Nutritional Factors in Treating Recalcitrant Dermatophytosis
Role of increasing protein intake and correction of micronutrients imbalance to promote desired autophagy in treating resistant dermatophyte infection: A cohort of 62 patients
Dr Suruchi Garg¹, Dr Carol Lobo², Dr Navnee Jain³
- Director and Chief Consultant, Aura Skin Institute, SCO 44-45, Sector 9D, Madhya Marg, Chandigarh,
- Assistant Professor, Department of Dermatology, St John’s Medical College, Bangalore 560034,
- Assistant Consultant Dermatologist, Aura Skin Institute, SCO 44-45, Sector 9D, Madhya Marg, Chandigarh
OPEN ACCESS
PUBLISHED: 30 September 2025
CITATION: Garg, S., Lobo, S., Jain, N., 2025. Role of increasing protein intake and correction of micronutrients imbalance to promote desired autophagy in treating resistant dermatophyte infection: A cohort of 62 patients. Medical Research Archives, Online 13(9). https://doi.org/10.18103/mra.v13 9.6939
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v139.6939
ISSN 2375-1924
Abstract
Background: Chronic and recurrent dermatophytosis is being increasingly reported worldwide, posing significant therapeutic challenges. Emerging evidence suggests a potential role of host factors, including nutritional status and autophagy, in disease persistence.
Objective: To evaluate the clinical, nutritional, and biochemical profiles of patients with recalcitrant dermatophytosis and assess treatment outcomes following systemic antifungal therapy combined with protein and micronutrient supplementation.
Methods: A retrospective chart review of 62 patients with recalcitrant dermatophytosis was conducted. A subgroup (n=17) underwent detailed nutritional and biochemical assessment. All patients received oral itraconazole, and those with deficiencies were provided dietary counselling, protein and micronutrient supplementation.
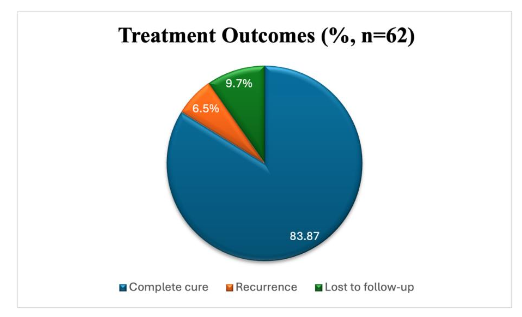
Results: Protein deficiency was observed in all patients assessed, with 58.8% consuming <50% of daily protein requirements. Vitamin D3 deficiency (82.3%) and low serum ferritin (76.5%) were prevalent. Comorbidities like diabetes and dyslipidemia were also noted. Clinical cure was achieved in 83.87% of patients, with recurrences in only 6.5% patients over period of one year linked to poor compliance or use of alternative therapies.
Conclusion: Host nutritional deficiencies, particularly in protein and iron, may contribute to deregulated autophagy and persistent dermatophytosis. Addressing these deficits alongside antifungal therapy improves treatment outcomes and reduces relapse risk. Evaluation for protein and micronutrient deficiencies and corrective measures should be integrated into the management protocols for recalcitrant dermatophytosis.
Keywords
Dermatophytosis, Autophagy, Proteins, Micronutrients.
Introduction
Superficial dermatophytosis is the fourth most common disease worldwide, with a surge in cases of recalcitrant and chronic dermatophytosis, particularly in the Indian subcontinent. Recalcitrant dermatophytosis is often refractory to standard antifungals and imposes a substantial socioeconomic burden by prolonging the disease course, impairing quality of life, increasing healthcare costs and facilitating transmission within communities. The persistence of this epidemic necessitates a revisit into the critical gaps in current understanding of host pathogen dynamics beyond fungal pathogenicity and drug resistance alone.
