WHO Cervical Cancer Guidelines: Insights from Argentina PMAs
Dissemination and adoption of the World Health Organization guidelines for cervical cancer screening, diagnosis, and treatment: perceptions of Professional Medical Associations from Argentina
Silvina Arrossi, MSc PhD¹, Cecilia Straw, PhD², Armando Baena, PhD³, Melisa Paolino, PhD¹, Mariluz Hernandez, PhD⁴, Paul Bloem, PhD⁴, Maribel Almonte, PhD⁴
Disclaimer: “Where authors are identified as personnel of the World Health Organization, the authors alone are responsible for the views expressed in this article, and they do not necessarily represent the decisions, policy or views of the World Health Organization.”
- Centro de Estudios de Estado y Sociedad / Consejo Nacional de Investigaciones Científicas y Técnicas, Buenos Aires, Argentina
- Centro de Estudios de Estado y Sociedad, Buenos Aires, Argentina
- Oak Ridge Institute for Science and Education (ORISE), Oak Ridge, TN, USA
- World Health Organization, Geneva, Switzerland
OPEN ACCESS
PUBLISHED: 30 April 2026
CITATION: Arrossi, S., et al., 2026. Dissemination and adoption of the World Health Organization guidelines for cervical cancer screening, diagnosis, and treatment: perceptions of Professional Medical Associations from Argentina. Medical Research Archives, [online] 14(4).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Background: The 2021 World Health Organization (WHO) guidelines for cervical cancer screening and treatment provide evidence-based recommendations to accelerate cervical cancer elimination. However, the adoption of these recommendations depends on how they are interpreted, disseminated, and endorsed within national health systems. Previous research in Argentina showed that health providers primarily rely on recommendations issued by Professional Medical Associations (PMAs) rather than consulting WHO guidelines directly. We conducted a qualitative study to examine how PMAs develop their cervical cancer screening recommendations, their perceptions of the 2021 WHO guidelines, and potential opportunities for collaboration to strengthen dissemination and adoption.
Methods: A qualitative study was conducted based on individual, semi-structured interviews with representatives of national and provincial PMAs involved in cervical cancer screening guideline development (n=17). Data were analyzed thematically using constructs from the Consolidated Framework for Implementation Research to explore perceptions regarding WHO recommendations, guideline development processes, and perceived barriers and facilitators for implementation.
Results: National PMAs reported formal processes for guideline development involving expert committees, literature review, and adaptation to local contexts. In contrast, provincial PMAs generally do not develop their own guidelines but adopt or adapt those produced by national PMAs, creating a cascade relationship in which national recommendations shape clinical practice nationwide. Across PMAs, international guidelines from high-income countries were the most frequently cited references for developing recommendations. Although WHO was widely perceived as a credible and authoritative institution, its guidelines were rarely used as primary references and were often viewed as broad frameworks requiring adaptation to Argentina’s clinical and health system context. Interviewees expressed strong support for HPV-based screening with triage, highlighting its higher sensitivity and efficiency compared with cytology. However, they largely rejected elements such as 10-year screening intervals and treatment without histological confirmation. The screen-and-treat strategy without triage was widely perceived as incompatible with current professional standards, although some participants considered it acceptable in populations with limited access to follow-up. Most PMAs reported limited engagement with WHO in guideline dissemination and little incorporation of quality assurance systems in their recommendations. Nevertheless, PMA representatives expressed willingness to collaborate with WHO in guideline dissemination, professional training, and implementation activities.
Conclusions: Professional Medical Associations play a central role in shaping clinical practice and mediating the adoption of global recommendations within decentralized health systems. The cascade relationship between national and provincial PMAs reinforces their influence on clinical standards nationwide. Strengthening collaboration between WHO, national health authorities, and PMAs particularly in guideline dissemination, training, and development of quality assurance systems may facilitate the integration of WHO recommendations into clinical practice and accelerate progress toward cervical cancer elimination.
Keywords: WHO cervical cancer guidelines, Implementation, Dissemination, Adoption, Argentina.
Introduction
In November 2020, the World Health Organization (WHO) launched the Global Strategy to Accelerate the Elimination of Cervical Cancer, establishing the 90 70 90 targets for 2030: 90% HPV vaccination coverage among girls by age 15, 70% screening coverage using a high-performance test, and 90% of women with precancer or cervical cancer appropriately managed. Achieving these targets requires not only expanded coverage but also the adoption of screening algorithms aligned with current evidence.
One key activity of this strategy is to issue evidence-based recommendations to guide countries in implementing effective cervical cancer (CC) prevention strategies. Therefore, in 2021 WHO updated its 2013 guidelines for CC screening and treatment (WHO CC Guidelines), with HPV testing recommended as the primary screening method as part of a screen, triage, and treat strategy or as part of a screen-and-treat strategy according to local needs, capacities, and resources. These recommendations target national, regional, and district-level decision-makers responsible for defining CC prevention strategies.
However, the existence of evidence-based guidelines does not guarantee their adoption. Studies consistently show low adherence by health providers (HPs) to CC screening recommendations. Moreover, discrepancies among guidelines developed by international, national, and professional institutions generate variability in clinical algorithms, screening initiation and cessation ages, intervals, and primary screening methods. Such variation has been associated with inefficient use of health resources and may exacerbate inequities in access to prevention services.
Adoption of guidelines largely depends on the strength of their dissemination efforts and on adopters’ acceptability and characteristics. At institutional and health-system levels, effective dissemination requires creating a supportive implementation environment and securing political consensus to translate recommendations into scalable health policies. Consequently, integrating WHO CC guidelines into routine public health practice hinges on how stakeholders interpret, disseminate, and endorse them, as well as on perceived barriers and facilitators to their adoption.
