Preventing Cancer-Related Lymphedema via Ketogenic Diet
Metabolic Health: The key to preventing cancer treatment-related lymphedema?
Leslyn Keith 1, Kristin Taylor 2, Siobhan Huggins 3, Christy Kesslering 4
- Lymphatic Lifestyle Solutions (corresponding author)
- Kristin Taylor Consulting.
- Lipedema Simplified
- KessRx
OPEN ACCESS
PUBLISHED 31 December 2025
CITATION Keith, L., Taylor, K., et al., 2025. Metabolic Health: The key to preventing cancer treatment-related lymphedema? Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7049
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i12.7049
ISSN 2375-1924
ABSTRACT
Lymphedema is a chronic and often irreversible complication of cancer treatment, marked by persistent tissue swelling, fibrosis, and adipose deposition in the affected body region. Although several risk factors have been identified, predicting which patients will develop lymphedema remains a significant clinical challenge. Growing evidence links poor metabolic health and insulin resistance to increased susceptibility to lymphedema, while interventions that improve metabolic function, including glucagon-like peptide-1 (GLP-1) receptor agonists and diabetes medications, have shown symptom-reducing effects. Notably, the ketogenic diet (KD), known for reversing insulin resistance and enhancing metabolic health, has demonstrated promise in managing lymphedema. A KD may confer additional advantages through ketone exposure. This paper explores the hypothesis that proactively improving metabolic and lymphatic health, specifically through ketogenic dietary strategies, could offer a novel means of preventing lymphedema in individuals undergoing cancer treatment. By examining the metabolic underpinnings of lymphedema and the beneficial targets of ketone exposure, we advocate for a paradigm shift: from managing lymphedema as a secondary complication to preventing it through early ketogenic dietary intervention.
Keywords: lymphedema, metabolic health, ketogenic diet, cancer treatment
Introduction
Lymphedema is a chronic, progressive condition marked by the pathological accumulation of protein-rich interstitial fluid, reactive fibrosis, and adipose tissue hypertrophy due to impaired lymphatic function. It may arise congenitally (primary lymphedema) or from secondary causes, such as a consequence of conventional treatments for cancer. While estimates vary due to inconsistent diagnostic criteria and reporting practices, recent analyses suggest that up to 250 million people globally are affected.
Although many individuals believe that they are following healthy habits long before a cancer diagnosis, current recommendations often overlook dietary quality and tend to inadvertently emphasize industrialized, high-carbohydrate (CHO) eating patterns. Heeding this mainstream advice may, in practice, elevate the risk of developing lymphedema after undergoing cancer treatment. A CHO-rich Western diet is strongly linked to poor metabolic health and recent studies have highlighted metabolic dysfunction, particularly insulin resistance and obesity, as a significant risk factor for post-treatment lymphedema. Given that only 6.8% of U.S. adults meet criteria for optimal metabolic health, the potential impact of diet on lymphedema risk warrants urgent clinical and public health attention.
When paired with a CHO-rich diet, the body’s natural tendency to develop insulin resistance during periods of physiological or psychological stress, such as occurs with cancer diagnosis and treatment, can intensify markers of poor metabolic health. This may include the development of type 2 diabetes and unfavorable shifts in body composition, such as increased visceral fat and total adiposity. The combination of innate vulnerability, suboptimal dietary patterns, and the metabolic disruptions caused by standard cancer therapies creates a “perfect storm” that may significantly increase the risk of developing cancer treatment-related lymphedema (CTRL). Without targeted intervention to reverse insulin resistance and restore metabolic balance, an elevated lymphedema risk will persist long after cancer treatment has ended, compromising long-term lymphatic health.
Encouragingly, interventions that target metabolic dysfunction, including GLP-1 receptor agonists and other insulin-sensitizing therapies, have shown benefits in reducing lymphedema symptoms. Among dietary approaches, the KD stands out for its effectiveness in improving metabolic parameters and reversing insulin resistance, both with and without weight loss. Beyond the well-established metabolic benefits of a KD, however, recent research suggests that ketone bodies themselves may confer therapeutic effects uniquely relevant to the prevention of CTRL.
Efforts to promote ketogenic strategies in oncology can be hindered by provider concerns regarding patient burden, dietary safety, and limited familiarity with emerging evidence, all factors that may inadvertently restrict patient access to a potentially beneficial intervention. In this paper, we present both a plausible mechanistic rationale for the therapeutic effects of a KD and evidence supporting its safety and efficacy in individuals affected by cancer.
Pathophysiological processes of cancer treatment-related lymphedema
A common form of secondary lymphedema arises after cancer treatment, initiated by injury to lymphatic vessels or lymph nodes during surgery (e.g., axillary lymph node dissection) and/or radiation therapy. Yet lymphatic injury alone is insufficient to explain why some patients develop lymphedema while others receiving identical treatments do not. Moreover, symptom onset may be delayed for months or even years after the initial intervention, indicating that additional factors contributing to lymphedema pathogenesis emerge over time. We propose that, although lymphedema has traditionally been viewed as a purely mechanical consequence of cancer therapies, metabolic dysfunction may represent a primary and previously underappreciated driver.
To clarify the biological mechanisms of lymphedema and support our prevention hypothesis, we use the analogy of a house representing the human body to contrast effective repair and resolution after lymphatic injury with the disrepair and dysfunction that result in lymphedema. In a healthy model of lymphatic repair, where a strong underpinning of metabolic health exists, the body is like a well-maintained house with a solid foundation. Imagine that a single stairway collapses due to a localized incident, representing the initial lymphatic injury from cancer surgery and/or radiation treatment. Promptly, the repair crew (the body’s adaptive response) arrives via the lymphatics, focusing on clearing debris (edema and metabolic waste), reinforcing the structure with quality materials (functional lymphatic vessel regrowth), and quickly restoring full usability. The house’s solid foundation of optimal metabolic health ensures the repairs hold without complications.
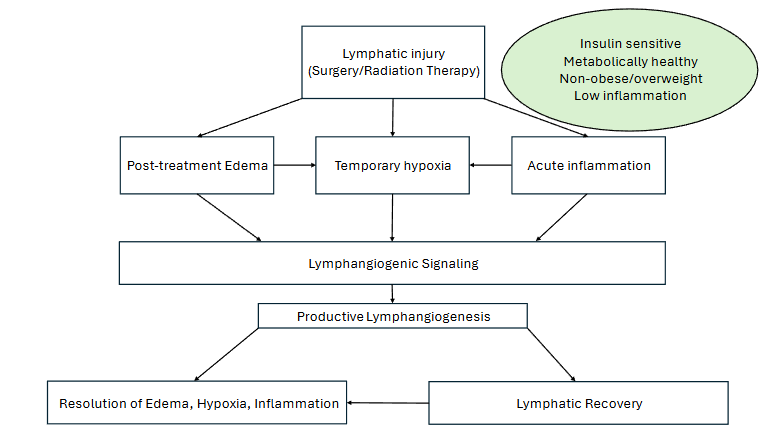
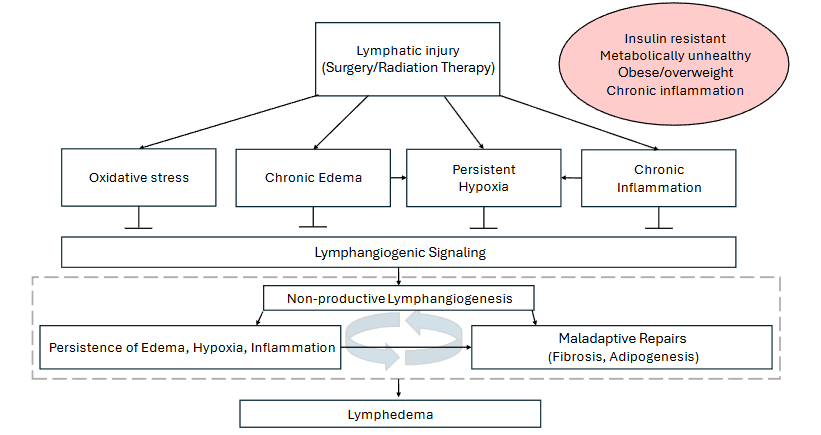
The complex cellular events that transform lymphatic injury into a chronic lymphedema in the unhealthy model begin in a house with deep, pre-existing structural flaws, such as a caved-in roof from years of neglect (metabolic dysfunction, such as insulin resistance, hyperglycemia, and chronic low-grade inflammation). When the stairway collapses, the already overwhelmed crew is unable to properly prioritize repair of this damage and instead uses haphazard, mismatched materials (non-functional lymph vessel growth) in a rushed repair attempt. This is partially due to a lack of necessary downtime needed to focus on the complete repair of damage caused by contradictory signals from chronic inflammation and poor underlying health. Haphazard fixes warp and fail over time, attracting maladaptive and unhealthy responses (chronic hypoxia, elevated oxidative stress, and excessive inflammation) that cause the stairs to bulge and crack further (e.g., vulnerability due to repeated localized tissue infection). The blocked-off, unusable stairway leads to deterioration in the surrounding areas of the house (persistent lymph stasis and tissue remodeling, such as fibrosis and adipogenesis). What could have been a quick, targeted repair spirals into an irreparable section of the house (chronic lymphedema), as the foundational problems corrupt every effort, turning adaptive responses into a vicious cycle of escalating damage.
