Maxillofacial Abnormal Bone Fusion: Timing of Surgery
Bits and pieces in Maxillofacial abnormal bone fusion and malformation
Othman Al-Tuwairgi1
- Doctor
OPEN ACCESS
PUBLISHED: 31 January 2025
CITATION: Al-Tuwairgi, O., 2025. Bits and pieces in Maxillofacial abnormal bone fusion and malformation. Medical Research Archives, [online] 13(1). https://doi.org/10.18103/mra.v13i1.6290
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i1.6290
ISSN 2375-1924
Abstract
Introduction: The Timing of surgical intervention when considering the crinofacial and cleft defect is somewhat contrevesical as such some practicians would rather delay the procedure to gain growth and prevent soft tissue scaring.
Argument and contra argument: In This note our argument is That earlier intervention is the best predicted and significant approach. on the other hand, the contra argument could not be beneficial anymore especially after the new knowledge of the theory of the cascade growth waves.
Conclusion: Earlier intervention and surgical repair is the Best approach as the growth cascade is significant and important for the patient social and financial outcome.
Keyword: Fusion
Introduction
The craniofacial boney fusion or craniofacial clefts (malfusion) of minor or major portions of the facial skeleton with its abnormal esthetic and functional outcome, represent a dilemma in the field of maxillofacial surgery. First of all, the cause of the process of malformation, is it a process of malnutrition verses metabolic insults. The second dilemma is timing of surgical interfering specially with the advances in intrauterine surgical intervention. Third is the multiple contrivers related to growth hindering and soft tissue restriction. Forth the planning for sequential surgical correction (Combining two stages verse the routine protocols)- with the considerations it period of the patient being abstaining from being fully embracing in social life.
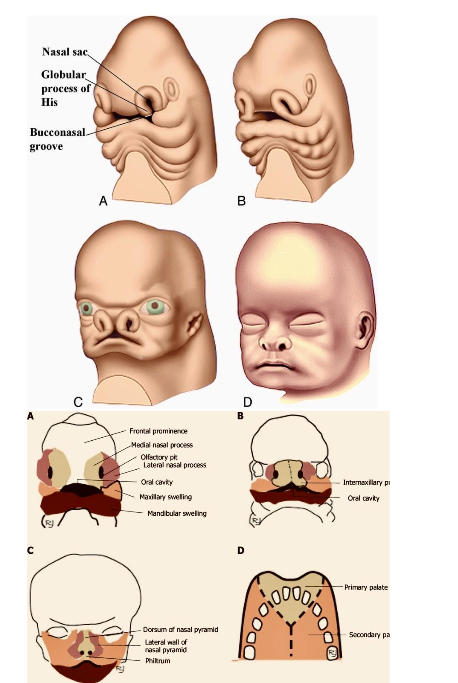
Natural development of the mammalian facial skeleton involves a complex and tightly coordinated series of biological processes involving cell migration, proliferation, growth, and differentiation as well as programmed cell death. Failures can occur during the different stages of the fetal development (Germinal, Embryonic, Fetal stages). The process of development and outgrowth, elevation, migration, or fusion of facial processes and palatal shelves, providing the best evidence that these developmental processes are under strict molecular control diagram (A&B).
