Ultrasound-Guided Lipoaspiration in Orthobiologics
Musculoskeletal Ultrasound in Orthobiologic Care—From Harvest to Injection: A Cadaveric Validation of Ultrasound-Guided Lipoaspiration for Orthobiologic Applications
Alberto Panero, DO¹*; Melissa S. Barber, ND, MSc²; Alan M. Hirahara, MD, FRCSC³; Terrance Manning II, ND, RMSK, MA⁴; Wyatt Andersen, MSHS, ATC⁵; Rahul Desai, MD⁶; Alyssa Speciale, MD⁷; Brice Blatz, MD, MA⁸
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Panero, A., Barber, M.S., et al., 2025. Musculoskeletal Ultrasound in Orthobiologic Care—From Harvest to Injection: A Cadaveric Validation of Ultrasound-Guided Lipoaspiration for Orthobiologic Applications. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6606
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6606
ISSN 2375-1924
ABSTRACT
Background: Musculoskeletal (MSK) ultrasound is increasingly used for point-of-care diagnosis, delivery of therapeutics like orthobiologics, and follow-up evaluations. However, it is underutilized for the harvest of orthobiologic tissue such as adipose, leading to concerns about safety and procedure accuracy. Adipose tissue is a valuable orthobiologic source due to its regenerative potential, high progenitor cell content, and resistance to age-related decline. Safe, targeted harvesting is critical to maintain graft integrity.
Objective: To describe and validate a novel, ultrasound-guided manual lipoaspiration technique for precise and safe harvest of abdominal adipose tissue for orthobiologic use.
Methods: High-frequency (4–13 MHz) interventional ultrasound was used to identify the mantle and deep layers of the anterior abdominal subcutaneous compartment in three cadaveric models. Manual lipoaspiration was performed using a blunt-tip cannula under real-time ultrasound guidance. Procedural validation included colored latex injections to the harvest sites to assess for anatomical accuracy, as well as dissection and microscopic evaluation of the aspirated tissue to confirm adipose identity.
Results: All six aspiration sites yielded adipose tissue without evidence of muscle or peritoneal involvement. Microscopy revealed classic adipocyte morphology, and density centrifugation confirmed low-density tissue, consistent with adipose. Colored latex remained confined to the intended adipose compartments on dissection, confirming harvest precision.
Conclusion: This cadaveric study demonstrates that manual lipoaspiration performed under ultrasound guidance allows for safe and anatomically accurate adipose harvesting, preserving adjacent structures and tissue integrity. MSK ultrasound supports a full orthobiologic workflow—from harvest to injection and post-treatment monitoring—enhancing procedural safety, biologic effectiveness, and standardization in regenerative care. Further clinical research is needed to assess graft viability and therapeutic outcomes in live patients.
Keywords
Ultrasound-Guided Lipoaspiration; Adipose Tissue Harvest; Orthobiologics; Regenerative Medicine; Musculoskeletal Ultrasound; Image-Guided Procedures
Introduction
The incorporation of musculoskeletal (MSK) ultrasound into orthobiologic therapies has transformed the landscape of orthopedic and sports medicine. MSK ultrasound informs multiple phases of patient care in a time-efficient manner: diagnosis, image-guided therapeutic delivery, and post-treatment reassessment. Point-of-care diagnostic ultrasound allows clinicians to provide same-day answers for musculoskeletal injuries, minimize reliance on more expensive imaging modalities, and circumvent patient challenges such as claustrophobia and implant contraindications with MRI.
In therapeutic applications, MSK ultrasound enables precise delivery of orthobiologic agents—including platelet-rich plasma (PRP), bone marrow concentrate (BMC), and micronized adipose tissue (m-adipose)—to target structures such as tendons, ligaments, muscles, and intra-articular spaces. MSK ultrasound also facilitates more advanced interventions, including ultrasound-guided nerve hydrodissections, implant placement, and assistance in procedures such as ligament reconstructions and carpal tunnel releases. While the value of ultrasound in improving injection accuracy and minimizing procedural side effects is well established, its role in tissue harvesting for orthobiologic purposes remains underrecognized.
Ultrasound-guided vascular access has long been employed in emergency departments and surgical settings and can aid interventional orthopedic procedures in patients with difficult venous access for PRP preparations. The use of ultrasound for bone marrow aspiration in orthobiologic settings has also been described. However, ultrasound remains underutilized for harvesting cellular therapies, particularly adipose tissue, which is most commonly obtained by palpation-based lipoaspiration. Palpation-guided methods raise concerns regarding safety, precision, and reproducibility, especially in specialties that do not routinely perform lipoaspiration, such as orthopedics or sports medicine.
