Breast Cancer Disparities in Brazil: Innovative Solutions
Addressing Breast Cancer Disparities in Brazil: A Roadmap for Innovative Strategies
Heloisa Resende MD, PhD¹, Vinicius de Queiroz Aguiar¹, Nataline Freitas de Azevedo Santos¹, Rafael Angelo Pinto de Souza¹, Bernardo Caetano Novaes¹, Frederico da Rocha Mello¹, Filippe Rocha Mello¹, Caio Miranda Oliveira¹, Carolina da Matta Pincowsky¹, André Mattar MD, PhD²
- Centro Universitário UniFOA, Volta Redonda, RJ, Brazil
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Resende, H., et al., 2024. Addressing Breast Cancer Disparities in Brazil: A Roadmap for Innovative Strategies. Medical Research Archives. doi:10.18103/mra.v12i5.2185
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i10.5846
ISSN 2375-1924
ABSTRACT
Breast cancer (BC) is the most common malignant disease among women globally, with an estimated 2.3 million new cases a year. It is a major burden for Latin America where the incidence rates have increased most rapidly with mortality rates steadily high in the last decade. In Brazil, it has been expected 723.600 new cases a year in the triennial period from 2023 to 2025, accounting the most common neoplasm in women. Although mortality rates have recently decreased in high-income countries (HICs), driven by the organized mammography screening, introduction of new medications, and advancements in treatment, the same cannot be said for low- and middle-income countries (LMICs). This article aims to present a roadmap for innovative strategies that could help in addressing breast cancer patient’s journey.
Keywords
Breast cancer, disparities, Brazil, innovative strategies, healthcare access
Introduction
Breast cancer (BC) is the most common cancer globally, representing a challenging health care matter worldwide, being expected by 2040 more than 3 million of new cases a year¹. It is a major burden for America Latina where the incidence rates have increased most rapidly with mortality rates steadily high in the last decades². In Brazil, it has been expected 73.600 new cases a year in the triennial period from 2023 to 2025³, accounting the most common neoplasm in women⁴. Although mortality rates have recently decreased in high-income countries (HICs), driven by the organized mammography screening, introduction of new medications, advances in tumor biology, and the application of this knowledge to specific molecular subtypes⁵, these improvements have yet to be reflected in better outcomes also for women in low- and middle-income countries (LMICs)⁶. Regardless of the incidence rates are lower in LMICs, than HICs (incidence rate is 66.5 in 100.000 in Brazil, 95.9 in USA and 94.0 in UK), the mortality rates are close (19.7 per 100.000 in Brazil v 18.7 in US v 14.0 in UK).
In Brazil, only about one-quarter of the population can afford private health insurance⁷, highlighting the government’s significant responsibility to provide access to cancer care. Good quality cancer care in the public health system will minimize disparities and utmost it will bring improvements in overall survival mitigating differences between private and public systems. Despite the fact, the Brazil has made progresses in public health, notably with creation of the public health system with universal coverage (Sistema Único de Saúde, SUS), mainly in notifiable diseases, the cancer care is yet to be upgraded⁸. “In the past decade, the Brazilian government established the National Cancer Control Policy”; however, several areas still require attention. The overwhelming demand on the three levels of care (primary, secondary, and tertiary), due to the high percentage of the population relying exclusively on the SUS, combined with a rapid epidemiological transition that results in a triple disease burden, fragmented care, and chronic underfunding of the SUS over the years, has led to delayed diagnosis and treatment initiation in breast cancer.
Within cancer units, the lack of strict oversight regarding which clinical procedures are actually being offered to patients has led to a scenario where three different levels of hospitals coexist within the SUS. Some hospitals provide only the minimum services required by the government, others offer less than the minimum, and some exceed expectations¹⁰. The scarcity of clinical research protocols, along with the concentration of the few available protocols in capital cities, has also hindered the participation of women with breast cancer in clinical trials. This significantly delays the development of academic research addressing critical local issues, which is essential for improving care.¹¹.
All these gaps have arguably contributed to poor outcomes in breast cancer patients in Brazil, which need for attention of government and other stakeholders. In this narrative review, we analyzed the timeline of main laws and ordinances by Brazilian government to assist breast cancer patients since the SUS creation, parallelly to results that have been gotten regarding screening coverage, staging distribution, framework to new treatments incorporation. Also, we have highlighted key access barriers and have proposed potential actions that could be implemented, which might improve the outcomes of breast cancer.
Methods
The current study is a narrative review, where authors applied descriptive approach analyzing laws and ordinances launched by Brazilian government, since the creation of its public health system, SUS, in 1990, that represent landmarks in the public oncological assistance in the country (Figure 1). Also, published data regarding screening breast cancer coverage, staging distribution at diagnosis and barriers to access to new medicines were analyzed. The authors drew the Brazilian public health system scenario, pointing out the need for timely access to diagnosis and treatment, need for widen access to new medicines, palliative care and telehealth tools. Following these steps, the authors proposed innovative strategies that could be implemented aiming to improve outcomes during breast cancer patient’s journey.
