Unusual Diffuse Large B-cell Lymphoma: Diagnostic Insights
Unusual Presentation of Diffuse large B-cell Lymphoma (DLBCL) with diagnostic challenges
Dr. Seema Devi1, Dr. Abhishek Kumar2, Dr. Prithvi Minkash3
- Additional Professor, Department of Radiation Oncology, IGIMS, Patna. Mobile no.: 7250925205
- Senior resident, Department of Radiation Oncology, IGIMS, Patna.
- PG 2nd Year, Department of Radiation Oncology, IGIMS, Patna.
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Devi, S., et al., 2024. Unusual Presentation of Diffuse large β-cell Lymphoma (DLBCL) Lymphoma with diagnostic challenges. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5657
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5657
ISSN 2375-1924
ABSTRACT
Introduction:
Worldwide diffused β cell lymphoma is the commonest type of non-Hodgkin lymphoma about 31% of all non-Hodgkin Lymphoma. Median age reported for diffuse large β-cell lymphoma 60 to 70 years of age. Extra nodal diffuse large β-cell lymphoma reported in about 30% cases and most common site were found gastrointestinal tract skin and soft tissue bone or Genito urinary tract, head and neck region. Extra nodal non-Hodgkin lymphoma only represented in 10% cases.
Case Study:
Patients presented with complaint of focal neurological deficit vomiting, headache nausea and status seizures with behavioural and mental change and 27-year-old male presented with difficulty pain while walking for last 1.5 months.
Discussion:
Musculoskeletal Lymphomas are predominantly a manifestation of lymphoma dissemination. Extra nodal Lymphomas commonly involved skin, head and neck and gastrointestinal tract. Lymphomas rarely involved skeletal system. Most common radiological finding with Extra-Nodal lymphoma are blastic lesion and lytic lesion, sometimes mixed lesion with lytic and blastic. Primary central nervous system lymphoma usually originates from cerebellum, spinal cord, Pia matter, Retina or optic nerve. Manifestation of lymphoma in skeletal muscles is rare, usually they involve gluteal muscles and pelvis.
Conclusion:
Primary Central Nervous System (CNS) Lymphoma often misdiagnosed as Brain tumour and this delays proper treatment and confirmation of diagnosis. Immunohisto-chemistry examination is mandatory to achieve confirm diagnosis.
Keywords:
Lymphoma, β-cell lymphoma, CNS Lymphoma.
Introduction
Worldwide diffused β-cell lymphoma is the commonest type of non-Hodgkin lymphoma about 31% of all non-Hodgkin Lymphoma¹.
Median age reported for diffuse large β-cell lymphoma (DLBCL) is 60 to 70 years of age. Extra nodal DLBCL reported in about 30% cases and most common site were found to be gastrointestinal tract skin and soft tissue bone or Genito urinary tract, head and neck region².
Extra nodal non-Hodgkin lymphoma only represented in 10% cases.
Primary central nervous system (CNS) lymphoma is rare non-Hodgkin lymphoma limited to central nervous system³.
They represent less than 1% of all non-Hodgkin lymphoma and 2% to 3% of all brain tumour⁴.
Primary central nervous system lymphoma mostly represented by DLBCL. It is characterised by single or multiple contrast enhancing lesion mainly at supratentorial region³.
In recent years, rising trend of incidence has been reported specially in older patients, incidence rate 0.5 per 100000 per year reported⁵.
These tumours usually have better response with chemotherapy and radiotherapy as compared to brain tumour, but survival reported inferior as compare to other lymphoma cases⁶.
Primary Central Nervous System Lymphoma commonly presents as sub-acute typical symptoms like headache, focal neurological deficit, nausea, vomiting, cognitive or personality changes seizure are less frequent (10-20%) as compared to brain tumour⁷. Sometimes lepto meningeal involvement also reported about in 15-20%⁸. Other symptoms may occur due to area of involvement like ocular leads to blurring of vision in 30% cases⁹.
Manifestation of Lymphoma in skeletal muscles is rare. Usually, they involve gluteal muscles and pelvis. Only 0.5% of extra nodal Lymphomas diagnosed as a primary skeletal muscle Lymphoma. Involvement of skeletal muscles and bone may mimic as a soft tissue sarcoma, bone tumours or metastasis cancerous which have different diagnostic criteria and treatment¹⁰. Involvement of muscle and metastasis to brain and other parts is extremely uncommon. Accurate diagnosis of these cases is a challenge for clinicians. Pathologist and Radiologist despite the advancement of radiological and pathological devices. Here we present two cases of DBCL with rare presentation with the belief that our cases will add evidence to diagnose DBCL with rate sites of presentation.