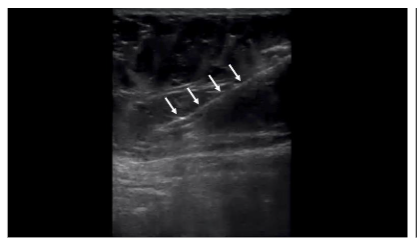
We therefore describe the use of high-frequency (4–13 MHz) interventional ultrasound to directly visualize tissue layers while performing manual adipose tissue harvest for orthobiologic applications. The anterior abdominal wall is widely considered an optimal donor site. Regions containing adipose in the anterior abdomen can be divided into visceral and subcutaneous compartments. The subcutaneous adipose can be further separated into the apical, mantle, and deep layers. High-frequency ultrasound (4–13 MHz) provides real-time visualization of tissue layers, enabling clinicians to localize adipose-rich zones and guide cannula placement while avoiding neurovascular structures.
The layers of the abdomen can be evaluated from superficial to deep: the epidermis and dermis appear thin and hyperechoic; the adipose layer presents as hypoechoic and lobular with hyperechoic fibrous septa. Deep to the adipose layer, muscle appears relatively hypoechoic, separated by fine hyperechoic perimysium, while the bowel wall appears relatively hyperechoic with a hypoechoic lumen and is easily identified by its movement while the patient is at rest.
A “safe zone” for harvest can be defined as the area between the superficial apical layer and the deeper aponeurosis of the abdominal musculature in the upper abdomen, and Scarpa’s fascia in the lower abdomen. Depth markings on the ultrasound screen allow for measurement of the harvest field size. Dynamic scanning can help identify abdominal wall defects or hernias, while Color Doppler is used to locate vascular structures. A linear transducer provides superior resolution at shallow depths, whereas a curved transducer may be selected for larger body habitus or when a wider field of view is preferred. This approach enhances targeted harvest and reduces the risk of complications such as hematoma, seroma, or inadvertent injury to deeper structures.
In contrast, some cosmetic and surgical procedures employ low-frequency ultrasound (<2 MHz) to assist with adipose emulsification and aspiration. However, these methods do not offer direct visualization of tissue removal and may compromise cellular viability due to mechanical or thermal disruption. Since orthobiologic outcomes are directly related to graft quality, manual lipoaspiration under high-frequency ultrasound guidance is preferable when tissue integrity must be preserved.
Taken together, these factors highlight the need for a reproducible, safe, and anatomically precise method for adipose tissue harvest in orthobiologic applications. Building on the diagnostic and therapeutic advantages of musculoskeletal ultrasound, the goal of this investigation was to validate a standardized, image-guided technique that can be incorporated into orthopedic and sports medicine practices. Real-time ultrasound guidance was hypothesized to enable precise extraction of viable adipose tissue while minimizing procedural risk and preserving graft integrity. The technique was applied to three cadaveric specimens, followed by gross microscopy to characterize the harvested tissue and cadaveric dissection to verify accurate targeting of the subcutaneous mantle and deep adipose layers.
Materials and Methods
Decedent protected health information was not accessed or released for the purpose of this study. All human subjects involved in this study provided consent premortem to be involved in cadaver research. A bioskills laboratory was accessed to conduct this study.
Ultrasound Equipment and Setup
All ultrasound evaluations were performed using a SonoSite Edge 2 musculoskeletal (MSK) ultrasound system (FUJIFILM SonoSite, Inc., Bothell, WA) with an HFL50x linear transducer (15–6 MHz). The ultrasound machine was positioned within the operator’s direct line of sight during the procedure. Standard MSK ultrasound settings were used, adjusting gain and depth to optimize visualization of subcutaneous.
Pre-Procedural Planning
Cadavers were positioned supine. A global ultrasound scan was performed, starting at the umbilicus in short axis to the rectus abdominis, extending laterally to visualize the external and internal oblique muscles and their fascial junctions. The dermis, adipose layer, muscle layers, and bowel were identified to demarcate a safe adipose harvest zone.
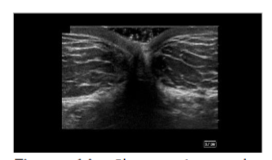
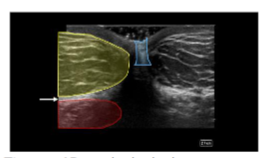
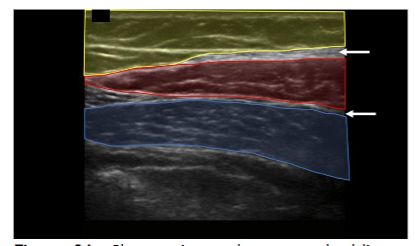
The apical, mantle, and deep layers of the subcutaneous adipose compartment were distinguished, with the mantle and deep layers selected as preferred harvest targets due to their greater thickness. Depth markers on the ultrasound screen were used to measure adipose thickness and define the field size. Areas with potential umbilical hernias or fascial defects were identified and avoided. In clinical practice, Color Doppler can be employed to exclude vascular structures; however, it was not utilized in this cadaveric study.