The Guides study was designed to analyze stakeholders’ knowledge, attitudes, and perceptions regarding WHO CC guidelines and to identify barriers and facilitators to their adoption. In the first phase of the study, local Professional Medical Associations (PMAs) emerged as the primary influencers of HPs screening, diagnosis, and treatment decisions. PMAs recommendations are valued because they are issued by scientifically prestigious entities with strong influence on training and clinical practice, are perceived as appropriate to patient characteristics and the local socioeconomic context, and provide legal backing for clinical practice. These results are consistent with evidence from the United States showing that gynecologists primarily rely on guidelines developed by professional societies in their screening practices. These findings underscore a critical knowledge gap: little is known about how PMAs develop their recommendations, how they engage with WHO guidance, and the extent to which their algorithms are aligned with current evidence. If PMAs recommendations are not updated or are based on algorithms designed for high-income settings, this may affect program quality leading to reduced coverage, over-screening of low-risk populations, and increased inequities in access to screening, diagnosis, and treatment.
In addition, HPV-based screening requires robust quality assurance systems (QAS) to ensure test accuracy, appropriate follow-up, and effective management. Yet, limited evidence exists regarding PMAs awareness of quality assurance requirements, the systems they endorse, and the barriers and facilitators they perceive in their implementation.
Methods
SETTING
Argentina is a federal country made up of 24 provinces, each of which is an autonomous entity responsible for the organization, management, and financing of its provincial health system. The National Ministry of Health (MoH) provides a regulatory framework for healthcare provision, as well as training and financing for specific programs through nationally and internationally funded initiatives. Provincial health ministries can choose whether to adhere to the proposed national health programs or activities; their adherence depends mainly on formal agreements that negotiate responsibilities and funding.
There are five national PMAs including the Argentinean Federation of Societies of Gynecology and Obstetrics (FASGO) that brings together 27 PMAs from different provinces and cities; and 21 provincial PMAs. Some cities also have their own PMAs. A list of PMAs whose representative was interviewed is presented in Box 1.
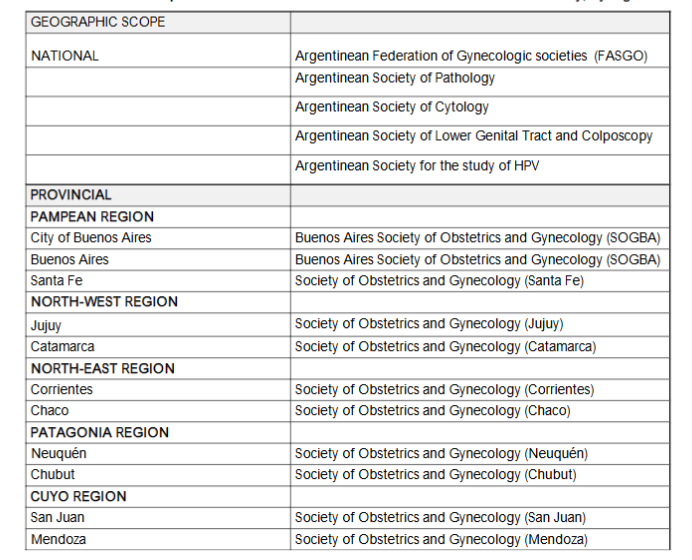
Since 2012, national authorities have established HPV testing as the primary CC screening test for women aged 30 years and older, to be performed every 5 years. In 2015, the Argentine National Cancer Institute set recommendations for HPV testing, triage, and treatment, which were developed with participation of the main national PMAs. These guidelines established that HPV-positive women should be triaged with cytology. Women with abnormal Pap smears are referred for colposcopy and, if needed, biopsies are collected. Women with histologically confirmed cervical intraepithelial neoplasia 2 or worse are referred for treatment. HPV-positive women with normal cytology are recommended to be re-screened in 18 months. We conducted a qualitative study with interviews with PMAs representatives. Following the methodology used in the first phase of the GUIDES Study we used the Consolidated Framework for Implementation Research (CFIR). This framework provides a menu of theoretical and multidisciplinary constructs that allows for a clear and consistent articulation of factors that may affect the results of implementation outcomes. From each domain, we selected the constructs that were most relevant to our study.
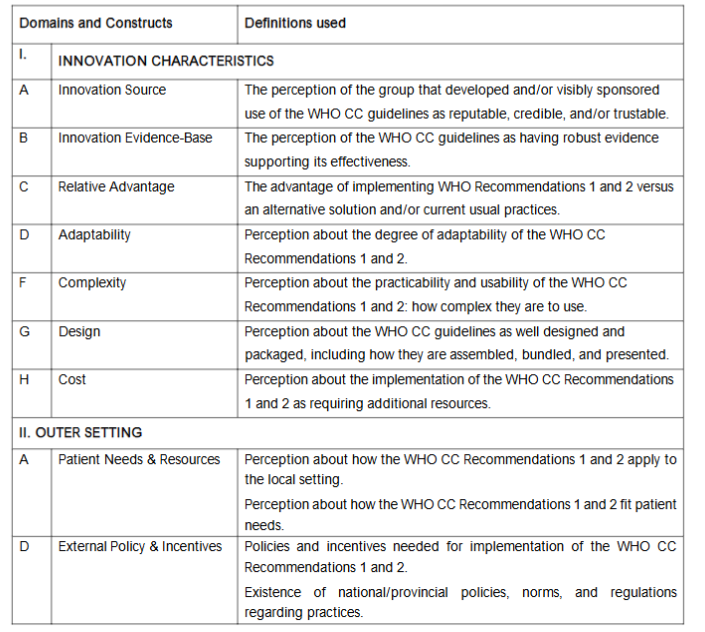
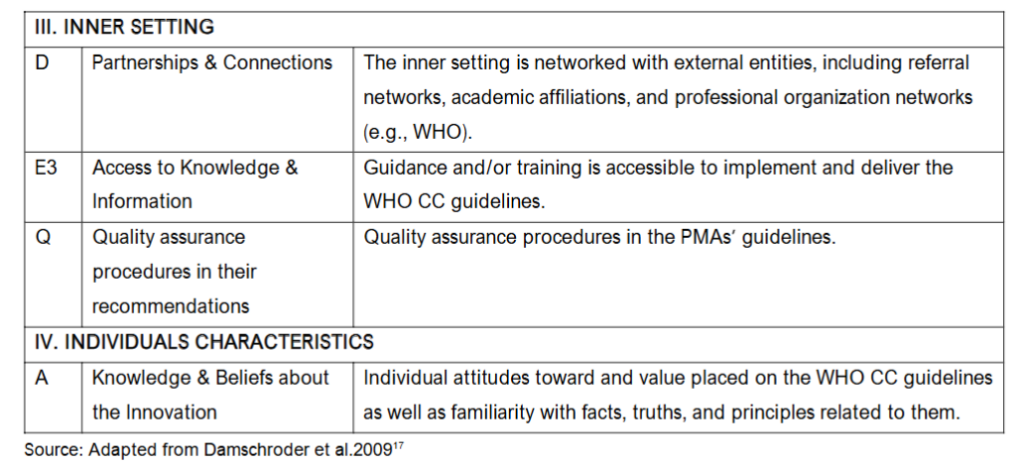
Table 1. Consolidated Framework for Implementation Research (CFIR) Domains and construct definitions used for data collection and analysis.