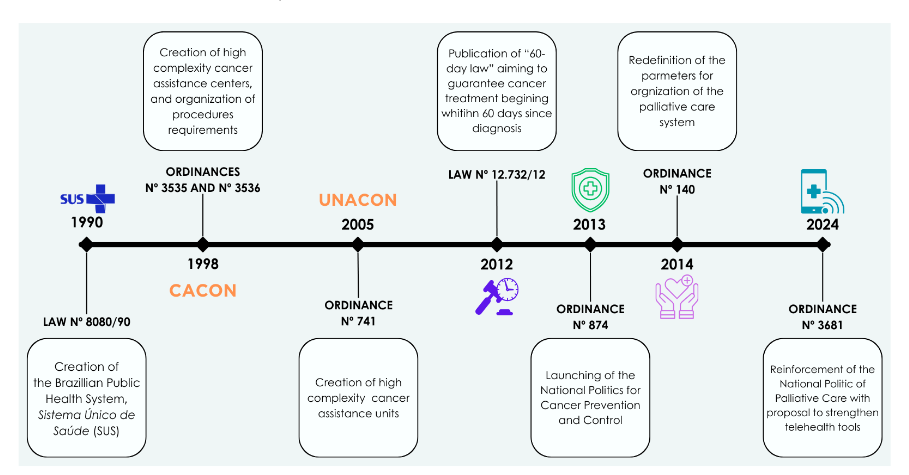
Fig. 1 – Landmarks of Brazilian Cancer Care within the Public Health System, Sistema Único de Saúde (SUS).
Timely access to cancer diagnosis
In Brazil, there is a high percentage of stage III breast cancer diagnosis, which is an immediate task for stakeholders to change. Advanced stage is correlated with higher risk of disease relapse and worse prognosis than early stages¹². Screening strategies have remained one of the necessary conditions to promote early detection, while most of breast cancers are attributed to non-modifiable risk factor and primary prevention is limited to lifestyle changes including exercise and healthy meals or avoidance of postmenopausal hormone replacement therapy¹³.The rate of breast cancer mortality in US, has decreased in the last four decades. Reasons that have promoted this incremental survival are mammography screening (range 25%), advances in treatment of patient’s stage II and III (range 47%) and advances in metastatic disease treatment (range 27%)⁵.Mammography screening in Brazil, has been widened, reaching coverage rates range to 65,3% in 2022³, although it keeps being opportunistic, and has been documented a major number of screening mammography during Pink October campaigns¹⁴. Also, these policies between 2001 and 2014 do not seem to have had any effect on the stage of breast cancer at diagnosis (figure 2)¹⁵. The AMAZONA trial, a big retrospective and multicentric trial that included patients from 2008 and 2009 has shown that 23,2% of these patients were stage III¹⁶, and prospective AMAZONA III, that recruited patients from 2016 to 2017 described 27,0% of patients stage III¹¹⁶,¹⁷. On one hand, early detection through screening should be encouraged, as its benefits are well-documented¹⁸–²⁰.
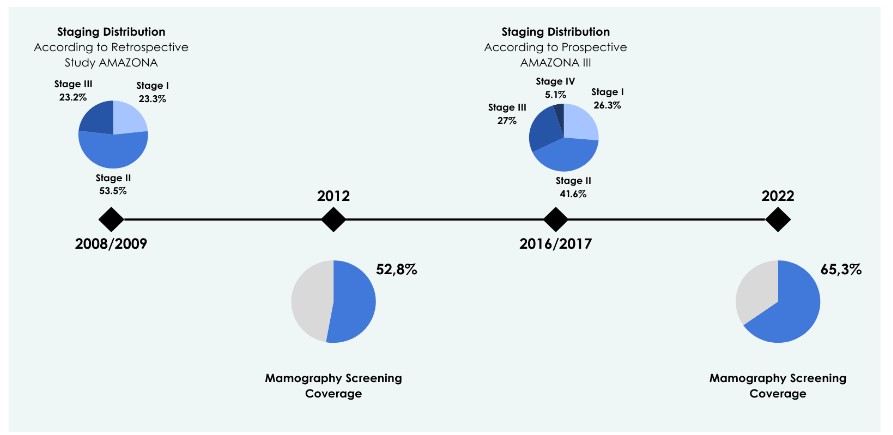
Fig. 2 – Relationship between mammography screening coverage and breast cancer staging.
The World Health Organization (WHO) has outlined two distinct strategies for promoting early breast cancer detection: screening (for asymptomatic patients) and early diagnosis (recognition of symptomatic cancer at an early stage)¹⁵. Additionally, the WHO recommends that countries with limited resources prioritize early diagnosis in symptomatic patients over mammographic screening for asymptomatic patients²¹,²². For instance, in Brazil, data from a nationwide hospital-based cancer registry estimated that 2,500 breast cancer deaths could have been prevented through effective mammography screening for women aged 50–69. If as many as 80%, and as low as 50%, of all women diagnosed with stage III or IV disease in the previous five years had been downstaged to stage II, an additional 8,000 deaths could have been prevented in 2012. This underscores that screening mammography may not be the optimal approach in countries where women are frequently diagnosed at late stages¹⁵.
Brazil struggles to provide timely access for symptomatic patients, with long waiting times reported at various stages of care: a median of 60 days from self-detected breast abnormalities to mammography and ultrasound, 41 days from mammography to biopsy, and 97 days from biopsy to the start of treatment²³. The reasons cited for these delays include fragmented care²⁴ and a lack of qualified healthcare professionals in primary care units to properly identify and facilitate timely assistance for these women. This chaotic scenario demands a commitment from the government at all three levels (municipal, state, and federal) and from healthcare professionals to integrate services within fewer facilities. It also requires the implementation of training programs for multidisciplinary teams, as well as the possibility of offering initial procedures such as mammography, ultrasound, and biopsy at primary care units. These strategies aim to shorten waiting times, ultimately leading to improved outcomes²⁵.
