kim, M. (2011) The efficacy of intravitreal bevacizumab for idiopathic central serous chorioretinopathy. Graefe’s Archive for Clinical Experimental Ophthalmology, 249: pp.969 – 974.