Lipoaspiration Technique
After sterile preparation and draping, the entry site was located approximately two fingerbreadths inferior and lateral to the umbilicus, superficial to the external oblique muscle.
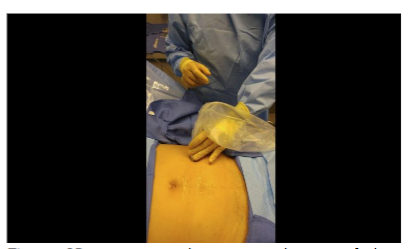
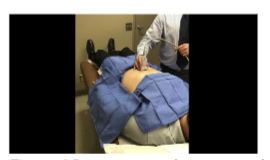
The site was anesthetized using 2–3 mL of local anesthetic. Trajectory markings were made at the skin surface: one perpendicular to the long axis of the body, and two additional markings oriented approximately 45 degrees cephalad and caudally relative to the midline marking.
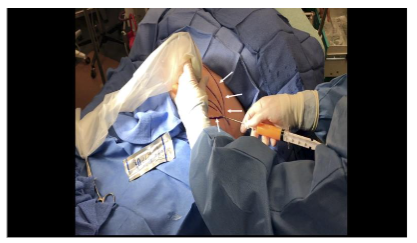
An optional tumescent technique was performed. A solution consisting of 500 mL sterile saline, 50 mL 2% lidocaine, and 1 mL of 1 mg/mL epinephrine was prepared. A small stab incision was made with an 11-gauge scalpel. Using a 17-gauge blunt-tipped infiltration cannula (Gallini Medical), 240 mL of tumescent solution was injected into the target adipose layer bilaterally under ultrasound guidance with the transducer positioned in short axis to the external oblique muscle. A 15–30 minute wait period would be allowed prior to lipoaspiration to facilitate anesthesia onset and vasoconstriction.
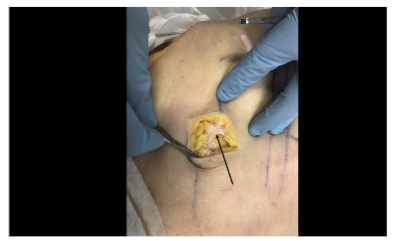
Once the tumescent technique was complete, a 13-gauge blunt-tipped lipoaspiration cannula (Gallini Medical, Mantova, Italy) attached to a 20 mL VacLok syringe (Merit Medical Systems, Inc., South Jordan, UT) was inserted using an in-plane technique under real-time ultrasound visualization. The cannula was introduced at an angle of less than 30 degrees to the skin surface. Negative pressure was created by retracting and locking the syringe plunger. Gentle lateral-to-medial and medial-to-lateral sweeping motions were used following the pre-planned trajectories to aspirate adipose tissue while always maintaining visualization of the cannula on ultrasound. If cannula visualization was lost, the ultrasound transducer was moved to reacquire the image rather than moving the cannula blindly. Approximately 5 mL of adipose tissue was harvested bilaterally from the abdomens of three cadavers for a total of six 5 mL collections (30 mL in total). Once aspirated, the tissue samples were sent to a laboratory for analysis.
Cadaveric Validation Following lipoaspiration, the cannula was maintained in the harvest site, and 3 mL of colored latex was injected under ultrasound guidance into the aspirated adipose layer. Subsequent cadaveric dissection was performed to assess latex distribution and to verify that the aspiration tracts remained confined to the adipose tissue, without penetration into muscular or peritoneal structures.
Laboratory Analysis
Aspirated samples were suspended in 0.9% saline and centrifuged at 300xg for 5 minutes. Gross microscopic analysis was performed at 20x and 40x magnification to identify characteristic adipose morphology. Density centrifugation analysis confirmed the harvested tissue as adipose based on buoyancy above the saline layer.
Results
Microscopic analysis of the aspirated tissue revealed “grape-like clusters” of very large cells (~100 micron) with no discernable nucleus in the center of the cytoplasm, characteristic of adipose tissue. All six samples following centrifugation had a compacted appearance. Any tissue dispersion would have been indicative of connective tissue rather than adipose tissue. All six samples post-centrifugation had tissue floating above the saline solution, indicating the tissue density was less than 1.0046 g/mL (the density of normal saline solution at 22° C). This lower density tissue is characteristic of adipose, which has a density between 0.925-0.970 g/mL. All samples appeared to contain adipose tissue with no evidence of peritoneal or muscular tissue. With cadaver dissection, all colored latex injections were found within the targeted adipose tissue layer of the anterior abdomen of the three cadavers.