Case Study
CASE-1
A 35-year-old male presented with sudden onset of headache and vertigo with loss of consciousness one month ago. After few minutes he gained consciousness but developed Hemiparesis and had multiple episodes of seizures. On Magnetic resonance imaging (MRI) 33×34×31 mm³ mass with perilesional oedema and mass effect.
Right frontal Parietal craniotomy with excisional Biopsy done which suggested Papillary meningioma. After immune-histochemistry confirmed as a diffuse B-cell. Lymphoma with CD 20, CD 45, KI 67.70-75%.
Positron emission tomography (PET) scan shows post-operative evidence of right frontal parietal craniotomy. Non-FDG avid mild to moderate vasogenic oedema seen in right frontal lobe with no significant mass effect noted. However, no abnormal focal fluorodeoxyglucose (FDG) avid/enhancing lesion seen in operated bed/right frontal lobe.
Patient has been planned for Radiotherapy, now he is on follow up.
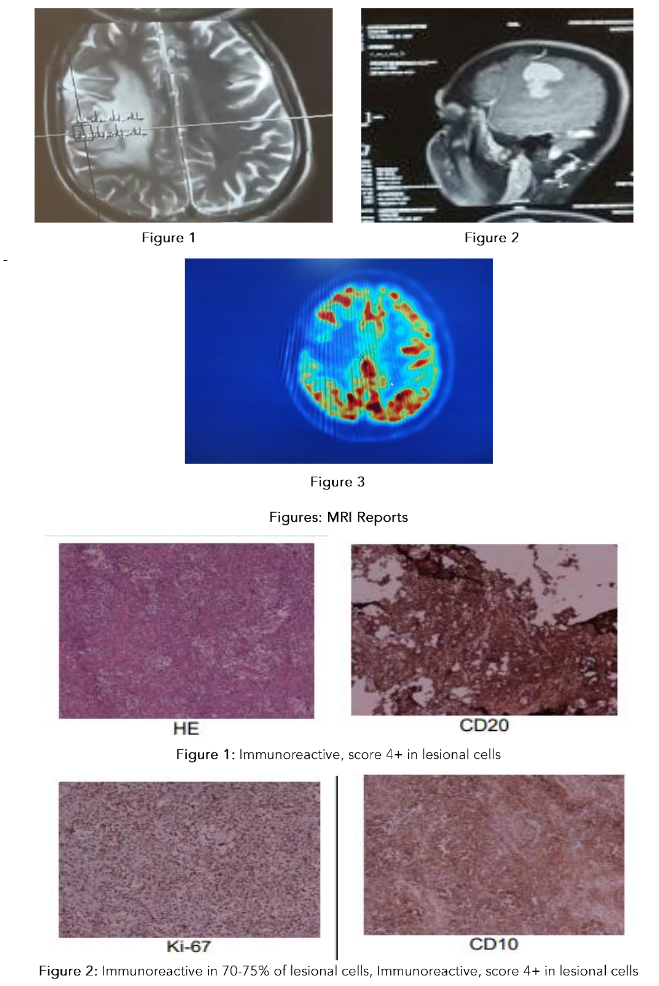
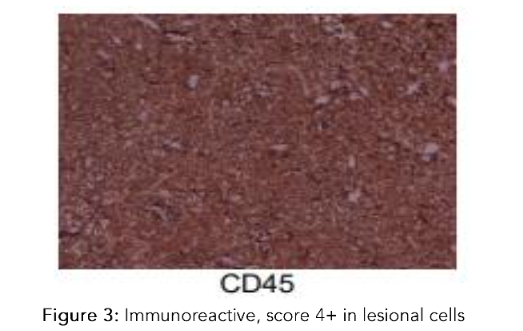
Table 2: HISTOPATHOLOGY FINAL DIAGNOSIS PANEL (Immunohistochemistry)
| IHC MARKER(S) | RESULT |
|---|---|
| CD20 | Immunoreactive, score 4+ in lesional cells. |
