Advancements in Anemia Treatment for Chronic Kidney Disease
Therapeutic Advancements in Anemia of Chronic Kidney Disease and End Stage Kidney Disease
Neeraj Sharma1, Marcus Gay1, Marina Tawadrous1, Tracy Lin1
- Department of Medicine, Division of Nephrology, Keck School of Medicine, University of Southern California, 2020 Zonal Avenue IRD #806, Los Angeles, California 90033, US
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Sharma, N., and Gay, M., et al., 2024. Therapeutic Advancements in Anemia of Chronic Kidney Disease and End Stage Kidney Disease. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5815
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5815
ISSN 2375-1924
ABSTRACT
Anemia is a frequent complication during the late stages of chronic kidney disease, reaching an almost universal prevalence among those with end stage kidney disease. There is a clear association between anemia and an increased risk for decline in the estimated glomerular filtration rate, cardiovascular events, and all-cause mortality. The main mechanisms for the development of anemia related to chronic kidney disease are a relative erythropoietin deficiency and disordered iron homeostasis related to chronic inflammation. Recent improvements in the understanding of the pathophysiology of anemia in chronic kidney disease have advanced the diagnosis and treatment of anemia in chronic kidney disease. While the cornerstone of the diagnosis of anemia related to chronic kidney disease remains excluding other etiologies of anemia, newer diagnostic resources include population-informed cutoffs for traditional measures of iron storage as well as evolving diagnostics of the reticulocyte hemoglobin index and percent hypochromic red blood cells in plasma. Intravenous iron has emerged as a novel tool for both the diagnosis and treatment of disordered iron homeostasis in the anemia of chronic kidney disease. Outside of iron replacement, the treatment of the anemia of chronic kidney disease has traditionally relied on erythropoiesis stimulating agents. A newer class of medications, the Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors, are an emerging therapy that work by stimulating the transcription of the erythropoietin gene in the Kidney and Liver. These agents are oral therapy, unlike erythropoiesis-stimulating agents, and are used in those who fail to respond to traditional therapy. Additional novel and experimental therapies include SGLT2 inhibitors, inhibitors of hepcidin production, and novel oral and intravenous iron formulations. This review encompasses the highlights of the epidemiology, pathophysiology, and the recent diagnostic and therapeutic advancements for anemia in chronic kidney disease.
Keywords
Anemia, Chronic Kidney Disease, End Stage Kidney Disease, Erythropoietin, Iron Homeostasis, Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors
I. Introduction
According to the Kidney Disease Improving Global Outcomes (KDIGO) guidelines, chronic kidney disease (CKD) is defined as abnormality of the kidney function or structure that is present for a minimum of 3 months, with implications for health. Anemia is a common and serious complication of CKD that presents at the early phase of CKD and worsens as the estimated glomerular filtration rate (eGFR) declines. The pathophysiology of anemia in CKD is multifaceted and complex but mainly involves the development of erythropoietin deficiency and resistance as well as a heightened inflammatory state. Anemia, defined by the World Health Organization as a hemoglobin concentration of <13 g/dL in men and <12 g/dL in women, is associated with decreased quality of life, as well as with increased morbidity and mortality. Prevalence of anemia increases as the eGFR declines due to development of erythropoietin deficiency, heightened inflammatory state, and the uremic milieu. The intricate interplay of sever concomitant factors affects the distribution and severity of anemia.
Sir Robert Christison in 1839 was the first to define anemia related to kidney disease. Christison observed that “by far the most remarkable character of the blood in the advanced stage of the Bright’s disease is a gradual and rapid reduction of its colouring” and “no other natural disease came as close to hemorrhage for impoverishing the red particles of the blood.” Similarly, Richard Bright had noticed that patients with kidney disease had paleness of the skin. The severity of anemia has been shown to be associated with poor outcomes in patients with CKD. Foley and colleagues demonstrated that mean hemoglobin was an independent risk factor for left ventricular dilatation, heart failure, and mortality in a cohort of 432 patients on dialysis followed prospectively for 41 months. In addition, anemia accelerates the progression of kidney disease through multiple mechanisms. First, low hemoglobin levels reduce oxygen delivery to the kidneys resulting in medullary hypoxia which favors interstitial fibrosis. Anemia may also stimulate renal sympathetic nerve activity and then induce an increase in glomerular pressure and proteinuria, which is another factor contributing to CKD progression.
Prior to the availability and approval of erythrocyte stimulating agents, it was common for patients on chronic dialysis to require regular blood transfusion. However, this treatment resulted in iron overload, human leukocyte antigen (HLA) sensitization, transfusion reactions, and transmission of viral agents. Within the last decade, there have been new insights into the regulation of iron homeostasis, discoveries of the hepcidin-ferroprotein axis, erythroferrone and the role of HIFs. The aim of the present review will therefore summarize the most recent findings, treatment and novel developments in the management of anemia in patients requiring dialysis.
