Akkermansia Muciniphila: A New Approach to Metabolic Disorders
Examining the use of Akkermansia muciniphila as a promising mitigation for metabolic disorders
Aava Joshi¹, Dan Li, MD, PhD²
- Aava Joshi; [email protected]
- Dan Li; [email protected]
OPEN ACCESS
PUBLISHED: 30 April 2026
CITATION Joshi, A., and Li, D., 2026. Examining the use of Akkermansia muciniphila as a promising mitigation for metabolic disorders. Medical Research Archives, [online] 14(4).
COPYRIGHT © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Research on the role of gut microbiota in metabolism has grown significantly over decades, with certain strains producing a huge scope of potential beneficial effects. This paper aims to determine the potential applications for Akkermansia muciniphila supplementation in humans as a treatment for metabolic disorders, such as insulin resistance, prediabetes, excess cholesterol, or high body fat to lean mass ratios. To understand the full effects and mechanisms of A. muciniphila supplementation, this review examined 43 reports from various databases on the topic of A. muciniphila supplementation using PRISMA guidelines before December 1, 2025, excluding irrelevant or external sources of Akkermansia. The narrative synthesis demonstrates substantial evidence for the improvement of insulin resistance, decrease in overall weight, fat mass, and cholesterol levels in the body as a result of supplementation. Akkermansia may also have effects in managing prediabetes, improving lipid profiles, regulating liver enzyme activity, diversifying gut microbiomes, and stimulating GLP-1 production. Although both live and pasteurized Akkermansia have been proven to be well tolerated in humans, pasteurized Akkermansia is able to increase beneficial effects. However, there are still many unanswered questions given the limited scope, inconsistencies across research, risk of bias, and varied certainty of results.
Keywords:
Akkermansia muciniphila; gut microbiome; metabolic disorders; insulin resistance; body composition; supplementation
1. Introduction
A. muciniphila is a mucin-degrading bacterium known to have a significant role in maintaining gut health. However, A. muciniphila has attracted attention in recent years for its role in the potential amelioration of metabolic diseases. It is found in the gut mucosa layer and has been proven to play a role in the body’s basal metabolism. Its contributions to maintaining a healthy weight and preventing metabolic syndrome have been studied widely in animal trials and are making its way to human research. In an age where lifestyle diseases and metabolic diseases take the center stage in preventative healthcare, A. muciniphila may provide a noninvasive, affordable, accessible way to reduce insulin resistance (IR), a key risk factor for metabolic syndrome. If found to be efficacious, safe, and have a long-term impact, Akkermansia supplementation has massive potential to open new doors for lifestyle disease interventions. A. muciniphila, discovered only in 2004, has an extensive potential for application that is yet to be explored. There has been a plethora of studies in mice, among other animal trials, but its role in humans has fewer studies. Many gaps exist in the direct correlation of A. muciniphila for humans, as many studies utilize natural compounds that contain trace amounts of A. muciniphila as opposed to direct supplementation. Supplementation can be facilitated only in an anaerobic medium. Specifically, the direct supplementation of A. muciniphila in humans is an area with research gaps due to its availability in live or pasteurized forms. In addition, a majority of the research conducted in humans has been focusing on results for body composition, obesity, lean body mass, or BMI, whereas the internal function is often overlooked. This has resulted in the prevalence of studies with correlation, but causation is still uncertain. Therefore, research geared towards the mechanisms of A. muciniphila on metabolic disorders, such as IR, liver enzymes, cholesterol, or the gut microbiome, is necessary. The aim of this review is to compile research regarding A. muciniphila and its role towards mitigating IR, among other metabolic functions. A. muciniphila has been observed to have a positive impact on IR. This is demonstrated by reduced blood glucose, among other parameters for measuring insulin sensitivity. Its role is not limited to IR however; supplementation has been correlated with decreased BMI, fat mass and blood glucose in numerous studies. Akkermansia supplementation has also been shown to increase liver enzyme efficacy and decrease the prevalence of prediabetes. There have not been significant associations between its supplementation and the diversity of gut microbiota or GLP-1 production, but further research is required to solidify this claim. These claims are largely produced by the pasteurized form of Akkermansia. Overall, A. muciniphila is a novel treatment for IR and metabolic syndrome with a wide array of promising results.
2. Methodology
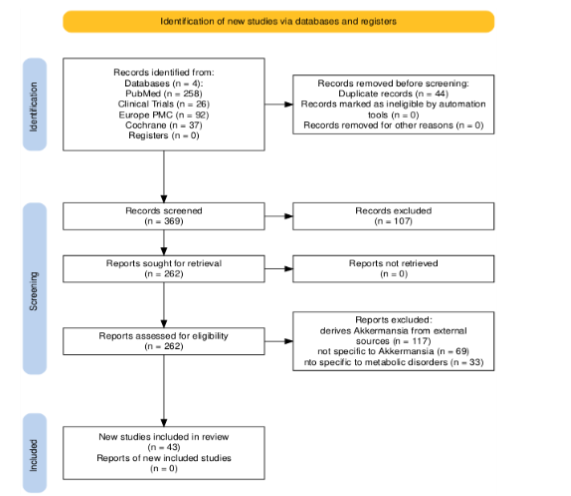
This review involved a thorough search of the literature regarding A. muciniphila supplementation and its correlation to metabolic disorders. Areas of particular focus included administration of Akkermansia in the body, its impact on IR, the correlated changes in body composition after supplementation, potential improvements in prediabetes or metabolic syndrome, GLP-1 production, and the correlation between Akkermansia and lipid profiles. In order to evaluate the full scope of information available, no papers were excluded on the basis of geographical context or population. A comprehensive literature search was performed using multiple electronic databases, including PubMed, Europe PMC, ClinicalTrials.gov, and Cochrane Library, which were the databases with the most prominent results, to identify relevant peer-reviewed articles and registered clinical trials related to Akkermansia. The most comprehensive results were found in the PubMed database. The PubMed database was used to facilitate search queries for articles published until December 1, 2025 using representative keywords, including the ones mentioned at the beginning of this paper, along with correlated topics and similar terms. The PubMed search strategy used was: “((“Insulin Resistance”[MeSH Terms] OR “Metabolic Syndrome”[MeSH Terms] OR “Glucose Intolerance”[MeSH Terms] OR “Prediabetic State”[MeSH Terms] OR “Insulin Resistance”[Title/Abstract] OR “Metabolic Syndrome”[Title/Abstract] OR “Glucose Intolerance”[Title/Abstract] OR “prediabetes”[Title/Abstract]) AND (“Akkermansia”[Title/Abstract] OR “Akkermansia muciniphila”[Title/Abstract] OR “a muciniphila”[Title/Abstract] OR “muciniphila”[Title/Abstract])) AND ((ffrft[Filter]) AND (fft[Filter]) AND (english[Filter]))” In addition to PubMed, Europe PMC was utilized to increase search sensitivity by identifying relevant full-text articles, preprints, and research outputs supported by European funding agencies, ensuring broader international coverage of the available evidence. The search strategy used for the Europe PMC database was: “TITLE_ABS:((Akkermansia muciniphila OR Akkermansia) AND (insulin resistance OR glucose metabolism OR insulin sensitivity))” Furthermore, ClinicalTrials.gov and the Cochrane Library were used to identify high-quality relevant articles and research trials with a global scope that are rigorously evaluated to fit the research question. These databases present a comprehensive view of Akkermansia and its relation to metabolic disorders with evidence-backed, reliable articles. The search for ClinicalTrials.gov was “Akkermansia muciniphila”. The search strategy used for Cochrane Library was “Akkermansia muciniphila AND “insulin””. Together, these databases provide a strong, reliable evidence base. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart, as shown in Figure 1, was used to visualize the numerical outputs from reviews and the inclusion decision process. In our flow chart, we illustrate the process for finding studies, removing duplicates, selecting the right research, retrieving the full article from the library and presenting the final analysis. Papers were screened in two rounds; the first round was screened using the title and abstract and the second round was screened on the contents of the full manuscript. Papers were excluded if they were not in the English language, irrelevant to the topic, or preprints. Common reasons for exclusion were probiotic supplementation not specific to Akkermansia, no linking to IR or other metabolic disorders, or using other plant derived supplements to trigger natural Akkermansia growth. At first, 413 papers were identified from the databases, out of which 44 duplicates were removed, leaving 369 papers to be screened using the title and abstract. From this process, 107 papers were excluded, and 262 papers were sought for retrieval for full text. Out of those 262, 43 were included in the review. Beyond the content of papers, the Joanna Briggs Institute critical appraisal tool was used to assess the selected papers’ potential biases, reliability, and credibility. Generative AI was used to categorize papers into low risk, moderate risk, and high risk studies according to each item on the checklist for the appropriate type of report. All results were manually checked by authors. Then, Generative AI was used to extract relevant quotations from selected texts. All extracted material using Generative AI was manually referenced and checked by authors. Using this information, a narrative synthesis was conducted to compile information into a review paper. Therefore, effect measures are not applicable for the qualitative nature of the data used. The effect measures and standard deviation for studies utilizing quantitative data can be found in the Data Extraction from supplementary materials. Certainty of evidence was assessed using the key factors of study quality, consistency, and precision. A full certainty of evidence table is presented in Supplementary Table S2. This review was not registered with PROSPERO or any other database.