Discussion
Adipose tissue has gained attention as a valuable orthobiologic source due to its abundant supply of mesenchymal progenitor cells, immunomodulatory factors, and growth factors, as well as its structural properties and relatively stable cellular yield across different age groups. Compared to bone marrow, adipose tissue often provides a larger volume of biologic material and demonstrates favorable regenerative properties. Regardless of the intended application—whether structural, reparative, or signaling—the quality and integrity of the adipose tissue graft are paramount. As such, accurate and safe harvesting techniques are critical to preserving graft viability and clinical utility.
This study describes a manual lipoaspiration technique performed under high-frequency, or interventional ultrasound guidance for the purpose of harvesting adipose tissue for orthobiologic use. The technique was demonstrated in three cadaveric models and validated through gross dissection and microscopic analysis. Our findings suggest that ultrasound-guided manual lipoaspiration enables precise extraction of adipose tissue from the mantle and deep layers of the subcutaneous abdominal compartment, without compromising adjacent anatomical structures. By focusing on these layers, we aim to optimize the harvest of progenitor cell-rich tissue while minimizing trauma to overlying dermis or underlying muscle.
The dual validation method used in this study—microscopic analysis to confirm tissue type, and colored latex dissection to verify anatomical targeting—adds rigor to the findings. The latex injections demonstrated that all six aspiration tracts remained within the adipose layers, without extension into muscle or peritoneal tissue. This mirrors validation methods previously used in studies of ultrasound-guided joint, nerve, and spinal procedures and offers proof of concept for the reproducibility of this technique.
Our findings reinforce existing literature on the utility of ultrasound in adipose harvesting. Lavler et al. demonstrated improved harvest precision in equine models using ultrasound, while Duran Vega et al. and Nagy et al. reported safer adipose and vascular access in human patients. Harutyunyan et al. found that preoperative ultrasound assessment of adipose thickness correlated strongly with lipoaspirate volumes, supporting ultrasound’s role in procedural planning. Our study is the first to validate the accuracy of manual ultrasound-guided lipoaspiration using both cadaveric dissection and microscopic confirmation—establishing a new benchmark for procedural precision in musculoskeletal and regenerative medicine.
The concept of “from harvest to injection” presented in this study adds to the growing body of work emphasizing ultrasound’s role across the full procedural continuum of orthobiologic care—from tissue harvest to targeted delivery to outcome monitoring. Musculoskeletal (MSK) ultrasound plays a pivotal role at each phase of this continuum: (1) guiding safe and targeted harvesting of biologic tissues; (2) enabling precise delivery of biologics into target structures; and (3) supporting post-procedure evaluation of tissue healing and biologic effect. This makes ultrasound not only a diagnostic tool but also a procedural companion throughout the entire orthobiologic workflow.
While various device-assisted lipoaspiration methods have been introduced—including those utilizing low-frequency ultrasound (<2 MHz), lasers, or water jets—these technologies often disrupt adipose tissue, potentially compromising its cellular viability. In contrast, manual lipoaspiration, when performed under real-time ultrasound guidance, allows for a more delicate and tissue-sparing technique better suited to orthobiologic use, where graft integrity and cellular function are critical to clinical success. This integrated use of ultrasound allows for procedural standardization and provides an important framework for clinical best practices.
Ultrasound guidance for difficult venous access during PRP preparation is becoming standard practice in many settings, improving success rates and reducing complications. Similarly, ultrasound-guided bone marrow aspiration has been described as a safer, more accurate alternative to landmark-based techniques. The application of ultrasound to adipose tissue harvesting, as explored in this study, represents a further evolution of the modality’s versatility. It allows for real-time differentiation of subcutaneous tissue layers, direct cannula visualization, and avoidance of vascular and muscular structures—all while enabling precise targeting of adipose-rich zones. The reproducibility and safety of ultrasound-guided techniques may also facilitate wider adoption of orthobiologic therapies across diverse clinical environments and improve consistency of biologic harvests.
A study limitation is the use of a cadaveric model. While ideal for anatomical validation, this model does not replicate clinical conditions such as perfusion, immune response, or patient feedback. Additionally, no comparison was made between ultrasound-guided and palpation-guided techniques, nor was the post-harvest biologic activity of the adipose graft assessed. While the downstream clinical use of harvested adipose tissue was beyond the scope of this study, clinicians should be familiar with current FDA guidelines regarding homologous use and minimal manipulation of human tissues. Future studies are warranted to assess clinical safety, graft viability, and long-term patient outcomes in live subjects undergoing ultrasound-guided adipose harvesting.