Timely access to cancer treatment
A significant issue in Brazil and other LMICs is the prolonged waiting time between cancer diagnosis and the start of treatment. The burden of new breast cancer cases, other malignancies, and non-communicable diseases represents a triple disease load, reflecting the rapid epidemiological transition Brazil has undergone. This has contributed to the current scenario, where waiting times from diagnosis to treatment exceed acceptable limits. Although global real-world data is scarce, some researchers have published studies highlighting extended waiting time intervals, further underscoring the heterogeneity of
outcomes. A subgroup analysis of AMAZONA III prospective study, described median of 56 days since diagnosis to treatment beginning in a public cohort²⁶, and Ferreira et al described that 80% of patients in a public health system have started their treatments within 60 days²⁷. However, there is not a systematically documentation of these times, especially regarding each region. Resende et al, for example, described a median of 97 days in a public cohort in Rio de Janeiro state²³. These discrepancies highlighted the heterogeneity of access related to the continental dimensions, and large socio-demographic variations, outstanding the existence of huge disparities within public health system in Brazil.
In 2012, the Brazilian government established the 60-day law (figure 1)²⁸, aimed at ensuring timely initiation of cancer treatment. However, despite the law, delays have continued to be frequently reported. Although the SUS was created in 1988, it has faced chronic underfunding⁹, with a relatively low allocation to the public health system²⁹. To provide timely treatment, it is necessary to strengthen the National Cancer Control Policy, which would undoubtedly require increased investment.
Access to new medicines
Significant progress has transformed the field of BC, leading to higher rates of early diagnosis, increased detection of curable cases, and longer overall survival in metastatic breast cancer (mBC)³⁰. However, these advancements have also been accompanied by rising healthcare costs, largely driven by (but not limited to) the high price of new medications³¹. This presents a challenge for both high-income countries (HICs) and low- and middle-income countries (LMICs), which are struggling to manage these escalating expenses³². It is actually a challenge for both, HICs and LMICs, which have struggled to incorporate these medications and maintain the sustainability of the systems. It was with perplexity that the world received in July 2024 the news regarding Trastuzumab deruxtecan (Enhertu®) not be incorporated in HER2 low mBC treatment by NICE (National Institute for Health and Care Excellence) due to high costs. The clinical data clearly demonstrate the benefits of the medication³³; however, in a public healthcare system with broad access, the cost becomes prohibitive. As a result, NICE deemed that pharmaceutical companies were unwilling to offer fair pricing for the medication and announced that it is not recommended in this context³⁴. As the costs have alarmingly increased in oncological care, a broader assessment is essential, considering value proposition. Some strategies have been considered to guide the new incorporations around the world. Ethereal and vague value definition can become obscure the theme and avoid a clear debate, so postponing solutions. Value based cancer care is defined by Leonard Saltz as a ratio of clinical benefit to toxicity, including financial toxicity³⁵. Deeping this concept, value proposition can be generated based on how the product will modify the system, how much benefit it will bring which ultimately depends on the treatment benefits already available³⁶). For example, in an adjuvant receptor hormonal positive, HER2 negative, breast cancer scenario, where endocrine therapy is the essential strategy³⁷–³⁹ to add cyclin inhibitor for high risk patients has improved overall survival⁴⁰–⁴³. Now we have two cyclin inhibitors available to apply in this scenario, Abemaciclib and Ribociclib. However, there is a difference between their costs, considering current prices in Brazil⁴⁴, and the length of treatment, 2 years for Abemaciclib with Number Needed to Treat (NNT) of 13, and 3 years for Ribociclib with NNT of 30⁴⁵,⁴⁶. This difference is favorable to abemaciclib. So, this kind of analysis need to be done to help us to choose the best and cost-effective medication.
Guidelines as ESMO has proposed a scale to evaluate the real magnitude of benefit provided by a new treatment to prioritizing funding particularly in countries with scarce resources⁴⁷. Although it is not perfect, it a framework that help us in the decision-making process. Other issues are being considered for inclusion of new drugs that bring benefit for a large proportion of the population, as cost-effectiveness alone, budget impact, or both. In countries with limited resources, budget impact
remains as a priority. So, what would be a worthy strategy for Brazilian government to stablish a guideline for new medicines incorporations? A panel of experts has recommended strengthening of national pricing strategies as well the price negotiation between government and pharmaceutical companies, as crucial points to broaden affordability of new drugs⁵⁶. Of note, these strategies will bring unique difficulties in each scenario considering the number of negotiations, discounts and price volume, which makes the transparency across all these processes essential⁴⁸. Aligned to these strategies, the judgment of cost effectiveness as offered by current guidelines will be useful for government to establish fair rules of new medicines incorporation.
Widen access to supportive cares in breast cancer journey
Recent information from the World Health Organization (WHO) indicates that although 70% of countries have established palliative care guidelines, only 62% report screening programs, at the same time, 40% report important management and treatment access restrictions and less than half have palliative care plans⁴⁹. In Brazil, the public control cancer politics have begun in 1990 with the law 8080 which stablishes public health system, SUS⁵⁰, whiting its principles, health is a citizen’s high and one state’s obligation.