3. Results
The 43 included studies were published between 2013 and 2025 and originated from diverse regions including Belgium, France, China, and the United States. Methodologies varied: one randomized controlled trial in overweight and obese adults (n=32; Depommier C., et al. evaluated live and pasteurized A. muciniphila supplementation; two human observational cohorts (Dao MC., et al., n=49; Zhou Q., et al., n≈900) examined associations between Akkermansia abundance and metabolic markers; several controlled animal experiments (Everard A., et al.; Wang Z., et al.; Yan S., et al.; Plovier H., et al.; Nian F., et al.; Zhao S., et al.) used mouse or rat models of obesity, diabetes, or glucose intolerance; some studies utilized metagenomic data to study the correlation between Akkermansia levels with metabolic disorders or to study the mechanisms of Akkermansia (Dao MC., et al.; Yang M., et al.; Ashrafian F., et al.); and the many other publications were narrative reviews or mechanistic overviews synthesizing preclinical and clinical evidence. Human participants were generally overweight, obese, or insulin-resistant adults, while animal studies predominantly employed diet-induced obese or genetically modified rodent models. Risk of bias was assessed for all included studies using the appropriate Joanna Briggs Institute (JBI) critical appraisal tools. However, some papers were categorized as unclear risk of bias due to their unpublished results and ongoing status. Registry-only trials were assessed as unclear risk of bias, as critical JBI domains, such as completeness of outcome data, appropriateness of analysis, and selective reporting, cannot be evaluated without peer-reviewed results. The randomized, double-blind, placebo-controlled trial by Depommier et al., 2019 demonstrates low risk of bias overall, with clearly defined eligibility criteria, intervention protocols, and prespecified outcomes. The primary limitation relates to the modest sample size and short intervention duration, which may limit precision and long-term inference but does not substantially undermine internal validity under JBI criteria. Another randomized controlled trial by Zhang et al., 2025 was assessed as low risk of bias, with adequate randomization, outcome completeness, and statistical analysis. Potential bias arises from a relatively narrow study population, which may limit generalizability rather than internal validity. An animal intervention study by Everard et al., 2013 presents moderate risk of bias, primarily due to small sample sizes and incomplete reporting of allocation concealment. Outcomes were biologically coherent and consistently measured, but translational relevance is limited by the exclusive use of murine models. A preclinical animal study by Plovier et al., 2017 exhibits moderate risk of bias due to inherent indirectness to human outcomes. While experimental conditions were well controlled and mechanistic pathways clearly demonstrated, randomization and blinding procedures were not fully described, resulting in unclear risk for selection and detection bias under JBI appraisal. Across remaining preclinical and animal studies, risk of bias was generally moderate, driven by small sample sizes, lack of explicit randomization or blinding, and indirectness to human metabolic outcomes. These studies are internally coherent but contribute primarily supportive rather than confirmatory evidence. An observational microbiome study by Yassour et al., 2018 shows moderate risk of bias, primarily related to variability in sequencing depth and analytical pipelines. While methods were transparently reported, outcome heterogeneity reduces comparability across studies. Additional observational studies consistently showed moderate risk of bias due to confounding and non-experimental design. While associations were reproducible, JBI appraisal indicates limited capacity for causal inference. Across the evidence base, individual risk of bias is lowest overall among randomized human trials, moderate among observational studies, and inherently high among preclinical models and narrative reviews due to indirectness, despite strong mechanistic consistency. A full risk of bias table can be found in Supplementary Table S1. Study characteristics are presented in Table 1.
| Study | Population/Design | Main Findings |
|---|---|---|
| Everard A., et al. (2013) | Animal Study (obese & T2DM mice) | Increased A. muciniphila abundance reduced diet-induced obesity; Improved gut barrier integrity and metabolic inflammation; Demonstrated direct host–microbe epithelial interactions |
| Dao MC., et al. (2016) | Human observational study (obese adults, dietary intervention) | Higher baseline A. muciniphila associated with better metabolic profiles; Increases in abundance correlated with improved insulin sensitivity; Linked A. muciniphila to greater gut microbiome richness |
| Plovier H., et al. (2017) | Animal Study (obese & diabetic mice) | Pasteurized A. muciniphila improved insulin sensitivity and glucose tolerance; Reduced fat mass and metabolic inflammation; Identified Amuc_1100 as a key active membrane protein |
| Zhao S., et al. (2017) | Animal Study (chow fed mice) | Supplementation reduced low-grade inflammation; Improved glucose metabolism despite standard diet; Effects observed independent of obesity |
| Van der Ark., et al. (2018) | In vitro/systems biology study | Confirmed strong mucus dependence of A. muciniphila metabolism; Defined minimal growth medium requirements; Supported ecological specialization in the gut mucosa |
| Depommier C., et al. (2019) | Human RCT (overweight/ obese volunteers) | Pasteurized A. muciniphila improved insulin sensitivity, body composition, and lipid profiles; Reduced markers of inflammation and insulin resistance; Demonstrated safety and tolerability in humans |
| Dao MC., et al. (2019) | Human observational study (obese patients before and after bariatric surgery) | A. muciniphila abundance increased post-surgery; No direct association between increased abundance and metabolic improvement; Suggested context-dependent metabolic effects |
| Ashrafian F., et al. (2019) | In vitro and ex vivo experimental study | Extracellular vesicles modulated Toll-like receptor signaling; Improved epithelial tight junction integrity |
| Yang M., et al. (2020) | Animal study (high-fat diet induced obese mice) | Newly isolated strains of Akkermansia reduced body weight and adiposity; Improved glucose tolerance and lipid metabolism; Demonstrated strain-specific metabolic efficacy |
| Zhou Q., et al. (2021) | Human Cross Sectional Study (adults with metabolic syndrome) | Higher Akkermansia abundance associated with lower metabolic syndrome severity; Dose-dependent relationship observed |
| Yoon HS., et al. (2021) | Animal Mechanistic Study (mouse models) | Identified secreted protein inducing GLP-1 secretion; Improved glucose homeostasis and insulin sensitivity; Provided mechanistic link between A. muciniphila and enteroendocrine signaling |
| Wang Z., et al. (2023) | Animal Study (genetically modified intestinal Ffar4 knockout mice) | Improved glucose tolerance during circadian transition periods; Effects depended on host lipid-sensing pathways; Highlighted host genotype–microbe interactions |
| Nian F., et al. (2023) | Animal Study (NAFLD mouse model) | Reduced hepatic steatosis and inflammation; Modulated FXR expression and bile acid metabolism; Synergistic effects with Bifidobacterium bifidum |
| Yan S., et al. (2024) | Animal Study (high-fat diet induced prediabetic rat model) | Improved pancreatic islet β-cell function; Enhanced insulin secretion capacity; Mitigated diet-induced β-cell dysfunction |
| Lin H., et al. (2025) | Animal Mechanistic Study (Mouse model of doxorubicin-induced cardiotoxicity) | Attenuated doxorubicin-induced cardiotoxicity; Improved mitochondrial biogenesis and energy metabolism; Effects mediated via PPARα signaling |
| Zhang Y., et al. (2025) | Human RCT (Overweight/ obese patients with T2DM) | Improved glycemic control and insulin sensitivity; Therapeutic response depended on baseline gut A. muciniphila levels |
3.1 The designing of supplementation with A. muciniphila