Conclusions
This study presents a novel ultrasound-guided manual lipoaspiration technique that enables safe and accurate harvesting of adipose tissue from the abdominal subcutaneous compartment for orthobiologic use. Interventional ultrasound allows for precise cannula placement, protects adjacent structures, and facilitates high-fidelity graft acquisition. These findings support the growing use of MSK ultrasound across the entire orthobiologic treatment continuum—from harvest to injection and re-evaluation. As orthobiologic medicine continues to expand, incorporating ultrasound throughout the full course of biologic treatment may standardize procedures, reduce complications, and enhance patient outcomes. Further research is warranted to assess clinical safety in live subjects and to evaluate the regenerative viability of ultrasound-guided adipose grafts.
Ethical Disclosure:
Decedent protected health information was not accessed or released for the purpose of this study. All human subjects involved in this study provided consent premortem to be involved in cadaver research. A bioskills laboratory was accessed to conduct this study.
Conflict of Interest Statement:
None.
Acknowledgements:
Pacific Medical Group provided access to a bioskills laboratory to conduct this study.
Funding Statement:
Alberto Panero – Arthrex Inc – Consultant Tenex Health – Speaker Honoraria Clarius Mobile Health – Stock Options Melissa S. Barber – N/A Alan M. Hirahara – Arthrex Inc – Royalties, consulting fees, and honoraria from Arthrex Inc; Patents. Clarius Mobile Health- Stock Options Terrance Manning II – Apex Biologics – Consultant Kuribl Premium Health – Consultant Wyatt Andersen – N/A Rahul Desai – N/A Alyssa Speciale – Royalties, Springer Publishing Company Brice Blatz – Curamedix – Honoraria ASIPP/TOBI: Honoraria for lectures and travel
References:
- Sivan M, Brown J, Brennan S, Bhakta B. A one-stop approach to the management of soft tissue and degenerative musculoskeletal conditions using clinic-based ultrasonography. Musculoskeletal Care. 2011;9(2):63-68.
- Howard ZD, Noble VE, Marill KA, Sajed D, Rodrigues M, Bertuzzi B, Liteplo AS. Bedside ultrasound maximizes patient satisfaction. J Emerg Med. 2014 Jan;46(1):46-53. doi: 10.1016/j.jemermed.2013.05.044. Epub 2013 Aug 12. PMID: 23942153.
- Panero AJ, Hirahara AM. A guide to ultrasound of the shoulder, Part 2: The diagnostic evaluation. Am J Orthop. 2015; 45(4):233-238.
- Hirahara AM, Panero AJ. A guide to ultrasound of the shoulder, part 3: interventional and procedural uses. Am J Orthop. 2016;45(7):440-445.
- Finnoff JT, Hall MM, Adams E, et al. American Medical Society for Sports Medicine (AMSSM) position statement: Interventional musculoskeletal ultrasound in sports medicine. Br J Sports Med. 2015;49(3):145-150.
- Partington P, Broome G. Diagnostic injection around the shoulder: Hit and miss? A cadaveric study of injection accuracy. J Shoulder Elbow Surg. 1998;7(2):147-150.
- Rutten M, Maresch B, Jager G, de Waal Malefijt M. Injection of the subacromial-subdeltoid bursa: Blind or ultrasound-guided? Acta Orthop. 2007;78(2):254-257.
- Kang M, Rizio L, Prybicien M, Middlemas D, Blacksin M. The accuracy of subacromial corticosteroid injections: A comparison of multiple methods. J Shoulder Elbow Surg. 2008;17(1 Suppl):61S-66S.
- Yamakado K. The targeting accuracy of subacromial injection to the shoulder: An arthrographic evaluation. Arthroscopy. 2002;19(8):887-891.
- Henkus HE, Cobben M, Coerkamp E, Nelissen R, van Arkel E. The accuracy of subacromial injections: A prospective randomized magnetic resonance imaging study. Arthroscopy. 2006;22(3):277-282.
- Sethi P, El Attrache N. Accuracy of intra-articular injection of the glenohumeral joint: A cadaveric study. Orthopedics. 2006;29(2):149-152.
- Naredo E, Cabero F, Beneyto P, et al. A randomized comparative study of short term response to blind injection versus sonographic-guided injection of local corticosteroids in patients with painful shoulder. J Rheumatol. 2004; 31(2):308-314.
- Royall NA, Farrin E, Bahner DP, Stanislaw PA. Ultrasound-assisted musculoskeletal procedures: A practical overview of current literature. World J Orthop. 2011;2(7):57-66.