II. Epidemiology
Chronic kidney disease is a global public health problem, involving approximately 10% of the world population. In the United States (U.S.), 14% of adults had a low eGFR, albuminuria, or both based on the National Health and Nutrition Examination Survey (NHANES) data from 2020. 5.1% of adults in the U.S., had stage 3 CKD. CKD was much more prevalent among individuals ages >65 years. In terms of healthcare expenditures, in 2021, 13.5% of Medicare fee-for-service (FFS) beneficiaries aged ≥66 years had a diagnosis of CKD, yet they accounted for about one-quarter of total Medicare FFS spending for this age group, at $76.8B. Regarding end stage kidney disease (ESKD), between 2001 and 2019, the number of patients with newly registered ESKD increased from 97,856 to 134,837, an increase of 37.8%. Overall, in 2021, the mean eGFR and hemoglobin at initiation of kidney replacement therapy was 9.9mL/min/1.73m2 and 9.4 g/dL, respectively. Given that patients with CKD often develop anemia, it is of no surprise that anemia of CKD presents a considerable healthcare burden due to increase costs of associated with disease management as well as a negative impact on patient health-related quality of life. According to 2007-2010 data from the NHANES, the prevalence of anemia among patients with CKD in the U.S. was previously estimated to be 15.4%. However, a more recent investigation by Kovesdy et al that included NHANES data from 1999 to 2018 showed that approximately 25% of individuals with stage 3-5 CKD in the U.S. had anemia. Among this group, about 2% had hemoglobin <10 g/dL, thus eligible to receive Erythropoiesis Stimulating Agent (ESA) treatment. Furthermore, the weighted prevalences of anemia and anemia with Hb<10 g/dL also increased with advancing CKD stage from 17.1% and 1.1%, respectively, in stage 3A to 66.1% and 12.7% respectively in stage 5 CKD. The same authors also demonstrated that anemia was significantly more likely to occur in patients aged ≥75 years, females, Black race, and those with CKD stage ≥3B with concomitant diabetes. The authors suggested that enhanced anemia surveillance may be needed for patients with those risk factors, which may assist in optimizing the screening of anemia by identifying high risk patients with CKD who require treatment. Evans et al. examined anemia management in patients with advanced chronic kidney (CKD ≥ stage 3B) and how the dialysis patients compared with patients with advanced CKD. This was an observational study from the Swedish Renal Registry evaluating the epidemiology of anemia across stage 3b CKD to dialysis (both peritoneal dialysis and hemodialysis). The study population was 11,370 patients with advanced CKD and 3,045 patients receiving dialysis. The results showed that anemia occurred in 60% of patients with advanced CKD and 93% of dialysis patients.
III. Pathophysiology
RELATIVE ERYTHROPOIETIN DEFICIENCY
Anemia of CKD is driven by a variety of mechanisms including a relative deficiency of erythropoietin (EPO), absolute and relative iron deficiency, possible uremic bone marrow suppression, and decreased RBC lifespan. Among the causes of anemia in CKD, the most important and most well-known is a relative EPO deficiency. In the mid-twentieth century, it was discovered that the kidneys are the main driver of erythropoiesis, as experimental animals with their kidneys removed could not mount an appropriate erythropoietic response to anemia. We now know that the kidneys maintain erythropoiesis by producing about 90% of the body’s EPO, a peptide hormone that mediates appropriate erythrocyte production in the setting of inadequate oxygenation. The details of EPO cellular physiology are explained elsewhere. Briefly, anemia or hypoxia stimulate the transcription factor Hypoxia Inducible Factor (HIF) to upregulate the transcription of EPO in Renal Pericytes, which goes on to promote the survival and proliferation of erythroblasts. Studies have shown that the kidneys of patients with CKD are unable to produce an appropriate level of EPO for a given level of anemia; for example, McGonigle and colleagues found that serum EPO concentrations increased exponentially as hematocrit decreased in anemic individuals without CKD, but that EPO did not increase with decreasing hematocrit in CKD patients. However, the absolute value of EPO in individuals with CKD is comparable to non-anemic people without CKD. The combination of these findings makes the basis for the “relative EPO deficiency” seen in individuals with CKD who are unable to mount an appropriate EPO production to anemia. The proposed mechanism behind inadequate EPO production in CKD is that renal inflammation driven by persistent renal injury causes a subset of renal pericytes, known as Renal Erythropoietin Producing Cells (REPCs), to undergo metaplasia into myofibroblasts, which have a decreased ability to produce EPO. The relative EPO deficiency in CKD is an important cause of the anemia of CKD, but it is not the only cause. Recent guidelines state that 10-20% of cases of anemia of CKD are resistant to supplemental EPO therapies. The resistance to EPO therapy highlights the importance of other pathophysiologic determinants of anemia in CKD outside of EPO deficiency.
