Hepatic Sclerosing Epitheloid Fibrosarcoma in Adolescents
Hepatic sclerosing epithelioid fibrosarcoma in an adolescent patient. Case report and literature review
Ghazal Shadmani1, MD
- Washington University in St Louis, School of Medicine, St Louis, MO
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Shadmani, G., 2025. Hepatic sclerosing epithelioid fibrosarcoma in an adolescent patient. Case report and literature review. Medical Research Archives, [online] 13(4).
https://doi.org/10.18103/mra.v13i4.6389
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6389
ISSN 2375-1924
ABSTRACT
Sclerosing epithelioid fibrosarcoma (SEF), first described by Meis-Kindblom et al., is a rare variant of fibrosarcoma that shows round or oval epithelioid cells within a dense collagenous matrix. Despite its benign histologic appearance, characterized by an absent or low mitotic rate, this tumor exhibits aggressive clinical behavior. It has a 37% likelihood of local recurrence and an 80% tendency for synchronous or metachronous distant metastasis, making it a clinically aggressive tumor.
Sclerosing epithelioid fibrosarcoma mainly occurs in middle-aged adults, but fewer than a handful of pediatric cases have been reported in the literature. Previous publications have primarily focused on the pathological aspects, with no documented descriptions of the imaging characteristics of hepatic Sclerosing epithelioid fibrosarcoma.
Keywords
Hepatic sclerosing epithelioid fibrosarcoma, adolescent, case report, imaging, literature review
Introduction
Sclerosing epithelioid fibrosarcoma (SEF), first described by Meis-Kindblom et al. in 1995, is a rare variant of fibrosarcoma that shows round or oval epithelioid cells within a dense collagenous matrix¹. Despite its benign histologic appearance, characterized by an absent or low mitotic rate, this tumor exhibits aggressive clinical behavior. It has a 37% likelihood of local recurrence² and an 80% tendency for synchronous or metachronous distant metastasis³ making it a clinically aggressive tumor.
Sclerosing epithelioid fibrosarcoma mainly occurs in middle-aged adults, and fewer than a handful of pediatric cases have been described in the literature. The locations of SEF in the pediatric cases presented in the literature include bone⁴, trunk (mediastinum, retroperitoneum, back, abdominal wall)⁵, kidney⁶, and liver⁸. Diagnosis is challenging due to the rarity of the disease and ultimately confirmed by pathology. Although the immunohistochemical and molecular genetic characteristics of pediatric hepatic SEF have been explored previously⁹, no studies have focused on its imaging features. Therefore, the imaging description of SEF remains poorly defined in the literature. The aim of this paper is to review US, MRI, Fluorodeoxyglucose Positron Emission Tomography scan (FDG PET) features of this rare pediatric liver malignancy.
Case Report
A 17-year-old female, previously healthy, presented with abdominal pain and pruritus for several months. She noticed jaundice in her eyes for the last two weeks but denied acholic stools. These symptoms were associated with a 10-pound weight loss and fatigue. She was not on any steroids or oral contraceptive pills. Initial lab data showed a total bilirubin of 4.7 mg/dL (0.0-1.2), alkaline phosphatase of 390 unit/L (40-130), AST of 101 unit/L (10-45), and ALT of 162 unit/L (7-45). Abdominal ultrasound (Figure 1) showed a large lobulated mixed echogenicity mass centered on the right hepatic lobe containing several hypoechoic components with posterior acoustic shadowing without a discernible central scar. Color Doppler showed scanty peripheral vascularity without significant internal vascularity. The spleen was moderate to markedly enlarged with a craniocaudal dimension of approximately 19 cm. Upon further laboratory evaluation, CA 19-9 was 68.8 units/mL (Nl: up to 35) with normal serum alpha-fetoprotein and beta-HCG. Abdominal MRI (Figure 2) showed a large, predominantly T1/T2 hypointense mass with delayed progressive enhancement (Figure 3). There was no signal drop throughout the lesion on out-of-phase images to suggest a fat component. DWI and ADC mapping demonstrated a predominantly hypointense signal (T2 blackout). No central scar was appreciated within the hepatic lesion. Mild, regular dilation of peripheral intrahepatic biliary ducts was also identified due to the mass effect on the biliary system. Splenomegaly and collateral vessels were present. There was no tumoral invasion or thrombosis of the inferior vena cava.