- Aly AR, Rajasekaran S, Ashworth N. Ultrasound-guided shoulder girdle injections are more accurate and more effective than landmark-guided injections: a systematic review and meta-analysis. Br J Sports Med. 2015;49(16):1042-1049.
- Hirahara AM, Panero A, Andersen WJ. An MRI Analysis of the Pelvis to Determine the Ideal Method for Ultrasound-Guided Bone Marrow Aspiration from the Iliac Crest. Am J Orthop (Belle Mead NJ). 2018;47(5):10.12788/ajo.2018.0038. doi:10.12788/ajo.2018.0038
- Asakura Y, Kinoshita M, Kasuya Y, Sakuma S, Ozaki M. Ultrasound-guided sternal bone marrow aspiration. Blood Res. 2017 Jun;52(2):148-150. doi: 10.5045/br.2017.52.2.148. Epub 2017 Jun 22. PMID: 28698857; PMCID: PMC5503898.
- Hirahara AM, Andersen WJ. Ultrasound-Guided Percutaneous Reconstruction of the anterolateral Ligament: Surgical Technique and Case Report. Am J Orthop. 2016;45(7):418-460.
- Hirahara AM, Andersen WJ. Ultrasound-Guided Percutaneous Repair of Medial Patellofemoral Ligament: Surgical Technique and Outcomes. Am J Orthop. 2017;46(3):152-157.
- Hirahara AM, Andersen WJ, Panero AJ. Superior Capsular Reconstruction: Clinical Outcomes After Minimum 2-Year Follow-Up. Am J Orthop. 2017;46(6):266-278.
- Saugel B, Scheeren TWL, Teboul JL. Ultrasound-guided central venous catheter placement: a structured review and recommendations for clinical practice. Crit Care. 2017 Aug 28;21(1):225. doi: 10.1186/s13054-017-1814-y. PMID: 28844205; PMCID: PMC5572160.
- Lawver J, Thaler R. Ultrasound-Guided Lipoaspiration for Mesenchymal Stromal Cell Harvest in the Horse. Equine Vet Educ (2016) 28 (1) 23-29. Doi: 10.1111/eve.12398
- Cs Nagy G, Verwiebe R, Wunsch M. Staged ultrasound-guided liposuction for hidden arteriovenous fistulas in obese patients. Vasa. 2018 Aug;47(5):403-407. doi:10.1024/0301-1526/a000719. Epub 2018 Jul 19. PMID: 30022718.
- Klein JA. Tumescent Technique: Tumescent Anesthesia & Microcannular Liposuction. 1st ed. Mosby; 2000.
- Jacobson JA. Fundamentals of Musculoskeletal Ultrasound. 2nd ed. Elsevier Saunders; 2013.
- Enriquez JL, Wu TS. An introduction to ultrasound equipment and knobology. Crit Care Clin. 2014;30(1):25-45, v. doi:10.1016/j.ccc.2013.08.006
- Viaro MSS, Danilla S, Cansanção AL, Viaro PS. Ultra HD Liposuction: Enhancing Abdominal Etching Using Ultrasound-Guided Rectus Abdominis Fat Transfer (UGRAFT). Plast Reconstr Surg Glob Open. 2020;8(5):e2818. doi:10.1097/GOX.0000000000002818
- Duscher D, Atashroo D, Maan ZN, et al. Ultrasound-Assisted Liposuction Does Not Compromise the Regenerative Potential of Adipose-Derived Stem Cells. Stem Cells Transl Med. 2016;5(2):248-257. doi:10.5966/sctm.2015-0064
- Shridharani SM, Broyles JM, Matarasso A. Liposuction devices: technology update. Med Devices (Auckl). 2014;7:241-251. doi:10.2147/MDER.S47322
- Collins PS, Moyer KE. Evidence-Based Practice in Liposuction. Ann Plast Surg. 2018;80(6S Suppl 6):S403-S405. doi:10.1097/SAP.0000000000001325
- Chia CT, Neinstein RM, Theodorou SJ. Evidence-Based Medicine: Liposuction. Plast Reconstr Surg. 2017;139(1):267e-274e. doi:10.1097/PRS.0000000000002859
- Habbema L. Safety of liposuction using exclusively tumescent local anesthesia in 3,240 consecutive cases. Dermatol Surg. 2009;35(11):1728-1735. doi:10.1111/j.1524-4725.2009.01284.x
- Hanke CW, Bernstein G, Bullock S. Safety of tumescent liposuction in 15,336 patients. National survey results. Dermatol Surg. 1995;21(5):459-462. doi:10.1111/j.1524-4725.1995.tb00213.x
- Estes BT, Diekman BO, Gimble JM, Guilak F. Isolation of adipose-derived stem cells and their induction to a chondrogenic phenotype. Nat Protoc. 2010;5(7):1294-1311. doi:10.1038/nprot.2010.81
- Tordjman J. Histology of Adipose Tissue. In: Bastard J-P, Bruno F, eds. Physiology and Physiopathology of Adipose Tissue. Springer; 2012.