| CD45 | Immunoreactive, score 4+ in lesional cells |
| Ki-67 | Immunoreactive in 70-75% of lesional cells |
| CD-10 | Immunoreactive, score 4+ in lesional cells |
| BCL-6 | Immunoreactive, score 2+ in lesional cells |
| MUM-1 | Immunoreactive, score 1+ in lesional cells |
| CMYC | Immunoreactive in 1-2% |
| BCL-2 | Immunoreactive, score 1+ in lesional cells |
| PAX-5 | Immunoreactive, score 4+ in lesional cells. |
CASE-2
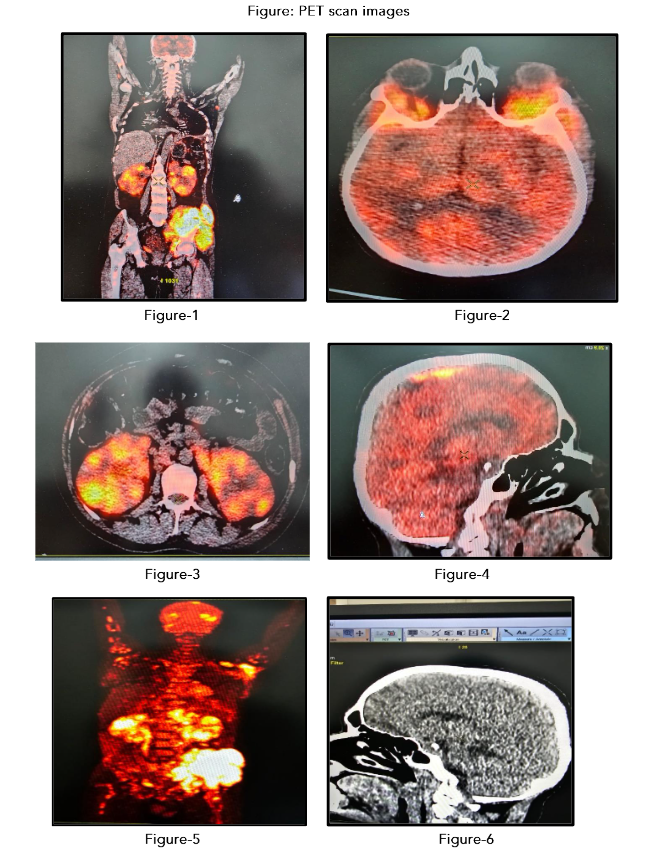
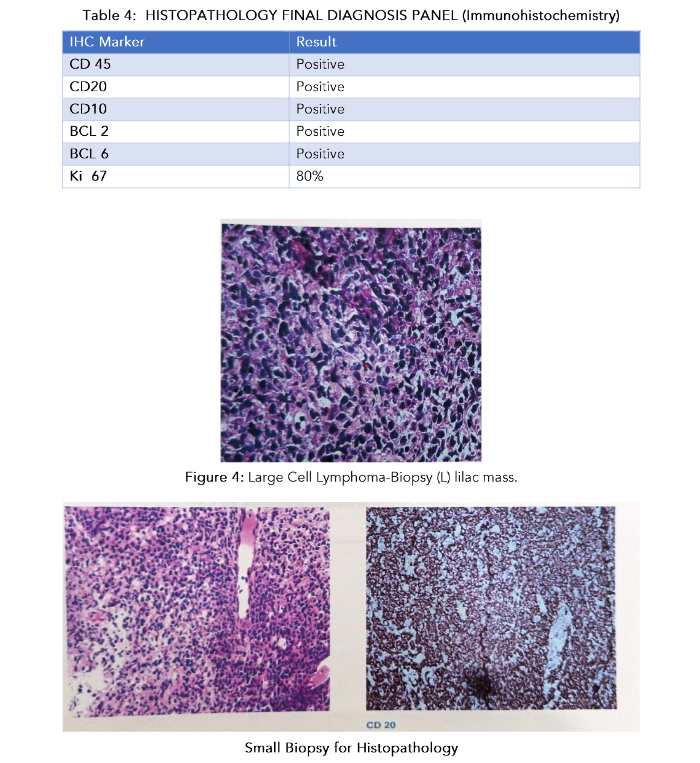
A 27-year-old male presented with difficulty and pain during walking for last one and half month. On examination, tender swelling was felt in left pelvis. Core biopsy from left iliac mass shown Large cell Lymphoma. On immune histochemistry examination it confirmed as diffuse B-cell lymphoma shown CD 45, CD 20, CD 10, BCL 2, BCL 6 positive. On MRI Brain and skeletal system shown. PET scan shown varying degree of Dural thickening and enhancement is seen overlying both cerebral hemispheres, predominantly overlying partial lobe and along posterior fix, Lesion in the posterior falx is compressing/invading posterior aspect of superior sagittal sinuses.