Liver biopsy confirmed the diagnosis of sclerosing epithelioid fibrosarcoma. F-FDG PET CT scan for initial staging showed mild to moderate uptake throughout the hepatic mass without evidence of distant metastatic disease (Figure 4). Subsequently, the patient underwent an orthotopic liver transplant. On the most recent surveillance imaging, 24 months after transplantation, no findings of disease recurrence were identified, and serum CA 19-9 remained normal.
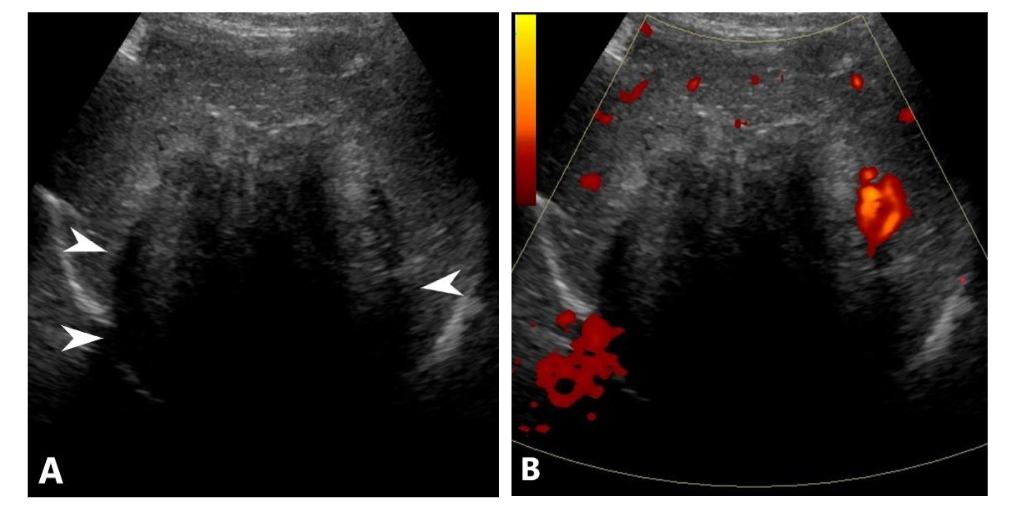
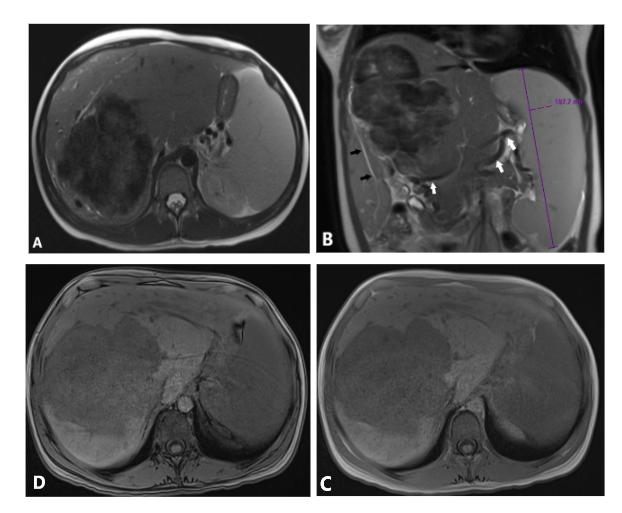
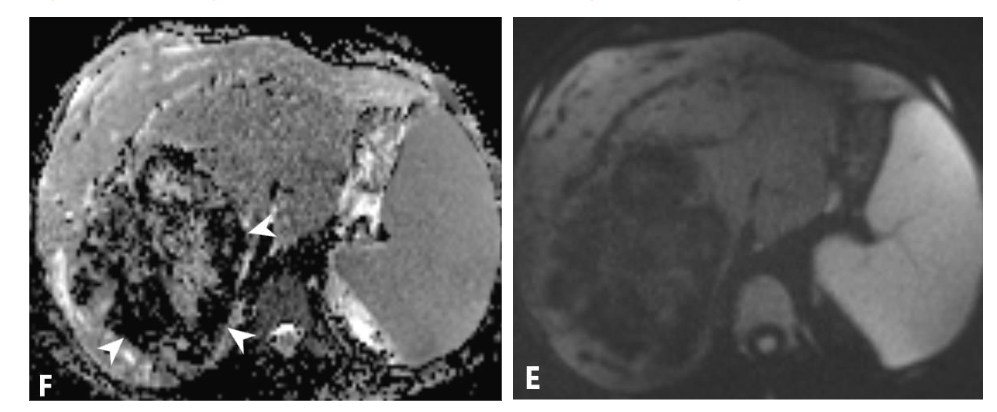
Figure 2. (A, B) Axial and coronal views of T2 weighted images (WI) show predominantly hypointense large hepatic mass. Note intrahepatic mild biliary dilation distal to the large mass (black arrows), splenomegaly, and multiple signal void collateral vessels (white arrows). In-phase (C) and out-phase (D) images do not show signal drop on out-phase image to suggest fat component. Diffusion weighted image (E) and Apparent Diffusion Coefficient (F) show predominantly T2 black out pattern (white arrowheads) and mild T2 shine-through centrally.