Therapeutic failure in dermatophytosis has been attributed to host non-compliance, inappropriate drug use, or fungal resistance in literature. The host immune response, nutritional status, and capacity for intracellular pathogen clearance remain underexplored in the context of chronic dermatophytosis. Autophagy, a key intracellular homeostatic and antimicrobial process, is essential in the clearance of pathogens and modulation of immune responses. Nutritional and micronutrient factors including protein intake, iron metabolism, and vitamin D status are known modulators of autophagy and immune function. Dysregulation of these pathways could potentially explain the persistence and chronicity of infection in certain individuals. However, the interplay between nutritional factors, autophagy, and treatment-resistant dermatophytosis has not been studied extensively in literature and there is a paucity of literature on the same. Addressing this lacuna is critical for identifying novel adjunctive strategies that could enhance treatment outcomes, especially in difficult to treat cases.
Through this study, we aim to assess the clinical, nutritional, and biochemical profiles of patients with recalcitrant dermatophytosis, with a particular focus on protein intake, iron homeostasis, vitamin D status, and autophagy-related dysfunction. By correlating these host factors with therapeutic response, the study seeks to explore the potential of host-directed interventions in improving management of chronic dermatophytosis.
Aims and Objectives
To evaluate the dietary habits, protein intake and micronutrient status in patients with recalcitrant dermatophytosis and to assess treatment outcomes following a combined regimen of systemic antifungals along with protein and micronutrient supplementation.
Materials and Methods
A retrospective chart review was conducted for all patients aged over 15 years who presented to a tertiary dermatology clinic in Chandigarh over the past four years (2019-2023) and were diagnosed with recalcitrant dermatophytosis (defined as persistent glabrous tinea, generally in settings like chronic, recurrent, corticosteroid-modified and resistant cases, with poor or no response to standard treatment) during this period. Demographic details, clinical characteristics (including disease duration, recurrence patterns, and prior treatment details), and associated comorbidities were noted and analysed. A subset of 17 patients underwent detailed nutritional and biochemical evaluation, which included the following:
- Dietary assessment: A 24-hour dietary recall was analysed using the ICMR food composition tables to calculate daily calorie and protein intake. The daily calorie (kcal), carbohydrate, and protein intake (g/kg/day) was then compared against the Recommended Dietary Allowances (RDA) as recommended by ICMR NIN (Hyderabad).
- Blood investigations: Serum protein, ferritin, iron, and hemoglobin levels.
- Inflammatory markers: Erythrocyte sedimentation rate (ESR) and red cell distribution width (RDW).
- Metabolic profile: Liver enzymes, lipid profile, serum vitamin D3 level and B12 level.
Treatment protocol: All adult patients received oral itraconazole 130 mg Super Bioavailable (SB) once daily for a duration of 6 weeks, along with topical azoles and antihistamines. The pediatric patient (Figure 3, panel A) was treated with itraconazole for 6 weeks at a dose adjusted for the body weight. Those diagnosed with onychomycosis were treated with an extended course of itraconazole pulse therapy in combination with topical antifungal nail lacquer.
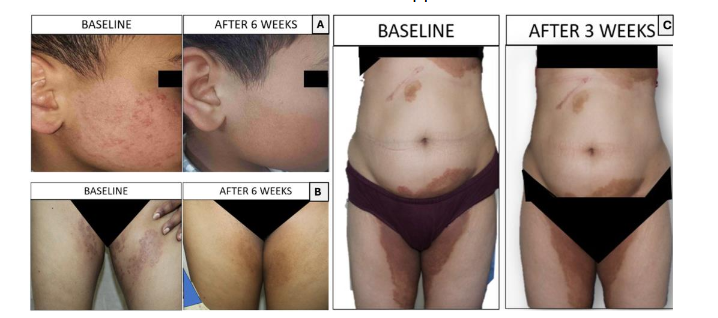
Patients who were identified with deficiencies were provided individualized dietary counselling, focusing on increased intake of protein-rich foods, along with an early morning plant-based protein supplement (1 1.5 g/kg/day). Although the RDA for protein is 0.8-1 g/kg/day for basic activity as per ICMR NIN (Hyderabad), we provided protein supplementation up to 1.5 g/kg/day based on the intensity of their physical activity. Those with normal hemoglobin and iron levels but low serum ferritin were recommended high protein supplementation to build up ferritin levels above 70 ng/mL. Specific micronutrient deficiencies were corrected with oral supplementation. Patients with low serum ferritin and iron deficiency received oral elemental iron therapy, while those with vitamin D insufficiency or deficiency were treated with oral cholecalciferol (60,000 international units (IU) once weekly for 6 8 weeks). Patients with Vitamin B12 deficiency were prescribed oral or intramuscular cyanocobalamin/methylcobalamin supplementation, depending on the severity of deficiency. Supplementation was continued until laboratory parameters normalized.