Risk factors
While not all individuals undergoing cancer treatment will develop lymphedema, the profound impact of the disease on quality of life has made the identification of risk determinants a critical focus of research. Current evidence indicates that CTRL arises from direct trauma to the lymphatic system secondary to oncologic therapies that are compounded by patient-specific vulnerabilities. Treatment-related risk factors include the extent of surgical intervention, the number of lymph nodes excised, administration of radiation therapy, certain chemotherapy regimens, advanced tumor stage, tumor-induced lymphatic obstruction, and postoperative complications such as infection and seroma formation. Although some cancers, such as those often treated by chemotherapy alone (e.g., lymphoma and leukemia), are much less likely to pose a lymphedema risk than others, the risk is not entirely absent.
Nontreatment-related factors associated with an elevated risk of CTRL include lower socioeconomic status and advanced age. Additional factors that may be modifiable include physical inactivity and comorbid conditions such as obesity, hypertension, vascular disease, metabolic syndrome, and insulin resistance. Specifically, chronic hyperglycemia and insulin resistance can cause lymphatic dysfunction and thus can be a significant contributor to the onset of lymphedema.
Conventional treatments for cancer treatment-related lymphedema
Current lymphedema treatments remain largely palliative, focusing on symptom control rather than disease modification or prevention. In the context of our metaphor, conventional treatments are like managing the symptoms of a house in disrepair. They can patch leaks and shore up collapsing sections, but they don’t address the foundational problems that exacerbated the damage in the first place. The focus is on reactive symptom management rather than proactive, structural repair.
Once diagnosed, the patient undergoes a treatment protocol that may include specialized manual techniques, skin care, compression therapy, and exercise. Surgical options can also be considered, including liposuction to remove excess adipose deposits and microsurgery to improve lymphatic drainage from the affected region. A procedure known as immediate lymphatic reconstruction can significantly reduce the occurrence rate, but 9.5% of non-obese patients still experienced lymphedema in one study. The patient is cautioned that although lymphedema can be managed, it is generally irreversible and will be a chronic, life-long condition.
Frequent infections in the unhealthy model can have serious consequences for individuals with CTRL, often exacerbating the condition and leading to a cycle of worsening lymphatic dysfunction. Recurrent infections can directly damage lymphatic endothelial cells, impairing their structure and function. Each episode of infection increases fibrosis and inflammation within the tissue, further contributing to lymphatic failure and increased tissue damage. The impact on immune functions is severe, as the lymphatic system becomes less efficient at trafficking immune cells and clearing pathogens due to sustained reduction in lymphatic vessel contractions post-infection. Additionally, the increased load of stagnant fluid in the affected body area creates a favorable environment for bacterial growth, which increases susceptibility to future infections. Frequent infections and often repeated courses of antibiotics, with severe cases resulting in hospitalizations, can increase the burden of the disease and negatively impact quality of life.
Managing lymphedema often requires time-consuming daily self-care, costly treatments and equipment, and carries the risk of hospitalization due to serious infections and other complications. This ongoing struggle, often with no guarantee of improvement, can lead to significant emotional distress, including shame, embarrassment, social isolation, depression, anxiety, anger, and profound fatigue. For many, particularly cancer survivors, lymphedema becomes a chronic, painful, and disfiguring reminder of their treatment experience. Despite its impact, the emotional toll of lymphedema remains poorly understood by many clinicians. The psychological burden is frequently underestimated in both clinical and research settings, yet it represents a critical component of patient well-being. Everyday occurrences, such as insect bites, minor injuries, or sunburn, can trigger medical emergencies, fostering a persistent sense of vulnerability and fear. This ongoing uncertainty contributes to frustration, despair, and emotional exhaustion, often progressing into chronic anxiety and depression. Many patients describe their experience as a constant effort to manage symptoms and prevent disease progression, with little hope for lasting relief. The visible disfigurement that often accompanies lymphedema further exacerbates body image concerns and diminishes quality of life.
These substantial physical and psychosocial challenges have driven patients, clinicians, and researchers alike to seek not only a cure but also effective risk-reduction strategies and improved long-term management options. A compassionate, comprehensive approach to care, one that addresses both physiological and emotional dimensions, is essential to truly support individuals living with lymphedema.
The ketogenic diet: A novel metabolic intervention for cancer treatment-related lymphedema prevention
A ketogenic diet can be seen as an intervention that addresses the underlying problems, as explained in the house metaphor. An effective preventive strategy would ideally target both the pathophysiological processes as well as the associated metabolic risk factors associated with CTRL. Here, we explore the potential of a therapeutic KD as a multifaceted intervention that may address these interconnected concerns. We provide plausible mechanisms of action through two main pathways: improved metabolic health and benefits associated with ketone exposure. We therefore suggest that a KD may perform as not just a treatment option, but as a formidable reduction in the risk of developing lymphedema.
Ketogenic Diet Definition
A KD is a structured nutritional regimen characterized by a substantial reduction in CHO intake, concomitant with a proportionate elevation in dietary fat to meet energy needs, as well as a moderate allocation of protein. The fundamental objective of a ketogenic dietary approach is to induce a metabolic state known as ketosis, wherein the body transitions from reliance on glucose as its primary energy substrate to the utilization of fatty acids and ketone bodies, which are themselves synthesized from the catabolism of fatty acids. A KD mandates a macronutrient distribution that deviates from conventional dietary guidelines. The caloric contribution from fats frequently ranges between 70% and 80%, while protein is generally about 20% of total caloric intake. CHO consumption is restricted to 5% of calories, or less than 50 grams per day. A reduction in CHO availability leads to a significant reduction in hepatic glycogen stores. Consequently, the liver ramps up the process of ketogenesis as a result of beta-oxidation of fatty acids, and these ketones then serve as an alternative and efficient fuel source for various tissues, including the brain. An adaptive metabolic shift from glucose to fatty acids is central to the therapeutic and physiological effects observed with the KD.
Hypothesis: Ketogenic diet as a prevention of cancer treatment-related lymphedema
We propose that the biological effects of a KD may represent an underrecognized strategy for improving lymphatic function and facilitating lymphatic healing and regeneration in individuals at risk for CTRL, particularly those undergoing radiation or surgical interventions known to disrupt lymphatic architecture. By shifting the clinical focus upstream toward metabolic optimization before or during cancer therapy, we argue for a reorientation of lymphedema care: from reactive symptom management to proactive risk reduction and possible prevention through early, ketogenic dietary intervention. Specifically, we highlight the importance of targeting the metabolic and inflammatory conditions that underlie lymphatic vulnerability and offer a novel, non-pharmacologic pathway for reducing the incidence and severity of lymphedema.