- Berry R, Church CD, Gericke MT, Jeffery E, Colman L, Rodeheffer MS. Imaging of adipose tissue. Meth Enzymol. 2014;537:47-73. doi:10.1016/B978-0-12-411619-1.00004-5
- Martin AD, Daniel MZ, Drinkwater DT, Clarys JP. Adipose tissue density, estimated adipose lipid fraction and whole body adiposity in male cadavers. Int J Obes Relat Metab Disord. 1994;18(2):79-83.
- Choi JS, Yang H-J, Kim BS, et al. Fabrication of porous extracellular matrix scaffolds from human adipose tissue. Tissue Eng Part C Methods. 2010;16(3):387-396. doi:10.1089/ten.TEC.2009.0276
- Choi JS, Kim BS, Kim JY, et al. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J Biomed Mater Res A. 2011;97(3):292-299. doi:10.1002/jbm.a.33056
- Aust L, Devlin B, Foster SJ, et al. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy. 2004;6(1):7-14. doi:10.1080/14653240310004539
- Bunnell BA, Flaat M, Gagliardi C, Patel B, Ripoll C. Adipose-derived stem cells: isolation, expansion and differentiation. Methods. 2008; 45(2):115-120. doi:10.1016/j.ymeth.2008.03.006
- Jang Y, Koh YG, Choi Y-J, et al. Characterization of adipose tissue-derived stromal vascular fraction for clinical application to cartilage regeneration. In Vitro Cell Dev Biol Anim. 2015; 51(2):142-150. doi:10.1007/s11626-014-9814-6
- Jurgens WJFM, Oedayrajsingh-Varma MJ, Helder MN, et al. Effect of tissue-harvesting site on yield of stem cells derived from adipose tissue: implications for cell-based therapies. Cell Tissue Res. 2008;332(3):415-426. doi:10.1007/s00441-007-0555-7
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. doi:10.1126/science.284.5411.143
- Roato I, Alotto D, Belisario DC, et al. Adipose Derived-Mesenchymal Stem Cells Viability and Differentiating Features for Orthopaedic Reparative Applications: Banking of Adipose Tissue. Stem Cells Int. 2016;2016:4968724. doi:10.1155/2016/4968724
- Schneider S, Unger M, van Griensven M, Balmayor ER. Adipose-derived mesenchymal stem cells from liposuction and resected fat are feasible sources for regenerative medicine. Eur J Med Res. 2017;22(1):17. doi:10.1186/s40001-017-0258-9
- Yoshimura K, Shigeura T, Matsumoto D, et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J Cell Physiol. 2006;208(1):64-76. doi:10.1002/jcp.20636
- Zhu Y, Liu T, Song K, Fan X, Ma X, Cui Z. Adipose-derived stem cell: a better stem cell than BMSC. Cell Biochem Funct. 2008;26(6):664-675. doi:10.1002/cbf.1488
- Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279-4295. doi:10.1091/mbc.E02-02-0105
- Durán Vega HC, Manzaneda R, Flores E, Manfrim C, Morelli H. Deep Back Liposuction: Ultrasound-Guided Deep Fat Liposuction of the Subiliac Crest. Aesthet Surg J. 2023 Jul 20:sjad236. doi: 10.1093/asj/sjad236. Epub ahead of print. PMID: 37474316.
- Harutyunyan R, Gilardino MS, Papanastasiou VW, Jeffries SD, Hemmerling TM. Ultrasound Calculation of Fat Volume for Liposuction: A Clinical Software Validation. Aesthet Surg J. 2023 Sep 14;43(10):1150-1158. doi: 10.1093/asj/sjad121. PMID: 37099036.