DISORDERED IRON HOMEOSTASIS AND OTHER CAUSES OF ANEMIA IN CHRONIC KIDNEY DISEASE
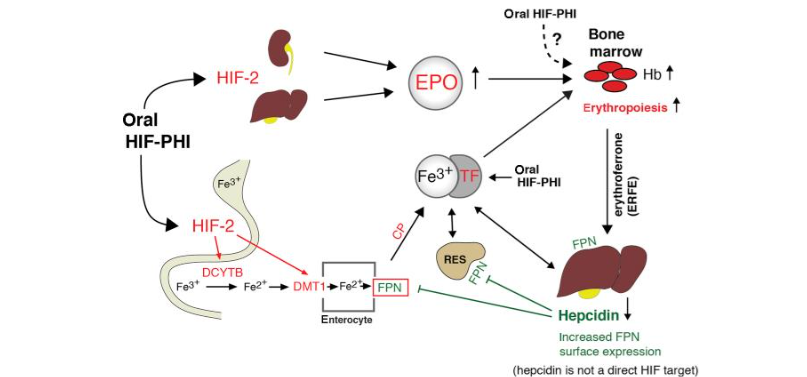
The second major cause of the anemia of CKD is disordered iron homeostasis, which occurs in the form of both absolute and functional iron deficiency. Absolute iron deficiency occurs from a depletion of the total body stores of iron. Individuals with CKD are also prone to developing a functional (or relative) iron deficiency, which is when the body’s iron stores are unable to be mobilized effectively for the generation of hemoglobin in the bone marrow. It is helpful to understand basic iron metabolism for discussions about relative iron deficiency; There are several excellent reviews of iron physiology. Hepcidin is a hormone produced by the liver that is the key player in mediating the sequestration of iron. Hepcidin is produced in response to increased iron intake, hypoxia, and inflammation. It inhibits the release of iron from storage cells by degrading ferroportin, the transporter that is necessary for iron to exit enterocytes and reticuloendothelial cells. Hepcidin also inhibits the intestinal absorption of iron. Studies have shown that hepcidin is increased in CKD, and Zaritsky et. al demonstrated that in their cohort of CKD patients, higher stages of CKD were associated with higher levels of circulating hepcidin. The increased levels of inflammation and hepcidin in CKD result in increased levels of ferritin and decreased levels available iron, measured as the transferrin receptor saturation, as well as impaired absorption of oral iron. The constellation of these changes results in a relative iron deficiency that causes decreased production of hemoglobin. Additionally, the mobilization of iron to may not be adequate to support the increased erythropoietic rate driven by ESAs in CKD patients. There has been a lot of interest in whether hypoxia inducible factor-prolyl hydroxylase inhibitors (HIF-PHI’s) improve availability of iron compared with Erythropoesis Stimulating Agents (ESAs). One mechanism we believe is that erythropoiesis stimulates release of erythroferrone from the bone marrow. Erythroferrone in turn decreases hepcidin production to make iron available for erythropoiesis. As such, HIF-PHIs reduce hepcidin indirectly by inducing erythropoiesis. Another mechanism is that HIF-PHI upregulates an iron transporter and ferroreductase, which would be expected to facilitate iron absorption from the gastrointestinal tract.
IV. Diagnosis of Anemia in Chronic Kidney Disease
The diagnosis of anemia in CKD involves periodic surveillance of hemoglobin levels to establish the diagnosis of anemia, evaluating for non-CKD related causes of anemia, and evaluating iron stores. In a landmark study, Astor et. al showed that among a large cohort of adult CKD patients, the prevalence and incidence of anemia increased as eGFR decreased, with an inflection point where the rate of anemia rapidly increased starting at an eGFR of 60mL/min/meters squared. Therefore, patients with lower renal function should be monitored more frequently for anemia. The 2012 KDIGO guidelines recommend monitoring hemoglobin at least annually in those with CKD 3, at least every 6 months in all CKD 4 patients and in CKD 5 patients not on dialysis, and at least every 3 months among patients on dialysis. The differential diagnosis of anemia is broad, and diagnosing anemia due to CKD requires excluding other causes of anemia. Therefore, the initial workup for anemia of someone with CKD is the same as for someone without CKD. KDIGO recommends obtaining, at a minimum, a complete blood count, absolute reticulocyte count, ferritin, and transferrin saturation (TSAT) at the time of diagnosis of anemia. Common additional tests include a fecal occult blood test, and measurements of serum Folate and Vitamin B12 levels. If all diagnostics do not suggest an alternative cause of anemia, and iron studies do not suggest an absolute iron deficiency anemia, then anemia due to CKD can be diagnosed. Although absolute iron deficiency can be reasonably excluded with laboratory tests, patients with anemia and CKD still have a relative iron deficiency driven by inflammation. The practical matter of predicting whether a patient will respond to iron therapy is difficult. A group found that even among those with adequate iron stores as measured by bone marrow biopsy, an erythropoietic response to intravenous iron is still seen in 30% of patients. In fact, those receiving EPO therapy may have a relative iron deficiency induced by the increased iron demands caused by EPO itself, which can make the use of traditional iron indices limited in their determination of iron-responders versus non-responders. Finally, there are other newer indices used to diagnose absolute and relative iron deficiency anemia in CKD, namely the percent hypochromic red blood cells and the reticulocyte hemoglobin index. These indices were evaluated in a meta-analysis performed within the 2016 National Institute of Health Care Excellence (NICE) Guidelines. The meta-analysis found that a percent hypochromic red blood cell greater than 6% and a reticulocyte hemoglobin index less than 29pg could be used to diagnose iron-responsiveness in anemics with CKD with a better negative predictive value compared to using a TSAT less than 20% and a ferritin less than 100 ng/dL. These tests are limited by the fact that they require flow cytometry, which is not as available as standard iron tests like TSAT and ferritin.