Follow up: Patients were followed up for one year post-treatment completion to assess clinical improvement and recurrence.
Statistical analysis: Data were analyzed using SPSS v16.0. Descriptive statistics (mean ± SD, median, frequencies, percentages) were used for demographic and clinical variables. Only descriptive analysis was performed. A p-value <0.05 was considered statistically significant.
Results:
A total of 62 patients were included in the study.
DEMOGRAPHICS:
The mean age was 32.5+/-13.1 years (range of 6-72 years). A male predominance was noted with 66.1% males (n=41) and 33.9% females (n=21), resulting in a male-to-female ratio of 1.9:1.
CLINICAL PROFILE:
The most common clinical presentation was tinea cruris et corporis, observed in 79% of patients (n=49). Other sites included tinea unguium (13%, n=8), tinea pedis/manuum (4.8%, n=3), and tinea faciei (3.2%, n=2).
The average duration of infection was 8.5 months (ranging from 2 months to 4 years).
TREATMENT DETAILS:
Prior to their visit to the clinic, all patients had received antifungal therapy, with 72.6%(n=45) having completed a 6-week course of oral and/or topical antifungals. All cases of onychomycosis (n=8) had received pulse therapy for a period of 4-6 months. Additionally, 14.4% of the study population (n=9) had used topical steroid-antifungal combinations before presenting to the clinic. Recurrence was noted in all the patients, experiencing a return of lesions within 6 weeks of completing prior treatment. Notably, 24.2% of patients (15/62) reported more than two episodes of recurrence.
NUTRITIONAL AND BIOCHEMICAL EVALUATION:
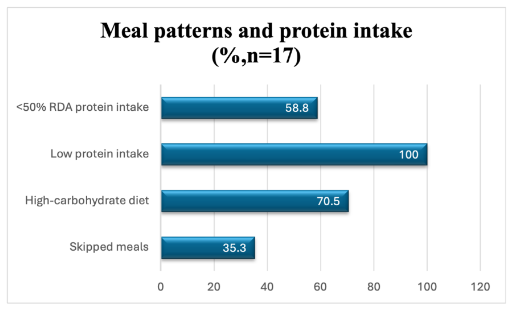
Of the 62 patients, a subgroup of 17 patients underwent detailed nutritional and biochemical evaluation via 24-hour dietary recall, Serum protein, ferritin, iron, and hemoglobin, ESR and RDW, liver enzymes, lipid profile, and vitamin D3 levels. Based on the dietary recall, all the patients had low protein intake, with majority of the patients (70.5%, n=12/17) consuming a high-carbohydrate diet with 35.3%(n=6/17) reporting skipped meals, and 58.8%(n=10/17) consuming less than 50% of their recommended daily protein intake.
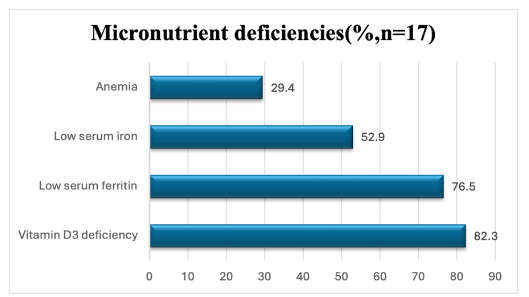
Vitamin D3 deficiency was seen in 82.3% (n=14/17) of this subgroup, low serum ferritin (< 70ng/ml) in 76.5% (n=13/17), low serum iron (< 70ug/dl) in 52.9% (n=9/17), and anemia in 29.4% (n=5/17). Raised ESR and RDW were observed in 35.3% (n=6/17).