STUDY PARTICIPANTS
Inclusion criteria were being a PMAs representative who led or was involved in the development of the PMAs cervical cancer screening guidelines. The selection of participants followed a purposive sampling approach to include a variety of PMAs, with both national and provincial representation. The sample size (n=17) was determined based on relevance and theoretical saturation. Interviews were carried out by a trained female social researcher. Participants were contacted by the interviewer using the contact details provided by the PI or through the PMAs webpages. The interviewer contacted the presidents of the PMAs, explained the study and invited them to participate. Interviewees signed an informed consent. Interviews were conducted via Zoom and lasted on average one hour; permission was obtained from participants to audio-record them. Two pilot interviews were conducted, and the interview guide was revised based on their results.
DATA ANALYSIS
Qualitative data were analyzed thematically using the CFIR dimensions. Interview audios were transcribed verbatim to carry out thematic analysis of the debates, based on an iterative and flexible process following six steps: 1. To ensure coding reliability, two researchers (CS and SA) became independently familiar with the data through transcriptions and the video recording. 2. We classified data using an initial codebook based on the CFIR constructs, and following our research objectives, we identified the professionals’ opinions. 3. We analyzed each category to generate new themes understood as a salient aspect of the data in a patterned way, regardless of whether that theme captures the majority experience of the participants. 4. Both researchers met to review themes, identify consistencies and resolve the inconsistencies. 5. We grouped the emergent themes according to their conceptual similarities to define and name the subthemes. 6. We sought examples that adequately graphed each theme. Several meetings were held to discuss the results and prepare the final manuscript. We used ATLAS.ti (version 7.5.4; Scientific Software Development GmbH, Berlin) to organize, code, and summarize patterns. To ensure coding reliability, we verified coding against a sample of transcripts and critically reviewed the data and themes to improve study trustworthiness. The study’s protocol was approved by the Gino Germani Institute’s Ethics Committee from the Faculty of Social Sciences, University of Buenos Aires, and by the Research Ethics Review Committee of the WHO. Participants provided written informed consent. Anonymity was guaranteed at every step of the study.
Results
PARTICIPANT’S CHARACTERISTICS
Seventeen stakeholders (11 women and six men) from 11/24 provincial and 5/5 national PMAs were interviewed. The mean age was 59 years (range 40 to 75), and the average time as PMA members was 22 years (range 2-36).
Context and development of local guidelines
All national-level PMAs, as well as the PMAs of the City of Buenos Aires, reported having their own guidelines, except for pathology and cytology PMAs. Notwithstanding the absence of standalone guidelines in these latter, all PMAs reported participation in inter-PMA consensus processes and some were involved in the development of the Argentine National Cancer Institute guidelines. None of the PMAs reported using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology in developing their guidelines. Across PMAs, guideline development was described as a formal process involving expert committees, literature review, and adaptation to local contexts.
Everything must be supported by national and international bibliography… aligned with the guidelines of SOGIBA, and FASGO. [E3] A committee was created, and smaller working groups were formed by topic… After a year of work, we had a document for each topic. [E9]
Across these PMAs, international guidelines from the United States (e.g. those of the American Society for Colposcopy and Cervical Pathology), Spain, and Australia were the most frequently cited sources for informing or updating local guidelines. These references were valued for incorporating current technological advances (e.g., HPV genotyping, liquid-based cytology) and/or constituting successful experiences in eliminating cervical cancer. They also use, to a lesser extent, the national guidelines developed by the Argentine NCI.
We rely heavily on the American guidelines… draw considerably from the Spanish and adapt the guidelines of the National Program on Cervical Cancer Prevention. [E9] Well, we also look closely at Australia, you see? Australia has really been the front-runner. [E3]
In contrast, in general, provincial-level PMAs do not develop their own guidelines, but they adopt those developed by national PMAs, and FASGO. In some cases, they have participated in adaptations of the national guidelines conducted by the provincial programs on cervical cancer prevention. These guidelines draw on both national and international PMAs.
Each provincial society does not generate its own guideline… we follow the guide of a national PMA which was endorsed by FASGO. [E7] Initially, we referred to the national guidelines… but found they did not adapt well… they did not incorporate genotypification. Therefore, we looked for guidelines better adapted to the provincial context. [E1]
Perceptions of the WHO guidelines
Knowledge, Innovation Source, Innovation Evidence-Base, and Peer Pressure
In general, the WHO was widely perceived by interviewees as a credible and authoritative institution. Respondents valued the WHO’s scientific rigor, global perspective, and capacity to mobilize expert and financial resources to support high-quality guideline development.