V. Management and novel therapies
If we take an overview of the optimal management of anemia between patients with CKD nondialysis and ESKD, they should be fundamentally different. CKD patients require individualized, patient focused care due to the limited use of ESA products in this population. There is a high rate of iron deficiency in the CKD patients and if that is treated then you delay the need for ESA very late into CKD stages and hopefully not need ESA at all. In addition, the risk benefit analysis does not strongly favor using ESA because the main value is reducing transfusions which is less common in the CKD population. And the competing risk of cancer progression or recurrence, risk of stroke. The target Hemoglobin in the CKD population should generally be decided based on the patient’s quality of life. Kidney Disease Improving Global Outcomes provides a grade 1A recommendation to target a hemoglobin <11 g/dl. In contrast to nondialysis patients, management of anemia in dialysis population is largely managed by protocols, which is appropriate because all patients on dialysis have a universal need for IV iron, either intermittently or continuously, and up to 90% need ESA. Anemia in CKD is usually mild. If the hemoglobin is below 9 g/dl in an otherwise stable stage 3 or stage 4 CKD patient, then other causes of anemia must be investigated. Such as dysproteinemias, gastrointestinal blood loss, drug-induced causes and or hemolysis or vitamin B12 deficiency superimposed on CKD-related anemia.
SEEK AND TREAT IRON DEFICIENCY
Both oral and intravenous formulations of iron are considered safe alternatives for management of anemia in CKD and ESKD. Evidence supporting a more liberal approach to iron supplementation came from the Randomized Trial Comparing Proactive, High dose versus Reactive, Low dose IV iron supplementation in Hemodialysis (PIVOTAL). The PIVOTAL trial, although done in the dialysis populations, warns of about the risks of iron deficiency. The study included 2,141 incidental hemodialysis patients and randomized them to a proactive arm and a reactive arm. The proactive arm repleted the iron stores with 400mg IV iron monthly if ferritin was <700 ng/ml and maintained ferritin around 700 ng/ml. The reactive arm, which was conservative approach, repleted iron stores with 100-200mg IV iron only if the ferritin was <200 ng/ml. Results showed that patients will achieve higher hemoglobin much faster, and this reduces the rate of transfusions significantly by 20%. The other important results showed that maintaining iron stores reduced major adverse cardiovascular events (Hazard ratio 0.85) and reduced death from any cause (Hazard ratio 0.84). This trial also analyzed infections rates and showed there was no difference infection rates. A study by Guedes et al. showed that iron deficiency, even in the absence of anemia, is associated with worse cardiovascular outcomes in nondialysis CKD patients. In the nondialysis CKD population, first and foremost, iron deficiency must be treated. In the NHANES data, 58 to 73% of patients with stages 3-5 CKD had iron deficiency. Many of those patients were not anemic. There is nothing wrong with treating iron deficiency before someone becomes anemic. To be effective, patients must be treated for iron deficiency much earlier to preclude the need for ESA therapy and to maintain higher hemoglobin. The general criteria for making the diagnosis are a transferrin saturation of <20% and a ferritin <200 to 500 ng/ml. Certainly, a ferritin<100 ng/ml is considered iron deficient in the CKD population. Treatment with oral iron works, that is if patients take it regularly. Patients will need to take around 200mg of elemental iron in split doses for a few months. The exception to this is Ferric Citrate, which offers higher oral iron levels per day, which leads to more iron absorption and a rise in ferritin significantly. IV iron is now considered a diagnostic and therapeutic test for CKD related iron deficiency anemia. There are several IV formulations that are available, but there are now three formulations where you can administer 1 gram in a single setting which is considered sufficient to treat iron deficiency.
| Iron Product | Max single dose | Inj./Infuse Time | Doses to equal 1g | Comment |
|---|---|---|---|---|
| Iron Sucrose | 200 mg (300 mg) | 2-5 minutes | 5 doses (3-4 doses) | Hypotension |
| Ferric Gluconate | 125 mg (250 mg) | 10 minutes | 8 doses (4 doses) | Hypotension |
| Iron Dextran | 100 mg (1g off label) | (1 g over 3-4 hr) | 10 (off label, 1 dose) | Anaphylaxis (requires test dose) |
| Ferumoxytol | 510 mg | 15 minutes | 2 doses | Anaphylaxis & hypotension |
| Ferric Carboxymaltose | 1000 mg | 15 minutes | 1 dose | Anaphylaxis, hypertension, flushing |
| Ferric Derisomaltose | 1000 mg | 20 minutes | 1 dose | Anaphylaxis |
ERYTHROPOIESIS STIMULATING AGENTS
ESA products have been considered standard of care for the treatment of anemia in CKD and ESKD. These products are reserved for a hemoglobin less than 10 g/dl. Epoetin alfa and beta were approved by the US Food and Drug Administration (FDA) in 1989. Some years later the first two ESAs (darbepoetin alfa and methoxy polyethylene glycol-epoetin beta) entered clinic practice. All ESA products share a common mechanism of action which is activation of the erythropoietin receptor. ESA products should be used in caution in patients with specific risk factors (cancer, diabetes, stroke, ischemic heart disease) given the risk of myocardial infarction, stroke, venous thromboembolism, thrombosis of vascular access and tumor progression or recurrence. As such, it is important to monitor patients and ensure ESAs are not over-dosed. To initiate ESA therapy in non-dialysis CKD, guidelines recommend to first demonstrate iron sufficiency and a hemoglobin less than 10 g/dl. The actual hemoglobin that triggers ESA use should be based on persistence of anemia, patient benefit from higher hemoglobin level (increase physical functioning, reducing fatigue and transfusions) and patient risk factors from ESA related side effects. Goal is to maintain the hemoglobin between 10 and 11 g/dl. Epoetin can be given subcutaneous weekly or bi-weekly with a dose between 2,000-10,000 units depending on initial hemoglobin and response, as well as patient size.