In the following years, several ordinances directed to the cancer care have been published, which have become possible, organizing the public cancer care in public and private hospitals accredited by government to offer such assistance, the units (UNACONs) or centers (CACONs) of high complexity in oncology (figure 1)⁵¹,⁵². However, these hospitals have as main goal the offering cancer treatment, which is reinforced by remuneration model, paying for treatment procedures as chemo, endocrine therapy, radiotherapy and surgery⁵¹. Arguably, this model, where hospitals and health professionals already have been overwhelmed with the burden of new oncological cases, has resulted in a lack of integral assistance. Brazilian government has published other ordinances aiming to complete these gaps, being considered a landmark the ordinance 874 in May 2013, which stablishes the National Politic of Prevention and Cancer Control⁵³. Complementing it, two other ordinances came into country, in 2014 and 2024 both reinforcing the obligation of provider services in oncology, to offer palliative care⁵³. According to the palliative care Atlas, in 2020, Brazil is a country with actions in the area, multiple sources of funding and able to provide opioids access and few educational initiatives⁵⁴. Actually, educational actions by government, have been occasional, being most of the courses offered by private sector⁵⁵,⁵⁶.
Despite these ordinances and governmental actions, only 66% of Brazilian oncologists that have assisted breast cancer patients, answered they have palliative team in their hospitals⁵⁷, which is in line with WHO data. The scarcity of supportive teams, including palliative care is crucial issue in oncology, but specially in metastatic disease, due to increasing incidence and prevalence. Many countries do not know number of patients living with metastatic disease, because their registries do not record relapses, however governments are expected to provide access to treatments and supportive strategies that alleviate suffering for all, ensuring that no one is forgotten or left behind⁵⁸.
To offer integral assistance is particularly challenging in models where there is no specific remuneration for supportive procedures and teams. These difficulties are gotten worse due to a scarcity of qualified human resources. To mitigating the lack of palliative assistance in UNACONS and CACONS, it would be worthy to offer continuously training programs funding by government, which certainly would broaden availability of this service. Currently, these programs have been offered predominantly by private institutions, which brings a need for health professionals paying for the training, hindering the palliative care team’s formation in the public system. Also to adopt a framework to deliver telehealth palliative care might will optimize resources and to provide access for a major number of patients⁵⁹.
Integration with digital tools
Telehealth is a digital tool that has been increasingly integrated to the in-person assistance in medical care⁶⁰. It is a helpful strategy to deal with growing number of new cases of cancer and survivorship⁶¹. Its utilization has permitted to assist major number of patients, improving quality of care. Specially during and after COVID pandemic its use has been adopted by most of medical services providers⁶².
The Brazilian government published in May 2024 an ordinance 3.681 (Programa Nacional de Cuidado Paliativo, PNCP) which reinforces the need for palliative care services with broad coverage and preconizes the telehealth use in the public health system⁶³. The effects of this newest ordinance yet have no measured. Certainly, the implantation of these tools in public health system will bring more equity, by becoming possible the navigation of patients, for instance in the management of scheduling treatments, registration and orientation about acute side effects of the chemotherapy and follow up of patients after the end of chemotherapy (figure 3). Also, during endocrine therapy phase, aiming to provide support in the chronic side effects. The long length of endocrine therapy in breast cancer, frequently 10 years, has been associated with low rates of adherence, with some records of 60% of patients have discontinued or have adherence below 80%, which is clearly related to the profile of side effects⁶⁴. The success of digital platforms, in this scenario, has been documented around the world⁶⁵. In Brazil, one notably example is THUMMI app, an applicative developed by Brazilian researchers including oncologists that is now operating in few Brazilian hospitals, including two public hospitals⁶⁶. Its utilization up to now, has demonstrated major treatment adherence and better management of side effects. The ideal scenario would be the large utilization of this kind of digital instrument, composition of teams with navigator nurses, as preconized by ordinance, that could broaden access to the health care system for patients in the public system.
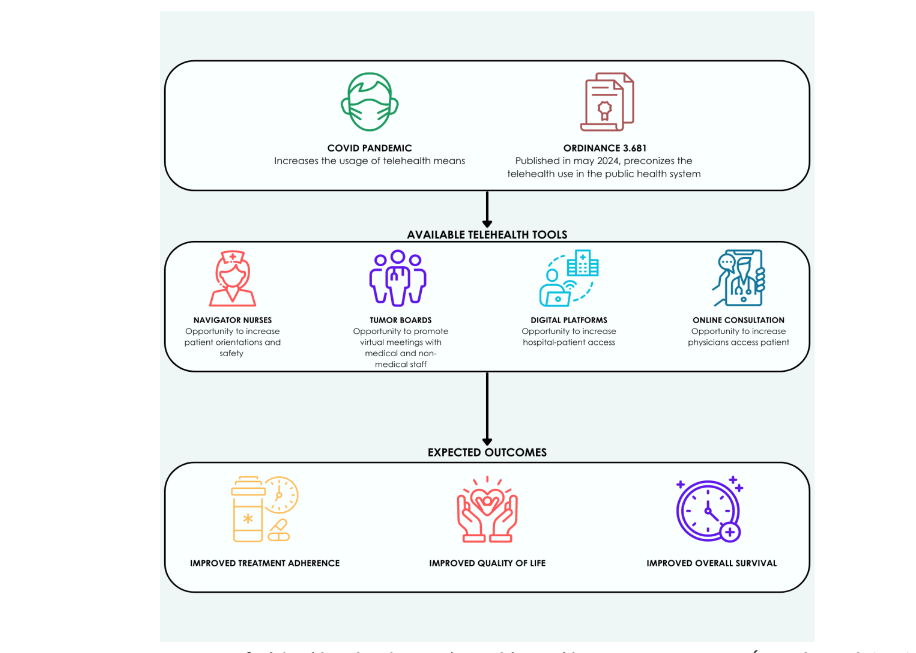
Fig. 3 – Increasing usage of telehealth within the Brazilian Public Healthcare System, Sistema Único de Saúde (SUS).