Regarding metabolic comorbidities, 29.4% (n=5/17) had diabetes or prediabetes, 23.5%(n=4/17) had dyslipidemia, 23.5% showed elevated liver enzymes, and 5.9% (n=1/17) had gout.
POST TREATMENT AND NUTRITIONAL SUPPLEMENTATION OUTCOME:
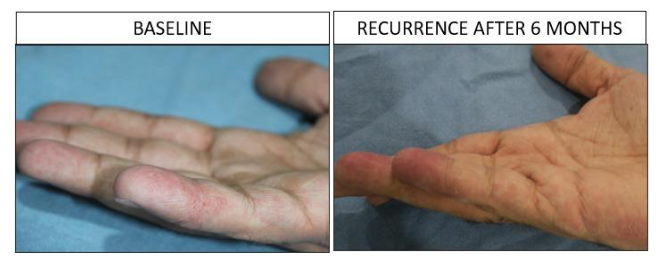
Following standardized treatment for all the 62 patients, 83.87% (55/62) of patients achieved complete clinical cure within 6-8 weeks. Recurrence over one year was documented in 6.5% (4/62), primarily in patients who defaulted on treatment or were using alternative therapies. Six patients (9.7%) were lost to follow-up and could not be assessed for outcomes.
Discussion:
Superficial dermatophytosis has emerged as the most common infectious dermatoses affecting almost 20-25% of the world population, with an alarming increase in chronic dermatophytosis in India. Despite being a superficial non-life threatening infection, it results in significant morbidity, financial and social burden for the patients. In light of the increasing cases of persistent or recurrent dermatophytosis, often modified by corticosteroid use and resistant to standard antifungal therapies we aimed to explore host factors, including immunity, autophagy, and micronutrient status, that may contribute to treatment resistance.
The interplay of host immunity, pathogen related, environmental, and barrier-related factors plays a crucial role in the pathogenesis of dermatophytosis.
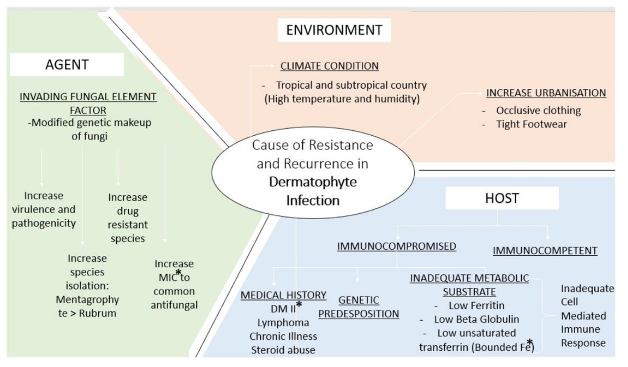
Environmental factors such as hot, humid climates, and lifestyle habits like wearing occlusive clothing or tight footwear, create a favourable niche for fungal colonization. Furthermore, epidermal barrier defects such as reduction of structural proteins, impaired keratinocyte turnover time, and delayed desquamation affect the skin’s ability to clear fungal elements, permitting persistent epidermal colonization. In our cohort, the observed male predominance, predilection for younger age group and frequent involvement of intertriginous were consistent with prior studies. This may be attributed to increased sweating, use of occlusive clothing, and occupational exposure in this group. The need for prolonged duration of antifungal treatment observed in our patients, further underscores the therapeutic challenges associated with recalcitrant tinea. Similar findings in recent literature have attributed the poor therapeutic response to emerging resistance, particularly from Trichophyton mentagrophytes complex, inappropriate use of fixed-dose corticosteroid combinations, and underlying host-related factors such as nutritional deficiencies and immune dysregulation.