Though there may be multiple pathways to achieving metabolic health, we emphasize the KD because it combines a low risk of serious side effects with a set of distinctive therapeutic properties that directly counteract the pathophysiological processes underlying CTRL. The benefits of a ketogenic approach are twofold. First, a KD has demonstrated effectiveness in improving metabolic health, particularly by reducing visceral adiposity, improving insulin sensitivity, and lowering systemic inflammation, all of which are associated with mitigating the risk of lymphedema following cancer treatment. Second, the exposure to ketone bodies produced during nutritional ketosis provides unique cellular and molecular benefits that may protect and repair lymphatic tissues by providing a protective buffer against the unavoidable lymphatic injury experienced with conventional cancer treatments. In mouse studies and in vitro models, ketone metabolism has been shown to enhance the body’s ability to recover and remain resilient in the face of such injuries. Additionally, emerging ketogenic diet research shows promise in humans that lymphangiogenesis correlates with ketone levels. By integrating metabolic optimization with tissue-level protection, a KD offers an effective approach to preventing or attenuating CTRL in at-risk populations.
Systemic metabolic health
Metabolic health refers to the optimal functioning of physiological processes that regulate energy production and utilization, encompassing glycemic control, lipid metabolism, insulin sensitivity, blood pressure, and body fat distribution. As we described in our introductory metaphor, the body’s foundation in metabolic health is a critical factor in averting the development of lymphedema. Unfortunately, disorders of metabolism, including obesity, insulin resistance, diabetes mellitus, and metabolic syndrome, afflict a substantial portion of the adult population. The findings of multiple studies underscore the importance of systemic metabolic health in influencing lymphatic function and lymphedema onset and progression. For instance, there is a strong association between obesity and lymphedema, both in patients with and without cancer treatment. Yusof et al. reported that breast cancer survivors with diabetes mellitus had nearly a 10-fold increased risk of early-onset lymphedema. Doruk Analan et al. similarly identified a significant association between insulin resistance, central obesity, and lymphedema related to breast cancer treatment. In a subsequent study, Yusof et al. confirmed that breast cancer survivors with lymphedema were more likely to have hypertension and/or diabetes mellitus compared to survivors who did not have lymphedema. Furthermore, a study of 40 women with breast CTRL found that conservative lymphedema treatments were more effective in participants without comorbid metabolic syndrome, suggesting that underlying metabolic health status may influence treatment efficacy. The role of metabolic health was highlighted in a case study report when complete resolution of chylothorax secondary to thoracic duct injury, compounded by insulin resistance, was achieved within three months of starting a KD.
Insulin resistance and metabolic derangement
Metabolic derangement refers to a disruption in the body’s normal metabolic processes, including energy production, nutrient use, tissue repair, and waste removal. When these processes become dysregulated, the body’s ability to maintain balance is impaired, setting off a cascade of adverse effects, the most significant of which may be insulin resistance. Insulin plays a central coordinating role in metabolism. It helps regulate blood glucose by reducing glucose production in the liver, promoting glucose uptake by tissues, and limiting fat breakdown in adipose tissue. Insulin resistance occurs when tissues such as skeletal muscle, liver, and fat no longer respond appropriately to insulin’s signals. This state can be triggered by several mechanisms, including chronic inflammation, mitochondrial dysfunction, and cellular stress.
Conventional dietary recommendations often emphasize low-fat, low-calorie eating patterns that frequently rely on poor-quality foods. Although the high refined CHO content of most modern industrialized diets is considered a primary driver of metabolic dysfunction, additional dietary components are also implicated in impaired metabolic health and obesity. These include industrial seed oils, high fructose corn syrup, and novel food additives or ingredients that may promote gut inflammation. Consequently, a secondary advantage of a KD may derive from its inherent guidance to limit or exclude these potentially deleterious components. For these reasons, although such high-CHO, low-fat (HCLF) dietary approaches are widely promoted for health and weight management, they may paradoxically contribute to weight gain and metabolic dysfunction. For many individuals who adhere to mainstream recommendations with diligence, progress toward improved health remains limited or ultimately stalls. This outcome reflects not a lack of motivation or adherence, but the predictable metabolic consequences of an industrialized HCLF dietary pattern.
Compelling evidence demonstrates that HCLF diets, particularly in metabolically compromised individuals, provoke exaggerated insulin and glucose responses that lead to subsequent postprandial glucose drops, intensifying hunger and cravings. This hormonal cascade is further compounded by the low satiety of such diets, as fat is a key macronutrient for promoting feelings of fullness and suppressing appetite. For those with metabolic disease, these widely promoted dietary recommendations are not merely ineffective; they are physiologically inadequate, setting the stage for a cycle of temporary success followed by inevitable frustration and relapse. The physiological consequences are sometimes termed the Insulin Roller Coaster Effect.
| Consequence | Description |
|---|---|
| Constant insulin demand | Eating 5-6 times per day, especially CHO-centric foods, means the body is almost constantly in a fed state, requiring continual insulin production. |
| No time for fasting or fat burning | A frequent eating pattern doesn’t allow the body enough time to dip into its fat stores for energy (which naturally happens during periods between meals or overnight fasting) as the body is constantly prioritizing glucose as fuel. |
| Hunger between meals | Despite frequent meals, the quick digestion of low-fat, high-CHO options can lead to more hunger and cravings between small meals, turning the “snack” into a necessity rather than an option. |
| Persistent hyperinsulinemia | High levels of insulin prevent access to stored fat and result in a vicious cycle of hunger, impaired glucose tolerance, more hunger, and chronic inflammation. |
Fluid balance and transport
One of the primary functions of the lymphatic system is to maintain tissue fluid homeostasis by collecting and returning excess interstitial fluid to the bloodstream. Fluid balance depends on the structural integrity and functional capacity of lymphatic vessels, which must adapt to physiological demands under varying conditions. Cancer treatments, such as surgery and radiation, can significantly impair lymphatic structure and function, thereby reducing transport capacity at a time when demands on the system are elevated due to inflammation and tissue damage from these very treatments. If present, comorbid metabolic disorders, such as liver disease, diabetes, and obesity, can further increase lymphatic load while simultaneously impairing transport mechanisms. As noted previously, metabolic disorders are associated with increased lymphatic vessel permeability and reduced contractile strength and frequency, both of which contribute to fluid accumulation. Impaired lymphatic transport also prevents the effective removal of macromolecules, metabolites, and other waste products, exacerbating local tissue damage. This means that disruptions to lymphatic function will contribute to a pro-inflammatory, hypoxic, and oxidative microenvironment that further impairs immune function and fluid balance, compounding vulnerability to CTRL.
Lymphangiogenesis is particularly crucial following cancer treatment-related lymphatic trauma. As fluid stasis becomes chronic, the body increasingly relies on lymphangiogenesis to manage the excess fluid burden. However, for repair to be effective, the new lymphatic vessels must be functional, well-integrated, and capable of supporting fluid transport under stress, which may not always be the case in the presence of metabolic disorders. KDs enhance metabolic health in ways that directly support lymphatic transport and fluid regulation in tissues vulnerable to or affected by lymphedema. Systemic metabolic improvements driven by a KD create a physiological environment that favors efficient lymphatic function. Furthermore, exposure to ketone bodies has been shown to promote lymphatic vessel integrity and stimulate lymphangiogenesis, reinforcing structural and functional lymphatic capacity. Recent evidence suggests that KDs may influence metabolic and growth factor pathways that promote effective and functional lymphangiogenesis. Animal studies indicate that fatty acid β-oxidation and ketone signaling can stimulate lymphatic sprouting, potentially improving transport capacity in damaged tissues. Clinical data with human subjects are still emerging, but early findings are promising. In an exploratory study, individuals with lymphedema who followed either a classic ketogenic or modified Atkins diet showed improvements in lymphatic function, as demonstrated by reduced dermal backflow on lymphatic imaging. Although average edema volume did not significantly decrease across the entire cohort, several participants experienced a clinically meaningful volume reduction, suggesting that the diet supported functional new vessels. Collectively, these effects suggest that the metabolic optimization achieved through ketogenic dietary interventions may play a central role in restoring lymphatic balance and preventing fluid overload in at-risk tissues.