[The WHO is] a highly respectable scientific organization. [E6] The WHO’s cervical cancer elimination strategy was inspiring, as it promoted high-quality HPV-based screening, and advancing broad population coverage. [E1]
Most interviewees reported being familiar with the WHO CC guidelines; however, this familiarity was often superficial or partial, and only a minority could clearly articulate their content or identify concrete examples of implementation. In a few cases, the experience of countries such as Australia and England, as well as the HPV-testing implementation in Argentina, were cited as examples of WHO-aligned strategies.
Australia… will probably eliminate cervical cancer around 2032 2033, … they follow the WHO CC guidelines. [E3] I knew about the experience in Jujuy [Argentina] with HPV testing. [E1]
Despite this familiarity, the WHO CC guidelines were not used as primary sources. Instead, they were viewed as a broad framework requiring substantial adaptation to Argentina’s clinical, economic, and epidemiological context. In that sense, interviewees felt that Argentine PMAs possess the know-how to develop a guide rooted in the national health system and clinical practice, and responsive to the population’s needs. Some isolated mentions reflected the perception that the screening age intervals were outdated and did not reflect demographic and social changes that occurred in recent years, with an impact on cervical cancer epidemiology.
The WHO guideline is a general orientation, not a sacred mandate… We have the know-how to elaborate a guide that is adapted to the available resources and to what can be done in Argentina. [E3]
Only a minority of participants reported familiarity with the methodology used by WHO to develop its 2021 guidelines. Nonetheless, even among those who did not know the process, there was a general belief that the guideline development steps were appropriate and robust.
I wasn’t familiar with the methodology. However, I believe the steps followed… are appropriate… to achieve a high-quality guideline. [E11]
Although the WHO CC guidelines were considered scientifically sound and clearly presented, interviewees expressed mixed views about their applicability in Argentina. Some described them as an ideal difficult to implement in many real-world settings, while others saw them as a broad, generalizable framework or as minimum standards better suited for low-resource countries.
They have scientific justification and credibility. But they are not feasible everywhere… they represent an ideal. [E6] It’s an important standard each country can make the necessary local adjustments. [E12]
There was strong consensus that national and provincial guidelines developed by Argentine PMAs were more appropriate for clinical decision-making in the local context, particularly considering specific implementation practices and healthcare infrastructure.
If you look at the Argentine guidelines, one of the screening methods is cytology and colposcopy at the same visit. That’s not included in the WHO guidelines or many global guidelines. [E12]
External Policy and Incentives
Multiple interviewees emphasized the importance of legal and regulatory support to guarantee long-term implementation of HPV testing. This includes incorporating the practice into the national budget law, ensuring funding for tests and reagents, and securing institutional backing from the MoH and the Argentine NCI.
It would be fantastic if there were a law, but not a fake law. A real law that could actually be enforced. [E2] If there’s a regulation it will be included in the general budget law. [E10] The most important factor would be the purchase of HPV tests nationwide, which has not yet been achieved. [E8]
Innovation Design
Participants evaluated the design of the WHO CC guidelines positively, particularly for visual clarity and digital accessibility. The format especially the inclusion of flowchart-style algorithms was seen as intuitive and easy to navigate.
Visually, they are user-friendly because the design is clear, and the algorithms are straightforward. [E12] It’s easy to understand the algorithms always guide you through the procedures from no to yes. [E6]
Several participants emphasized the importance of digital formats over physical manuals, especially among younger generations of healthcare professionals. A commonly suggested improvement was the development of a mobile application version of the guidelines to facilitate point-of-care access.
Maybe if you have it on your phone you can consult it quickly… especially if there were an app that is constantly updated. [E12]
Partnerships & Connections
Interviewees described active engagement between national-level PMAs and international, European, and Latin American scientific networks. Collaborations include participation in subspecialty research committees, academic training forums, and international congresses.
In 2025, we will jointly host the congress in Argentina with the Latin American Society. We participate as speakers and attendees from all over Latin America. [E14] Many of our members are also members of international and European PMAs [E2]
Despite robust regional and international interaction, most interviewees reported little or no collaboration with the WHO, either in guideline development or dissemination. Only a minority recalled involvement in the dissemination of the 2013 WHO CC guidelines, but not with the 2021 version.
Honestly, we have no experience with joint activities with the WHO. [E9] Yes, I participated in multiple seminars [on dissemination of the 2013 WHO TDT-CC guideline]. [E3]
There was general agreement on the need for the WHO to assume a more proactive role in coordinating dissemination with national PMAs, particularly for the 2021 guidelines.
The WHO should take the lead in sending the guidelines or summaries to scientific societies. [E2] I haven’t seen any specific training on this guideline… FASGO would be a great space for discussion. [E8]
Implementation climate
Almost all interviewees stated that national PMAs would be willing to collaborate with the WHO on implementation efforts tailored to the Argentine context. Suggested areas of collaboration included professional training, dissemination of guides, and implementation of interventions.
Definitely yes [to collaborative activities with the WHO], especially in human resource training and guideline implementation. [E3] We could give them space in our congresses to promote the WHO guidelines. [E9] After this conversation, I’m going to propose something basic: having a direct link to the WHO recommendations on our website. [E10]
Perceptions regarding WHO Recommendations
Recommendation 1: Screening with HPV testing starting at age 30, every 5 to 10 years, with triage, and treatment of lesions with or without histological confirmation.
Relative Advantage
There was broad consensus among interviewees regarding the advantages of Recommendation 1 of the WHO CC guidelines. HPV testing was described as superior to cytology due to its higher sensibility, its ability to safely extend screening intervals, and its high negative predictive value.