Fairly large soft tissue component is seen superolateral quadrant of both orbits involving lacrimal gland also altered signal intensity of lateral wall of both orbits are seen, soft tissue component is also seen in periorbital region and superficial tissue of scalp overlying left parietal region. Both cerebral hemispheres are normal in volume and signal intensity on all pulse sequence.
Altered marrow signal intensity lesion is seen involving the left with large associated soft tissue mass measuring 13.5×11.6×7.4cm size seen involving the gluteus muscle as well as left ileo psoas muscle. There is extension until the anterior aspect of left sacroiliac joint. The lesion is hypointense on T1 and hyperintense on T2 STIR and post contrast showing moderate heterogeneous enhancement. The soft tissue thickening showing loss of fat plane with femoral and external ilia vessels however flow voids are maintained. Altered marrow signal intensity lesion with patchy enhancement in the left sacral ala around bilateral hip joints, proximal femurs, bilateral acetabulum, right iliac bone. On STIR images patchy hyperintensity is also seen in few lumbar vertebrates. Mild patchy enhancement is seen in the CVJ (Carme vertebral junction).
Patient has been planned for chemotherapy with R-CHOP – 6 Cycle.
Figure: PET scan images
Discussion
Musculoskeletal Lymphomas are predominantly a manifestation of lymphoma dissemination¹¹˒¹². Extra nodal Lymphomas commonly involved skin, head and neck and gastrointestinal tract. Lymphomas rarely involve skeletal system¹³˒¹⁴. Most common radiological finding with Extra-Nodal lymphoma are Blastic lesion and lytic lesion, sometimes mixed lesion with lytic and blastic¹¹˒¹⁵˒¹⁶. Involvement of muscle and metastasis to Brain and other part is extremely uncommon. Accurate diagnosis of these cases is a challenge for Clinicians, Pathologists and Radiologists despite the advancement of Radiological and Pathological devices¹⁶. Primary central nervous system lymphoma usually originates from cerebellum, spinal cord, Pia matter, Retina or optic nerve¹⁷˒¹⁸. Usually, Primary CNS Lymphoma characterized by supratentorial localization and clinical manifestation are very similar to other intracranial tumour, with the symptoms of intracranial tumours like headache, vomiting and focal neurological deficit¹⁷.
Commonest location in MRI images found periventricular white matter, basal ganglia, or corpus callosum and less commonly areas are cerebellum, brainstem, and spinal cord¹⁴. In 70% cases single lesion presented multifocal lesion presented more frequently in immune compromised patient. Commonly differential diagnosis found with high grade glioma and less often metastasis or infectious granuloma¹⁸. Tumour diagnostic finding of primary intracranial lymphoma are hypointense T1 or hyperintense T2 signals. On diffusion weighted imagining there may be homogeneous or heterogeneous and high circular signals and PET scan findings may reveal single or multiple high metabolic signals¹⁹˒²⁷. Role of surgery in primary intracranial lymphoma is debatable²⁸˒²⁹. Diagnosis is confirmed by pathological analysis. MRI stereotactic biopsy can be considered by confirming the diagnosis³⁰. Radiation and chemotherapy can be considered for treatment. A previous study reported 90% response rate with high dose methotrexate and procarbazine and can provide survival up to 5 years²⁴. Other combination are used in these cases are Temozolomides high dose of methotrexate, Rituximab³¹˒³². Temozolomides can cross blood brain barrier and have a high oral bioavailability. The national comprehensive cancer network has recommended the use of Temozolomides in refractory intracranial lymphoma³³˒³⁴. Radiotherapy in Intracranial lymphoma usually prescribed as 40 to 50 GY to whole brain followed by boost of 10gy in 5 fraction to localized area³⁵. With thorough intracranial lymphoma survival is 3-6 month but with chemotherapy survival can be progression free and overall survival can be improved. 5-year survival in these cases remains 20-25%³⁶. Recurrence reported 35-60% within 2 years and overall survival reported about 8-18 months³⁷.