A. muciniphila is a bacteria that has gained substantial traction and investigation for its abilities to regulate metabolism, improve IR, and alter body composition. The bacteria itself is one of many Gram negative bacteria that reside in the intestines of many living organisms, including humans and most mammals. A. muciniphila thrives by degrading the mucus lining of the intestines. This, in turn, stimulates the intestinal lining to produce new mucin, which is often healthier and stronger. The intestinal lining is strengthened by an abundance of Akkermansia through this process, allowing for eubiosis in the gut. Eubiosis supports gut barrier function, which prevents large molecules, toxins, or harmful substances from entering the bloodstream. Additionally, the eubiosis attained through A. muciniphila promotes the absorption of beneficial nutrients, vitamins, and minerals, as well as regulating digestive functions. Supplementation with Akkermansia aims to harness the numerous metabolic effects of A. muciniphila to be used for improving IR, metabolic syndrome, prediabetes, and more. Of note, A. muciniphila is an obligate anaerobic bacteria, which provides certain obstacles in terms of formulating a stable supplementation formula. Therefore, culturing of A. muciniphila must take place in an anaerobic medium, such as the one used by a recent supplementation study in mice. The Akkermansia is usually cultured in a mucin base in order for the bacteria to be able to degrade mucin and gain energy for reproduction. However, one of the most notable differences in formulation is between the usage of live and pasteurized Akkermansia. Pasteurized Akkermansia is treated with consistent heat for a specific duration in order to kill the bacteria. This has been shown to increase the efficacy of the bacteria in overweight and obese human trials, particularly influencing the gut-brain signaling axis. However, the characteristics of trial participants are also crucial for understanding the effects of Akkermansia based on the baseline levels of these participants. Firstly, A. muciniphila supplementation was tested on mice before clinical human trials. The effects of Akkermansia on induced diabetic mice are comparatively similar to that of human trials. In human trials, most studies include participants who are screened to be overweight or obese, and diagnosed with different components of metabolic syndrome. Therefore, the considerable effects observed may not reflect or ameliorate all the conditions that make up metabolic syndrome, including abdominal obesity, high blood glucose, high blood sugar, high LDL levels, and low HDL levels. Nonetheless, supplementation of A. muciniphila has been administered orally across all subjects.
Table 1. Overview of Completed Studies Examining the Relation Between A. Muciniphila and Metabolic Effects.
3.2 A. muciniphila’s application for regulating gluconeogenesis
One of the leading applications for A. muciniphila supplementation is for the mitigation of IR. IR is one of the many cardiometabolic diseases that have become increasingly prevalent in the world we live in today. Recently, interventions for IR have shifted focus to the role of gut microbiota in metabolic function. Specifically, A. muciniphila has recently gained traction for its abilities to regulate metabolism and maintain gut health. Through its role in intestinal wall strengthening, it is able to reduce gluconeogenesis, which is a vital function for managing blood glucose. IR hinders the body’s ability to control and suppress gluconeogenesis, and therefore, A. muciniphila provides a restraint on excessive gluconeogenesis when the body is less responsive to insulin. This application of Akkermansia has been widely studied in animals and humans. In a study of 50 prediabetic rats conducted by Yan S. et al., significant improvements were observed between baseline and supplemented HOMA-IR measurements. The effects of Akkermansia were able to increase insulin production to the level that IR was alleviated. Palliative management of IR is a practicable application of Akkermansia as shown by this study. In another study conducted in 2019 by Depommier C. et al., of 32 people, HOMA-IR measurements and insulinemia indices were dramatically improved in groups supplemented with A. muciniphila compared to a placebo. The study was conducted using overweight and obese volunteers who were instructed to maintain their usual diets, exercise, and not change any lifestyle factors. Therefore, the effects of this study can be attributed primarily to Akkermansia’s intervention. Interestingly, pasteurized A. muciniphila provided even more changes to IR markers than live Akkermansia. In fact, pasteurized Akkermansia supplementation reduced plasma insulin level markers by about 30% compared to a placebo and insulin sensitivity indices by more than 10%. This finding is consistent with studies done by Wang Z. et al., who have studied the effects of pasteurized Akkermansia in mice and seen dramatic improvements in glucose tolerance and utilization, and Everard A. et al., who studied the effects of both live and heat-inactivated A. muciniphila in obese mice. This indicates that pasteurized Akkermansia supplementation has more promising effects towards the amelioration of IR. Moreover, having a higher baseline amount of Akkermansia present in the intestinal lining is also associated with lower insulin sensitivity. Nonetheless, both live and pasteurized Akkermansia have substantial potential for the management of IR. There is a high level of consistency in studies affirming this effect, as almost all studies analyzed show improvement in IR. The predominant study types have low risk, but a moderate level of precision due to small sample sizes, correlational observations, and limited independent studies. However, due to their clear outcome definitions, high volume, and statistical analysis, the overall certainty of this effect is high.
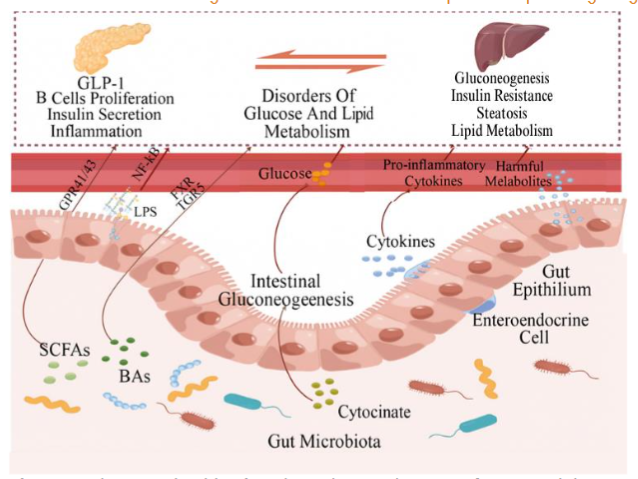
A. muciniphila’s effects for managing gluconeogenesis are not limited to IR; it has a potential role in managing blood sugar in relation to prediabetes as well. Prediabetes, another prevailing lifestyle disease, is characterized by elevated blood sugar above normal levels, but not yet at levels to be classified as Type 2 Diabetes Mellitus (T2DM). Through its role in gluconeogenesis illustrated in Figure 2, A. muciniphila has been considered as a mitigation for reducing blood glucose levels. In a 2024 study of 50 prediabetic rats by Yan S. et al., the effects of Akkermansia supplementation were studied in relation to blood glucose and as a comparison to dietary interventions. A. muciniphila was shown to delay the progression of prediabetes at similar rates to a dietary intervention. Whether paired with a dietary intervention or a high-fat diet, A. muciniphila had effects on insulin sensitivity and body composition. However, there was no significant difference in blood glucose levels after supplementation compared to baseline levels. In similar studies, including those for T2DM, parameters for measuring glucose levels, such as blood glucose and HbA1c, are not lowered significantly by A. muciniphila supplementation. However, in individuals with a naturally high composition of Akkermansia in the gut microbiota, body weight and Type 1 Diabetes were inversely correlated. There is also research that suggests that the efficacy of Akkermansia supplementation as a mitigation for T2DM is dependent on the baseline levels of Akkermansia present in the gut microbiota. Nevertheless, A. muciniphila abundance is positively correlated with T2DM. This finding proves that there are a plethora of roles A. muciniphila may play in the gut microbiota and metabolic regulation, and this area of application needs more investigation and research to make a definitive association between A. muciniphila and impaired glucose tolerance. Currently, the certainty of evidence regarding Akkermansia’s correlation to glycemic control and prediabetes is moderate to high, due to the high volume of RCTs, registered clinical trials, and observational studies that corroborate this effect.