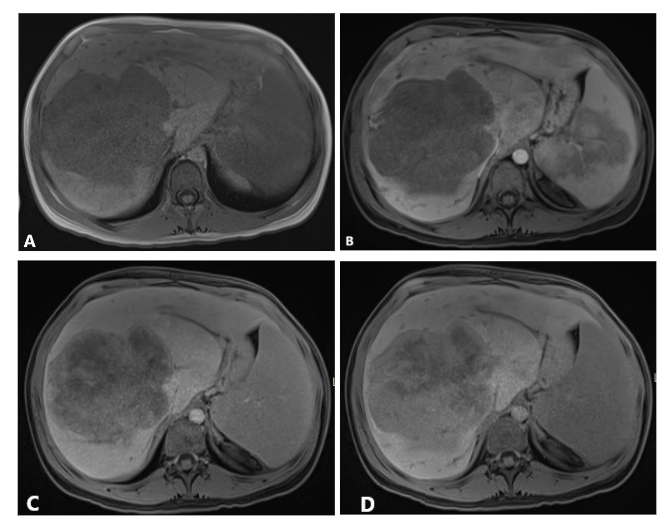
Figure 3. Axial T1 WI without contrast (A) and T1 WI immediate post contrast (B) show lack of early arterial enhancement. Axial T1WI 5 minutes post contrast (C) shows foci of enhancement which become more confluent and pronounced on axial T1WI 20 minutes post contrast (D).
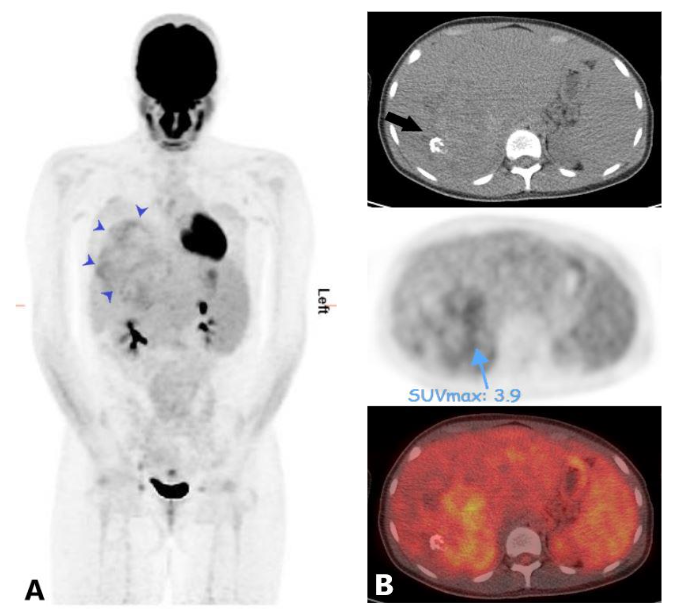
Figure 4. Maximum intensity projection (MIP) of FDG PET (A) shows mild to moderate uptake within the hepatic mass (blue arrow heads) and otherwise normal biodistribution of the tracer throughout the body with no PET findings of distant metastasis. Fused axial images of FDG PET and CT scan (B) show the most FDG avid areas of the mass. Note a focus of calcification in the mass on the non-contrast CT scan image (arrow).
Discussion
Two largest studies that evaluated SEF belong to Warmke et al³ and Ossendorff et al² who evaluated 51 and 89 cases, respectively, with two pediatric/adolescent patients in the former and one adolescent patient in the latter study. They did not report a notable sex predilection. The reported median tumor size at presentation was around 8 cm and it is most commonly seen in the soft tissue of lower extremity, trunk, upper extremity, and head and neck region. The primary sites of metastases are lung and bone²,⁹.
In this case, several imaging features suggest the tumor’s high fibrous content:
-
Ultrasonography (US) Findings:
-
The tumor appears hypoechoic with marked posterior acoustic shadowing, despite the absence of internal calcification, indicating a dense collagenous composition.