The host immunity against dermatophytosis involves contribution by both the innate and adaptive immune system. Langerhans cells form the first line of defence of the skin barrier, by recognising the dermatophytes. Pattern recognition receptors (PRRs) such as Dectin-1, Dectin-2 and Mincle, recognize the pathogen-associated molecular patterns (PAMPs) such as β-glucans, triggering inflammatory signalling. These receptors signal through downstream adaptor proteins such as caspase recruitment domain-containing protein 9 (CARD9), which initiate intracellular pathways like Nuclear factor kappa B (NF-κB) activation, leading to the production of inflammatory mediators critical for an effective innate immune response. This cascade activates the adaptive immune response resulting in differentiation of T helper 17 (Th17) cell subset and cytokine release, reinforcing the host defence against fungal invasion. A study by Rai et al demonstrated a higher expression of Th17 and T regulator (Treg) cell markers on CD4+ cells of patients with chronic dermatophytosis as compared to controls, suggesting that an altered Th17/Treg balance contributed towards disease chronicity and higher fungal burden.
Dermatophytes colonize and persist within the stratum corneum due to secretion of proteolytic enzymes such as fungalysins, subtilisins, and metalloproteases that degrade keratin. The production of cell wall mannans further inhibits the proliferation of keratinocyte, delaying epidermal desquamation, permitting long-term survival within the epidermis. Additionally, they escape immune detection by masking the PAMPs. The downregulation/structural alteration of β-glucans expression prevents recognition by DECTIN-1 and impairs immune recognition and activation. In immunodeficient states, including CTLA-4 haploinsufficiency, impaired DECTIN-1 function has been demonstrated to increase susceptibility to fungal infections, potentially through autophagy dysfunction. This complex evasion of the host immunity by the dermatophytes contributes significantly to the persistence of infection despite adequate antifungal therapy.
AUTOPHAGY IN SKIN IMMUNITY AND DEFENCE:
Autophagy is the natural mechanism of lysosome-dependent degradation of proteins and dysfunctional organelles, crucial for regulating cellular functions and cytoprotective responses during states of nutrient deficiencies and stress. It plays a significant role in skin homeostasis, immunity, and pathogen defence. Dysregulation of autophagy has been linked with various inflammatory, autoimmune and skin infections such as streptococcal, staphylococcal and viral infections.
The role of autophagy in antifungal host defence has been highlighted in previous murine studies, where proteins such as autophagy-related genes 5 (ATG5) and ATG7 were shown to support macrophage and epithelial responses against Candida albicans. Additionally, exposure to antifungals was shown to induce the formation of autophagic vacuoles in Trichophyton mentagrophytes and Cryptococcus neoformans in animal models, indicating a key role of autophagy in fungal survival and treatment response. While these studies provide insights into the role of autophagy in fungal infections, no substantial human studies are available. In line with these findings, our observations in nutritionally deficient patients with chronic dermatophytosis suggest that inadequate host autophagy may contribute to persistent infection and influence treatment outcomes.
In the context of growing antifungal resistance, altered host immunity and barrier defects, the persistence of chronic dermatophytosis may be partially attributed to deregulated autophagy, which is influenced by host nutrition and micronutrient status.
ROLE OF PROTEIN IN PROMOTING AUTOPHAGY
Protein plays a critical role in tissue repair and immunity, with the availability of amino acids like leucine directly regulating the mammalian target of rapamycin (mTOR) pathway. mTOR is a nutrient sensor regulating autophagy against fungal infections depending on the host nutritional status. In states of prolonged protein deficiency or stress, downregulation of mTOR and impaired cytokine signalling, results in deregulated autophagy, weakening the host immunity. While short term starvation may transiently boost immunity, prolonged starvation and protein deficient diet, as noted in our cohort, is likely to deplete nutrients leading to deregulated autophagy which in turn predisposed to chronic infection and immune dysregulation.
Authors have previously noted reversal and prevention of deregulated autophagy in autoimmune disorders such as vitiligo by correcting micronutrient deficiencies and introducing early morning low carb-high protein nutrition, eventually reducing disease burden and recurrence.
FUNGAL AUTOPHAGY IN THE EVENT OF LOW PROTEIN AND MICRONUTRIENT STATUS
Among the 17 patients who were assessed in detail for dietary and biochemical parameters in our study, a consistent pattern of nutritional deficiency was observed in almost all patients. All the patients (100%) had protein intake below the recommended dietary allowance (RDA), with over half (n=10) of them consuming less than 50% of daily requirements. Dietary patterns such as high-carbohydrate diets (70.5%) and meal skipping; especially breakfast (35.3%), both of which may influence stress responses.