Another need for broadening high quality assistance access is continuous commitment of the whole team during patient’s treatment and making decision. Multidisciplinary teams (MDTs) or tumor boards have been implemented in the medical teams for many years, and numerous studies have demonstrated its contribution in improving patient management⁶⁷. However, MDTs has happened more frequently in academic centers⁶⁸. In Brazil, it has been frequently underutilized and misunderstood, mainly in the public health system⁶⁹. There are several points at the time, responsible for this low reproducibility of MDTs, among those the lack of protocol previously stablished, low representativity of some medical specialties and non-medical staff ⁷⁰. Nowadays one recent difficult is to gather the team at a same hospital at a same time, due to new models of work, where oncologists and other members of multidisciplinary teams have worked in more than one institution. The proposition is the use of digital platforms, online meetings, which would offer the possibility to gather them virtually, even if they are not in that hospital in that day. It would be a first step toward construct a culture of participation in MDTs. After this first step, it would highlight other challenges and certainly would help to improve institutional process and quality of management care.
Conclusion
Breast cancer care in Brazil has evolved over recent decades, particularly since the creation of the SUS. However, mortality rates have remained consistently high, in contrast to HICs, where rates have declined during the same period. Addressing barriers such as reducing waiting times from self-detected abnormalities to diagnosis, and from diagnosis to treatment initiation, is an urgent priority. A possible strategy would be to centralize care in one or two facilities to reduce bureaucracy. Additionally, providing medical training for primary care physicians and multidisciplinary teams to ensure timely diagnostic steps is essential. Equally important is the development of sustainable models for incorporating new treatments, including medications, within a value-based care framework, alongside training programs for supportive teams. All of these efforts must be recognized and prioritized by the government, healthcare providers, and medical societies to move forward into a new era of breast cancer care in Brazil.
Conflict of Interest:
None
Acknowledgements:
None
References
1. Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3): 209-249. doi:10.3322/caac.21660.
2. Torres-Román JS, Ybaseta-Medina J, Loli-Guevara S, et al. Disparities in breast cancer mortality among Latin American women: trends and predictions for 2030. BMC Public Health. 2023;23(1):1449. doi:10.1186/s12889-023-16328-w.
3. Santos MO, Lima FCS, Martins LFL, et al. Estimativa de Incidência de Câncer no Brasil, 2023-2025. Revista Brasileira de Cancerologia. 2023;69(1): p.e-213700. doi.org/10.32635/2176-9745.RBC.2023v69n1.3700
4. Age-Standardized Rate (World) per 100 000, Incidence and Mortality, Both sexes, in 2022, Breast. Global Cancer Observatory. 2022. Accessed July 28, 2024. https://gco.iarc.fr/today/en/dataviz/bars?types=0_1.
5. Caswell-Jin JL, Sun LP, Munoz D, et al. Analysis of Breast Cancer Mortality in the US-1975 to 2019. JAMA. 2024;331(3):233-241.
doi:10.1001/jama.2023.25881.
6. Barrios C, Freitas-Junior R, Martins S, Bines J, Estevez-Diz MDP, Caleffi M. Challenge of Incorporating New Drugs for Breast Cancer in Brazil: A Proposed Framework for Improving Access to Innovative Therapies. JCO Glob Oncol. 2021;7: 474-485. doi:10.1200/GO.20.00566.
7. Agência Nacional de Saúde ANS: General data beneficiaries of private health plans, by assistance coverage (Brazil: 2014-2024). GOV.BR. Published September 27, 2024. Accessed September 27, 2024. https://www.gov.br/ans/pt-br/acesso-a-informacao/perfil-do-setor/dados-gerais
8. Castro MC, Massuda A, Almeida G, et al. Brazil’s unified health system: the first 30 years and prospects for the future. Lancet. 2019;394(1019 5):345-356. doi:10.1016/S0140-6736(19)31243-7.
9. Padilha ARS. Ordinance 874 May 2013. Ministério da Saúde. Published May 16, 2013.
Accessed July 27, 2024. https://bvsms.saude.gov.br/bvs/saudelegis/gm/2013/prt0874_16_05_2013.html
10. Proposta da SBOC para garantir o efetivo acesso a antineoplásicos incorporados ao SUS. Sociedade Brasileira de Oncologia Clínica. Published May 25, 2023. Accessed July 27, 2024. https://sboc.org.br/images/pdf/075-Proposta-SBOC-Iniquidade-Pos-Conferencia.pdf
11. Resende H, Arai RJ, Barrios CH, et al. Improving access to cancer clinical research in Brazil: recent advances and new opportunities. Expert opinions from the 4th CURA meeting, São Paulo, 2023. Ecancermedicalscience. 2024;18:1698. doi:10.3332/ecancer.2024.1698.