3.3 A. muciniphila’s correlation to body composition
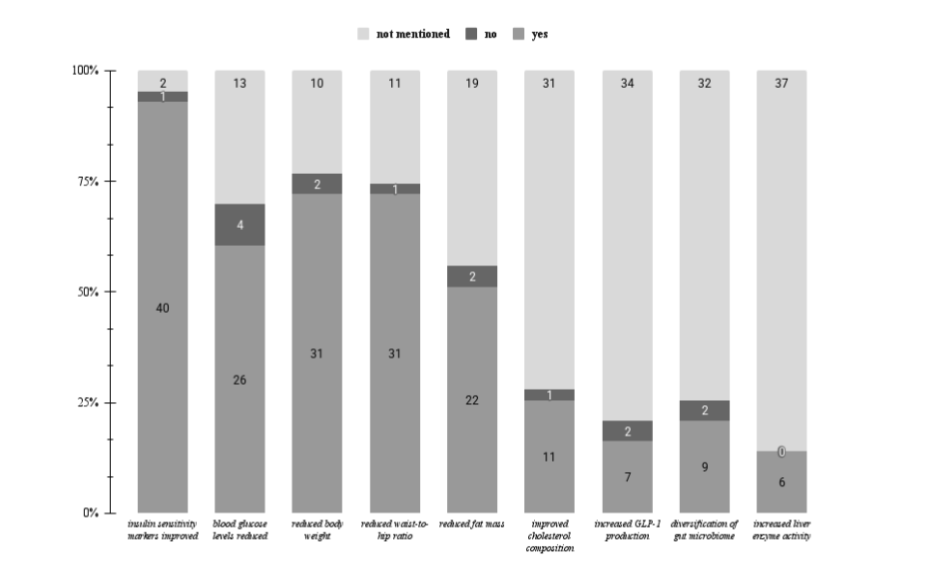
A. muciniphila also has a notable function for inducing positive change in body composition. Lipogenesis is the metabolism of extra energy from carbohydrates into lipids that can be stored for future use. However, when the body is in conditions such as IR, calorie restriction, inflammation, or eating a high-fat diet, lipogenesis is hindered, causing a higher rate of fat deposition in adipose tissue that can lead to obesity. Therefore, regulating lipogenesis has been a topic of study for decades. A potential solution has been A. muciniphila’s induced eubiosis in the intestinal lining, which allows for the regulation of lipogenesis. There is data from metagenomic studies that suggests that a natural higher composition of A. muciniphila in the gut microbiota is inversely correlated with obesity. Many studies suggest that supplementation with A. muciniphila has the ability to reduce body weight in overweight and obese individuals. In addition, Akkermansia supplementation has been shown to reduce waist-to-hip ratios, waist circumference, and hip circumference, key markers of abdominal obesity. Overall fat mass percentage is also reduced by supplementation and lean body mass is increased. In fact, 31 out of 43 papers examined directly validate the correlation between A. muciniphila and improved body composition, as demonstrated by Figure 3. One study by Depommier C., et al. of 32 overweight and obese adults from 2019 saw substantial results over 12 weeks of supplementation with pasteurized Akkermansia: “decreased body weight by about -2.27 kg, fat mass by about -1.37 kg and hip circumference by -2.63 cm.” This use of Akkermansia supplementation has been widely studied and has enormous potential for the future. There is also research that suggests that the regulation of lipogenesis induced by high levels of Akkermansia can allow for better maintenance of body weight as well. Although there is a high volume of studies supporting these conclusions, the overall certainty of evidence is moderate because there are very heterogeneous outcome measures across studies, variable factors that control body composition, and the duration of interventions is often relatively short.
3.4 Potential Applications for A. muciniphila
Recently, GLP-1 medications have taken the spotlight for their abilities to manage T2DM and reduce body weight in overweight and obese individuals. Being initially used for the management of T2DM, GLP-1 medications use semaglutide, liraglutide, dulaglutide, or tirzepatide, marketed under the brand names of Ozempic, Victoza, Trulicity, and Mounjaro, respectively. Its use for weight loss is a more recent application, with brand names like Wegovy, Saxenda, and Zepbound dominating markets in the current health landscape. Although GLP-1 medications operate in a different mechanism, mimicking the natural hormone that controls hunger, appetite, and blood sugar regulation, they have a relatively similar skillset to A. muciniphila supplementation. Therefore, researchers have tried to study potential links between A. muciniphila and natural GLP-1 production. As a relatively new topic of interest compared to other applications of Akkermansia, there is conflicting data on the correlation between Akkermansia levels and GLP-1 production. In a study of mice by Everard A., et al. conducted in 2013, higher Akkermansia levels were correlated with higher natural GLP-1 production, but the mechanisms of this association are unknown. This phenomenon is concurred by Zhao Y., et al.; Wang Z., et al.; Si J., et al.; and Sanjiwani., et al. However, in a supplementation study of 32 overweight and obese individuals by Depommier C., et al. from 2019, there were no significant changes to plasma GLP-1 levels post-intervention. The contrasting research and lack of understanding for the underlying process in which Akkermansia is related to GLP-1 production proves the necessity for further research, exploration, and examination of this topic. Nonetheless, the quality, precision, and consistency across studies is very varied and the overall volume of studies on this topic is also very low, revealing that these results have low certainty.
There have been predictions that supplementation with A. muciniphila would allow for diversification of the gut microbiome. However, this has yet to be positively correlated. Although A. muciniphila is able to strengthen the gut barrier, limited associations have been made between its supplementation and other gut microbiota. Overall, a diverse gut microbiome is generally considered beneficial for gut health and has associations with better digestion, nutrient absorption, and regulation of metabolism. However, Akkermansia only plays a part in the plethora of bacteria actively working towards strengthening the intestinal barrier. A. muciniphila typically makes up 1-5% of the gut microbiota in humans. Studies using supplementation of bacteria from the genus of Lactobacillus and Bifidobacterium also have similar results, pointing to the benefits of an overall diverse gut microbiota. However, many of these studies have limited data precision so far due to varied diversity metrics and sequencing methods across studies, despite there being a moderate volume of studies, meaning this result has moderate certainty in the current research.
A. muciniphila supplementation may also have the ability to decrease activity in liver enzymes. Liver enzymes, such as gamma-glutamyl transferase (γGT), aspartate-aminotransferase (AST) and alanine aminotransferase (ALT), play a key role in amino acid and nitrogen metabolism. Liver enzymes are crucial for the processing of nutrients, detoxification of harmful substances, and maintaining metabolic balance. However, excess activity has negative effects on metabolic balance and regulation. In a supplementation study of mice, Akkermansia reduces ALT and AST levels, mitigating future potential liver damage caused by overactivity of these enzymes. In a human trial of overweight and obese adults carried out by Depommier C., et al. (2019), γGT and AST levels were drastically reduced, with little to no change in AST levels. However, it is noteworthy that, although the reasoning behind this is unknown, only pasteurized Akkermansia supplementation was able to yield these effects and live supplementation had no effect on liver enzyme activity. These effects are moderately varied across studies with mixed quality levels and precision, demonstrating that these effects have a low level of certainty. Again, this proves the limited scope of our understanding of how gut microbiota function in tandem with the rest of the body systems and requires more research in this area.