There are limited human studies directly linking iron/protein deficiency to chronic dermatophytosis. Low-protein states inhibit the mTOR signalling pathway, activating the autophagy-related (ATG) genes and formation of autophagosomes. This deregulated autophagy favours fungal proliferation by degradation and recycling of damaged organelles, misfolded proteins, and other cytoplasmic components into essential substrates like amino acids and fatty acids to aid in synthesis of ergosterol and keratin-degrading enzymes in the fungi. In high-carbohydrate and low-protein dietary conditions, the abundant glucose and free iron, particularly in the absence of adequate ferritin fosters an ideal environment for the dermatophytes and promotes fungal growth. Additionally, the activation of serine subtilisins, proteases and fungalysin results in invasion of keratinocyte cell membrane. These conditions result in deregulated autophagy, leading to immune evasion, increased virulence of the pathogens and resistance to antifungals which could explain the persistence of fungal infection in our patients.
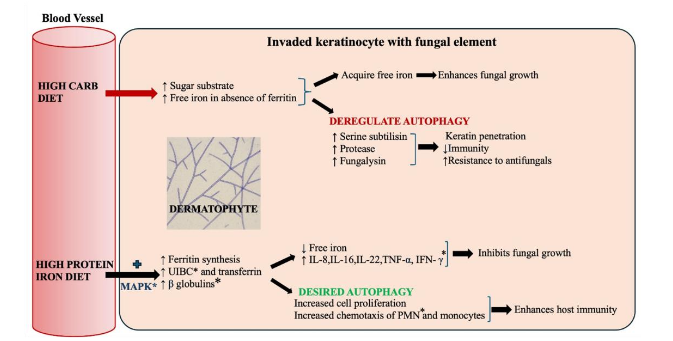
In contrast, a high-protein intake increases the synthesis of ferritin, unsaturated iron-binding capacity (UIBC), transferrin, and β-globulins, which results in iron storage and limited iron availability, thereby limiting fungal growth. The increased epidermal proliferation in the parakeratotic keratinocytes results in elimination of the fungal hyphae in the scales. Pro-inflammatory cytokines such as IL-8, IL-1β, IL-22, TNF-α, and IFN-γ are upregulated occurs via the MAPK (Mitogen-Activated Protein Kinase) signalling pathway, leading to chemotaxis of polymorphonuclear cells (PMNs) and monocytes. This promotes autophagy which facilitates effective fungal clearance and augments host defence mechanisms, as was demonstrated by resolution of infection in our patients following a high protein diet and protein supplementation.
ROLE OF MICRONUTRIENTS IN PROMOTING IMMUNITY:
In our cohort, significantly low levels of serum ferritin were seen in majority of the patients, with concomitant low serum iron and anemia. Iron is an essential micronutrient crucial for both host and pathogen. Nutritional immunity is a mechanism of host defence against microbial iron acquisition, wherein iron is tightly sequestered by host ferritin and transferrin. Ferritin, an iron storage protein present in macrophages, binds to free iron and limits the bioavailability to fungi, subsequently inhibiting dermatophytic growth. Low ferritin levels impair macrophage and neutrophil function, compromise T-cell proliferation, and disrupt skin barrier integrity and increase vulnerability to oxidative stress resulting in undesired deregulated autophagy via ferritophagy. Other key proteins such as transferrin, lactoferrin and siderocalin sequester iron, thereby preventing microbial access causing microbial starvation. Protein intake improves iron absorption and increases the synthesis of ferritin and transferrin. In protein-deficient states, impaired ferritin synthesis results in reduced iron sequestration, lowered immunity and increased susceptibility to chronic or recurrent dermatophytosis.
Conversely, iron overload or inflammatory states with elevated ferritin levels can increase oxidative stress and suppress effective autophagy. This was noted in 35.3% of our cases where inflammatory markers like erythrocyte sedimentation rate (ESR), red cell distribution width (RDW) were raised. Of note, a single patient in our cohort exhibited elevated serum iron with low ferritin. Elevated iron in this context may, paradoxically, enhance fungal growth and impair immune responses through oxidative damage and mTOR suppression.