Mamorska-Dyga et al. reported a case which was initially diagnosed as a sarcoma on physical and radiological examination, after biopsy it was diagnosed as DLBCL³⁸. Another case was diagnosed as a sarcoma reported by Mayo at al., who presented as a subcutaneous swelling on shoulder, diagnosed as a case of DLBCL after surgical excision³⁹. Gupta et al. reported a case of cutaneous Lymphoma which was presented as a swelling in inguinal swelling and mimicked as a sarcoma⁴⁰. Primary extra nodal Lymphoma is rare about 0.1 to 0.5 of all non-Hodgkin’s Lymphoma cases⁴¹˒⁴². Generally skeletal muscle lesions involves in lower extremities and thighs⁴³. It can occur after treatment at lower extremities⁴⁴. Sometime skeletal muscle Lymphoma shows triphasic lesion which are most aggressive and arises from T-cell and have poor prognosis⁴⁵. Sometimes DLBCL shows aggressive growth pattern and present as a large necrotic masses that may infiltrate and surround’s structure¹³˒⁴⁶˒⁴⁷. A recent study shown association of SUV (max) on FDC 18 PET and survival⁴⁸˒⁴⁹. SUV (max) can be considered as a good Prognostic factor (20) for progression and treatment response⁴⁸˒⁴⁹. For confirmation of diagnosis, histological examination of specimen and imaging studies are required. A recently published studies shown PET-CT fusion is more. Sensitive and Specific (88% and 100%) as compared to CT scan (50% and 90%)⁵⁰˒⁵². Utkan et al. reported a 68-year-old male presented with mass in (R) bullock for last 2 month. After Biopsy and histopathological examination of this Inguinal Lymph nodes and mass shown tuberous sclerosing Hodgkin disease⁵³. Present a case of 52-year-old female who presented with gluteus for last 21 days, CT scan shows mass in (L) gluteus muscle. After Biopsy, she was diagnosed a case of DLBCL.
Scally et al reported 51-year-old female presented with pain in left hip and diagnosed as Sciatica, a lump approved on her left hip with severe pain, CT showed bullock mass and after biopsy she was diagnosed as B cell NHL⁵⁵.
Diagnostic Challenges
Immune response of Lymphoid tissue causes Proliferation and differentiation of various immune cells associated with non-Hodgkin Lymphoma. Malignant transformation of immune system usually presented in the form of solid tumours of Lymphoid organs and tissues like Lymph nodes, Tonsil, spleen and bone marrow. These tumours can originate from any part of Lymphoid tissue and other organs⁵⁶.
Primary lesion occurs in any extra nodal is called as extra nodal Lymphoma. Commonly involved extra nodal tissues are gastrointestinal tract, Waldeger’s ring, nasal cavity. It rarely involves mediastinal soft tissues⁵³˒⁵⁵.
Conclusion
Neoplasm of extremity many times misdiagnosed as a sarcoma and confirmation of diagnosis delay the treatment and chances to treat and survival decreases as the time prolongs for confirmation of diagnosis. Aggressive disease reduces the chances of survival. Primary CNS Lymphoma often misdiagnosed as Brain tumour and this delays proper treatment and confirmation of diagnosis. Immuno histo-chemistry examination is mandatory to achieve confirm diagnosis.
Conflict of Interest:
None
Acknowledgements:
None
References:
1. Martelli M, Ferreri AJM, Agostinelli C, Di Rocco A, Pfreundschuh M, Pileri SA. Diffuse large B-cell lymphoma. Crit Rev Oncol Hematol. 2013;87 (2):146-171. doi:10.1016/j.critrevonc.2012.12.009
2. Ting C, Gan G, Bee‐Lan Ong D, Tan S, Bee P. Extranodal site of diffuse large B‐cell lymphoma and the risk of R‐CHOP chemotherapy resistance and early relapse. Int J Clin Pract. 2020;74(10). doi:10.1111/ijcp.13594
3. Bathla G, Hegde A. Lymphomatous involvement of the central nervous system. Clin Radiol. 2016;71(6):602-609. doi:10.1016/j.crad.2016.02.006
4. Swerdlow SH, Campo E, Harris NL. Bosman FT, Jaffe ES, Lakhani Ohgaki H, et al. WHO classification of tumours of haematopoietic SR, and lymphoid tissues. World Health Organization Classification of Tumours Lyon, France: IARC; 2008.