HYPOXIA INDUCIBLE FACTOR PROLYL HYDROXYLASE INHIBITORS, A NOVEL THERAPY
Hypoxia inducible factor prolyl hydroxylase inhibitors (HIF-PHIs) are an emerging therapy that work by preventing the degradation of Hypoxia Inducible Factor (HIF) to ultimately stimulate the transcription of the erythropoietin gene. These agents thus cause increased levels of endogenous erythropeitin. As mentioned earlier, HIF PHIs may also increase the levels of available iron through increasing intestinal absorption of iron. Recent Phase 3 clinical trials have shown that Roxadustat, Vadadustat, and Daprodustat are noninferior to conventional EPO therapy in terms of achieving and maintaining goal hemoglobin concentrations and cardiovascular safety outcomes. Moreover, a meta-analysis of six randomized control trials found that Roxadustat increased hemoglobin values by 0.52 g/dl compared to epoetin-alfa in dialysis-dependent patients. This same meta-analysis found that Daprodustat decreased hepcidin levels compared to placebo and epoetin-alfa. This finding bolsters the idea that Daprodustat and other HIF PHIs may address the underlying cause of disordered iron homeostasis that is central to the anemia of CKD. While the efficacy of HIF PHIs in the treatment of anemia of CKD is promising, these agents are not as ubiquitous as ESAs due to clinical familiarity with HIF PHIs and a relative lack of long-term safety data for HIF PHIs. Trials have suggested that Daprodustat, Vadadustat, and Roxadustat have noninferior rates of adverse events compared to ESAs in the short-term for the treatment of anemia in dialysis-dependent CKD patients. The types of adverse effects of HIF PHIs are similar to those of ESAs, given that the two share a similar biologic mechanism. However, the longest follow-up period in these trials was 52 weeks. Thus, there remains a concern that HIF PHIs may have unknown long-term adverse effects that are yet to be uncovered. The United States FDA issued regulatory directives to evaluate the long-term safety of Daprodustat and Vadadustat, the two HIF HPIs that have been approved in the United States. Daprodustat and Vadadustat are only approved in the United States for the treatment of anemia in dialysis-dependent CKD patients, as there is more evidence to support their safety in this population compared to non-dialysis CKD patients. In clinical practice, these agents are mostly used for the 10-20% of patients who have anemia refractory to iron and ESA therapy. One rationale for the use of HIF PHIs in ESA hyporesponders is that chronic inflammation is thought to be a cause of ESA hyporesponsiveness, and that HIF PHIs may decrease chronic inflammation through modulating hepcidin and other inflammatory mediators. A study investigated the use of Daprodustat in patients with anemia refractory to ESA and iron treatment and found that 29% percent of participants achieved hemoglobin concentrations within the target range. It should be noted that this study only enrolled 15 participants. Further investigations will clarify the role for HIF PHIs in those with anemia refractory to ESA therapy. One advantage of HIF PHIs over ESAs is that HIF PHIs are oral agents, which may be preferred among those on peritoneal dialysis who cannot receive IV ESA therapy during dialysis. Finally, HIF PHIs are a reasonable option for those with an allergy to ESAs. HIF PHI agents are approved for management of anemia in nondialysis CKD only outside the United States and have not received approval within the United States for this indication due to concerns for overall safety in the nondialysis CKD population. Studies have suggested that HIF PHIs are as effective for the treatment of anemia in nondialysis CKD compared to ESAs. However, several studies have raised concerns that there may be increased safety risks with the use of HIF PHIs in the nondialysis CKD population. An investigation of a pooled analysis between two clinical trials of nondialysis CKD patients found that Vadadustat had a higher risk of major adverse cardiovascular events compared to Darbepoetin Alfa (hazard ratio 1.17). In another study, Daprodustat was associated with an increased risk of heart failure hospitalizations compared to conventional ESA therapy in nondialysis CKD patients (risk ratio 1.46). The same study did not observe an association between heart failure hospitalizations and Daprodustat use in dialysis-dependent patients. As discussed earlier, HIF-PHI agents have all shown to reduce Hepcidin expression indirectly as a result leading to a decrease in hepcidin and ferritin and an increase in transferrin and total iron binding capacity. There are studies ongoing investigating hepcidin antagonists that are in development. Daprodustat and Vadadustat are the only HIF-PHI to receive FDA approval for treatment of CKD-related anemia in patients on dialysis > 4 months. There are some notable treatment considerations that clinicians should be aware of when using these agents. First, their use is contraindicated in patients taking Cytochrome P450 inhibitors. Second, the initial Daprodustat or Vadadustat dose is based on prior ESA dose that the patient was taking, and not Hemoglobin level. Finally, it is common in current clinical practice for the dose to be adjusted every 4 weeks as needed to maintain goal Hemoglobin.