Vitamin D3 deficiency was also highly prevalent in our cohort, a finding consistent with previous studies which highlight the potential role of hypovitaminosis D in inflammatory dermatoses. Raja SK et al also noted vitamin D3 deficiency in majority of their patients, with dermatophytosis, which aligns with our observation. The other common micro-nutrient deficits in our cohort included low ferritin, iron, and protein intake. This constellation of nutritional deficiencies low ferritin, protein, and vitamin D may provide a plausible explanation for the chronicity and poor therapeutic response observed in dermatophytosis.
In addition to nutritional deficits, the study population demonstrated a high prevalence of comorbidities, which are known to impair host immune function and compromise the integrity of the skin barrier. Notably, 29.4% of the patients (5/17) were diagnosed with type 2 diabetes mellitus or pre-diabetes, while 23.5% (4/17) exhibited dyslipidaemia, and an equal proportion had elevated liver enzymes, suggesting hepatic dysfunction. A high protein diet addresses the root cause of metabolic syndrome resulting in better control of diabetes, hyperlipidemia, obesity and cardiac health. Additionally, it protects liver health and rescues it from non-alcoholic fatty liver disease. Owing to the improved health, the liver may also get a buffer from Itraconazole induced liver injury due to repeated courses in recalcitrant cases. These findings highlight the potential role of underlying metabolic dysfunction in perpetuating chronic dermatophytosis and the need to address these systemic factors for therapeutic success.
Our therapeutic approach incorporated systemic antifungal agents in combination with topical antifungals, along with targeted correction of nutritional deficits, particularly protein and micronutrients. This strategy resulted in clinical resolution in the majority of patients with relapses over one year occurring mainly in those who defaulted on treatment or resorted to alternative therapies. While the IADVL Taskforce guidelines recommend adding a different class of topical antifungals (such as allylamines) in recalcitrant dermatophytosis to minimize resistance, our patients (83.87%) responded well to azole-based topicals when combined with oral itraconazole and nutritional supplementation. This suggests that correction of micronutrient and protein deficiencies may play an important role in improving therapeutic outcomes in recalcitrant dermatophytosis. These findings add to the growing body of evidence supporting host-directed adjunctive strategies in resistant tinea to achieve better long-term control. Notably, patients with onychomycosis required prolonged treatment, including itraconazole pulse therapy and adjunctive topical agents, which is consistent with current therapeutic guidelines.
Overall, the favorable outcomes in our cohort reinforce the hypothesis that restoring the protein-ferritin-autophagy balance is crucial for both treatment success and relapse prevention in chronic dermatophytosis.
Conclusion:
While there is a paucity of literature on the effect of protein intake and ferritin levels in dermatophytosis, parallels with other dermatoses such as alopecia areata and atopic dermatitis suggest that protein intake and iron homeostasis are crucial to skin immunity. Further studies are warranted to explore the role of autophagy in dermatophyte infections.
Adjunctive therapies to address the host nutritional status are important, given the increasing rates of chronic and recalcitrant dermatophytosis. In our experience, early morning high-protein, low-carbohydrate diets and micronutrient supplementation, particularly iron and vitamin D, need to be incorporated into the management of dermatophytosis.
Limitations:
Although the results of our study are promising, they are limited by the smaller subgroup that underwent detailed nutritional evaluation, absence of a control group, and lack of direct assessment of autophagy markers. These constraints restrict the direct causality between micronutrient deficiencies and autophagy. Future prospective studies with expanded cohorts, measurement of autophagy-related biomarkers, and comprehensive nutritional profiling will be essential to confirm these results and further elucidate the host pathogen interactions underlying recalcitrant dermatophytosis.
Conflicts of Interest Statement:
The authors have no conflicts of interest to declare.
Funding Statement:
This research received no external funding.
Acknowledgments:
None.