4. Discussion
A. muciniphila is an emerging bacterium with several studied effects on human metabolism, with some being strongly linked while others being only ambiguously correlated. Its supplementation seeks to utilize these benefits for supporting the treatment of metabolic disorders, such as IR, prediabetes, or obesity. The supplementation of A. muciniphila has been shown to be well tolerated in humans. Several reports have correlated A. muciniphila supplementation with strengthening of the gut lining, which, in turn, has the results of improving insulin sensitivity, prediabetes, body composition, and cholesterol levels. In terms of insulin sensitivity, many studies corroborate Akkermansia supplementation with improved HOMA-IR and plasma insulin. This well-studied application has made strides in the treatment of IR, serving as a new and upcoming innocuous supplement that, when coupled with subtle lifestyle changes, can ameliorate IR. In addition, it has been found that there is an inverse correlation between A. muciniphila levels in the body and prediabetes. This use of Akkermansia supplementation has been shown to be effective at similar levels as a dietary intervention and can pave a path for the prevention of prediabetes. Moreover, A. muciniphila has had measurable effects for improving body composition according to numerous reports. Supplementation has been proven to reduce body weight, fat mass, and waist-to-hip ratios, while also promoting an increase in lean body mass. Moreover, supplementation with Akkermansia has been proven to reduce overall cholesterol levels, while also increasing HDL ratios. In an era when GLP-1 medications are quickly taking over the weight management landscape, Akkermansia supplementation provides a novel way to achieve similar results with less side effects.
However, as this is a topic backed with only about two decades of research, there are many studies that conflict with findings made previously. The mechanism behind Akkermansia supplementation itself is loosely understood, and therefore, its effects have contrasting evidence for and against it. Firstly, there are contrasting reports about the safety of Akkermansia supplementation. Although it has been administered widely in trials for both mice and humans, there are concerns that its mucin degrading properties may make oversupplementation a problem, raising concerns about potential inflammation. In addition, for reasons that are unclear so far, several studies point to the finding that pasteurized Akkermansia supplementation has better effects on IR, prediabetes, and body composition than live Akkermansia. This leads to uncertainty behind the actual mechanism of Akkermansia supplementation, as far as whether Akkermansia actually stays in the body when administered, whether supplementation has only temporary or long-lasting effects, and if supplementation can aid the natural composition of Akkermansia or not. Also, some of the links between Akkermansia supplementation have not been quantified by numbers. The most notable example is the lower prevalence of prediabetes with A. muciniphila supplementation without an actual substantial decrease in blood glucose levels in most studies. Likewise, some older studies claim Akkermansia supplementation has the ability to increase and stimulate the production of the GLP-1 hormone. However, more recent studies have not found a link between Akkermansia supplementation and plasma GLP-1 levels. This leads to the question of whether Akkermansia supplementation is actually effective, or if effects are dependent on the natural gut composition and prevalence of Akkermansia in the body. Metagenomic studies have found that natural levels of Akkermansia within the body are positively correlated with improved body composition, lower levels of prediabetes, and less insulin sensitivity, but it is still unclear how or even if Akkermansia supplementation is able to enhance these effects. As Akkermansia is naturally found in higher prevalence in individuals at healthy weight levels, studies done on overweight or obese participants, as most studies regarding A. muciniphila are, may not fully reflect the effects on the general population as a whole.
This review has several limitations that should be acknowledged. First, the literature search was restricted to English-language articles from PubMed, Europe PMC, Cochrane Library, and ClinicalTrials.gov, which, although they constitute the most comprehensive biomedical database regarding the subject of the review, may not have captured all relevant studies indexed in other sources, such as Scopus or Web of Science. This introduces a risk of selection bias and incomplete retrieval of evidence. Screening and data extraction were not performed in duplicate, further increasing risk of selection bias. Moreover, although we applied the PRISMA framework and screened studies systematically, the body of available evidence included heterogeneous study types, ranging from randomized controlled trials and observational studies to animal experiments and narrative reviews. The use of different critical appraisal tools allowed us to evaluate each according to its design, but direct comparison of findings across these diverse methodologies remains challenging. Therefore, a meta-analysis was not conducted. Although JBI checklists and certainty tables were applied, other frameworks (e.g., GRADE) were not used. Additionally, most of the human evidence is derived from small randomized controlled trials and a number of observational studies with moderate risk of bias, which limits the certainty and generalizability of conclusions. Finally, as this review did not include a quantitative meta-analysis, the synthesis remains qualitative and is subject to interpretation. Taken together, these factors highlight that while A. muciniphila shows promise as a therapeutic adjunct for metabolic disorders, the current evidence base remains preliminary and should be interpreted with caution. Future reviews should aim to overcome these issues in their methodologies.
Certainty of evidence varied across outcomes. For IR, study quality was moderate based on JBI appraisal, with high consistency across findings but limited precision due to small sample sizes; therefore, overall certainty was judged as moderate. For body composition and cholesterol, certainty was also moderate for similar reasons. In contrast, evidence for GLP-1 modulation and gut microbiome diversity was inconsistent, drawn primarily from small animal experiments or narrative reviews, and appraised as low certainty. Liver enzyme outcomes were supported by limited studies, yielding low–moderate certainty. These ratings underscore that while Akkermansia supplementation shows promise for metabolic outcomes, the strength of evidence remains limited by study design, sample size, and inconsistency in less well-studied endpoints.
A pioneering report in the study of Akkermansia supplementation in mice, a 2013 study by Everard A., et al. demonstrates the mechanisms of Akkermansia by examining the intestinal epithelium strengthening induced by supplementation. Concurrently, it endeavors to use this method of supplementation to manage obesity. In accordance with the present results, the study provides insight for the mechanisms behind Akkermansia supplementation by proving the bacteria strengthens the intestinal lining and regulates metabolism. However, contradictory to current findings, the study saw results from only live Akkermansia and no results from heat-killed Akkermansia. This result would suggest that bacterial activity, not just presence, is crucial for outcomes. This discrepancy to today’s research may be attributed to its focus on the intestinal epithelium rather than overall metabolic markers. Nonetheless, this finding proves the gaps in the understanding of the mechanisms of Akkermansia supplementation. A 2016 study of overweight and obese adults by Dao MC., et al. provided a baseline for Akkermansia supplementation in humans. Although this study did not supplement Akkermansia, it was able to examine and study the correlations between natural Akkermansia levels and metabolic markers. It was found that higher prevalence of Akkermansia levels in the gut microbiota are inversely related to insulin sensitivity, fasting glucose, waist-to-hip ratio, body fat mass, and plasma triglycerides, corroborating previous evidence in mice of the effects of Akkermansia for regulating metabolism. The study also provided the results of a dietary intervention for its 49 participants and correlated the effectiveness of the dietary changes with higher baseline levels of Akkermansia. A note of caution is due here, however, since the study did not include a control group and only provided before-and-after figures. Moreover, the study only focused on correlation, not causation, and therefore, these findings cannot be extrapolated to all patients and are somewhat limited. This study raises the question of whether or not Akkermansia supplementation would actually make a difference in the metabolic profile of people with an already high abundance of Akkermansia or if results are limited by baseline levels of Akkermansia, as Akkermansia was not directly administered in this study. An animal study by Plovier H., et al. (2017) aimed to find the mechanisms behind Akkermansia supplementation’s benefits to ameliorate gut barrier dysfunction. The study found that pasteurized Akkermansia improved insulin sensitivity and glucose tolerance in obese and diabetic mice. Moreover, metabolic inflammation and fat mass were reduced, corroborating previous studies. Specifically, this study found that Amuc_1100 was the key active membrane protein that facilitated these effects. However, this study is limited by the purification of the membrane protein, which may differ translationally in the supplementation of the whole bacterium. As an animal study, the effects of the purified membrane protein may also not apply fully to human supplementation. A 2017 animal study by Zhao S., et al. focused on the effects of Akkermansia supplementation on chow-fed mice. It was found that supplementation reduced inflammation and improved glucose metabolism. The effects observed in this study were independent of obesity status and, therefore, have limited translational relevance to metabolic syndrome, but are still important supporting evidence for Akkermansia’s relevance outside metabolic syndrome.