-
T2 Blackout Pattern:
-
The tumor exhibits low signal intensity on both T2-weighted and diffusion-weighted imaging (DWI), a
characteristic attributed to its high collagen content. The dense fibrous tissue shortens the T2 relaxation time, resulting in a markedly dark appearance—a phenomenon commonly observed in tumors with extensive fibrous components¹¹. The “T2 blackout” phenomenon has also been observed in hematomas, attributed to the paramagnetic properties of deoxyhemoglobin in the blood¹².
-
Delayed Contrast Enhancement: Delayed contrast enhancement, a hallmark imaging feature of fibrous tumors¹³,¹⁴, is also evident in this case.
Differential consideration in the presented case includes benign and malignant causes; benign lesions include hepatic adenoma and focal nodular hyperplasia¹⁵. There was no signal drop on out-of-phase images to suggest internal fat component. Moreover, there was no T1 hyperintensity indicative of internal hemorrhage. Malignant differential considerations in this patient include metastasis, Hepatocellular carcinoma (HCC), fibrolamellar HCC, undifferentiated embryonal sarcoma, cholangiocarcinoma, and angiosarcoma.
HCC and hepatoblastoma are very unlikely the absence of underlying liver disease and considering patient’s age, respectively. Furthermore, progressive contrast enhancement on delayed images rather than early arterial enhancement argues against HCC. Moreover, there was no central scar to support the diagnosis of fibrolamellar HCC¹⁵.
Undifferentiated embryonal sarcoma is another differential consideration that usually presents in the right hepatic lobe and is large at the time of presentation, similar to this case. However, this tumor is rare in patients older than 15 years old and, on imaging, lack of cystic component and T2 bright matrix do not support this diagnosis¹⁵. The lack of robust internal vascularity argues against angiosarcoma. Delayed progressive enhancement raises concern for cholangiocarcinoma, but in the absence of predisposing underlying diseases such as choledochal cyst, primary sclerosing cholangitis, and inflammatory bowel disease, cholangiocarcinoma is very unlikely. Moreover, cholangiocarcinoma is usually T2 hyperintense¹⁶.
Xia et al. described the imaging features of SEF in an adult patient with pancreatic SFE with similar findings T1/T2 hypointense pancreatic mass and progressive centripetal enhancement on delayed images¹⁷. Several prior case reports described T2 hypointense appearance of intracranial metastases, ethmoid sinus, and intramuscular SEF¹⁸–²¹. Tomimaru et al. presented the first adult case of hepatic SEF in a 35-year-old patient with a 7 cm right hepatic lobe mass and inferior vena cava invasion at the time of presentation with delayed centripetal enhancement on contrast enhanced CT images²². Ding et al. described the MRI findings of SEF in the right fibula of a 19-year-old patient as a T1/T2 hypointense mass with faint peripheral contrast enhancement during arterial phase and progressive enhancement during venous phase⁴. SEF has been described as a very slow growing nature based on observation of superficial soft tissue tumors²³. In the presented case, the presence of cavernous venous formation at the porta hepatis and splenomegaly support the slow growing nature of SEF.
Conclusion
Pediatric sclerosing epithelioid fibrosarcoma in the liver is extremely rare with no described imaging appearance in the current literature. In this presented case, significant posterior acoustic shadowing of the hypoechoic component of the hepatic mass on ultrasonography corresponding to low signal T2 appearance, and progressive delayed enhancement on MRI are imaging clues that can help radiologists to arrive a narrow differential consideration and differentiate this tumor from other hepatic malignancies.
Acknowledgement:
The author appreciates Dr. Joel Thomas’s comment regarding the T2-blackout MRI pattern in this case.
-
References
2. Ossendorf C, Studer GM, Bode B, Fuchs B. Sclerosing epithelioid fibrosarcoma: Case presentation and a systematic review. Clin Orthop Relat Res. 2008; 466(6):1485–91.
3. Warmke L, Meis JM. Sclerosing Epithelioid Fibrosarcoma A Distinct Sarcoma With Aggressive Features. Am J Surg Pathology. 2021 Mar;45(3): 317–28.
4. Ding Q, Shao X, Liu X, Fu Y, Huang F, Wang C. Case Report: Radiological Features of Sclerosing Epithelioid Fibrosarcoma in the Right Fibula. Front Oncol. 2020 Nov 18;10.
5. Suster DI, Gross JM, Fayad L, Wenokor C, Goldsmith JD, Ward A, et al. Sclerosing epithelioid fibrosarcoma of bone with hybrid features: clinicopathologic, radiologic, and molecular analysis of three cases. Skeletal Radiol. 2024 Feb 1;53(2): 387–93.