12. Richards MA, Westcombe AM, Love SB, Littlejohns P, Ramirez AJ. Influence of delay on survival in patients with breast cancer: a systematic review. Lancet. 1999;353(9159):1119-1126. doi:10.1016/s0140-6736(99)02143-1.
13. Rivera-Franco MM, Leon-Rodriguez E. Delays in Breast Cancer Detection and Treatment in Developing Countries. Breast Cancer (Auckl). 2018;12:1178223417752677. doi:10.1177/1178223417752677.
14. Antonini M, Pinheiro DJPDC, Salerno GRF, et al. Does Pink October really impact breast cancer screening?. Public Health Pract (Oxf). 2022;4:100316. doi:10.1016/j.puhip.2022.100316.
15. Dos-Santos-Silva I, De Stavola BL, Renna NL Junior, et al. Ethnoracial and social trends in breast cancer staging at diagnosis in Brazil, 2001-14: a case only analysis. Lancet Glob Health. 2019; 7(6):e784-e797. doi:10.1016/S2214-109X(19)30151-2.
16. Simon SD, Bines J, Werutsky G, et al. Characteristics and prognosis of stage I-III breast cancer subtypes in Brazil: The AMAZONA retrospective cohort study. Breast. 2019;44:113-119. doi:10.1016/j.breast.2019.01.008.
17. Rosa DD, Bines J, Werutsky G, et al. The impact of sociodemographic factors and health insurance coverage in the diagnosis and clinicopathological characteristics of breast cancer in Brazil: AMAZONA III study (GBECAM 0115). Breast Cancer Res Treat. 2020;183(3):749-757. doi:10.1007/s10549-020-05831-y.
18. Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353(17): 1784-1792. doi:10.1056/NEJMoa050518.
19. Duffy SW, Tabár L, Yen AM, et al. Mammography screening reduces rates of advanced and fatal breast cancers: Results in 549,091 women. Cancer. 2020;126(13):2971-2979. doi:10.1002/cncr.32859.
20. Duffy SW, Tabár L, Yen AM, et al. Beneficial Effect of Consecutive Screening Mammography Examinations on Mortality from Breast Cancer: A Prospective Study. Radiology. 2021;299(3):541-547. doi:10.1148/radiol.2021203935.
21. Yip CH. Downstaging is more important than screening for asymptomatic breast cancer. Lancet Glob Health. 2019;7(6):e690-e691. doi:10.1016/S2214-109X(19)30190-1.
22. WHO. World health statistics 2019: monitoring health for the SDGs, sustainable development goals. WHO. Published May 21, 2019. Accessed July 27, 2024. https://www.who.int/publications/i/item/9789241565707
23. Resende H, Aguiar V, Jacob L, et al. The journey of breast cancer patient from self-perception of breast abnormalities to first cancer treatment- a sectional study in Sul Fluminense region-RJ-Brazil. Medical Research Archives. 2023;11(10). doi:10.18103/mra.v11i10.4577.
24. INCA. A situação do câncer de mama no Brasil: síntese de dados dos sistemas de informação. Instituto Nacional de Câncer. 2019. Published July 22, 2022. Accessed July 27, 2024.
https://www.inca.gov.br/publicacoes/livros/situacao-do-cancer-de-mama-no-brasil-sintese-de-dados-dos-sistemas-de-informacao
25. Bretas G, Renna NL, Bines J. Practical considerations for expediting breast cancer treatment in Brazil. Lancet Reg Health Am. 2021;2: 100028. doi:10.1016/j.lana.2021.100028.
26. Franzoi MA, Rosa DD, Zaffaroni F, et al. Advanced Stage at Diagnosis and Worse Clinicopathologic Features in Young Women with Breast Cancer in Brazil: A Subanalysis of the AMAZONA III Study (GBECAM 0115). J Glob Oncol. 2019;5:1-10. doi:10.1200/JGO.19.00263.
27. Ferreira NAS, Schoueri JHM, Sorpreso ICE, Adami F, Dos Santos Figueiredo FW. Waiting Time between Breast Cancer Diagnosis and Treatment in Brazilian Women: An Analysis of Cases from 1998 to 2012. Int J Environ Res Public Health. 2020;17(11):4030. doi:10.3390/ijerph17114030.
28. Rousseff DCJ, Padilha ARS. LEI No 12.732, DE 22 DE NOVEMBRO DE 2012. Presidência da República. Published November 22, 2012. Accessed July 27, 2024.
https://legislacao.presidencia.gov.br/atos/?tipo=LEI&numero=12732&ano=2012&ato=276cXUq1kMVpWT8c5
29. Barrios C, de Lima Lopes G, Yusof MM, Rubagumya F, Rutkowski P, Sengar M. Barriers in access to oncology drugs – a global crisis. Nat Rev Clin Oncol. 2023;20(1):7-15. doi:10.1038/s41571-022-00700-7.