With a negative test, we can separate that patient and not evaluate her again for several years. [E6] The most important thing about HPV testing is its negative predictive value. [E14] HPV-based screening is ideal… it’s mandatory to perform a second triage test. [E9]
Conversely, a strong majority opposed treating without histological confirmation, expressing concerns about overdiagnosis and the inability to accurately stratify lesion severity.
In Argentina, we do not treat lesions without histological confirmation. [E4] Histological confirmation is fundamental for our specialty. [E10]
A minority acknowledged that treatment without biopsy might be exceptionally acceptable in settings with limited resources, poor access to follow-up, or in cases involving high-risk HPV genotypes.
It depends on the region… the guideline allows with or without histological confirmation to adapt to regional and resource constraints. [E5]
Interviewees also questioned the safety of a 10-year screening interval, emphasizing that it would increase the risk of missing new infections or lesion progression.
Every 10 years is crazy. It’s not safe… the patient could contract another HPV infection. [E4]
Patient Needs and Resources
The modified version of Recommendation 1 HPV testing every 5 years, with triage and histological confirmation was widely perceived as aligned with patient needs. Participants noted that it ensured safe, periodic preventive care, appropriate risk stratification, and access to timely treatment, potentially reaching broader segments of the target population.
It recommends using a high-quality HPV test and ensures that patients reach treatment. This is the key to cervical cancer prevention. [E1] It would allow us to reach a larger proportion of the target population… we often fail to do this with Pap smears. [E7]
Adaptability
The majority of participants considered that modifications to Recommendation 1 were necessary to ensure its feasibility and clinical appropriateness in the Argentine context, such as: reducing the interval to every 5 years, maintaining mandatory histological confirmation, and, in some cases, lowering the age of initiation to 25 years due to observed cases of invasive cancer in younger women.
The HPV test should be performed every 5 years, not every 10. [E7] Starting HPV screening at age 30 is absurd. [E16] I see women in their 20s with invasive cervical carcinomas. [E2]
Complexity
The recommendation was generally perceived as in agreement with standard clinical practices, particularly when adapted to include histological confirmation and a 5-year screening interval. However, access to HPV testing was presented as the main operational challenge due to irregular supply and insufficient procurement at the national level.
It would be easy, but with histological confirmation… that’s what all gynecologists are accustomed to. [E3] We would need access to the HPV test, which currently is not uniformly available. [E7]
Cost
The main reported cost was the HPV tests, as resource constraints faced by the country in the last years resulted in interruptions in the national provision of tests. Several interviewees expressed frustration with the lack of sustained investment in HPV-based screening by national health authorities.
The Ministry of Health has not purchased HPV tests for two years. [E3] At this moment, it seems almost like a utopia we don’t have the resources for HPV testing. [E1]
Recommendation 2: Screening with HPV test from age 30, with screening every 5 to 10 years, without triage, and treatment for all HPV+ individuals.
Relative Advantage
This strategy was widely rejected by interviewees, who viewed it as incompatible with national clinical guidelines and professional standards.
We do not follow this approach of treating all HPV-positive individuals because it would lead to many unnecessary treatments. [E1] That would result in overtreating many women. [E9]
Participants emphasized that HPV infection alone does not equate to disease and underscored the need for colposcopic or cytological results prior to intervention.
Triage relates to ensuring that if there’s no cytological or colposcopic lesion, we do not treat solely based on HPV presence… All of this is part of the colposcopist’s assessment. [E10]
Patient Needs and Resources
Most interviewees concluded that Recommendation 2 does not meet the needs of the general population because of the high likelihood of unnecessary treatment. Nevertheless, a few participants suggested that the screen-and-treat approach may be suitable in resource-limited or high-risk populations, where follow-up is unlikely.
It’s very good for a specific type of population I’d rather overtreat than miss a cervical cancer. [E8]
Adaptability
The vast majority of interviewees deemed Recommendation 2 non-adaptable to the Argentine healthcare system. They noted that applying this strategy to the general population would contradict current practice and clinical reasoning.
It’s not adaptable. I would discard it completely, forget about it. [E4] A minority supported limited adaptation of Recommendation 2 in selected populations, particularly in geographically remote areas or among women unlikely to return for follow-up, where the risk of loss to care may justify a less stringent approach.
We don’t treat all HPV-positive patients unless it’s for populations with no regular access to healthcare or who can’t maintain continuity of care. [E10]
Complexity
Although technically simple eliminating the need for triage or histological confirmation Recommendation 2 was perceived as highly complex in terms of professional acceptance and ethical implications. Respondents anticipated strong resistance from clinicians due to concerns about overtreatment and lack of diagnostic confirmation.
This recommendation would create complications within professional societies A positive HPV test alone is not synonymous with having a lesion. [E8] Physicians won’t agree to treat without a biopsy. [E14]
Cost
Perceptions of the cost of Recommendation 2 were mixed. Some interviewees argued that it would lower costs by reducing the need for diagnostic steps such as colposcopy or biopsy. While others highlighted the unsustainable burden of treating all HPV-positive individuals, many of whom do not have clinically relevant disease.
This strategy should require far fewer resources. [E9] In our context it would not be cost-effective the patient volume would be unmanageable. [E7]
External Policy and Incentives
Participants emphasized that legal frameworks and national institutional endorsement would be necessary to legitimize the treatment of HPV-positive individuals without histological confirmation. They also noted that any such change would require policy-level endorsement by the National Cancer Institute or the Ministry of Health.
This recommendation needs high legal backing because we would be treating a woman based solely on a positive test. [E14] A recommendation from the National Cancer Institute supporting this approach would be convenient and very helpful. [E1]
Quality assurance procedures in Professional Medical Association recommendations
Most interviewees reported that the current Argentine guidelines do not include recommendations on QAS. The absence of such guidance was often attributed to perceived limitations in scientific societies’ enforcement capacity. In contrast, some interviewees acknowledged their lack of knowledge on the subject. Only in isolated instances was it mentioned that developing QAS recommendations is a technically complex task that the PMAs have not yet addressed.