Microenvironment of at-risk and/or affected tissue
The initial analogy of the house with a collapsed stairway can illustrate the impact of lymphatic injury on the microenvironment of at-risk and/or affected tissue. Although the initial lymphatic trauma is the damage to the stairway, a multitude of pre-existing issues can compromise the repairs. These issues are the foundational flaws of the house, specifically metabolic dysfunction, insulin resistance, and chronic inflammation that create a chaotic environment for repair. Physiological stressors, both internal (e.g., inflammation, hypoxia, oxidative stress, metabolic dysregulation) and external (e.g., surgery, radiation therapy, poor diet, infection), can disrupt homeostasis and initiate complex cellular and systemic responses. While acute activation of these stress pathways triggers and aids in tissue repair, chronic or unresolved stress contributes to immune dysregulation and potentially irreversible tissue remodeling. In the development of CTRL, these stressors are not only induced by standard cancer therapies but also amplified by pre-existing metabolic dysfunction, which positions them as central drivers of CTRL pathophysiology. Failure to restore lymphatic function in a timely fashion leads to progressive degradation of the tissue microenvironment, marked by further fluid accumulation, chronic inflammation, fibrosis, and maladaptive tissue remodeling. This ultimately results in lymphedema and, in our analogy, an irreparably damaged section of the house.
INFLAMMATION
While inflammation is a necessary part of the healing process, chronic inflammation poses serious risks to the lymphatic system’s formation, structure, and function. In our house analogy, this is the moisture that gets trapped by shoddy repair work, leading to degradation and further damage. Prolonged inflammatory signaling promotes excessive or abnormal lymphangiogenesis, largely driven by elevated growth factors. Over time, this dysregulated growth, combined with sustained oxidative stress, can lead to lymphatic valve dysfunction and loss of vessel contractility, critically impairing lymph transport and immune surveillance. These pathological changes are commonly observed in individuals with CTRL, where a persistently inflamed and oxidative microenvironment worsens lymphatic damage. Reducing systemic inflammatory drivers not only helps preserve existing lymphatic structures but may also improve their function and support overall health. Multiple lines of evidence suggest that a KD may be beneficial for promoting a non-inflammatory environment. As discussed previously, KDs can help reverse or resolve metabolic disorders, many of which have been associated with a state of low-grade chronic inflammation. In addition, trials comparing KDs to high CHO diets show that systemic inflammatory markers are decreased with a CHO-restricted intervention, suggesting a lower inflammatory burden. KDs have also been shown to reduce levels of excess visceral fat, thus potentially reducing another source of chronic inflammation. Along with the described systemic effects, KDs also increase the production of β-hydroxybutyrate (BHB), a ketone body known to inhibit nucleotide-binding domain-like receptor protein 3 (NLRP3) inflammasome, which is a major contributing pathway in chronic inflammation. These study findings demonstrate how a KD creates a more favorable environment for regenerative processes to occur in lymphatic tissue. This is akin to removing the source of degradation and disrepair so that the house can be properly rebuilt.
HYPOXIA
Tissue hypoxia, in which oxygen demand exceeds supply, is common in lymphedema, particularly in the context of obesity and metabolic syndrome. Lymph stasis, inflammation, and fibrosis create a state of local oxygen deprivation, a pervasive, low-level hypoxia that is akin to the lack of proper ventilation that causes the house to rot. Evidence from both human and animal studies confirms that hypoxia is present in CTRL, suggesting that interventions targeting hypoxic signaling may support lymphatic repair. Hypoxia-inducible factors (HIFs) are key regulators of the cellular response to low oxygen and play a central role in promoting lymphangiogenesis during wound healing and tissue remodeling, making HIFs a promising target for intervention. However, direct studies of HIF expression or function in human lymphedematous tissue, especially in the context of CTRL, are limited or lacking. Early research indicates that KDs and the ketone body BHB can modulate HIF activity in cancer models by either reducing or stabilizing particular variations of HIFs. Additionally, BHB has demonstrated protective effects in hypoxic environments by increasing adenosine triphosphate (ATP) efficiency and cellular energy resilience. This improved energy yield may help tissues withstand oxygen deprivation, reducing damage and supporting recovery. Although further study is needed, these findings point to a plausible mechanism through which a ketogenic intervention could improve outcomes in hypoxia-related lymphatic dysfunction, as may occur with conventional cancer treatments.
OXIDATIVE STRESS
Oxidative stress occurs when the production of reactive oxygen species (ROS) exceeds the body’s antioxidant defenses, leading to damage to deoxyribonucleic acid (DNA), proteins, and lipids, and ultimately contributing to cellular dysfunction or death. This imbalance is a known contributor to a range of chronic diseases, including cancer, cardiovascular disease, diabetes, metabolic syndrome, and autoimmune conditions. Within the setting of post-cancer treatment lymph stasis and CTRL, oxidative stress plays a particularly damaging role as another contributor to delayed repairs. Studies have shown that higher volumes of lymphedematous fluid correlate with increased oxidative stress and that reducing limb volume may help lower oxidative burden. Further, during episodes of cellulitis, prolonged oxidative stress may damage the lymphatic vessels, contributing to a cycle of inflammation, relapse, and increased lymphatic dysfunction. Importantly, ROS are not solely harmful; they also function as signaling molecules in physiological processes, including lymphangiogenesis. At controlled levels, ROS support growth factor signaling and vascular remodeling. However, when ROS levels become excessive, they suppress lymphangiogenesis by inducing apoptosis (programmed cell death) in lymphatic endothelial cells and disrupting tissue repair. Although specific studies on the effects of KDs on oxidative stress in patients at risk for CTRL are lacking, related research offers promising insights. KDs have been shown, for example, to reduce ROS generation and improve mitochondrial health and resilience by limiting glucose availability and shifting metabolism toward fat oxidation and ketone production. Shimazu et al. found that pre-treatment with BHB protected mice from oxidative damage, suggesting a potential preventive role for ketogenic strategies if implemented before cancer therapies. These findings highlight the need to explore KDs as a means to mitigate the oxidative and inflammatory damage that contributes to CTRL.
In summary, chronic inflammation, persistent hypoxia, and sustained oxidative stress create a hostile biochemical environment that impairs lymphatic repair and function, potentially driving the development of CTRL. Chronic inflammation promotes abnormal lymphangiogenesis, valve dysfunction, and loss of contractility; hypoxia exacerbates tissue injury through dysregulated HIF signaling; and oxidative stress damages lymphatic endothelium while perpetuating cycles of dysfunction. Evidence suggests that KDs may address these drivers through multiple mechanisms: reducing systemic inflammation and visceral adiposity, modulating HIF activity to improve hypoxic resilience, and lowering oxidative burden via improved mitochondrial efficiency and ketone-mediated antioxidant effects. While direct studies in patients at risk for CTRL are limited, existing metabolic, vascular, and inflammatory data point to KDs as a promising nutritional intervention to create a more regenerative, less damaging tissue environment conducive to lymphatic repair.
Tissue remodeling in response to lymphatic injury
Conventional cancer treatments cause direct injury to the lymphatic system, triggering a cascade of tissue remodeling processes that affect the skin, adipose tissue, lymphatic vasculature, and extracellular matrix. Structural and functional alterations are not only a consequence of the initial insult but also evolve progressively once lymphedema develops. Even in the earliest clinical stages, affected tissues exhibit signs of pathological remodeling, including fibrosis and adipose tissue hypertrophy. This remodeling contributes to the persistence and progression of lymphedema, highlighting the importance of preventive measures and early intervention that both support lymphatic repair and improve the health of the tissue microenvironment. Two of the primary tissue changes that occur with lymphedema, discussed here, are fibrosis and adipogenesis.
FIBROSIS
Fibrosis is a central pathological feature of lymphedema, contributing to impaired lymphatic vessel function and chronic disease progression. Some researchers have even described lymphedema as a form of end-stage organ failure of the lymphatic system, akin to fibrotic failure in organs such as the heart, liver, and kidneys. Returning to our house analogy, fibrosis is the biological equivalent of the stairs bulging and cracking, as the imperfect fixes generated by chronic inflammation and tissue hypoxia stimulate excessive collagen deposition in affected tissues. KDs have shown anti-fibrotic potential in several preclinical models, including cardiac, renal, and adipose tissue fibrosis. Although not yet studied directly in the context of patients at risk for CTRL, these findings raise the possibility that a KD could modulate fibrotic pathways relevant to lymphatic dysfunction. A proposed mechanism for the anti-fibrotic effects of KDs is the suppression of HIFs, as detailed previously. Also discussed previously, KDs may reduce systemic and tissue-specific inflammation and improve oxygenation, both of which may indirectly mitigate lymphatic trauma-related fibrosis. One particularly relevant molecular target is transforming growth factor-beta (TGF-β), a cytokine involved in wound healing, immune regulation, and fibrogenesis. TGF-β has been implicated in lymphedema-related fibrosis among patients with head and neck cancer. In animal models, inhibition of TGF-β signaling reduced fibrosis, improved lymphatic function, and promoted lymphangiogenesis. Significantly, the ketone BHB has been shown to downregulate TGF-β expression in models of pulmonary fibrosis and reduce fibrotic signaling in adipose tissue. KDs have been shown to reduce oxidative stress in the kidneys, enhance antioxidant enzyme activity, and lower pro-inflammatory cytokines. Although direct evidence is still lacking, the convergence of anti-fibrosis mechanisms and anti-inflammatory effects suggests that KDs may offer a novel therapeutic avenue for reducing or preventing fibrosis and the subsequent development of CTRL, especially if adopted before or soon after lymphatic injury caused by cancer treatments.