30. Carioli G, Malvezzi M, Rodriguez T, Bertuccio P, Negri E, La Vecchia C. Trends and predictions to 2020 in breast cancer mortality in Europe. Breast. 2017;36:89-95. doi:10.1016/j.breast.2017.06.003.
31. Howlader N, Forjaz G, Mooradian MJ, et al. The Effect of Advances in Lung-Cancer Treatment on Population Mortality. N Engl J Med. 2020;383 (7):640-649. doi:10.1056/NEJMoa1916623.
32. Dolgin E. Bringing down the cost of cancer treatment. Nature. 2018;555(7695):S26-S29. doi:10.1038/d41586-018-02483-3.
33. Modi S, Jacot W, Yamashita T, et al. Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. N Engl J Med. 2022;387(1):9-20. doi:10.1056/NEJMoa2203690.
34. National Institute for Health and Care Excellence. NICE disappointed companies unwilling to offer fair price to make Enhertu available for advanced breast cancer. NICE. Published July 29, 2024. Accessed July 30, 2024.
https://www.nice.org.uk/news/articles/nice-disappointed-that-companies-unwilling-to-offer-fair-price-to-the-nhs-to-make-enhertu-available
35. Saltz LB. The Value of Considering Cost, and the Cost of Not Considering Value. J Clin Oncol. 2016;34(7):659-660. doi:10.1200/JCO.2015.64.7867.
36. WHO. Health Technology Assessment. World Health Organization. Published May 29, 2024. Accessed July 30, 2024. https://www.who.int/health-topics/health-technology-assessment#tab=tab_1
37. Cuzick J, Sestak I, Baum M, et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol. 2010;11(12):1135-1141. doi:10.1016/S1470-2045(10)70257-6.
38. NCCN. Breast Cancer (Version 4.2024). National Comprehensive Cancer Network. Published July 03, 2024. Accessed July 30, 2024. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1419
39. Ruhstaller T, Giobbie-Hurder A, Colleoni M, et al. Adjuvant Letrozole and Tamoxifen Alone or Sequentially for Postmenopausal Women With Hormone Receptor-Positive Breast Cancer: Long-Term Follow-Up of the BIG 1-98 Trial. J Clin Oncol. 2019;37(2):105-114. doi:10.1200/JCO.18.00440.
40. Giordano SH, Freedman RA, Somerfield MR; Optimal Adjuvant Chemotherapy and Targeted Therapy Guideline Expert Panel. Abemaciclib With Endocrine Therapy in the Treatment of High-Risk Early Breast Cancer: ASCO Optimal Adjuvant Chemotherapy and Targeted Therapy Guideline Rapid Recommendation Update. J Clin Oncol. 2022;40(3):307-309. doi:10.1200/JCO.21.02677.
41. Johnston SRD, Toi M, O’Shaughnessy J, et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 2023;24(1):77-90.
doi:10.1016/S1470-2045(22)00694-5.
42. Slamon DJ, Fasching PA, Hurvitz S, et al. Rationale and trial design of NATALEE: a Phase III trial of adjuvant ribociclib + endocrine therapy versus endocrine therapy alone in patients with HR+/HER2- early breast cancer. Ther Adv Med Oncol. 2023;15 :17588359231178125. doi:10.1177/17588359231178125.
43. Slamon D, Lipatov O, Nowecki Z, et al. Ribociclib plus Endocrine Therapy in Early Breast Cancer. N Engl J Med. 2024;390(12):1080-1091. doi:10.1056/NEJMoa2305488.
44. Agência Nacional de Vigilância Sanitária Anvisa. Listas de preços de medicamentos. GOV.BR. Published April 26, 2024. Accessed July 30, 2024. https://www.google.com/url?sa=i&url=https%3A%2F%2Fwww.gov.br%2Fanvisa%2Fpt-br%2Fassuntos%2Fmedicamentos%2Fcmed%2Fprecos&psig=AOvVaw0YjqU-vJMLVYJZa2IILdKR&ust=1727648065457000&source=images&cd=vfe&opi=89978449&ved=0CAQQn5wMahcKEwiQ3eP91eaIAxUAAAAAHQAAAAAQBA
45. Perez-Padilla R, Patino CM, Ferreira JC. Number needed to treat: a useful statistic to evaluate the impact of an intervention. J Bras Pneumol. 2019;45(6):e20190371. Published 2019 Dec 9. doi:10.1590/1806-3713/e20190371.
46. Fletcher GS. Epidemiologia Clínica – 6.ed.: Elementos Essenciais.Artmed Editora; 2021.
47. Cherny NI, de Vries EGE, Dafni U, et al. Comparative Assessment of Clinical Benefit Using the ESMO-Magnitude of Clinical Benefit Scale Version 1.1 and the ASCO Value Framework Net Health Benefit Score. J Clin Oncol. 2019;37(4):336-349. doi:10.1200/JCO.18.00729.
48. WHO. Informal Advisory Group on the Availability and Affordability of Cancer Medicines. World Health Organization. Published April 18, 2018. Accessed July 30, 2024.
https://www.who.int/publications/i/item/WHOEMPIAU201804
49. Duggan C, Dvaladze A, Rositch AF, et al. The Breast Health Global Initiative 2018 Global Summit on Improving Breast Healthcare Through Resource-Stratified Phased Implementation: Methods and overview. Cancer. 2020;126 Suppl 10(Suppl 10):2339-2352. doi:10.1002/cncr.32891.