Our guidelines do not include anything about quality control. I don’t see how a scientific society could enforce this. [E9] It’s still a pending issue. Quality control hasn’t been properly developed is really cumbersome and very difficult. [E14]
Some respondents narrowed the concept of QAS to HPV test sample taking and handling, and their storage in HPV laboratories. Thus, they considered that formal QA mechanisms were unnecessary, since only institutionally validated, automated HPV tests are used in Argentina, and these inherently meet regulatory standards set by the National Administration of Drugs, Foods, and Medical Devices (ANMAT).
We recommend institutionally validated tests approved by ANMAT. Test validation is automatic done by the validated machines themselves. [E3]
Several participants expressed a need for technical assistance and WHO support in developing more comprehensive QAS recommendations.
Yes, we would need WHO expert advice on that subject. [E10]
The only structured QAS initiative cited by interviewees involved a regional project led by Latin American Federation of Lower Genital Tract Pathology and Colposcopy (FLPTGIC). This effort, organized under the umbrella of the regional Federation and the International Federation for Cervical Pathology and Colposcopy, resulted in the development of a guideline focused on quality control in colposcopy clinics and training of colposcopists.
We formed a group with presidents of Latin American Colposcopy Societies and decided the guide would focus on two key areas: clinical quality control and colposcopist training. [E17] Spanish national guidelines served as the primary reference, while IARC materials were consulted specifically for colposcopic image documentation protocols.
For colposcopy, we used the Spanish guide. From WHO [IARC], we only took the image registration methodology. [E17]
This initiative was motivated by the absence of quality standards adapted to the clinical and epidemiological context of Latin America, where colposcopy is often used as a de facto screening method, in contrast to its role in high-income countries.
There’s a lot of cervical cancer in Latin America we almost use colposcopy as a screening method we need our own [guidelines] based on our training and reality. [E17]
Discussion
In 2021, the WHO updated their CC screening and treatment guidelines to accelerate elimination. To our knowledge, the GUIDES study is the first to examine the process by which WHO CC guidelines are adopted in health services and clinical practices. While the first phase of the study showed that HPs primarily rely on recommendations issued by Argentinean PMAs rather than consulting WHO guidelines directly or following those of national health authorities, the present findings illuminate the upstream institutional dynamics shaping this pattern. Specifically, this study shows that PMAs function not merely as passive recipients of global guidance but as active mediators and producers of normative standards that structure clinical practice.
Our results suggest that PMAs operate as epistemic gatekeepers within Argentina’s decentralized health system. Implementation research has consistently shown that the legitimacy of an innovation’s source influences its uptake. However, legitimacy alone does not guarantee adoption. According to CFIR, adoption is shaped by the interaction between intervention source, evidence strength, and perceived compatibility that is, the degree of fit between the intervention and the norms, values, and workflows of those involved, as well as patient needs.
Although WHO was perceived as a credible source, PMAs prioritized guidelines from high-income countries PMAs, which were regarded as more clinically actionable and aligned with the gold standard in CC prevention. The main national PMAs only changed their recommendations to include HPV testing every 5 years around 2022, following guidelines of the main high-income PMAs. And this, even though the national MoH had changed its recommendation to HPV-testing as early as 2012. This temporal alignment suggests that high-income PMAs exert a normative influence on national professional standards, which coexist with recommendations from national health authorities.
Our results also show that the relationship between PMAs is a cascade-type, as national PMAs develop their guidelines based on high-income countries PMAs recommendations, which are then generally adopted by provincial PMAs. Thus, recommendations developed by PMAs in high-income countries have an influence that permeates clinical practices nationwide.
A key aspect to consider when analyzing factors that influence adoption of an intervention is the webs of social networks and formal and informal communications of individuals and organizations. In our study, interviewees were actively engaged with international, European, and Latin American professional organizations, including participation in research committees, academic training, and international congresses, whereas they reported little or no collaboration with the WHO. These external relationships imply dimensions of shared vision and information sharing and strongly influence how quickly organizations adopt interventions. Therefore, participation of PMAs in these networks likely strongly influences their positions on using/adopting WHO guidelines. In other words, PMAs operate within transnational professional networks in which WHO may have comparatively lower visibility and influence.
A key finding of our study was that, in decentralized health systems, meso-level actors such as PMAs play a crucial role in mediating between national and provincial policies and frontline practice. Argentina’s federal structure, characterized by weak enforcement of national norms, amplifies the normative authority of PMAs. Both national and provincial PMAs function as governance actors and opinion leaders, shaping the implementation of recommendations beyond national policy mechanisms. This aligns with implementation research, which emphasizes that new practices become embedded through interactions across organizational and system levels rather than through simple top-down transmission.
Our study also shows that the reluctance to adopt elements such as ten-year screening intervals or screen-and-treat strategies without histological confirmation was not rooted in a lack of awareness or cost considerations. A survey of 29 stakeholders from 18 countries found that the choice of the screening algorithm made by health authorities and HPs was associated with perceived trade-offs in demand, training needs, and follow-up. Our study found that PMAs choices reflected professional norms and values, diagnostic certainty, medico-legal responsibility, and specialty training. Implementation science highlights compatibility with professional identity and existing practice as a critical determinant of adoption. Extended intervals and the elimination of histological confirmation were perceived as misaligned with deeply embedded standards of care, strongly influenced by those of high-income countries, which were considered the highest level of care. In this sense, high-income countries’ recommendations are perceived as better aligned with women’s needs and with how professionals should provide preventive care for their patients’ preventive health. Although some of the interviewees expressed that alternative algorithms could be used in areas with limited resources, the prevailing view was that all women should have an HPV test every 5 years, with histological confirmation before treatment. Ultimately, this reflects a tension between a pragmatic implementation logic focused on adapting strategies to existing resource constraints and a professional logic prioritizing the provision of what is perceived as the highest standard of care, even when access to such standards is uneven.