5. O’Neill BP, Decker PA, Tieu C, Cerhan JR. The changing incidence of primary central nervous system lymphoma is driven primarily by the changing incidence in young and middle-aged men and differs from time trends in systemic diffuse large B-cell non-Hodgkin’s lymphoma. Am J Hematol. 2013;88(12):997-1000. doi:10.1002/ajh.23551
6. Grommes C, DeAngelis LM. Primary CNS Lymphoma. J Clin Oncol. 2017;35(21):2410-2418. doi:10.1200/JCO.2017.72.7602
7. Bataille B, Delwail V, Menet E, Vandermarcq P, Ingrand P, Wager M, et al. Primary intracerebral malignant lymphoma: report of 248 cases. J Neurosurg. 2000;92(2):261-266. doi:10.3171/jns.2000.92.2.0261
8. Fischer L, Martus P, Weller M, Klasen HA, Rohden B, Röth A, et al. Meningeal dissemination in primary CNS lymphoma. Neurology. 2008;71 (14):1102-1108. doi:10.1212/01.wnl.0000326958.52546.f5
9. Chan CC, Rubenstein JL, Coupland SE, Davis JL, Harbour JW, Johnston PB, et al. Primary vitreoretinal lymphoma: a report from an International Primary Central Nervous System Lymphoma Collaborative Group symposium. Oncologist. 2011; 16(11):1589-1599. doi:10.1634/theoncologist.2011-0210
10. Alamdari A, Naderi N, Peiman S, Shahi F. Non-Hodgkin lymphoma with primary involvement of skeletal muscle. Int J Hematol Oncol Stem Cell Res. 2014;8(3):55-57.
11. Thomas AG, Vaidhyanath R, Kirke R, Rajesh A. Extranodal Lymphoma From Head to Toe: Part 2, The Trunk and Extremities. Am J Roentgenol. 2011;197(2):357-364. doi:10.2214/ajr.11.6738
12. Ilica AT, Kocacelebi K, Savas R, Ayan A. Imaging of Extranodal Lymphoma With PET/CT. Clin Nucl Med. 2011;36(10):e127-e138. doi:10.1097/rlu.0b013e31821c99cd
13. Zucca E. Extranodal lymphoma: a reappraisal. Ann Oncol. 2008;19:iv77-iv80. doi:10.1093/annonc/mdn204
14. Chua SC, Rozalli FI, O’Connor SR. Imaging features of primary extranodal lymphomas. Clin Radiol. 2009;64(6):574-588. doi:10.1016/j.crad.2008.11.001
15. Kashyap R, Rai Mittal B, Manohar K, Balasubramanian Harisankar CN, Bhattacharya A, Singh B, et al. Extranodal manifestations of lymphoma on [18F]FDG-PET/CT: a pictorial essay. Cancer Imaging. 2011;11(1):166-174. doi:10.1102/1470-7330.2011.0023
16. Braunstein EM, White SJ. Non-Hodgkin lymphoma of bone. Radiology. 1980;135(1):59-63. doi:10.1148/radiology.135.1.6892659
17. Gerstner ER, Batchelor TT. Primary Central Nervous System Lymphoma. Arch Neurol. 2010; 67(3). doi:10.1001/archneurol.2010.3
18. Senocak E, Oguz KK, Ozgen B, Mut M, Ayhan S, Berker M, et al. Parenchymal lymphoma of the brain on initial MR imaging: A comparative study between primary and secondary brain lymphoma. Eur J Radiol. 2011;79(2):288-294. doi:10.1016/j.ejrad.2010.01.017
19. Haque S, Law M, Abrey LE, Young RJ. Imaging of Lymphoma of the Central Nervous System, Spine, and Orbit. Radiol Clin North Am. 2008;46(2):339-361. doi:10.1016/j.rcl.2008.04.003
20. Server A, Josefsen R, Kulle B, Mæhlen J, Schellhorn T, Gadmar Ø, et al. Proton magnetic resonance spectroscopy in the distinction of high-grade cerebral gliomas from single metastatic brain tumors. Acta radiol. 2010;51(3):316-325. doi:10.3109/02841850903482901
21. Barajas Jr RF, Rubenstein JL, Chang JS, Hwang J, Cha S. Diffusion-weighted MR imaging derived apparent diffusion coefficient is predictive of clinical outcome in primary central nervous system lymphoma. AJNR Am J Neuroradiol. 2010;31(1): 60-66. doi:10.3174/ajnr.A1750
22. Mohile NA, Deangelis LM, Abrey LE. The utility of body FDG PET in staging primary central nervous system lymphoma. Neuro Oncol. 2008;10(2):223-228. doi:10.1215/15228517-2007-061
23. Karantanis D, O’Neill BP, Subramaniam RM, Witte RJ, Mullan BP, Nathan MA, et al. 18F-FDG PET/CT in primary central nervous system lymphoma in HIV-negative patients. Nucl Med Commun. 2007;28(11):834-841. doi:10.1097/mnm.0b013e328264ae7f
24. Kawai N, Okubo S, Miyake K, Maeda Y, Yamamoto Y, Nishiyama Y, et al. Use of PET in the diagnosis of primary CNS lymphoma in patients with atypical MR findings. Ann Nucl Med. 2010; 24(5):335-343. doi:10.1007/s12149-010-0356-z
25. Raoux D, Duband S, Forest F, Trombert B, Chambonnière ML, Dumollard JM, et al. Primary central nervous system lymphoma: Immunohistochemical profile and prognostic significance. Neuropathology. 2009;30(3):232-240. doi:10.1111/j.1440-1789.2009.01074.x
26. Kinoshita M, Hashimoto N, Izumoto S, Okita Y, Kagawa N, Maruno M, et al. Immunohistological profiling by B-cell differentiation status of primary central nervous system lymphoma treated by high-dose methotrexate chemotherapy. J Neurooncol. 2010;99(1):95-101. doi:10.1007/s11060-010-0112-1