The etiology of this anomaly ranges from genetic to environmental to idiopathic. The anomalies can be of different clinical presentation, but can be divided into overzealous earlier growth and early fusion (synostosis). On the other spectrum, failure of the facial plates and facial process to reach to their final destination and commitment of growth (facial cleft) paragraph (C)
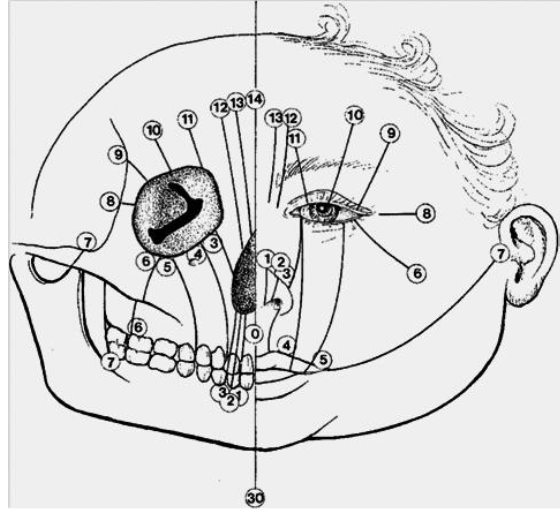
Classification of the craniofacial clefts according to Tessier; line of the bone tissue cleft is shown on the left side and line of the soft tissue cleft is shown on the right side.
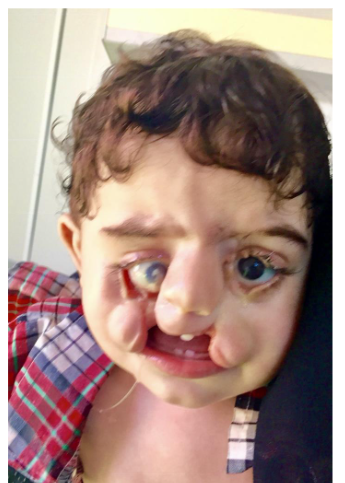
Recently The FGFR2 gene and the right combination, appears to induces for making a protein called fibroblast growth factor receptor 2 (FGFR2). FGFR2 protein is crucial for embryonic development bone growth. It appears that maxillofacial bony fusion is an overzealous activity of the FGFR2, as such we had published a four case of maxillary mandibular syngnathaia- the primary thought is that early activity of the FGFR2 is the primary cause.
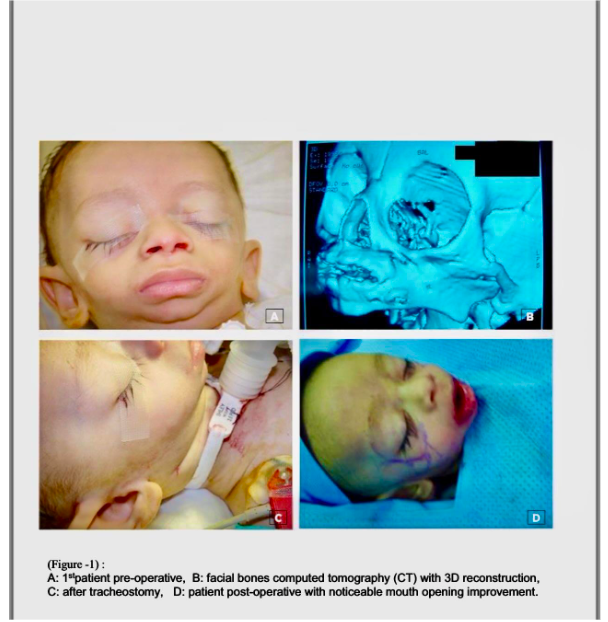
FGFR2 gene multiple mutations were found in different craniofacial- skeletal syndromes one of which is Apert syndrome. Such a mutation appears to causes premature closure/ fusion of the bones of the skull (craniosynostosis). As such presenting the mutating FGFR2 gene and its varieties (isoforms) of FGFR2 protein as the causative agent as those mutation produces the phenomena “gain-of-function” by increasing the activity of the protein, leading to stronger signaling, causing cells to mature too quickly. leading the premature and the abnormal facial bone fusion. This led us to includes FGFR2 gene in our genetic study for the different cases of the facial boney fusion. The treatment would be much simpler if we target early genetic treatment during development, since we have the FGFR2 gene mutation and we developed the knowledge of the most common sight of mutation which could be targeted in our treatments.