VI. Conclusion
Anemia remains a commonly encountered problem in the nondialysis CKD and ESKD populations. In CKD, anemia and iron dysregulation are prevalent and have a significant adverse outcome. Since the 2012 KDIGO guideline on anemia management, new trials and data have emerged. First, the importance of iron-related disorders should be considered prior to initiating other therapies. Secondly, treatment of anemia in CKD is dependent on both ESA and iron repletion. If hemoglobin levels do not respond after therapy with ESA, investigation for bleeding or other causes of anemia should be considered, since higher doses of ESA are associated with inflammation and higher risk of major adverse cardiovascular event. Future research should explore the role of combination therapies which target different pathogenetic mechanisms underlying the development of CKD anemia. In addition, future research should take advantage of agents or treatment that are being studied in other clinical settings.
Conflicts Of Interest:
The author(s) have no conflicts of interest to disclose.
Funding Statement:
This study was not supported by any sponsor or funder.
Works Cited
- Kidney Disease Improving Global Outcomes (KDIGO). KDIGO Clinical Practice Guideline for Anemia in Chronic Kidney Disease. Kidney Int Suppl. 2012;2(4).
- Ryu SR, Park SK, Jung JY, et al. The Prevalence and Management of Anemia in Chronic Kidney Disease Patients: Result from the KoreaN Cohort Study for Outcomes in Patients With Chronic Kidney Disease (KNOW-CKD). J Korean Med Sci. 2017;32(2):249-256. doi:10.3346/jkms.2017.32.2.249
- Stauffer ME, Fan T. Prevalence of Anemia in Chronic Kidney Disease in the United States. PLOS ONE. 2014;9(1):e84943. doi:10.1371/journal.pone.0084943
- Babitt JL, Lin HY. Mechanisms of Anemia in CKD. J Am Soc Nephrol JASN. 2012;23(10):1631-1634. doi:10.1681/ASN.2011111078
- Nutritional anaemias. Report of a WHO scientific group. World Health Organ Tech Rep Ser. 1968;405:5-37.
- Chisholm H. Encyclopædia Britannic. In: Christison, Sir Robert. Vol 6. 11th ed. ; 1911:292-293.
- On Granular Degeneration of the Kidneys, and Its Connection with Dropsy, Inflammation, and Other Diseases. Edinb Med Surg J. 1840;54(144):234-244.
- Foley RN, Parfrey PS, Harnett JD, Kent GM, Murray DC, Barre PE. The impact of anemia on cardiomyopathy, morbidity, and and mortality in end-stage renal disease. Am J Kidney Dis Off J Natl Kidney Found. 1996;28(1):53-61. doi:10.1016/s0272-6386(96)90130-4
- Liang R, Ghaffari S. Advances in understanding the mechanisms of erythropoiesis in homeostasis and disease. Br J Haematol. 2016;174(5):661-673. doi:10.1111/bjh.14194
- Babitt JL, Eisenga MF, Haase VH, et al. Controversies in optimal anemia management: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int. 2021;99(6):1280-1295. doi:10.1016/j.kint.2021.03.020
- Jadoul M, Aoun M, Masimango Imani M. The major global burden of chronic kidney disease. Lancet Glob Health. 2024;12(3):e342-e343. doi:10.1016/S2214-109X(24)00050-0
- United States Renal Data System. 2023 USRDS Annual Data Report: Chronic Kidney Disease. Published online 2023.