In 2018, van der Ark., et al. studied the conditions required to make a growth medium for Akkermansia, and in doing so, confirmed Akkermansia’s strong mucus dependence. Using systems biology and in vitro experiments, this study defined minimal growth requirements, supporting its ecological specialization in the gut mucosa. However, the work did not assess metabolic or clinical outcomes and lacks in vivo validation, limiting its direct relevance to therapeutic applications. Nonetheless, this medium provides strides towards supplementation methods for future use and has proved to be very useful and insightful in establishing the mechanisms of Akkermansia. Another in vitro experiment was accompanied by an ex vivo experimental study by Ashrafian F., et al. (2019). This experimental study showed that A. muciniphila and its extracellular vesicles modulated Toll-like receptor signaling and improved epithelial tight junction integrity in cellular and ex vivo models. While supportive of immunomodulatory mechanisms, the lack of in vivo confirmation limits its applicability to supplementation, but nonetheless, provides insights into the mechanisms driving gut microbiome changes by Akkermansia.
The first randomized, double-blind, placebo-controlled study examining the use of A. muciniphila supplementation in humans was conducted by Depommier C., et al. This study’s rigorous design was aimed at eliminating bias and to support prior findings from previous research. This study confirms the relationship between supplementation with Akkermansia and regulating gluconeogenesis, as well as lipogenesis. By measuring insulin sensitivity, lipid profiles, inflammation markers, and body composition, the study succeeded at providing a broad metabolic snapshot of the results of Akkermansia supplementation. These results further support the idea that Akkermansia supplementation is able to mitigate IR, prediabetes, and improve body composition. However, with an emphasis on safety and tolerability in humans, it did not aim to conclude with any significant new findings, but rather to support earlier ones. Although it provided evidence for the novel approach of using pasteurized Akkermansia supplementation, its results can be limited due to its short duration of only 3 months and limited participant pool (32 individuals who were overweight or obese). It can be argued that significant effects were seen largely due to the homogeneous participant pool that started out with decreased metabolic health, and that results may not apply to healthy weight individuals.
A 2019 study by Dao MC., et al. expanded on the bounds of Akkermansia abundance in the specific context of before and after bariatric surgery. This longitudinal observational study monitored Akkermansia levels in the gut microbiome of obese patients before and after an intervention of bariatric surgery. The study found that A. muciniphila abundance increased following bariatric surgery but was not independently associated with improvements in metabolic health. The results suggest context-dependent effects, based on health status over time, baseline Akkermansia, and more. This ties into the question brought up by earlier research by Dao MC., et al. of whether the effects of Akkermansia are limited by baseline levels and provides a discernment between natural Akkermansia growth and supplementation’s effects proven by other studies. This includes the study done by Yang M., et al. (2020), which examined the effects of newly identified Akkermansia strains on mice with high-fat diet induced obesity. Administration of the newly isolated A. muciniphila strains to obese mice reduced body weight, adiposity, and improved glucose and lipid metabolism, highlighting strain-specific effects. This shows the potential for strain specific, highly-targeted treatments for future amelioration of metabolic dysregulation. This study particularly opens doors for the commercialization of “personalized” treatments in the future, as strain specific treatments can be of future importance. However, there is still uncertainty regarding the generalizability of specific strains to a population as a whole, as the study used a population of homogenous mice. There is also promising, but limited, evidence that effects will be translational to a human population. Nonetheless, the study’s focus on newly isolated Akkermansia strains provides enormous potential for future research directions. In a cross-sectional human study by Zhou Q., et al. (2021), higher gut Akkermansia abundance was associated with lower metabolic syndrome severity. The study examined the correlation of Akkermansia abundance and the individual factors of metabolic syndrome. Models were then used to find the dose-dependent responses and microbial interactions present from Akkermansia, demonstrating that there was a nonlinear relationship between the factors of metabolic syndrome and A. muciniphila. The study found correlations between Akkermansia and obesity, waist circumference, hypertriglyceridemia, HDL, and blood pressure. However, the study found no correlation between Akkermansia and hyperglycemia, fasting blood glucose, and hypertension. The discrepancy between this study’s results and previous findings about hyperglycemia and fasting blood glucose are stark, as acknowledged by the authors. The authors provide some reasons for the discrepancy to be heterogeneity of the host population or variability in the gut microbiome. However, given the large sample size of over 6,000 participants and balancing of factors such as age, sex, etc., these results do provide a valid divergence from other studies, and require more research in future studies. Another research discrepancy was found in the study done by Yoon HS., et al. (2021). This mechanistic mouse study identified a secreted A. muciniphila protein that stimulated GLP-1 secretion, improving glucose homeostasis and insulin sensitivity. Although mechanistically compelling, the findings are limited to animal models, and the clinical relevance of protein dosing and delivery remains uncertain. Moreover, this finding has very limited evidence in any other studies due to a very low volume of work on the topic. Other studies, such as the RCT by Depommier C., et al., have found no correlation between Akkermansia and GLP-1 production. This may be an area of substantial future research directions due to the rising popularity of GLP-1 medications.
In 2023, Wang Z., et al. studied the relation between Akkermansia supplementation and glucose tolerance from the unique perspective of the daily light to dark transition in mice. This study used targeted genetic models to pinpoint the mechanisms of Akkermansia in mice genetically engineered to lack intestinal FFAR4 which is known to regulate metabolism, lipogenesis, and gluconeogenesis. This work confirms that pasteurized Akkermansia supplementation surpasses the results of live Akkermansia. However, these findings may be somewhat limited due to the extremely specific species type and genetic alterations present in the study’s subjects. A 2024 study by Yan S., et al. examined the role of Akkermansia supplementation in prediabetic rats with an emphasis on pancreatic islet β-cell function for mitigating the development of T2DM. The study is consistent with previous findings that Akkermansia supplementation is able to regulate gluconeogenesis, and in turn, manage or even reverse prediabetes. This study is especially novel in its approach, as it directly compares Akkermansia supplementation to a dietary intervention. The study also examined the role of Akkermansia alongside a dietary intervention, not just instead of it, providing supplementary evidence for the role of Akkermansia in a healthy gut microbiota. However, the study’s multiple groups allotted for only 8 rats per group, meaning the small sample size can cause inaccuracies in data trends. While an informative study, this data must be interpreted with caution because results may not directly correlate or translate to humans. In addition, the long-term implications of Akkermansia supplementation were not studied, creating a gap of knowledge for the eventual progression and potential relapse of the rats’ conditions. In 2023, Nian F., et al. conducted an animal study using a mouse model of NAFLD to explore the effects of combined supplementation with A. muciniphila and Bifidobacterium bifidum. It was found that this combination reduced hepatic steatosis and inflammation through FXR-related pathways. However, the interpretation of these results in regards to its application for Akkermansia supplementation is limited by the combined intervention, which precludes attribution of effects to A. muciniphila alone. In 2024, another animal study by Yan S., et al. studied mice with high-fat-diet-induced prediabetes. This rat study demonstrated that A. muciniphila improved pancreatic islet β-cell function and insulin secretion capacity in a high-fat-diet-induced prediabetes model. The report’s focus on the pathways by which Akkermansia affects prediabetes provides huge insight into its mechanisms. Furthermore, the study compared Akkermansia with a dietary intervention, providing important benchmarks of comparison. However, the study’s results are limited by the short-term outcome measurement, which leaves long-term effects and mechanisms unknown. Although improved insulin sensitivity was an outcome of this study, this cannot be generalized to a human population due to the use of mice, as well as the lack of whole-body insulin sensitivity measures. In a 2025 animal mechanistic study, Lin H., et al. used a mouse model of doxorubicin-induced cardiotoxicity to explore the possibility of Akkermansia’s use for treatment. The study found that Akkermansia administration restructured the microbiome present, causing protective effects against doxorubicin-induced cardiotoxicity. A. muciniphila supplementation also improved mitochondrial biogenesis and energy metabolism via PPARα signaling. Although the primary outcome was cardiotoxicity rather than metabolic disease, which limits its relevance to obesity or insulin resistance, this study provided insights into the effect pathways along which Akkermansia acts.