The second line of treatment is the early surgical intervention. This may be logical if we detect such abnormalities earlier in the embryonic live. The window of surgical intervention stars after the second trimester, seven months (33/34 weeks) of pregnancy is may be the best time to intervene for the cases where essential function, like ability for feeding, as the case of clefts and maxillary- mandibular fusion.
The Fetal surgery found its history emerging out of the University of California, San Francisco in the 1980s in the laboratory of Dr. Michael Harrison. Initial research hypothesis focused in utero repair of myelomeningocele (MMC) with improve outcomes in the ovine model, this effort followed by Management of Myelomeningocele Study (MOMS), which was a multicenter randomized controlled trial comparing the prenatal versus postnatal MMC repair. The study was stopped earlier as a result of the prenatal repair was with impressive outcomes, establishing the open fetal MMC repair as the standard of care. three operative approaches were proposed, including open, endoscopic and a hybrid approach combining open and endoscopic.
To that prospective, cleft lip and palate would be an excellent filed for earlier prenatal repair specialty if it’s combined with the new advances in stem cell therapy with biological induction and tissue engineering with 3D Osteogenic scaffold for the repair and induction of the committed cells in the field to reach its final site and produce the tissue that needed to close the gap between the different layers of the orofacial line of fusion.
Regenerating oral and maxillofacial structures utilizing stem cell therapy, is gaining momentum in treating clefts and facial defects. for the least 10 years. Many studies have shown that bone marrow, dental pulp, umbilical cord blood, and adipose tissue can be a source of osteoblastic, adipogenic, and chondrogenic cell lines.
Implanting Mesenchymal stem cells (MSCs) into the defect site, with or without employing appropriate scaffolds to support the cells. As such those MSCs are drifted by an osteoconductive scaffold and differentiated into osteogenic cells using osteo-inductive growth factors in bone tissue engineering.
A study by Tanikawa et al. was done in 2020 using the deciduous dental pulp stem cells (DDPSC) associated with a hydroxyapatite-collagen sponge (Bio-Oss Collagen® 250 mg, Geistlich) in repairing alveolar defects during the dental eruption. They isolated the DDPSC from each patient and then associated those cells with the biomaterial. They used this bone tissue-engineered set to fill the alveolar defect and evaluated the outcome with cone beam computed tomography at a six-month and 12-month follow-up. The researchers observed progressive alveolar bone union in all patients. DDPSC therapy resulted in adequate healing of alveolar defects with excellent feasibility and safety, and the researchers concluded that stem cell therapy resulted in satisfactory bone regeneration with dental eruption and reduced morbidity.
The prenatal intervention would leave us with less complicated cases of cleft and facial deformities to deal with, as such it would promote better surgical and repair outcomes, as well less growth intervention, as the cases of all the craniofacial and different facial cleft carry with its early intervention.
The growth factors and the biological induction proteins, are another edged advance in the management of facial defects and obtaining the full tissue growth potential. Though Autologous bone is the golden standard for treating the facial defects and considered as natural scaffold that is both osteoconductive and osteo-inductive as well as favorable for promoting osteoblast functions. Recent research and retrospective comparisons studies in which 70 patients, 45 received ICBG and 25 received ICBG with BMP-2 showed that the recombinant human bone morphogenetic protein (BMP-2) treated patients had better success than those whom the only get the iliac bone graft-(16% versus 33%. Needed seconded procedure to produce the desired outcome).
This left us with final prospective of treatment and care, which an earlier surgical treatment as soon as we recognize the facial defects and the patient stability to conducts our surgical management.
The protocol and the timing for surgical intervention is sold but rather flexible to give the Surgeon the decision to choose between earlier versus late intervention. As the arguments continue between earlier intervention with possibility of restrictions of growth and limiting the full-grown potential with possible deformation that would result more final deformation to deal with at later stages of the treatment.
Earlier intervention having its support from obtaining early function and coordination between different tissue and facial parts. Delayed intervention on the other hand get it support from being careful and minimizes the external influences on restricting the growth potential of the different organs and parts of the facial skeleton.