- Nissenson AR, Wade S, Goodnough T, Knight K, Dubois RW. Economic burden of anemia in an insured population. J Manag Care Pharm JMCP. 2005;11(7):565-574. doi:10.18553/jmcp.2005.11.7.565
- Kovesdy CP, Davis JR, Duling I, Little DJ. Prevalence of anaemia in adults with chronic kidney disease in a representative sample of the United States population: analysis of the 1999-2018 National Health and Nutrition Examination Survey. Clin Kidney J. 2023;16(2):303-311. doi:10.1093/ckj/sfac240
- Evans M, Bower H, Cockburn E, Jacobson SH, Barany P, Carrero JJ. Contemporary management of anaemia, erythropoietin resistance and cardiovascular risk in patients with advanced chronic kidney disease: a nationwide analysis. Clin Kidney J. 2020;13(5):821-827. doi:10.1093/ckj/sfaa054
- Jacobson LO, Goldwasser E, Fried W, Plzak L. Role of the kidney in erythropoiesis. Nature. 1957;179(4560):633-634. doi:10.1038/179633a0
- Fried W. Erythropoietin and the kidney. Nephron. 1975;15(3-5):327-349. doi:10.1159/000180519
- Shih HM, Wu CJ, Lin SL. Physiology and pathophysiology of renal erythropoietin-producing cells. J Formos Med Assoc Taiwan Yi Zhi. 2018;117(11):955-963. doi:10.1016/j.jfma.2018.03.017
- Vuori K, Pihlajaniemi T, Marttila M, Kivirikko KI. Characterization of the human prolyl 4-hydroxylase tetramer and its multifunctional protein disulfide-isomerase subunit synthesized in a baculovirus expression system. Proc Natl Acad Sci U S A. 1992;89(16):7467-7470. doi:10.1073/pnas.89.16.7467
- McGonigle RJ, Wallin JD, Shadduck RK, Fisher JW. Erythropoietin deficiency and inhibition of erythropoiesis in renal insufficiency. Kidney Int. 1984;25(2):437-444. doi:10.1038/ki.1984.36
- Rege AB, Brookins J, Fisher JW. A radioimmunoassay for erythropoietin: serum levels in normal human subjects and patients with hemopoietic disorders. J Lab Clin Med. 1982;100(6):829-843.
- KDOQI, National Kidney Foundation. KDOQI Clinical Practice Guidelines and Clinical Practice Recommendations for Anemia in Chronic Kidney Disease. Am J Kidney Dis Off J Natl Kidney Found. 2006;47(5 Suppl 3):S11-145. doi:10.1053/j.ajkd.2006.03.010
- Gafter-Gvili A, Schechter A, Rozen-Zvi B. Iron Deficiency Anemia in Chronic Kidney Disease. Acta Haematol. 2019;142(1):44-50. doi:10.1159/000496492
- Tsukamoto T, Matsubara T, Akashi Y, Kondo M, Yanagita M. Annual Iron Loss Associated with Hemodialysis. Am J Nephrol. 2016;43(1):32-38. doi:10.1159/000444335
- Shepshelovich D, Rozen-Zvi B, Avni T, Gafter U, Gafter-Gvili A. Intravenous Versus Oral Iron Supplementation for the Treatment of Anemia in CKD: An Updated Systematic Review and Meta-analysis. Am J Kidney Dis Off J Natl Kidney Found. 2016;68(5):677-690. doi:10.1053/j.ajkd.2016.04.018
- Leung AKC, Lam JM, Wong AHC, Hon KL, Li X. Iron Deficiency Anemia: An Updated Review. Curr Pediatr Rev. 2024;20(3):339-356. doi:10.2174/1573396320666230727102042
- Santos-Silva A, Ribeiro S, Reis F, Belo L. Hepcidin in chronic kidney disease anemia. Vitam Horm. 2019;110:243-264. doi:10.1016/bs.vh.2019.01.012
- Camaschella C, Nai A, Silvestri L. Iron metabolism and iron disorders revisited in the hepcidin era. Haematologica. 2020;105(2):260-272. doi:10.3324/haematol.2019.232124
- Madu AJ, Ughasoro MD. Anaemia of Chronic Disease: An In-Depth Review. Med Princ Pract. 2017;26(1):1-9. doi:10.1159/000452104
- J Z, B Y, Hj W, et al. Hepcidin–a potential novel biomarker for iron status in chronic kidney disease. Clin J Am Soc Nephrol CJASN. 2009;4(6). doi:10.2215/CJN.05931108
- Hanudel MR, Rappaport M, Gabayan V, et al. Increased serum hepcidin contributes to the anemia of chronic kidney disease in a murine model. Haematologica. 2017;102(3):e85-e88. doi:10.3324/haematol.2016.150433
- Guedes M, Muenz DG, Zee J, et al. Serum Biomarkers of Iron Stores Are Associated with Increased Risk of All-Cause Mortality and Cardiovascular Events in Nondialysis CKD Patients, with or without Anemia. J Am Soc Nephrol. 2021;32(8):2020-2030. doi:10.1681/ASN.2020101531
- Astor BC, Muntner P, Levin A, Eustace JA, Coresh J. Association of kidney function with anemia: the Third National Health and Nutrition Examination Survey (1988-1994). Arch Intern Med. 2002;162(12):1401-1408. doi:10.1001/archinte.162.12.1401
- Válka J, Čermák J. Differential diagnosis of anemia. Vnitr Lek. 2018;64(5):468-475.