50. Collor FGA. Law No. 8,080, of September 19, 1990. Presidência da República. Published September 19, 1990. Accessed July 30, 2024. https://www2.camara.leg.br/legin/fed/lei/1990/lei-8080-19-setembro-1990-365093-publicacaooriginal-1-pl.html
51. Serra J. Ordinance No 3,535, of September 2, 1998. Ministério da Saúde. Published September 2, 1998. Accessed July 30, 2024. https://bvsms.saude.gov.br/bvs/saudelegis/gm/1998/prt3535_02_09_1998_revog.html
52. Temporao JG. Ordinance No 741, of December 19, 2005. Ministerio da Saúde. Published December 19, 2005. Accessed July 30, 2024. https://bvsms.saude.gov.br/bvs/saudelegis/sas/2005/prt0741_19_12_2005.html
53. Junior HMM. Ordinance No140, of February 27, 2014. Ministério da Saúde. Published February 27, 2014. Accessed July 30, 2024. https://bvsms.saude.gov.br/bvs/saudelegis/sas/2014/prt0140_27_02_2014.html
54. PAHO. Global Atlas of Palliative Care, 2nd Edition. Pan American Health Organization. Published October 21, 2020. Accessed July 30, 2024. https://www.paho.org/en/node/75063
55. Universidade Federal de Pelotas. Abordagem Domiciliar em Cuidados Paliativos Interdisciplinar. UNASUS. Published July 21, 2024. Accessed July 30, 2024. https://www.unasus.gov.br/cursos/curso/45389
56. Fundação Oswaldo Cruz. Abordagem das síndromes geriátricas e cuidados paliativos na Atenção Primária à Saúde. UNASUS. Published July 21, 2024. Accessed July 30, 2024. https://www.unasus.gov.br/cursos/curso/46624
57. Resende H, Soares I, Reno A, et al. Abstract PO1-09-09: The journey of HR positive, HER2 negative metastatic breast cancer’s patients: heterogeneities and barriers in Brazilian public health system- a national survey. Cancer Research. 2024; 84(9_Supplement): p. PO1-09-09-PO1-09-09. DOI:10.1158/1538-7445.SABCS23-PO1-09-09
58. Coles CE, Earl H, Anderson BO, et al. The Lancet Breast Cancer Commission. Lancet. 2024;403(10439):1895-1950. doi:10.1016/S0140-6736(24)00747-5.
59. Greer JA, Trotter C, Jackson V, et al. Comparative effectiveness trial of early palliative care delivered via telehealth versus in person among patients with advanced lung cancer. JCO. 2024; 42(17_suppl): p. LBA3-LBA3.
DOI:10.1200/JCO.2024.42.17_suppl.LBA3
60. Sirintrapun SJ, Lopez AM. Telemedicine in Cancer Care. Am Soc Clin Oncol Educ Book. 2018; 38:540-545. doi:10.1200/EDBK_200141.
61. Uemoto Y, Yamanaka T, Kataoka Y, et al. Efficacy of Telemedicine Using Videoconferencing Systems in Outpatient Care for Patients With Cancer: A Systematic Review and Meta-Analysis. JCO Clin Cancer Inform. 2022;6:e2200084. doi:10.1200/CCI.22.00084.
62. Mann DM, Chen J, Chunara R, Testa PA, Nov O. COVID-19 transforms health care through telemedicine: Evidence from the field. J Am Med Inform Assoc. 2020;27(7):1132-1135. doi:10.1093/jamia/ocaa072.
63. Lima NT. Ordinance No 3681, of May 7, 2024. Diário Oficial da União. Published May 7, 2024. Accessed July 30, 2024. https://www.in.gov.br/web/dou/-/portaria-gm/ms-n-3.681-de-7-de-maio-de-2024-561223717
64. Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011;126(2):529-537. doi:10.1007/s10549-010-1132-4.
65. Wright H, Martin F, Clyne W, et al. A Digital Self-management Program (Help to Overcome Problems Effectively) for People Living With Cancer: Feasibility Randomized Controlled Trial. J Med Internet Res. 2021;23(11):e28322. doi:10.2196/28322.
66. Thummi – Seu aliado no tratamento contra o câncer. Thummi Global. Published May 7, 2024. Accessed July 30, 2024. https://thummi.global/
67. Perlmutter B, Said SA, Hossain MS, et al. Lessons learned and keys to success: Provider experiences during the implementation of virtual oncology tumor boards in the era of COVID-19. J Surg Oncol. 2022;125(4):570-576.
doi:10.1002/jso.26784.
68. Basta YL, Bolle S, Fockens P, Tytgat KMAJ. The Value of Multidisciplinary Team Meetings for Patients with Gastrointestinal Malignancies: A Systematic Review. Ann Surg Oncol. 2017;24(9):26 69-2678. doi:10.1245/s10434-017-5833-3.
69. Menezes TU, Serra MM, Barrios CH, et al. Results of a survey study on health professionals’ perceptions of tumor boards in Brazil. Future Oncol. 2023;19(14):1013-1020. doi:10.2217/fon-2023-0158.
70. Mano MS, Çitaku FT, Barach P. Implementing multidisciplinary tumor boards in oncology: a narrative review. Future Oncol. 2022;18(3):375-384. doi:10.2217/fon-2021-0471.