- Duscher D, Atashroo D, Maan ZN, et al. Ultrasound-Assisted Liposuction Does Not Compromise the Regenerative Potential of Adipose-Derived Stem Cells. Stem Cells Transl Med. 2016;5(2):248-257. doi:10.5966/sctm.2015-0064
- Shridharani SM, Broyles JM, Matarasso A. Liposuction devices: technology update. Med Devices (Auckl). 2014;7:241-251. doi:10.2147/MDER.S47322
- Collins PS, Moyer KE. Evidence-Based Practice in Liposuction. Ann Plast Surg. 2018;80(6S Suppl 6):S403-S405. doi:10.1097/SAP.0000000000001325
- Chia CT, Neinstein RM, Theodorou SJ. Evidence-Based Medicine: Liposuction. Plast Reconstr Surg. 2017;139(1):267e-274e. doi:10.1097/PRS.0000000000002859
- Habbema L. Safety of liposuction using exclusively tumescent local anesthesia in 3,240 consecutive cases. Dermatol Surg. 2009;35(11):1728-1735. doi:10.1111/j.1524-4725.2009.01284.x
- Hanke CW, Bernstein G, Bullock S. Safety of tumescent liposuction in 15,336 patients. National survey results. Dermatol Surg. 1995;21(5):459-462. doi:10.1111/j.1524-4725.1995.tb00213.x
- Estes BT, Diekman BO, Gimble JM, Guilak F. Isolation of adipose-derived stem cells and their induction to a chondrogenic phenotype. Nat Protoc. 2010;5(7):1294-1311. doi:10.1038/nprot.2010.81
- Tordjman J. Histology of Adipose Tissue. In: Bastard J-P, Bruno F, eds. Physiology and Physiopathology of Adipose Tissue. Springer; 2012.
- Berry R, Church CD, Gericke MT, Jeffery E, Colman L, Rodeheffer MS. Imaging of adipose tissue. Meth Enzymol. 2014;537:47-73. doi:10.1016/B978-0-12-411619-1.00004-5
- Martin AD, Daniel MZ, Drinkwater DT, Clarys JP. Adipose tissue density, estimated adipose lipid fraction and whole body adiposity in male cadavers. Int J Obes Relat Metab Disord. 1994;18(2):79-83.
- Choi JS, Yang H-J, Kim BS, et al. Fabrication of porous extracellular matrix scaffolds from human adipose tissue. Tissue Eng Part C Methods. 2010;16(3):387-396. doi:10.1089/ten.TEC.2009.0276
- Choi JS, Kim BS, Kim JY, et al. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J Biomed Mater Res A. 2011;97(3):292-299. doi:10.1002/jbm.a.33056
- Aust L, Devlin B, Foster SJ, et al. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy. 2004;6(1):7-14. doi:10.1080/14653240310004539
- Bunnell BA, Flaat M, Gagliardi C, Patel B, Ripoll C. Adipose-derived stem cells: isolation, expansion and differentiation. Methods. 2008; 45(2):115-120. doi:10.1016/j.ymeth.2008.03.006
- Jang Y, Koh YG, Choi Y-J, et al. Characterization of adipose tissue-derived stromal vascular fraction for clinical application to cartilage regeneration. In Vitro Cell Dev Biol Anim. 2015; 51(2):142-150. doi:10.1007/s11626-014-9814-6
- Jurgens WJFM, Oedayrajsingh-Varma MJ, Helder MN, et al. Effect of tissue-harvesting site on yield of stem cells derived from adipose tissue: implications for cell-based therapies. Cell Tissue Res. 2008;332(3):415-426. doi:10.1007/s00441-007-0555-7
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. doi:10.1126/science.284.5411.143
- Roato I, Alotto D, Belisario DC, et al. Adipose Derived-Mesenchymal Stem Cells Viability and Differentiating Features for Orthopaedic Reparative Applications: Banking of Adipose Tissue. Stem Cells Int. 2016;2016:4968724. doi:10.1155/2016/4968724
- Schneider S, Unger M, van Griensven M, Balmayor ER. Adipose-derived mesenchymal stem cells from liposuction and resected fat are feasible sources for regenerative medicine. Eur J Med Res. 2017;22(1):17. doi:10.1186/s40001-017-0258-9
- Yoshimura K, Shigeura T, Matsumoto D, et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J Cell Physiol. 2006;208(1):64-76. doi:10.1002/jcp.20636
- Zhu Y, Liu T, Song K, Fan X, Ma X, Cui Z. Adipose-derived stem cell: a better stem cell than BMSC. Cell Biochem Funct. 2008;26(6):664-675. doi:10.1002/cbf.1488
- Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279-4295. doi:10.1091/mbc.E02-02-0105
- Durán Vega HC, Manzaneda R, Flores E, Manfrim C, Morelli H. Deep Back Liposuction: Ultrasound-Guided Deep Fat Liposuction of the Subiliac Crest. Aesthet Surg J. 2023 Jul 20:sjad236. doi: 10.1093/asj/sjad236. Epub ahead of print. PMID: 37474316.
- Harutyunyan R, Gilardino MS, Papanastasiou VW, Jeffries SD, Hemmerling TM. Ultrasound Calculation of Fat Volume for Liposuction: A Clinical Software Validation. Aesthet Surg J. 2023 Sep 14;43(10):1150-1158. doi: 10.1093/asj/sjad121. PMID: 37099036.