27. Kleihues P, Cavenee WK. Pathology and Genetics of Tumours of the Nervous System. IARC Press, Lyon. 2000:198-203,.
28. Angelov L, Doolittle ND, Kraemer DF, Siegal T, Barnett GH, Peereboom DM, et al. Blood-brain barrier disruption and intra-arterial methotrexate-based therapy for newly diagnosed primary CNS lymphoma: a multi-institutional experience. J Clin Oncol. 2009;27(21):3503-3509. doi:10.1200/JCO.2008.19.3789
29. Zhang D, Hu LB, Henning TD, Ravarani EM, Zou LG, Feng XY, et al. MRI findings of primary CNS lymphoma in 26 immunocompetent patients. Korean J Radiol. 2010;11(3):269-277.
doi:10.3348/kjr.2010.11.3.269
30. Basso U, Brandes AA. Diagnostic advances and new trends for the treatment of primary central nervous system lymphoma. Eur J Cancer. 2002;38 (10):1298-1312. doi:10.1016/s0959-8049(02)00031-x
31. Glantz MJ, Cole BF, Recht L, Akerley W, Mills P, Saris S, et al. High-dose intravenous methotrexate for patients with nonleukemic leptomeningeal cancer: is intrathecal chemotherapy necessary? J Clin Oncol. 1998;16(4):1561-1567. doi:10.1200/jco.1998.16.4.1561
32. Guha-Thakurta N, Damek D, Pollack C, Hochberg FH. Intravenous Methotrexate as Initial Treatment for Primary Central Nervous System Lymphoma: Response to Therapy and Quality of Life of Patients. J Neurooncol. 1999;43(3):259-268. doi:10.1023/A:1006210703827
33. Murakami M, Fujimaki T, Asano S, et al. Combination therapy with rituximab and temozolomide for recurrent and refractory primary central nervous system lymphoma. Yonsei Med J. 2011;52(6):1031-1034. doi:10.3349/ymj.2011.52.6.1031.
34. Makino K, Nakamura H, Hide T ichiro, Kuratsu J ichi. Salvage treatment with temozolomide in refractory or relapsed primary central nervous system lymphoma and assessment of the MGMT status. J Neurooncol. 2011;106(1):155-160. doi:10.1007/s11060-011-0652-z.
35. Yang Y, He J, Dang Y, Xia X, Dai Y and Xu R. One case report and literature review of primary central nervous system non Hodgkin’s lymphoma. Lin Chuang Shen Jing Wai Ke Za Zhi. 2014; 5: 373-375 (In Chinese).