Furthermore, barriers to screening and follow-up are not exclusive to low-resource settings. Incorporating alternative approaches such as screen-and-treat into high-income PMAs recommendations may help to reframe these strategies as context-responsive models of care rather than as substandard options reserved for resource-constrained settings. A step forward toward this is the American Society of Clinical Oncology (ASCO) resource-stratified guidelines which recommend different approaches according to resources availability, recognizing social and gender disparities in the access to CC prevention within the United States. Such reframing may also benefit underserved populations within high-income countries.
Despite the selective acceptability of WHO CC recommendations, PMAs representatives expressed willingness to collaborate in their dissemination and implementation. Implementation research underscores that co-production and participatory adaptation enhance ownership and legitimacy. Rather than relying solely on top-down dissemination, effective implementation may require strategic engagement with PMAs as co-producers of contextually legitimate standards. A study that evaluated the acceptability of a mobile intervention to increase adherence to triage among women with HPV-tests on self-collected samples showed that the intervention, having been proposed by prestigious external institutions and then designed and implemented since its early stages with local stakeholders, was a main factor for their positive evaluation of the intervention. This approach aligns with emerging models of dynamic adaptation and knowledge translation, which emphasize iterative negotiation between global evidence and local systems. Engaging PMAs as active partners in dissemination and implementation processes may expand professional networks and accelerate adoption of WHO guidelines. The regional WHO offices such as the Pan American Health Organization for the Americas Region can have a key role in establishing these interactions.
The effectiveness of interventions in real-world settings is influenced by fidelity, defined as the degree to which an intervention was implemented as intended according to the established protocol. Thus, implementing screening recommendations cannot be dissociated from quality assurance processes. Regarding this, a critical gap identified in our study concerns quality assurance. Most PMAs did not include explicit QAS frameworks in their recommendations, often viewing enforcement as outside their mandate. QAS was often narrowly understood as laboratory validation of HPV tests, rather than a comprehensive system including monitoring of coverage, follow-up, diagnosis quality, treatment outcomes, and use of information systems and audits. This reveals a disconnect between guideline production and system-level performance monitoring. PMAs can play a key role in establishing and promoting the incorporation of QAS into health services processes and individual clinical practices. A huge step in a positive direction is the FLPTGIC’s effort to establish QAS standards for colposcopy.
Given the strong influence of PMAs in clinical practice, incorporating a broad QAS concept as a key part of their recommendations can be essential for changing how HPV screening is monitored and implemented, at both the individual and organizational levels. Furthermore, leveraging the FLPTGIC initiative on QAS assurance in colposcopy could serve as a blueprint for developing QAS mechanisms across the whole continuum of care, ensuring that HPV testing is supported by robust, institutionally endorsed monitoring systems.
Conclusions
In summary, these findings extend the insights of the first phase of the GUIDES study by shifting the analytical lens from individual providers to PMAs. If HPs adherence is highly shaped by PMAs guidance, then WHO guideline uptake ultimately depends not only on health authorities’ adoption and inclusion in national policies, but also on the institutional processes through which PMAs evaluate, reinterpret, and institutionalize global recommendations. In decentralized systems lacking strong enforcement mechanisms, PMAs become critical intermediaries in the governance of HPV-testing implementation. Incorporation of PMAs into the design, implementation, and dissemination of guidelines will greatly contribute to the adoption of the WHO CC guidelines, thereby contributing to accelerated progress toward cervical cancer elimination.
From an implementation science perspective, this study highlights the importance of engaging meso-level actors in translating global guidelines. Adoption is less a matter of provider awareness than of negotiated legitimacy within professional networks. Understanding these institutional dynamics and incorporating PMAs into WHO guidelines production and dissemination is essential to accelerate the integration of evidence-based strategies and advance cervical cancer elimination.
Conflict of Interest: The authors declare that they have no competing interests.
Funding: This article presents independent research financed by the WHO.
Acknowledgments: We thank the interviewees for their generosity in dedicating their time to complete the interviews.
References
- World Health Organization. Global strategy to accelerate the elimination of cervical cancer as a public health problem. Geneva, Switzerland: World Health Organization; 2020.
- Bouvard V, Wentzensen N, Mackie A, et al. The IARC perspective on cervical cancer screening. N Engl J Med. 2021;385(20):1908-1918. doi:10.1056/NEJMsr2030640
- World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention. 2nd ed. Geneva, Switzerland: World Health Organization; 2021.