References:
- Rengasamy M, Shenoy MM, Dogra S, Asokan N, Khurana A, Poojary S, et al. Indian Association of Dermatologists, Venereologists and Leprologists (IADVL) Task Force against Recalcitrant Tinea (ITART) Consensus on the Management of Glabrous Tinea (INTACT). Indian Dermatol Online J. 2020 Jul; 11(4):502-19.
- Indian Council of Medical Research National Institute of Nutrition. (2017). Indian food composition tables. Hyderabad: National Institute of Nutrition, ICMR.
- Indian Council of Medical Research National Institute of Nutrition, Expert Group. (2020). Nutrient requirements for Indians: Recommended dietary allowances and estimated average requirements. Hyderabad: National Institute of Nutrition, ICMR.
- Medindia. Protein Intake Calculator [Internet]. Chennai: Medindia; 2024 [cited 2025 Aug 29]. Available from: https://www.medindia.net/patients/calculators/protein-intake-calculator.asp
- Havlickova B, Czaika VA, Friedrich M. Epidemiological trends in skin mycoses worldwide. Mycoses. 2008 Sep;51 Suppl 4:2-15.
- Verma SB, Panda S, Nenoff P, Singal A, Rudramurthy SM, Uhrlass S, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: I. Epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021;87(2):154-75.
- Deng R, Wang X, Li R. Dermatophyte infection: from fungal pathogenicity to host immune responses. Front Immunol. 2023 Nov 2;14:1285887.
- Ferwerda B, Ferwerda G, Plantinga TS, Willment JA, van Spriel AB, Venselaar H, et al. Human Dectin-1 deficiency and mucocutaneous fungal infections. N Engl J Med. 2009 Oct 29;361(18):1760-7.
- Lanternier F, Mahdaviani SA, Barbati E, Koumar Y, Levy R, Ghandil P, et al. Frequency and geographic distribution of CARD9 mutations in patients with severe fungal infections. Front Microbiol. 2018 Oct 9;9:2196.
- Rai G, Das S, Ansari MA, Singh PK, Pandhi D, Tigga RA, et al. The interplay among Th17 and T regulatory cells in the immune dysregulation of chronic dermatophytic infection. Microb Pathog. 2020 Feb;139:103921.
- García-Romero MT, Arenas R. New insights into genes, immunity, and the occurrence of dermatophytosis. J Invest Dermatol. 2015 Mar;135(3):655-7.
- Chai LYA, Netea MG, Vonk AG, Kullberg BJ. Fungal strategies for overcoming host innate immune response. Med Mycol. 2009 May;47(3):227-36.
- Collette JR, Lorenz MC. Mechanisms of immune evasion in fungal pathogens. Curr Opin Microbiol. 2011 Dec;14(6):668-75.
- Mizushima N, Levine B, Cuervo AM, Klionsky DJ. Autophagy: renovation of cells and tissues. Cell. 2008 Nov 28;132(4):697-710.
- Edinger AL, Thompson CB. Death by design: apoptosis, necrosis and autophagy. Curr Opin Cell Biol. 2004 Dec;16(6):663-9.
- Kroemer G, Levine B. Autophagy: dual roles in life and death? Nat Rev Mol Cell Biol. 2008 Jun;9(6):505-14.
- White E. The role of autophagy in cancer development and response to therapy. Nat Rev Cancer. 2012 Jun;12(6):401-10.
- Garg S, Sangwan A. Dietary protein deficit and deregulated autophagy: a new clinico-diagnostic perspective in pathogenesis of early aging, skin, and hair disorders. Indian Dermatol Online J. 2019; 10(2):115-24.
- Nicola AM, Albuquerque P, Martinez LR, Dal-Rosso RA, Saylor C, De Jesus M, et al. Macrophage autophagy in immunity to Cryptococcus neoformans and Candida albicans. Infect Immun. 2012 Sep;80(9):3065-76.
- Kanayama M, Inoue M, Danzaki K, Hammer G, He YW, Shinohara ML. Autophagy enhances NF-κB activity in specific tissue macrophages by sequestering A20 to boost antifungal immunity. Nat Commun. 2015 Jan;6:5779.