36. Weaver JD, Vinters H V, Koretz B, Xiong Z, Mischel P, Kado D. Lymphomatosis Cerebri Presenting as Rapidly Progressive Dementia. Neurologist. 2007;13(3):150-153. doi:10.1097/01.nrl.0000254706.85609.95
37. Suh C, Kim JE, Yoon DH. Relapse pattern and prognostic factors for patients with primary CNS lymphoma. Korean J Hematol. 2012;47(2) :155-156. doi:10.5045/kjh.2012.47.2.155
38. Mamorska-Dyga A, Ronny FMH, Puccio C, Islam H, Liu D. A rare case of the upper extremity diffuse large B-cell lymphoma mimicking soft tissue sarcoma in an elderly patient. Stem cell Investig. 2016;3:25. doi:10.21037/sci.2016.06.06
39. Mayo J, Bogenberger K, Raj T, Reha J. Subcutaneous mass concerning for sarcoma: a peculiar presentation of diffuse large B-cell lymphoma. BMJ Case Rep. 2019;12(7):e229327. doi:10.1136/bcr-2019-229327
40. Gupta P, Agarwal P, Ahuja A, Durga CK. Primary cutaneous non-Hodgkin’s lymphoma, clinically mimicking a soft tissue sarcoma. Cytojournal. 2018;15:2. doi:10.4103/cytojournal.cytojournal_41_17
41. Gao YH, Xu Q, Wei G, Liu HS, Wu X, Liu LH, et al. Primary giant lymphoma of the right thigh: A case report and brief review of the literature. Oncol Lett. 2012;4(5):1023-1026. doi:10.3892/ol.2012.848
42. Elsamaloty H, Zenooz NA, Alam T. Aggressive muscular non-Hodgkin lymphoma. Eur J Radiol Extra. 2006;58(2):27-30. doi:10.1016/j.ejrex.2006.02.009
43. Faraji-Goodarzi M. Sepsis after non-perforated acute appendicitis. Clin case reports. 2019;7(3):520-523. doi:10.1002/ccr3.2030
44. Masaoka S, Fu T. Malignant lymphoma in skeletal muscle with rhabdomyolysis: a report of two cases. J Orthop Sci. 2002;7(6):688-693. doi:10.1007/s007760200122
45. Laffosse JM, Gomez-Brouchet A, Molinier F, Chiron P, Roché H, Puget J. A case of malignant primary non-Hodgkin’s lymphoma in skeletal muscle treated by exclusive chemotherapy. Jt Bone Spine. 2009;76(1):86-88. doi:10.1016/j.jbspin.2008.02.022
46. Ferry JA. Extranodal Lymphoma. Arch Pathol & Lab Med. 2008;132(4):565-578.
doi:10.5858/2008-132-565-el
47. Siddiqui MT, Pitelka LA, Gattuso P. Extranodal lymphomas: Review of clinicopathologic and cytologic features. Diagn Cytopathol. 2009;37(3):220-229. doi:10.1002/dc.21045
48. Watanabe R, Tomita N, Takeuchi K, Sakata S, Tateishi U, Tanaka M, et al. SUVmax in FDG-PET at the biopsy site correlates with the proliferation potential of tumor cells in non-Hodgkin lymphoma. Leuk & Lymphoma. 2009;51(2):279-283. doi:10.3109/10428190903440953
49. Miyazaki Y, Nawa Y, Miyagawa M, Kohashi S, Nakase K, Yasukawa M, et al. Maximum standard uptake value of 18F-fluorodeoxyglucose positron emission tomography is a prognostic factor for progression-free survival of newly diagnosed patients with diffuse large B cell lymphoma. Ann Hematol. 2013;92(2):239-244.
doi:10.1007/s00277-012-1602-3
50. Khiewvan B, Macapinlac HA, Lev D, McCutcheon IE, Slopis JM, Al Sannaa G, et al. The value of 18F-FDG PET/CT in the management of malignant peripheral nerve sheath tumors. Eur J Nucl Med Mol Imaging. 2014;41(9):1756-1766. doi:10.1007/s00259-014-2756-0
51. Schaefer NG, Hany TF, Taverna C, Seifert B, Stumpe KDM, von Schulthess GK, et al. Non-Hodgkin Lymphoma and Hodgkin Disease: Coregistered FDG PET and CT at Staging and Restaging—Do We Need Contrast-enhanced CT? Radiology. 2004; 232(3):823-829. doi:10.1148/radiol.2323030985
52. Wang X. PET/CT: appropriate application in lymphoma. Chin Clin Oncol 2015;4:4.
53. Utkan G, Buyukcelik A, Yalcin B, Tek I, Doruk H, Dincol D, et al. Extranodal Hodgkin Disease Presenting with Gluteal Mass and Hypercalcemia. South Med J. 2006;99(10):1149-1150.
doi:10.1097/01.smj.0000240720.24001.77
54. Katsura M, Nishina H, Shigemori Y, Nakanishi T. Extranodal lymphoma originating in the gluteal muscle with adjacent bone involvement and mimicking a soft tissue sarcoma. Int J Surg Case Rep. 2015;7C:161-164.
doi:10.1016/j.ijscr.2015.01.024
55. Scally J, Garrett A. Primary extranodal lymphoma in muscle. Br J Radiol. 1989;62(733):81. doi:10.1259/0007-1285-62-733-81
56. Vitolo U, Seymour JF, Martelli M, Illerhaus G, Illidge T, Zucca E, et al. Extranodal diffuse large B-cell lymphoma (DLBCL) and primary mediastinal B-cell lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27:v91-v102. doi:10.1093/annonc/mdw175