Conclusion
Recently (July, 2024), the Kyoto university hospital, Japan (https://mainichi.jp/english/articles/20230609/p2a/00m/0sc/026000c)-13. Showed a different prospective to our understanding and believe of the theory of defect or suppression of the stimulation of growth or the process of developing a tissue is by the gene or product of certain growth protein that being going out off function or production. To that believe Katsu Takahashi (head of the research), showed that suppression of USAG-1 protein by certain medications would rather induces tooth growth.
Such findings indicates that it’s rather our delayed intervention to induce the appropriate facial norm is the main cause of having patients with facial defects not being treated with its sequential functional and social withdrawals effects.
Bibliography
- Principles of Nutrigenetics and Nutrigenomics Fundamentals of Individualized Nutrition Book • 2020; Edited by: Raffaele DE Caterina, J. Alfredo Martinez and Martin Kohlmeier
- https://medlineplus.gov/genetics/gene/fgfr2/, Entry – 176943 – FIBROBLAST GROWTH FACTOR RECEPTOR 2; FGFR2 – OMIM
- Congenital maxillomandibular fusion: Case series and review of the literature congenital syngnathia treatment approach – Advances in Oral and Maxillofacial Surgery Volume 6, April–June 2022, 100251 Othman Altuwairgia,b,c,, Abdulsalam Aljabab d, Ali Makrami e, Ahmed Alomar f,Yasir Alturkistany g, Abu Bakarsharwani e, Bushra Alhazmi h, laila layqah , Ghufran Alayfan j,Waleed Baharoon
- Fetal myelomeningocele repair: a narrative review of the history, current controversies and future directions – Kaeli J. Yamashiro, Diana L. Farmer/ Vol 10, No 5 (May 27, 2021)
- Bobis S, Jarocha D, Majka M. Mesenchymal stem cells: characteristics and clinical applications. Folia Histochem Cytobiol. 2006;44:215–230.
- Bueno DF, Kerkis I, Costa AM, et al. New source of muscle-derived stem cells with potential for alveolar bone reconstruction in cleft lip and/or palate patients. Tissue Eng Part A. 2009;15:427–435. doi:10.1089/ten.tea.2007.0417.
- Bueno DF, Kerkis I, Costa AM, et al. New source of muscle-derived stem cells with potential for alveolar bone reconstruction in cleft lip and/or palate patients. Tissue 2018;10:66–77. doi:10.4252/wjsc.v10.i6.66.
- de Mendonça Costa A, Bueno DF, Martins MT, et al. Reconstruction of large cranial defects in nonimmunosuppressed experimental design with human dental pulp stem cells. J Craniofac Surg. 2008;19:204–210. doi:10.1097/scs.0b013e31815c8a54.
- Moos S, Marcolin F, Tornincasa S, et al. Cleft lip pathology diagnosis and foetal landmark extraction via 3D geometrical analysis. Int J Interact Des Manuf. 2017;11:1–8. doi:10.1007/s12008-014-0244-1
- Tanikawa D, Pinheiro CC, Almeida MC, et al. Deciduous dental pulp stem cells for maxillary alveolar reconstruction in cleft lip and palate patients. Stem Cells Int. 2020;2020:1–9. doi:10.1155/2020/6234167.
- Tanikawa D, Pinheiro CC, Almeida MC, et al. Deciduous dental pulp stem cells for maxillary alveolar reconstruction in cleft lip and palate patients. Stem Cells Int. 2020;2020:1–9. doi:10.1155/2020/6234167.
- Innovative Molecular and Cellular Therapeutics in Cleft Palate Tissue Engineering, Jeremie D Oliver, Shihai Jia, Leslie R Halpern, Emily M Graham, Emma C Turner, John S Colombo, David W Grainger, Rena N D’Souza Tissue Eng Part B Rev. 2021 Jun 16;27(3):215–237. doi:10.1089/ten.teb.2020.0181.