- Stancu S, Bârsan L, Stanciu A, Mircescu G. Can the Response to Iron Therapy Be Predicted in Anemic Nondialysis Patients with Chronic Kidney Disease? Clin J Am Soc Nephrol. 2010;5(3):409-416. doi:10.2215/CJN.04280609
- Ratcliffe LEK, Thomas W, Glen J, et al. Diagnosis and Management of Iron Deficiency in CKD: A Summary of the NICE Guideline Recommendations and Their Rationale. Am J Kidney Dis Off J Natl Kidney Found. 2016;67(4):548-558. doi:10.1053/j.ajkd.2015.11.012
- Macdougall IC. What is the most appropriate strategy to monitor functional iron deficiency in the dialysed patient on rhEPO therapy? Merits of percentage hypochromic red cells as a marker of functional iron deficiency. Nephrol Dial Transplant Off Publ Eur Dial Transpl Assoc – Eur Ren Assoc. 1998;13(4):847-849. doi:10.1093/ndt/13.4.847
- Macdougall IC, White C, Anker SD, et al. Intravenous Iron in Patients Undergoing Maintenance Hemodialysis. N Engl J Med. 2019;380(5):447-458. doi:10.1056/NEJMoa1810742
- Hu X, Xie J, Chen N. Hypoxia-Inducible Factor-Proline Hydroxylase Inhibitor in the Treatment of Renal Anemia. Kidney Dis Basel Switz. 2021;7(1):1-9. doi:10.1159/000510587
- Singh AK, Carroll K, Perkovic V, et al. Daprodustat for the Treatment of Anemia in Patients Undergoing Dialysis. N Engl J Med. 2021;385(25):2325-2335. doi:10.1056/NEJMoa2113379
- Coyne DW, Singh AK, Lopes RD, et al. Three Times Weekly Dosing of Daprodustat versus Conventional Epoetin for Treatment of Anemia in Hemodialysis Patients: ASCEND-TD: A Phase 3 Randomized, Double-Blind, Noninferiority Trial. Clin J Am Soc Nephrol CJASN. 2022;17(9):1325-1336. doi:10.2215/CJN.00550122
- Akizawa T, Iwasaki M, Yamaguchi Y, Majikawa Y, Reusch M. Phase 3, Randomized, Double-Blind, Active-Comparator (Darbepoetin Alfa) Study of Oral Roxadustat in CKD Patients with Anemia on Hemodialysis in Japan. J Am Soc Nephrol JASN. 2020;31(7):1628-1639. doi:10.1681/ASN.2019060623
- Liu J, Zhang A, Hayden JC, et al. Roxadustat (FG-4592) treatment for anemia in dialysis-dependent (DD) and not dialysis-dependent (NDD) chronic kidney disease patients: A systematic review and meta-analysis. Pharmacol Res. 2020;155:104747. doi:10.1016/j.phrs.2020.104747
- Eckardt KU, Agarwal R, Aswad A, et al. Safety and Efficacy of Vadadustat for Anemia in Patients Undergoing Dialysis. N Engl J Med. 2021;384(17):1601-1612. doi:10.1056/NEJMoa2025956
- KDOQI, National Kidney Foundation. KDOQI Clinical Practice Guidelines and Clinical Practice Recommendations for Anemia in Chronic Kidney Disease. Am J Kidney Dis Off J Natl Kidney Found. 2006;47(5 Suppl 3):S11-145. doi:10.1053/j.ajkd.2006.03.010
- Cizman B, Sykes AP, Paul G, Zeig S, Cobitz AR. An Exploratory Study of Daprodustat in Erythropoietin-Hyporesponsive Subjects. Kidney Int Rep. 2018;3(4):841-850. doi:10.1016/j.ekir.2018.02.009
- Chen P, Nirula A, Heller B, et al. SARS-CoV-2 Neutralizing Antibody LY-CoV555 in Outpatients with Covid-19. N Engl J Med. Published online October 28, 2020. doi:10.1056/NEJMoa2029849
- Shutov E, Sułowicz W, Esposito C, et al. Roxadustat for the treatment of anemia in chronic kidney disease patients not on dialysis: a Phase 3, randomized, double-blind, placebo-controlled study (ALPS). Nephrol Dial Transplant Off Publ Eur Dial Transpl Assoc – Eur Ren Assoc. 2021;36(9):1629-1639. doi:10.1093/ndt/gfab057
- Fishbane S, El-Shahawy MA, Pecoits-Filho R, et al. Roxadustat for Treating Anemia in Patients with CKD Not on Dialysis: Results from a Randomized Phase 3 Study. J Am Soc Nephrol JASN. 2021;32(3):737-755. doi:10.1681/ASN.2020081150
- Chertow GM, Pergola PE, Farag YMK, et al. Vadadustat in Patients with Anemia and Non-Dialysis-Dependent CKD. N Engl J Med. 2021;384(17):1589-1600. doi:10.1056/NEJMoa2035938
- Cunningham JW, Claggett BL, Lopes RD, et al. Daprodustat and Heart Failure in CKD. J Am Soc Nephrol JASN. 2024;35(5):607-617. doi:10.1681/ASN.0000000000000321
- Kanbay M, Tapoi L, Ureche C, et al. Effect of sodium-glucose cotransporter 2 inhibitors on hemoglobin and hematocrit levels in type 2 diabetes: a systematic review and meta-analysis. Int Urol Nephrol. 2022;54(4):827-841. doi:10.1007/s11255-021-02943-2
- The EMPA-KIDNEY Collaborative Group. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2023;388(2):117-127. doi:10.1056/NEJMoa2204233