- World Health Organization. WHO guidelines for screening and treatment of precancerous lesions for cervical cancer prevention. Geneva, Switzerland: World Health Organization; 2013. Available at: https://iris.who.int/handle/10665/94830
- Min CJ, Massad LS, Dick R, Powell MA, Kuroki LM. Assessing physician adherence to guidelines for cervical cancer screening and management of abnormal screening results. J Low Genit Tract Dis. 2020;24(4):337-342. doi:10.1097/LGT.0000000000000558
- Holland-Barkis P, Forjuoh SN, Couchman GR, Capen C, Rascoe TG, Reis MD. Primary care physicians awareness and adherence to cervical cancer screening guidelines in Texas. Prev Med. 2006;42(2):140-145. Doi:10.1016/j.ypmed.2005.09.010
- Han PK, Klabunde CN, Breen N, et al. Multiple clinical practice guidelines for breast and cervical cancer screening: perceptions of US primary care physicians. Med Care. 2011;49(2):139-148. doi:10.1097/MLR.0b013e318202858e
- Wang W, Arcà E, Sinha A, Hartl K, Houwing N, Kothari S. Cervical cancer screening guidelines and screening practices in 11 countries: a systematic literature review. Prev Med Rep. 2022;28:101813. doi:10.1016/j.pmedr.2022.101813
- Boone E, Lewis L, Karp M. Discontent and confusion: primary care providers’ opinions and understanding of current cervical cancer screening recommendations. J Womens Health (Larchmt). 2016;25(3):255-262. doi:10.1089/jwh.2015.5326
- Herrero R, Ferreccio C, Salmerón J, et al. New approaches to cervical cancer screening in Latin America and the Caribbean. Vaccine. 2008;26(suppl 11):L49-L58. doi:10.1016/j.vaccine.2008.05.025
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA. 1999;282(15):1458-1465. doi:10.1001/jama.282.15.1458
- Arrossi S, Paolino M, Sankaranarayanan R. Challenges faced by cervical cancer prevention programs in developing countries: a situational analysis of program organization in Argentina. Rev Panam Salud Publica. 2010;28(4):249-257. doi:10.1590/s1020-49892010001000003
- Woolf SH, Grol R, Hutchinson A, Eccles M, Grimshaw J. Clinical guidelines: potential benefits, limitations, and harms of clinical guidelines. BMJ. 1999;318(7182):527-530. doi:10.1136/bmj.318.7182.527
- Arrossi S, Straw C, Sanchez Antelo V, Paolino M, Baena A, Forestier M, Rol M, Almonte M. Implementation of WHO guidelines for cervical cancer screening, diagnosis and treatment: knowledge and perceptions of health providers from Argentina. BMC Cancer. 2024;24(1):996. doi:10.1186/s12885-024-12650-7
- Argentine Federation of Societies of Gynecology and Obstetrics (FASGO). Available at: https://www.fasgo.org.ar/
- Arrossi S, Thouyaret L, Paul L. Prevención del cáncer cervicouterino: recomendaciones para el tamizaje, seguimiento y tratamiento de mujeres en el marco de programas basados en el test de VPH. Actualización 2015. Buenos Aires: Instituto Nacional del Cáncer; 2015. Available at: http://www.msal.gob.ar/images/stories/bes/graficos/0000000017cnt-manual_recomendaciones_tamizaje_2015_baja.pdf
- Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50. doi:10.1186/1748-5908-4-50
- Damschroder LJ, Reardon CM, Widerquist MAO, Lowery J. The updated Consolidated Framework for Implementation Research based on user feedback. Implement Sci. 2022;17(1):75. doi:10.1186/s13012-022-01245-0
- Damschroder LJ. Clarity out of chaos: use of theory in implementation research. Psychiatry Res. 2020;283:112461. doi:10.1016/j.psychres.2019.06.036
- Braun V, Clarke V, Hayfield N, Terry G. Thematic analysis. In: Liamputtong P, ed. Handbook of research methods in health social sciences. Singapore: Springer; 2019:843-860. doi:10.1007/978-981-10-5251-4_103
- Lincoln YS, Guba EG. Naturalistic inquiry. Beverly Hills, CA: Sage; 1985.
- Rogers EM. Diffusion of innovations. 5th ed. New York, NY: Free Press; 2003.
- Kitson A, Harvey G, McCormack B. Enabling the implementation of evidence based practice: a conceptual framework. Qual Health Care. 1998;7(3):149-158. doi:10.1136/qshc.7.3.149
- Straw C, Sanchez-Antelo V, Kohler R, Paolino M, Viswanath K, Arrossi S. Implementation and scaling-up of an effective mHealth intervention to increase adherence to triage of HPV-positive women (ATICA study): perceptions of decision-makers and providers. BMC Health Serv Res. 2023;23(1):47. doi:10.1186/s12913-023-09022-5
- Fixsen DL, Naoom SF, Blase KA, Friedman RM, Wallace F. Implementation research: a synthesis of the literature. Tampa, FL: University of South Florida; 2005.
- May C, Finch T. Implementing, embedding, and integrating practices: an outline of Normalization Process Theory. Sociology. 2009;43(3):535-554. doi:10.1177/0038038509103208
- World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention. 2nd ed. Geneva, Switzerland: World Health Organization; 2021. Web annex A: Syntheses of evidence, supplementary material 11:148-164.
- Kim JJ. Opportunities to improve cervical cancer screening in the United States. Milbank Q. 2012;90(1):38-41. doi:10.1111/j.1468-0009.2011.00653.x
- Lucas E, Rol ML, Muwonge R, Basu P. Cancer screening in five continents. Lyon, France: International Agency for Research on Cancer; 2025. Available at: https://canscreen5.iarc.fr (accessed March 9, 2026).
- Shastri SS, Temin S, Almonte M, et al. Secondary prevention of cervical cancer: ASCO resource-stratified guideline update. JCO Glob Oncol. 2022;8:e2200217. doi:10.1200/GO.22.00217
- Greenhalgh T, Robert G, Macfarlane F, Bate P, Kyriakidou O. Diffusion of innovations in service organizations: systematic review and recommendations. Milbank Q. 2004;82(4):581-629. doi:10.1111/j.0887-378X.2004.00325.x
- Chambers DA, Norton WE. The adaptome: advancing the science of intervention adaptation. Am J Prev Med. 2016;51(4 suppl 2):S124-S131. doi:10.1016/j.amepre.2016.05.011
- Proctor E, Silmere H, Raghavan R, et al. Outcomes for implementation research: conceptual distinctions, measurement challenges, and research agenda. Adm Policy Ment Health. 2011;38(2):65-76. doi:10.1007/s10488-010-0319-7
- Fleider LA, Celis Amórtegui M, Díaz López E, et al. Consensus on quality standards for colposcopy and training from the Latin American Federation of Lower Genital Tract Pathology and Colposcopy. J Low Genit Tract Dis. 2025;29(4):329-334. doi:10.1097/LGT.0000000000000908