A 2025 human randomized-controlled trial by Zhang Y., et al. aimed to fill some of the gaps present in current human research regarding Akkermansia supplementation. This randomized controlled trial evaluated Akkermansia muciniphila supplementation in overweight and obese individuals with type 2 diabetes. Using precise statistical analysis, this study demonstrated significant improvements in glycemic control and insulin sensitivity compared with placebo. Notably, treatment efficacy was strongly modified by baseline gut A. muciniphila abundance, with participants harboring higher initial levels experiencing more pronounced metabolic benefits. This stratified response supports the concept of personalized microbiome-based interventions, as brought up by previous research, and suggests that host microbial context is a key determinant of probiotic efficacy rather than supplementation acting as a universally effective therapy. Despite its strengths as a well-designed human intervention study, the follow-up period was relatively short, limiting conclusions regarding durability of glycemic improvements, long-term insulin resistance trajectories, or effects on clinically meaningful outcomes such as diabetes progression, medication requirements, or cardiovascular risk. The baseline-dependent response, while mechanistically informative, also constrains generalizability, as the intervention may be ineffective in individuals with low endogenous A. muciniphila levels, which is arguably the population most in need of metabolic improvement. Although this trial does not evaluate long-term efficacy or safety, it provides some of the strongest direct human evidence supporting A. muciniphila’s metabolic potential.
There are many areas regarding Akkermansia supplementation that are not fully understood or unexplored and warrant further research and studies. The long-term effects of Akkermansia supplementation have not been studied yet. There is still very little variation studied between the supplementation of different strains within the A. muciniphila genus, which could potentially have different effects. Moreover, although it has been demonstrated that pasteurized Akkermansia has more significant effects than live Akkermansia on IR, as well as body composition, the mechanisms behind this occurrence are only inferred, but not completely understood currently. Despite these promising results, questions remain unanswered. Many studies offer hypotheses, but a concrete explanation for this phenomenon is not agreed upon. Furthermore, additional testing is required to solidify the optimal dosage, form, and frequency of supplementation. There is abundant room for further progress in determining the minutia of Akkermansia supplementation with the aim of optimizing its effects.
5. Conclusions
With many of the leading causes of death in the world being attributed to lifestyle disease, the global pandemic of metabolic disorders is becoming increasingly important to address. An upcoming field of study aims to mitigate metabolic disorders through gut microbiome-based treatments, specifically pasteurized A. muciniphila. This noninvasive, well-tolerated approach has shown potential in improving metabolic health by strengthening the gut barrier. Studies strongly correlate Akkermansia with reduced IR, improved body composition, and improved cholesterol. There is also emerging research for Akkermansia’s ability to reduce the prevalence of prediabetes, stimulate GLP-1 production, regulate liver enzyme activity, and diversify the gut microbiome through supplementation, highlighting its immense range of promising potential applications. Although more research is required to fully understand the mechanisms behind these effects, the current studies provide substantial evidence for its use in mitigating metabolic disorders. Given the complexity of the gut microbiome and growing scrutiny of GLP-1 medications, Akkermansia offers a promising ‘natural’ alternative. The next steps in research are to optimize the form, dosage, frequency, and specific strain of supplementation, along with researching long-term safety and efficacy. Akkermansia supplementation supports a foundational element of overall health and may become part of therapies for complex, multi-system diseases beyond just metabolism in the future. A. muciniphila supplementation gives hope for a future where chronic diseases are prevented, not just managed by pharmaceuticals and drugs, by reshaping and harnessing the power of the ecosystems already inside us.
Supplementary Materials:
The following supporting information can be downloaded: Table S1: Risk of Bias Table; Table S2: Certainty of Evidence Table
Author Contributions: All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Data Availability Statement: The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author(s).
Acknowledgments: During the preparation of this manuscript/study, the author(s) used OpenAI GPT-4 and Perplexity AI GPT-4 for the purposes of extracting relevant quotations from full text articles, analyzing risk of bias for studies, generating a summary table, and generating a graphical abstract. All Generative AI output was manually reviewed by the authors. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest: The authors declare no conflicts of interest.
References
- Dao MC, Everard A, Aron-Wisnewsky J, et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. 2016;65(3):426-436. doi:10.1136/gutjnl-2014-308778
- Wang Y, He J, Chen S, et al. Bibliometric analysis of research trends and prospective directions of Akkermansia muciniphila from 2010 to 2024. Front Microbiol. 2025;16:1569241. doi:10.3389/fmicb.2025.1569241
- Yan S, Chen L, Li N, et al. Effect of Akkermansia muciniphila on pancreatic islet β-cell function in rats with prediabetes mellitus induced by a high-fat diet. Bioresour Bioprocess. 2024;11(1):51. doi:10.1186/s40643-024-00766-4
- Salles BIM, Cioffi D, Ferreira SRG. Probiotics supplementation and insulin resistance: a systematic review. Diabetol Metab Syndr. 2020;12(1):98. doi:10.1186/s13098-020-00603-6
- Depommier C, Everard A, Druart C, et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. 2019;25(7):1096-1103. doi:10.1038/s41591-019-0495-2
- Jian H, Liu Y, Wang X, Dong X, Zou X. Akkermansia muciniphila as a Next-Generation Probiotic in Modulating Human Metabolic Homeostasis and Disease Progression: A Role Mediated by Gut-Liver-Brain Axes? Int J Mol Sci. 2023;24(4):3900. doi:10.3390/ijms24043900
- Chang CJ, Lin TL, Tsai YL, et al. Next generation probiotics in disease amelioration. J Food Drug Anal. 2019;27(3):615-622. doi:10.1016/j.jfda.2018.12.011
- Everard A, Belzer C, Geurts L, et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci U S A. 2013;110(22):9066-9071. doi:10.1073/pnas.1219451110
- He K, An F, Zhang H, et al. Akkermansia muciniphila: A Potential Target for the Prevention of Diabetes. Foods Basel Switz. 2024;14(1):23. doi:10.3390/foods14010023
- Zhao Y, Yang H, Wu P, et al. Akkermansia muciniphila: A promising probiotic against inflammation and metabolic disorders. Virulence. 2024;15(1):2375555. doi:10.1080/21505594.2024.2375555
- Zhou Q, Pang G, Zhang Z, et al. Association Between Gut Akkermansia and Metabolic Syndrome is Dose-Dependent and Affected by Microbial Interactions: A Cross-Sectional Study. Diabetes Metab Syndr Obes Targets Ther. 2021;14:2177-2188. doi:10.2147/DMSO.S311388
- Wang Z, Cui S, Zhang T, et al. Akkermansia muciniphila supplementation improves glucose tolerance in intestinal Ffar4 knockout mice during the daily light to dark transition. mSystems. 2023;8(5):e0057323. doi:10.1128/msystems.00573-23
- Plovier H, Everard A, Druart C, et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat Med. 2017;23(1):107-113. doi:10.1038/nm.4236
- Nian F, Wu L, Xia Q, Tian P, Ding C, Lu X. Akkermansia muciniphila and Bifidobacterium bifidum Prevent NAFLD by Regulating FXR Expression and Gut Microbiota. J Clin Transl Hepatol. 2023;11(4):763-776. doi:10.14218/JCTH.2022.00415
- Zhao S, Liu W, Wang J, et al. Akkermansia muciniphila improves metabolic profiles by reducing inflammation in chow diet-fed mice. J Mol Endocrinol. 2017;58(1):1-14. doi:10.1530/JME-16-0054
- Dao MC, Belda E, Prifti E, et al. Akkermansia muciniphila abundance is lower in severe obesity, but its increased level after bariatric surgery is not associated with metabolic health improvement. Am J Physiol Endocrinol Metab. 2019;317(3):E446-E459. doi:10.1152/ajpendo.00140.2019
- Yang M, Bose S, Lim S, et al. Beneficial Effects of Newly Isolated Akkermansia muciniphila Strains from the Human Gut on Obesity and Metabolic Dysregulation. Microorganisms. 2020;8(9):1413. doi:10.3390/microorganisms8091413
- Ashrafian F, Behrouzi A, Shahriary A, et al. Comparative study of effect of Akkermansia muciniphila and its extracellular vesicles on toll-like receptors and tight junction. Gastroenterol Hepatol Bed Bench. 2019;12(2):163-168.