ADIPOGENESIS
The proliferation of fat cells, or adipogenesis, is another key component of pathological remodeling in lymphedema. In our house analogy, this is the body’s attempt to use unreliable materials to patch up the damage. Substantial evidence demonstrates that the swelling observed in lymphedema is not solely due to fluid accumulation but is significantly driven by adipose tissue deposition. Both animal and human studies consistently show a strong relationship between lymphatic dysfunction and fat accumulation, particularly in the affected limb. The subcutaneous tissue compartment is the primary site of lymphedema fat accumulation, although other tissue layers may also be involved. Importantly, lymphedema-induced excess adipose tissue is not reversed through traditional conservative therapies, such as manual lymph drainage and compression therapy, driving some patients to have it surgically removed via liposuction. At the cellular level and histologically, adipose tissue in a lymphedematous limb exhibits distinct pathological features when compared to healthy fat tissue, showing increased basal lipolytic activity, elevated expression of lymphangiogenic markers, and enhanced secretion of pro-inflammatory cytokines, as well as hypertrophic and heterogeneous adipocyte size, suggesting an actively remodeling and inflamed tissue environment. Moreover, stem cells isolated from lymphedematous adipose tissue have demonstrated greater adipogenic potential than stem cells from unaffected adipose tissue, indicating a shift toward adipose tissue expansion in response to lymph stasis. Interventions targeting metabolic dysfunction may help mitigate adipose deposition in lymphedema. In a small pilot study, participants with obesity and lymphedema who followed a KD experienced a significant correlation between reduced body fat percentage and decreased limb volume, an effect not observed in participants who did not adopt the diet. Similar findings have been reported in related disorders, such as lipedema, where KDs have led to meaningful reductions in body fat and symptom burden. These findings support the hypothesis that chronic lymph stasis following cancer treatment promotes adipose tissue hypertrophy and that targeting this pathway through dietary or metabolic interventions may offer a novel strategy to arrest or reverse the progression of lymphedema.
Ketogenic diets: An opportunity for metabolic optimization
Realizing the potential benefits of a KD for CTRL prevention or management will depend largely on the presence of informed, supportive healthcare providers who can guide patients through the transition to ketogenic strategies. However, despite its therapeutic promise, the KD is not without controversy or concern, particularly within conventional dietary and medical frameworks. It is common to hear arguments against using a KD for cancer patients, often centered on concerns about unfounded fears of its restrictive nature, risk of weight loss, and/or nutrient deficiencies. But for those battling cancer, a well-implemented ketogenic lifestyle can indeed be a sustainable and highly beneficial long-term approach. Its success and sustainability in the cancer population are greatly enhanced when individuals receive proper dietary education, have strong support systems for making dietary changes, and adopt a gradual, personalized dietary approach. This strategy not only makes a KD more manageable amid the challenges of cancer treatment but also empowers patients by giving them a beneficial sense of personal agency in their health journey.
There is a lack of large-scale trials testing the efficacy of KDs for all cancer types; however, we are unaware of any studies that suggest that a well-formulated KD causes a detrimental amount of unintentional weight loss or contributes to nutrient deficiencies. When done thoughtfully and under expert supervision, the potential for improved metabolic health, weight management, and enhanced well-being can make it a sustainable and empowering choice, directly challenging the notion that a KD is universally unsuitable or unsustainable for cancer patients. Several commonly expressed concerns are discussed here.
Navigating lipid metabolism and ketogenic diets
Concerns regarding the impact of a KD on lipid profiles, particularly cholesterol levels, are frequently raised. However, a comprehensive examination can reveal a more intricate physiological response than a sole focus on elevated low-density lipoprotein cholesterol (LDL-C) might suggest. While an increase in LDL-C is observed in some individuals following a KD, it is important to consider a broader spectrum of lipid markers. KDs often lead to a significant elevation in high-density lipoprotein cholesterol (HDL-C), a marker consistently associated with cardioprotective effects, alongside a notable reduction in triglycerides, another key indicator of cardiovascular risk. Furthermore, while a KD is often associated with an increase in LDL-C, this rise typically reflects a shift in particle size. Specifically, the LDL profile tends to shift toward larger, more buoyant particles, which are generally considered less atherogenic than the smaller, denser LDL particles, which have been more strongly linked to cardiovascular risk. Individual variability in response to a KD is a critical factor, as not all individuals experience a substantial increase in LDL-C; genetic predispositions and specific dietary compositions play significant roles in modulating lipid responses. The intentional selection of dietary fats that emphasize sources such as olive oil, avocado, and animal-sourced fats like dairy fat, lard, and tallow can exert a positive influence on lipid profiles. In addition, recent analyses show that LDL-C response to CHO-restricted diets depends on two factors. First, greater CHO restriction is linked to larger LDL-C increases, which reverse with moderate CHO reintroduction. Second, higher body mass index (BMI) is associated with little or no LDL-C change on a KD. These findings highlight that LDL-C responses to CHO restriction are highly individual, with mechanisms discussed more in-depth in Norwitz et al. Beyond isolated lipid markers, the broader metabolic benefits conferred by a KD, including weight loss (when desired or warranted), enhanced insulin sensitivity, and reduced systemic inflammation, represent substantial contributions to overall cardiovascular health. The multifaceted improvements observed when adhering to a KD often supersede the implications of isolated cholesterol measurements. Therefore, a personalized assessment, guided by a knowledgeable healthcare professional and encompassing a comprehensive lipid panel that includes HDL-C, triglycerides, and ideally LDL particle size, is paramount for individuals adhering to a KD. Additional monitoring, such as through serial imaging, can also be utilized to evaluate the actual presence of disease and identify rapid progression if it occurs. Research is ongoing on the risk of isolated diet-induced elevated LDL-C in individuals utilizing CHO-restricted diets, but it is still in the early stages. As such, if concerns arise from an increase in LDL-C or evidence of rapid cardiovascular disease progression, moderate CHO reintroduction or pharmacotherapy to lower LDL-C levels could be utilized to reverse the effect.
ADDRESSING WEIGHT LOSS CONCERNS IN PATIENTS WITH CANCER
The concern that a KD might lead to undesirable weight loss in cancer patients is also a common apprehension. However, a well-managed KD does not inherently result in detrimental weight loss; rather, it can be strategically implemented to preserve muscle mass and optimize body composition. The emphasis should shift from merely monitoring scale weight to assessing changes in body composition. While an initial reduction in water weight is typical, a properly formulated KD aims to facilitate the reduction of fat mass while safeguarding lean muscle tissue. In many patients, particularly those with obesity, a reduction in fat mass can be therapeutically beneficial. Protein intake plays a pivotal role in muscle sparing within the context of a therapeutic KD. Adequate to high protein consumption is essential for preventing muscle wasting, and emerging research also suggests that ketones themselves may exert a muscle-sparing effect. Furthermore, the high-fat content characteristic of a KD often contributes to appetite stabilization and sustained satiety, potentially supporting consistent caloric intake despite initial appetite fluctuations. The risk of unintended and detrimental weight loss is significantly mitigated through a personalized and supervised approach. This involves individualized dietary plans and close monitoring by dietitians or physicians specializing in metabolic therapies for cancer. When implemented strategically and under professional guidance, a KD can optimize metabolic health and body composition for the patient with cancer without inducing harmful, uncontrolled weight loss, and may even enhance treatment tolerance and overall well-being.
OVERCOMING PHYSICIAN BIAS AGAINST THE IMPLEMENTATION OF KETOGENIC DIETS
A significant hurdle in cancer care is the unexamined bias among some healthcare providers against suggesting a KD, especially for patients at risk of CTRL. While oncologists often have good intentions and wish to steer clear of information overload and to avoid any perceived risks, withholding information inadvertently disempowers individuals, denying them agency and access to all potential options. The consequence is that patients may miss out on an approach that could offer significant benefits for both cancer management and lymphedema prevention/treatment. Evidence from a Norwegian cross-sectional study illustrates the gap between provider hesitancy and patient willingness: while 97% of cancer patients reported making dietary changes after a cancer diagnosis, guidance from healthcare providers was underutilized, with most patients relying instead on the internet or informal sources for information. These findings underscore the inclination of patients to modify their diet and highlight a critical missed opportunity for clinicians to provide evidence-based nutritional guidance during and after cancer treatment.
Suggestions for future research
Future investigations hold considerable promise for advancing the understanding of lymphatic diseases and improving care for an expanding patient population. We encourage researchers to leverage resources such as the Lymphatic Education & Research Network (LE&RN) Global Registry for Lymphatic Diseases, which provides a valuable repository of biological specimens, including blood and tissue samples, to support both clinical and laboratory studies. Defining the natural history of lymphedema and identifying reliable biomarkers are essential steps toward building predictive models of disease progression.
Several specific research directions merit attention. First, although CTRL has been most extensively studied in the context of breast cancer, targeted research is still needed to investigate CTRL across diverse cancers and treatment modalities to determine whether comparable pathways are involved and to ensure that incidence, risk factors, and mechanisms are accurately characterized to guide appropriate clinical management. Another interesting line of inquiry could examine how KDs influence lymphangiogenesis and lymphatic repair following cancer treatment. A critical area is the role of macrophage polarization: clarifying how distinct macrophage subtypes contribute to tissue repair and vessel growth across disease stages, and whether KDs modulate these processes, could reveal new therapeutic targets. Likewise, the potential of KDs to beneficially influence HIF signaling in lymphedematous tissue remains largely theoretical, yet may carry important implications for lymphatic recovery. Research should also explore the role of carnitine in lymphatic endothelial cell (LEC) metabolism. Because LEC proliferation and fatty acid oxidation depend on acylcarnitine, and carnitine deficiency frequently accompanies poor metabolic health, such deficiencies may impair lymphatic repair. Addressing this link could open avenues for nutritional or metabolic interventions to enhance lymphatic function. Further research could build on the current understanding of fibrosis; future studies should investigate how BHB and KD interventions affect TGF-β signaling and fibrotic pathways within the specific context of lymphedema. Particular attention should be given to modifying factors such as oxidative stress and glycemic control, as these may critically shape fibrotic responses in affected tissues. Finally, it is essential that future studies rigorously test the null hypothesis by exploring scenarios in which KDs may be harmful in lymphedema. Such balanced inquiry will ensure that clinical recommendations are grounded in a comprehensive assessment of both potential benefits and risks.
Conclusion
CTRL remains a substantial and frequently underrecognized morbidity of contemporary oncology care, with existing management strategies aimed primarily at mitigating downstream symptoms rather than intervening on upstream drivers of lymphatic susceptibility. This review advances the conceptual framework that metabolic dysfunction constitutes a key pathogenic factor in lymphatic impairment and that ketogenic dietary interventions may offer a viable, non-pharmacologic modality for reducing CTRL risk. By enhancing insulin sensitivity, modulating inflammatory pathways, and improving systemic fluid regulation, a KD may augment metabolic resilience, while ketone bodies themselves exert direct cytoprotective and immunomodulatory effects relevant to lymphatic repair and tissue homeostasis.
Given the high burden of metabolic dysfunction among individuals undergoing cancer therapy, the integration of evidence-based nutritional strategies represents an urgent and underutilized opportunity for prevention. Incorporating ketogenic approaches into oncology practice could reorient survivorship from reactive management to proactive risk reduction by addressing fundamental metabolic determinants of lymphatic compromise. To advance this paradigm, future work should prioritize rigorously designed clinical trials and mechanistic investigations that delineate the effects of KDs on lymphatic biology and establish clear, oncology-specific guidelines for their implementation.
References
1. Greene AK. Epidemiology and Morbidity of Lymphedema. In: Greene AK, Slavin SA, Brorson H, eds. Lymphedema: Presentation, Diagnosis, and Treatment. Springer International Publishing; 2015:33-44. doi:10.1007/978-3-319-14493-1_4
2. Rahavi EB, Altman JM, Stoody EE. Dietary Guidelines for Americans, 2015–2020: National Nutrition Guidelines. In: Lifestyle Medicine, Third Edition. 3rd ed. CRC Press; 2019.
3. Kopp W. How Western Diet And Lifestyle Drive The Pandemic Of Obesity And Civilization Diseases. Diabetes Metab Syndr Obes. 2019;12:2221-2236. doi:10.2147/DMSO.S216791
4. Brown S, Tadros AB, Montagna G, et al. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) may reduce the risk of developing cancer-related lymphedema following axillary lymph node dissection (ALND). Front Pharmacol. 2024;15:1457363. doi:10.3389/fphar.2024.1457363
5. O’Hearn M, Lauren BN, Wong JB, Kim DD, Mozaffarian D. Trends and Disparities in Cardiometabolic Health Among U.S. Adults, 1999–2018. J Am Coll Cardiol. 2022;80(2):138-151. doi:10.1016/j.jacc.2022.04.046
6. Wei M, Wang L, Liu X, et al. Metformin Eliminates Lymphedema in Mice by Alleviating Inflammation and Fibrosis: Implications for Human Therapy. Plast Reconstr Surg. 2024;154(6):1128-1137. doi:10.1097/PRS.0000000000011363
7. Hyde PN, Sapper TN, Crabtree CD, et al. Dietary carbohydrate restriction improves metabolic syndrome independent of weight loss. JCI Insight. 4(12):e128308. doi:10.1172/jci.insight.128308
8. Chen S, Su X, Feng Y, et al. Ketogenic Diet and Multiple Health Outcomes: An Umbrella Review of Meta-Analysis. Nutrients. 2023;15(19):4161. doi:10.3390/nu15194161
9. García-Caballero M, Zecchin A, Souffreau J, et al. Role and therapeutic potential of dietary ketone bodies in lymph vessel growth. Nat Metab. 2019;1(7):666-675. doi:10.1038/s42255-019-0087-y
10. Petrek JA, Senie RT, Peters M, Rosen PP. Lymphedema in a cohort of breast carcinoma survivors 20 years after diagnosis. Cancer. 2001;92(6):1368-1377. doi:10.1002/1097-0142(20010915)92:6<1368::AID-CNCR1459>3.0.CO;2-9
11. Johansson K, and Branje E. Arm lymphoedema in a cohort of breast cancer survivors 10 years after diagnosis. Acta Oncol. 2010;49(2):166-173. doi:10.3109/02841860903483676
12. Li CY, Kataru RP, Mehrara BJ. Histopathologic Features of Lymphedema: A Molecular Review. Int J Mol Sci. 2020;21(7):2546. doi:10.3390/ijms21072546
13. Jones D, Meijer EFJ, Blatter C, et al. Methicillin-resistant Staphylococcus aureus causes sustained collecting lymphatic vessel dysfunction. Sci Transl Med. 2018;10(424):eaam7964. doi:10.1126/scitranslmed.aam7964
14. Yuan Y, Arcucci V, Levy SM, Achen MG. Modulation of Immunity by Lymphatic Dysfunction in Lymphedema. Front Immunol. 2019;10. doi:10.3389/fimmu.2019.00076
15. Dominick SA, Madlensky L, Natarajan L, Pierce JP. Risk factors associated with breast cancer-related lymphedema in the WHEL Study. J Cancer Surviv. 2013;7(1):115-123. doi:10.1007/s11764-012-0251-9
16. Koelmeyer LA, Gaitatzis K, Dietrich MS, et al. Risk factors for breast cancer–related lymphedema in patients undergoing 3 years of prospective surveillance with intervention. Cancer. 2022;128(18):3408-3415. doi:10.1002/cncr.34377
17. Shen A, Lu Q, Fu X, et al. Risk factors of unilateral breast cancer-related lymphedema: an updated systematic review and meta-analysis of 84 cohort studies. Support Care Cancer. 2022;31(1):18. doi:10.1007/s00520-022-07508-2
18. Letellier ME, Ibrahim M, Towers A, Chaput G. Incidence of lymphedema related to various cancers. Med Oncol Northwood Lond Engl. 2024;41(10):245. doi:10.1007/s12032-024-02441-2
19. Izawa T, Kobayashi A, Kawashima M, et al. Quantitative analysis of the effect of docetaxel-induced edema on quality of life in patients with breast cancer and related factors: a prospective cohort study. BMC Womens Health. 2024;24:165. doi:10.1186/s12905-024-03003-4
20. Stout NL, Dierkes M, Oliveri JM, Rockson S, Paskett ED. The influence of non-cancer-related risk factors on the development of cancer-related lymphedema: a rapid review. Med Oncol. 2024;41(11):274. doi:10.1007/s12032-024-02474-7
21. Leray H, Malloizel-Delaunay J, Lusque A, et al. Body Mass Index as a Major Risk Factor for Severe Breast Cancer-Related Lymphedema. Lymphat Res Biol. 2020;18(6):510-516. doi:10.1089/lrb.2019.0009
22. Chakraborty S, Zawieja S, Wang W, Zawieja DC, Muthuchamy M. Lymphatic system: a vital link between metabolic syndrome and inflammation. Ann N Y Acad Sci. 2010;1207(s1):E94-E102. doi:10.1111/j.1749-6632.2010.05752.x
23. Doruk Analan P, Kaya E. Is There a Relationship Between Insulin Resistance and Breast Cancer-Related Lymphedema? A Preliminary Study. Lymphat Res Biol. 2022;20(1):76-81. doi:10.1089/lrb.2019.0072
24. Lee Y, Chakraborty S, Meininger CJ, Muthuchamy M. Insulin resistance disrupts cell integrity, mitochondrial function, and inflammatory signaling in lymphatic endothelium. Microcirculation. 2018;25(7):e12492. doi:10.1111/micc.12492
25. Rockson SG, Rivera KK. Estimating the population burden of lymphedema. Ann N Y Acad Sci. 2008;1131:147-154. doi:10.1196/annals.1413.014
26. Executive Committee of the International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2020 Consensus Document of the International Society of Lymphology. Lymphology. 2020;53(1):3-19. doi:10.2458/lymph.4649
27. Granzow JW. Lymphedema surgery: the current state of the art. Clin Exp Metastasis. 2018;35(5):553-558. doi:10.1007/s10585-018-9897-7
28. Coriddi M, Mehrara B, Skoracki R, Singhal D, Dayan JH. Immediate Lymphatic Reconstruction: Technical Points and Literature Review. Plast Reconstr Surg – Glob Open. 2021;9(2):e3431. doi:10.1097/GOX.0000000000003431
29. Ge LP, Tang L, Zuo W, et al. Psychological Stress and Its Correlations to Patients with Acute Lymphedema After Breast Cancer Surgery. Breast Cancer Targets Ther. 2024;16:867-876. doi:10.2147/BCTT.S485827
30. Zhou X, Su X, Ma G, Tang Y, Wu J, Liu B. Depression and Psychological Distress in Patients with Lower Extremity Lymphedema: A Mixed-Method Study. Lymphat Res Biol. 2025;23(2):115-122. doi:10.1089/lrb.2024.0071
31. Monteiro AJ, de Labra C, Losa-Iglesias ME, et al. Depressive symptoms and their severity in a sample with lymphedema: a case–control investigation. Front Psychiatry. 2023;14. doi:10.3389/fpsyt.2023.1202940
32. Jager G, Doller W, Roth R. Quality-of-life and body image impairments in patients with lymphedema. Lymphology. 2006;39(4). Accessed July 17, 2025. http://journals.librarypublishing.arizona.edu/lymph/article/id/3568/
33. Diana R, Raditya Atmaka D. Ketogenic diet for weight loss and its implications on health: A literature study. Media Gizi Indones. 2020;15(3):184-193. doi:10.20473/mgi.v15i3.184-193
34. Bisschop PH, Pereira Arias AM, Ackermans MT, et al. The effects of carbohydrate variation in isocaloric diets on glycogenolysis and gluconeogenesis in healthy men. J Clin Endocrinol Metab. 2000;85(5):1963-1967. doi:10.1210/jcem.85.5.6573
35. Bachar A, Birk R. Ketogenic Diet Intervention for Obesity Weight-Loss- A Narrative Review, Challenges, and Open Questions. Curr Nutr Rep. 2025;14(1):43. doi:10.1007/s13668-025-00634-3
36. Ji J, Fotros D, Sohouli MH, Velu P, Fatahi S, Liu Y. The effect of a ketogenic diet on inflammation-related markers: a systematic review and meta-analysis of randomized controlled trials. Nutr Rev. 2025;83(1):40-58. doi:10.1093/nutrit/nuad175
37. Kosinski C, Jornayvaz FR. Effects of Ketogenic Diets on Cardiovascular Risk Factors: Evidence from Animal and Human Studies. Nutrients. 2017;9(5):517. doi:10.3390/nu9050517
38. Paoli A, Rubini A, Volek JS, Grimaldi KA. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur J Clin Nutr. 2013;67(8):789-796. doi:10.1038/ejcn.2013.116
39. Zampell JC, Aschen S, Weitman ES, et al. Regulation of Adipogenesis by Lymphatic Fluid Stasis Part I: Adipogenesis, Fibrosis, and Inflammation. Plast Reconstr Surg. 2012;129(4):825-834. doi:10.1097/PRS.0b013e3182450b2d
40. Wong BW, Wang X, Zecchin A, et al. The role of fatty acid β-oxidation in lymphangiogenesis. Nature. 2017;542(7639):49-54. doi:10.1038/nature21028
41. Lodewijckx I, Matthys C, Verheijen J, et al. Potential therapeutic effect of a ketogenic diet for the treatment of lymphoedema: Results of an exploratory study. J Hum Nutr Diet. 2024;37(4):885-891. doi:10.1111/jhn.13330
42. Helyer LK, Varnic M, Le LW, Leong W, McCready D. Obesity is a Risk Factor for Developing Postoperative Lymphedema in Breast Cancer Patients. Breast J. 2010;16(1):48-54. doi:10.1111/j.1524-4741.2009.00855.x
43. Sudduth CL, Greene AK. Lymphedema and Obesity. Cold Spring Harb Perspect Med. 2022;12(5):a041176. doi:10.1101/cshperspect.a041176
44. Yusof KM, Avery-Kiejda KA, Ahmad Suhaimi S, et al. Assessment of Potential Risk Factors and Skin Ultrasound Presentation Associated with Breast Cancer-Related Lymphedema in Long-Term Breast Cancer Survivors. Diagnostics. 2021;11(8):1303. doi:10.3390/diagnostics11081303
45. Pirincci CS, Mete O, Yasa ME, Dalyan M. A comparative evaluation of the efficacy of complete decongestive therapy in the treatment of unilateral breast cancer–related lymphedema with and without metabolic syndrome. Support Care Cancer. 2024;32(7):473. doi:10.1007/s00520-024-08676-z
46. Redick JC, Kesslering C. Resolution of Persistent Chylothorax With a Ketogenic Diet: A Case Report. Cureus. 2024;16(7):e64144. doi:10.7759/cureus.64144
47. de la Monte SM. Metabolic derangements mediate cognitive impairment and Alzheimer’s disease: role of peripheral insulin resistance diseases. Panminerva Med. 2012;54(3):171-178.
48. Campbell DB, Sobol CG, Stacy MR, et al. Revascularization Outcomes Stratified by Glycemic Control in Patients with Diabetes Mellitus and Chronic Limb-Threatening Ischemia. Ann Vasc Surg. 2024;100:91-100. doi:10.1016/j.avsg.2023.10.018
49. Sharma MD, Garber AJ, Farmer JA. Role of Insulin Signaling in Maintaining Energy Homeostasis. Endocr Pract. 2008;14(3):373-380. doi:10.4158/EP.ep.14.3.373
50. Chandrasekaran P, Weiskirchen R. Cellular and Molecular Mechanisms of Insulin Resistance. Curr Tissue Microenviron Rep. 2024;5(3):79-90. doi:10.1007/s43152-024-00056-3
51. Ransom T. Are Vegetable Seed Oils Fueling the Obesity Epidemic? Social Science Research Network. Preprint posted online October 28, 2024. Accessed October 28, 2025. https://papers.ssrn.com/abstract=4997712
52. Graham DS, Liu G, Arasteh A, Yin XM, Yan S. Ability of high fat diet to induce liver pathology correlates with the level of linoleic acid and Vitamin E in the diet. PLOS ONE. 2023;18(6):e0286726. doi:10.1371/journal.pone.0286726
53. Acosta M. High Fructose Corn Syrup’s Role in Obesity Among Hispanic Adolescent Females in the Texas-Mexico Border. DHR Proc. 2025;4(1):30-44. doi:10.47488/dhrp.v4iS1.3
54. Bancil AS, Sandall AM, Rossi M, Chassaing B, Lindsay JO, Whelan K. Food Additive Emulsifiers and Their Impact on Gut Microbiome, Permeability, and Inflammation: Mechanistic Insights in Inflammatory Bowel Disease. J Crohns Colitis. 2021;15(6):1068-1079. doi:10.1093/ecco-jcc/jjaa254
55. Noakes TD. Hiding unhealthy heart outcomes in a low-fat diet trial: the Women’s Health Initiative Randomized Controlled Dietary Modification Trial finds that postmenopausal women with established coronary heart disease were at increased risk of an adverse outcome if they consumed a low-fat ‘heart-healthy’ diet. Open Heart. 2021;8(2). doi:10.1136/openhrt-2021-001680
56. Gardner CD, Trepanowski JF, Del Gobbo LC, et al. Effect of Low-Fat vs Low-Carbohydrate Diet on 12-Month Weight Loss in Overweight Adults and the Association With Genotype Pattern or Insulin Secretion: The DIETFITS Randomized Clinical Trial. JAMA. 2018;319(7):667-679. doi:10.1001/jama.2018.0245
57. Sacks FM, Bray GA, Carey VJ, et al. Comparison of Weight-Loss Diets with Different Compositions of Fat, Protein, and Carbohydrates. N Engl J Med. 2009;360(9):859-873. doi:10.1056/NEJMoa0804748
58. Ebbeling CB, Feldman HA, Klein GL, et al. Effects of a low carbohydrate diet on energy expenditure during weight loss maintenance: randomized trial. BMJ. Published online November 14, 2018:k4583. doi:10.1136/bmj.k4583
59. Müller M j., Bosy-Westphal A. Adaptive thermogenesis with weight loss in humans. Obesity. 2013;21(2):218-228. doi:10.1002/oby.20027
60. Ludwig DS, Ebbeling CB. The Carbohydrate-Insulin Model of Obesity: Beyond “Calories In, Calories Out.” JAMA Intern Med. 2018;178(8):1098-1103. doi:10.1001/jamainternmed.2018.2933
61. Rubin J, Pollack B, Coleman-Belin J, et al. Metformin Use and Risk of Breast Cancer-Related Lymphedema: A Retrospective Analysis. J Am Coll Surg. Published online January 19, 2023:10.1097/XCS.0000000000001434. doi:10.1097/XCS.0000000000001434
62. Jeong J, Tanaka M, Iwakiri Y. Hepatic lymphatic vascular system in health and disease. J Hepatol. 2022;77(1):206-218. doi:10.1016/j.jhep.2022.01.025
63. Kataru RP, Park HJ, Baik JE, Li C, Shin J, Mehrara BJ. Regulation of Lymphatic Function in Obesity. Front Physiol. 2020;11:459. doi:10.3389/fphys.2020.00459
64. Rockson SG. Advances in Lymphedema. Circ Res. 2021;128(12):2003-2016. doi:10.1161/CIRCRESAHA.121.318307
65. Hossain L, Gomes KP, Safarpour S, Gibson SB. The microenvironment of secondary lymphedema. The key to finding effective treatments? Biochim Biophys Acta Mol Basis Dis. 2025;1871(3):167677. doi:10.1016/j.bbadis.2025.167677
66. Liao S, von der Weid PY. Inflammation-induced lymphangiogenesis and lymphatic dysfunction. Angiogenesis. 2014;17(2):325-334. doi:10.1007/s10456-014-9416-7
67. Cataldi A. Cell responses to oxidative stressors. Curr Pharm Des. 2010;16(12):1387-1395. doi:10.2174/138161210791033969
68. Bowman C, Rockson SG. The Role of Inflammation in Lymphedema: A Narrative Review of Pathogenesis and Opportunities for Therapeutic Intervention. Int J Mol Sci. 2024;25(7):3907. doi:10.3390/ijms25073907
69. Siems WG, Brenke R, Beier A, Grune T. Oxidative stress in chronic lymphoedema. QJM Int J Med. 2002;95(12):803-809. doi:10.1093/qjmed/95.12.803
70. Ridner SH. Pathophysiology of lymphedema. Semin Oncol Nurs. 2013;29(1):4-11. doi:10.1016/j.soncn.2012.11.002
71. Wang P, Guo X, Zhou Y, et al. Monocyte-to-high-density lipoprotein ratio and systemic inflammation response index are associated with the risk of metabolic disorders and cardiovascular diseases in general rural population. Front Endocrinol. 2022;13. doi:10.3389/fendo.2022.944991
72. Forsythe CE, Phinney SD, Fernandez ML, et al. Comparison of low fat and low carbohydrate diets on circulating fatty acid composition and markers of inflammation. Lipids. 2008;43(1):65-77. doi:10.1007/s11745-007-3132-7
73. Gyorkos A, Baker MH, Miutz LN, Lown DA, Jones MA, Houghton-Rahrig LD. Carbohydrate-restricted Diet and High-intensity Interval Training Exercise Improve Cardio-metabolic and Inflammatory Profiles in Metabolic Syndrome: A Randomized Crossover Trial. Cureus. 2019;11(9):e5596. doi:10.7759/cureus.5596
74. Kawai T, Autieri MV, Scalia R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am J Physiol-Cell Physiol. 2021;320(3):C375-C391. doi:10.1152/ajpcell.00379.2020
75. Cunha GM, Guzman G, Correa De Mello LL, et al. Efficacy of a 2-Month Very Low-Calorie Ketogenic Diet (VLCKD) Compared to a Standard Low-Calorie Diet in Reducing Visceral and Liver Fat Accumulation in Patients With Obesity. Front Endocrinol. 2020;11. doi:10.3389/fendo.2020.00607
76. Amini MR, Aminianfar A, Naghshi S, Larijani B, Esmaillzadeh A. The effect of ketogenic diet on body composition and anthropometric measures: A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2022;62(13):3644-3657. doi:10.1080/10408398.2020.1867957
77. Castro-Barquero S, Casas R, Rimm EB, et al. Loss of Visceral Fat is Associated with a Reduction in Inflammatory Status in Patients with Metabolic Syndrome. Mol Nutr Food Res. 2023;67(4):2200264. doi:10.1002/mnfr.202200264
78. Kong G, Wang J, Li R, Huang Z, Wang L. Ketogenic diet ameliorates inflammation by inhibiting the NLRP3 inflammasome in osteoarthritis. Arthritis Res Ther. 2022;24(1):113. doi:10.1186/s13075-022-02802-0
79. Nguyen LK, Cavadas MAS, Scholz CC, et al. A dynamic model of the hypoxia-inducible factor 1α (HIF-1α) network. J Cell Sci. 2013;126(Pt 6):1454-1463. doi:10.1242/jcs.119974
80. Lee P, Chandel NS, Simon MC. Cellular adaptation to hypoxia through hypoxia inducible factors and beyond. Nat Rev Mol Cell Biol. 2020;21(5):268-283. doi:10.1038/s41580-020-0227-y
81. Jiang X, Tian W, Granucci EJ, et al. Decreased lymphatic