6. Dubrus E., Bisig B, Jengler C. A rare pediatric case of primary renal sclerosing epithelioid fibrosarcoma. Pediatric and developmental pathology. 2019;22(2):256–63.
7. Wu KH, Huang YS, Li CC. Primary sclerosing epithelioid fibrosarcoma of the kidney: A rare case report. Urol Case Rep. 2024 Mar 1;53.
8. Armstrong A, Boulos F, Kulkarni S, He M. Sclerosing Epithelioid Fibrosarcoma of the Liver in a Pediatric Patient. Pediatr Dev Pathol 2023 Mar-Apr;26(2):153-160. 2023 Mar;26(2):153–60.
9. Woods AD, Purohit R, Mitchell LC, Collier JR, Collier KA, Lathara M, et al. Metastatic pediatric sclerosing epithelioid fibrosarcoma. Cold Spring Harb Mol Case Stud. 2021 Dec 1;7(5).
10. Hussain MRK, Kulasekeran N, Anand AM, Dhinadhyalan R. All that glitters are not gold: holds true in female adnexal lesions. Vol. 52, Egyptian Journal of Radiology and Nuclear Medicine. Springer Science and Business Media Deutschland GmbH; 2021.
11. Takeuchi M, Matsuzaki K, Nishitani H. Diffusion-Weighted Magnetic Resonance Imaging of Ovarian Tumors: Differentiation of Benign and Malignant Solid Components of Ovarian Masses [Internet]. Available from: www.jcat.org
12. Lv B, Tian CL, Cao XY, Liu XF, Wang J, Yu SY. Role of diffusion-weighted imaging in the diagnosis of cerebral venous thrombosis. Journal of International Medical Research. 2020 Jun 1;48(6).
13. Moser T, Nogueira TS, Neuville A, Riehm S, Averous G, Weber JC, et al. Abdominal Imaging Moser et al. Fibrous Tumor of the Liver Case Report Delayed Enhancement Pattern in a Localized Fibrous Tumor of the Liver [Internet]. 2005. Available from: www.ajronline.org
14. Savvidou OD, Korkolopoulou P, Tsifountoudis I, Lakiotaki E, Agrogiannis G, Papagelopoulos PJ. Giant sclerosing epithelioid fibrosarcoma of the thigh. JBJS Case Connect. 2021 May 6;11(2).
15. Chung EM, Cube R, Lewis RB, Conran RM. From the archives of the AFIP: Pediatric liver masses: Radiologic-pathologic correlation part 1. Benign tumors. Radiographics. 2010 May;30(3):801–26.
16. Dong Y, Cekuolis A, Schreiber-Dietrich D, Augustiniene R, Schwarz S, Möller K, et al. Review on Pediatric Malignant Focal Liver Lesions with Imaging Evaluation: Part I. Vol. 13, Diagnostics. Multidisciplinary Digital Publishing Institute (MDPI); 2023.
17. Xia W, Yang Y, Huang Y. Imaging Features of Sclerosing Epithelioid Fibrosarcoma of the Pancreas: A Case Report. Front Oncol. 2020 Jun 17;10.
18. Goel N, Aryan S, Sivaraju L, Ghosal N. Sclerosing epithelioid fibrosarcoma with brain metastases. Vol. 36, Clinical Neuropathology. Dustri-Verlag Dr. Karl Feistle; 2017. p. 86–9.
19. Shankar DS, Phan N, Shirsat H, Westra WH, Trosman SJ, Colley PM. A 27-year-old female with sclerosing epithelioid fibrosarcoma of the left ethmoid sinus. Otolaryngology Case Reports. 2020 Sep 1;16.
20. Rizk RC, Yasrab M, Chu LC, Weisberg EM, Fishman EK. Metastatic sclerosing epithelioid fibrosarcoma. Radiol Case Rep. 2024 May 1;19(5): 1815–8.
21. Christensen DR, Ramsamooj R, Gilbert TJ, Gilbert TJ, Christensen DR, Ramsamooj R. Sclerosing epithelioid fiberosarcoma: short T2 on MR imaging. Vol. 26, Skeletal Radiol. International Skeletal Society; 1997.
22. Tomimaru Y, Nagano H, Marubashi S, Kobayashi S, Eguchi H, Takeda Y, et al. Sclerosing epithelioid fibrosarcoma of the liver infiltrating the inferior vena cava. World J Gastroenterol. 2009;15 (33):4204–8.