- A-Mansia Biotech S.A. A Randomized, Double-Blind, Placebo-Controlled Study to Investigate the Effects of Next Generation Beneficial Microbes, Akkermansia Muciniphila (AMF-01), on Long-Term Improvements in Insulin Resistance in Otherwise Healthy Hyperglycaemic Adults. clinicaltrials.gov; 2025. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT05114018
- Cani PD. Evaluation of the Effects Associated With the Administration of Akkermansia Muciniphila on Parameters of Metabolic Syndrome Related to Obesity. clinicaltrials.gov; 2019. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT02637115
- Chen Y. A Randomized, Double-Blind, Placebo-Controlled Clinical Study to Evaluate the Effects of Akkermansia Muciniphila and Berberine on Prediabetes Among Obese Subjects. clinicaltrials.gov; 2025. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT05720299
- Maastricht University Medical Center. Effect of Pasteurized Akkermansia Muciniphilia on Maintenance of Body Weight After a Low Calorie Diet. clinicaltrials.gov; 2023. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT05417360
- National Taiwan University Hospital. Development of Microbial Therapeutics for Metabolic-Associated Fatty Liver Disease: From Mechanistic Investigations to Clinical Trials. clinicaltrials.gov; 2025. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT06873165
- Wecare Probiotics Co., Ltd. Effect of Akkermansia Muciniphila Akk11 on Weight Loss in Obese Patients: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Study. clinicaltrials.gov; 2025. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT06780007
- Weiqing W. A Randomized, Double-Blind, Placebo-Controlled, Multicenter Clinical Trial to Evaluate the Effect of Akkermansia Muciniphila WST01 Strain in Patients With Type 2 Diabetes. clinicaltrials.gov; 2023. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT04797442
- van der Ark KCH, Aalvink S, Suarez-Diez M, Schaap PJ, de Vos WM, Belzer C. Model-driven design of a minimal medium for Akkermansia muciniphila confirms mucus adaptation. Microb Biotechnol. 2018;11(3):476-485. doi:10.1111/1751-7915.13033
- Lin H, Shao X, Gu H, et al. Akkermansia muciniphila ameliorates doxorubicin-induced cardiotoxicity by regulating PPARα-dependent mitochondrial biogenesis. Npj Biofilms Microbiomes. 2025;11(1):86. doi:10.1038/s41522-025-00712-y
- Yoon HS, Cho CH, Yun MS, et al. Akkermansia muciniphila secretes a glucagon-like peptide-1-inducing protein that improves glucose homeostasis and ameliorates metabolic disease in mice. Nat Microbiol. 2021;6(5):563-573. doi:10.1038/s41564-021-00880-5
- Zhang Y, Liu R, Chen Y, et al. Akkermansia muciniphila supplementation in patients with overweight/obese type 2 diabetes: Efficacy depends on its baseline levels in the gut. Cell Metab. 2025;37(3):592-605.e6. doi:10.1016/j.cmet.2024.12.010
- Corb Aron RA, Abid A, Vesa CM, et al. Recognizing the Benefits of Pre-/Probiotics in Metabolic Syndrome and Type 2 Diabetes Mellitus Considering the Influence of Akkermansia muciniphila as a Key Gut Bacterium. Microorganisms. 2021;9(3):618. doi:10.3390/microorganisms9030618
- Kobyliak N, Falalyeyeva T, Kyriachenko Y, et al. Akkermansia muciniphila as a novel powerful bacterial player in the treatment of metabolic disorders. Minerva Endocrinol. 2022;47(2):242-252. doi:10.23736/S2724-6507.22.03752-6
- Abuqwider JN, Mauriello G, Altamimi M. Akkermansia muciniphila, a New Generation of Beneficial Microbiota in Modulating Obesity: A Systematic Review. Microorganisms. 2021;9(5):1098. doi:10.3390/microorganisms9051098
- ChiCTR2400090949. A Study on the Effectiveness of Akkermansia muciniphila in Improving Insulin Resistance in Obese Patients with Polycystic Ovary Syndrome | Cochrane Library. Published online May 31, 2018. doi:10.1002/central/CN-02782407
- Zeng Z, Chen M, Liu Y, et al. Role of Akkermansia muciniphila in insulin resistance. J Gastroenterol Hepatol. 2025;40(1):19-32. doi:10.1111/jgh.16747
- Sanjiwani MID, Aryadi IPH, Semadi IMS. Review of Literature on Akkermansia muciniphila and its Possible Role in the Etiopathogenesis and Therapy of Type 2 Diabetes Mellitus. J ASEAN Fed Endocr Soc. 2022;37(1):69-74. doi:10.15605/jafes.037.01.13
- Yuan L, Li Y, Chen M, et al. Therapeutic applications of gut microbes in cardiometabolic diseases: current state and perspectives. Appl Microbiol Biotechnol. 2024;108(1):156. doi:10.1007/s00253-024-13007-7
- VERONICA O. Efficacy Evaluation of Inulin Supplementation on Metabolic Control and Akkermansia Muciniphila Levels in Patients With Type 1 Diabetes: A Pilot Study. clinicaltrials.gov; 2023. Accessed December 7, 2025. https://clinicaltrials.gov/study/NCT05795972
- Bednarska NG, Håberg AK. Understanding Patterns of the Gut Microbiome May Contribute to the Early Detection and Prevention of Type 2 Diabetes Mellitus: A Systematic Review. Microorganisms. 2025;13(1):134. doi:10.3390/microorganisms13010134
- Xu Y, Wang N, Tan HY, Li S, Zhang C, Feng Y. Function of Akkermansia muciniphila in Obesity: Interactions With Lipid Metabolism, Immune Response and Gut Systems. Front Microbiol. 2020;11:219. doi:10.3389/fmicb.2020.00219
- Shaheen N, Khursheed W, Gurung B, Wang S. Akkermansia muciniphila: A key player in gut microbiota-based disease modulation. Microbiol Res. 2025;301:

