COVID-19 Severity: Risk Factors in Saudi Arabia
A Retrospective Study of the factors associated with deterioration severity of Coronavirus Disease-2019 illness at a tertiary hospital in Saudi Arabia
Mohammad AlJabr¹, MD, ABFM; Areej AlDossary², MD, ABFM; Kanan Alkanani², MD; Turky Al Zahrani², MD, CCFP, ABFM; Sofian Almulim², MD, CCFP; Hatim Kheir³, MD, MRCGP, CCFP; Assim AlAbulkader⁴, MD, MPH, FAAFP; Hayat Mushcab⁵, Ph.D.; Yaser Alreshidi⁶, MBBS; Nouf Albalawi², MD; Wedyan Alabdullatif⁷, MBBS; Abrar Almarzooq⁸, MD; and Saeed Al Qahtani², MD, CCFP, FCFP, ACPE
-
Population Health Department, Johns Hopkins Aramco Healthcare, Dhahran, Eastern Province, Saudi Arabia
-
Primary Care, Johns Hopkins Aramco Healthcare, Dhahran, Eastern Province, Saudi Arabia
-
Health Plus Medical, Calgary, Canada
-
Family Medicine and Community Department, Imam Abdulrahman bin Faisal University, Dammam, Eastern Province, Saudi Arabia
-
Research Office, Johns Hopkins Aramco Healthcare, Dhahran, Eastern Province, Saudi Arabia
-
Hail Cluster, Ministry of Health, Hail, Saudi Arabia
-
Dr. Suliaman Al-Habib Hospital, Al Khobar, Saudi Arabia
-
Eastern Cluster, Ministry of Health, Dammam, Saudi Arabia
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: AlJabr, M., AlDossary, A., et al., 2024. A Retrospective Study of the factors associated with deterioration severity of Coronavirus Disease-2019 illness at a tertiary hospital in Saudi Arabia. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.6083
COPYRIGHT :© 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i11.6083
ISSN: 2375-1924
Keywords
COVID-19, Chronic illness, severity, deterioration, risk factors, comorbidity
Abstract
Background: Coronavirus Disease-2019 (COVID-19) has rapidly progressed to a pandemic causing a severe burden on healthcare systems worldwide. COVID-19 has seriously threatened specific populations, such as the elderly and those with chronic illnesses.
Objective: In this study, we aim to determine risk factors contributing to and affecting COVID-19 severity and deterioration during illness.
Methods: This is a retrospective cohort study of patients with a confirmed COVID-19 diagnosis and admitted to JHAH from March to July 2020.
Results: A total of 639 patients were included in this study. The vast majority were Saudi patients (87.3%). 465 (76%) were symptomatic with abnormal x-ray imaging diagnosis (56.2%). The predetermined clinical risk factors include obesity (46.3%), hypertension (42.3%), diabetes mellitus (41.8%), dyslipidemia (39%), ischemic heart disease (13.2%), chronic lung disease (11%), chronic kidney disease (10.3%), cancer (7.9%), congestive heart failure (6.1%); and immunocompromised patients (4.6%). However, most of these patients were stable upon admission (82.1%), while 9.7% were severely or critically ill. Ordinal regression analysis revealed that age, chest x-ray finding upon diagnosis, and presenting symptoms upon admission significantly impacted the deterioration pattern.
Conclusion: This study found that elderly and chronically ill patients are at higher risk for more severe COVID-19 infection and risk of deterioration, hospitalization, intensive care unit admission, and probably death when compared to younger and healthier patients.
Keywords: COVID-19, Chronic illness, severity, deterioration, risk factors, comorbidit
Abbreviations
| Abbreviation | Stands for |
|---|---|
| COVID-19 | Corona Virus Disease 2019 |
| CVD | cardiovascular disease |
| COPD | chronic obstructive pulmonary disease |
| PCR | polymerase chain reaction |
| IHD | Ischemic heart disease |
| CHF | congestive heart failure |
| DM | Diabetes Mellitus |
| HTN | Hypertension |
| MoH | Ministry of Health |
| ICU | Intensive Care Unit |
| BMI | Body Mass Index |
| CI | confidence interval |
| CXR | Chest x-ray |
Introduction
As of April 2021, there are nearly 138 million Coronavirus Disease 2019 (COVID-19) cases and nearly three million deaths worldwide. Initially, COVID-19 symptoms were not specific; patients presented with flu-like symptoms such as fever and cough that can progress to shortness of breath and pneumonia and may lead to acute respiratory distress syndrome, multi-organ failure, and death.
Researchers have been unifying and intensifying their efforts globally to identify people at most significant risk of developing the infection and progressing from asymptomatic to symptomatic, mild to moderate, and severe to a critical level causing them to die. Risk factors can vary from demographical attributes, such as patients’ age, sex, ethnicity, diet, and lifestyle, to underlying chronic medical conditions such as obesity, diabetes, hypertension, cardiovascular disease (CVD), kidney disease, and chronic obstructive pulmonary disease (COPD).
It is helpful for clinicians and healthcare providers to have a clinical prediction model by understanding the possible risk factors within their population in combination with medical conditions associated with COVID-19 severity. Such a prediction model will enable them to classify clinical severity early, provide appropriate disposition, prioritize treatment for patients at a higher risk of disease progression, prevent deterioration, and reduce mortality.
Our hospital is a tertiary hospital located in Dhahran, Eastern Province of Saudi Arabia. It serves a population of 300,000 people. Patients from other parts of the Saudi Arabian Kingdom may have different demographic, epidemiologic, and clinical characteristics. Determining these indicators and factors could help predict the severity of the illness.
In this study, we report all diagnosed with COVID-19 and admitted to our hospital from March to July 2020 for hospitalization. This study presents our patients’ demographic, epidemiological, clinical, laboratory, and imaging characteristics. Moreover, we aim to identify and determine risk factors contributing to and affecting COVID-19 severity and deterioration during their illness course.
Material and Methods
STUDY DESIGN AND POPULATION
This is a retrospective cohort study of patients admitted to JHAH hospital with a confirmed diagnosis of COVID-19 from March to July 2020. The Institutional Review Board’s Approval (#20-33) was received on the 27th of October 2020. In Saudi Arabia, the Covid-19 confirmed cases reached their peak in mid-June 2020. The highest record of more than 2000 COVID-19 critical cases was mid-April 2020.
The diagnosis of COVID-19 was confirmed by a positive polymerase chain reaction (PCR) test, in addition to presenting symptoms, epidemiological, and contact history. We excluded patients with no symptoms and those without risk factors such as pregnancy, chronic diseases, or immunocompromising medication.
OUTCOME MEASURES
The primary outcome of this study is to assess and study risk factors that contribute to illness severity and patients’ deterioration during their hospitalization in our hospital. Patients were categorized as either symptomatic or asymptomatic. Symptomatic patients were then subcategorized into 1) mild, 2) moderate, 3) severe, and 4) critical. Asymptomatic cases were determined as having a positive PCR without any associated clinical symptoms. Mild cases were defined as having fever, malaise, cough, and upper respiratory symptoms. Moderate cases were defined with infiltration on lung imaging without hypoxia (Oxygen saturation in resting state ≥ 94%). Severe cases were defined as having dyspnea and hypoxia with oxygen saturation of less than 94%. Finally, critical cases were defined with at least one of these presentations: respiratory failure, septic shock, or multi-organ dysfunction.
DATA COLLECTION
Data were collected using patients’ electronic health records (EPIC) system. EPIC is a fully integrated electronic health records system that provides more versatile and customized reports about patients with any given criteria. Data collected include demographic data: age, gender, nationality, travel history, and mode of exposure; medical history: Ischemic heart disease (IHD), congestive heart failure (CHF), diabetes mellitus (DM), hypertension (HTN), dyslipidemia, obesity, cancer, kidney disease, lung disease, immunocompromised condition, smoking, and pregnancy; clinical data: recent flu shot, hospitalization, PCR swab test, x-ray findings, symptoms upon admission, severity and deterioration of illness, admission date, and discharge date.
Data were collected by Family Medicine residents and reviewed by their consultants, who determined the severity of the illness. Separately, 10% of the data were then validated by the other co-investigators.
RISK OF BIAS
We included all patients admitted to our hospital from March to July 2020 with a confirmed COVID-19 diagnosis in the study without selection bias.
SAMPLE SIZE
The total number of patients included in the study is 639. All included patients were diagnosed with COVID-19 before hospitalization. At the beginning of the pandemic, all COVID-19 positive cases were hospitalized based on the Saudi Arabian Ministry of Health’s (MoH) guidelines. With a sufficient understanding of the disease and its behavior, the MoH modified its guidelines on the 8th of June. A home monitoring program was launched for home isolation to specific COVID-19 positive patients. This service was offered to patients with asymptomatic or mildly symptomatic (i.e., low-risk). Thus, our sample has excluded seven asymptomatic patients during their hospitalization.
STATISTICAL ANALYSIS
The data analysis was done using the statistical package IBM SPSS Statistics for Windows, Version 25.0 Armonk. Categorical variables were described as frequencies and percentages. The chi-square and Fisher’s exact tests were used to assess the deterioration severity level and other categorical variables. A test was considered significant if the p-value <0.05. Ordinal logistic regression was performed to predict risk factors for deterioration severity with the baseline characteristics.
Results
The total number of confirmed COVID-19 cases was 3400 patients with a positive PCR test and confirmed diagnosis of COVID-19. Of the total number, 656 patients were hospitalized for medical assistance and monitoring. Out of the 656 patients, 17 patients were excluded for not meeting the inclusion criteria (
).
The vast majority of the hospitalized patients were Saudis (87.3%), older than 50 years of age (60.3%), males (53.2%), and non-smokers (92%). The mode of exposure to the virus was primarily unknown to the healthcare providers (58%), followed by contact transmission (36.2%). International and domestic traveling (4.9%), and (1%) of the patients have acquired it during their hospitalization. Most study participants did not receive the influenza vaccine during the previous year of 2019 (80.2%), and only (6.1%) had a travel history within the 14 days before admission; table 1 describes the demographical data in detail.
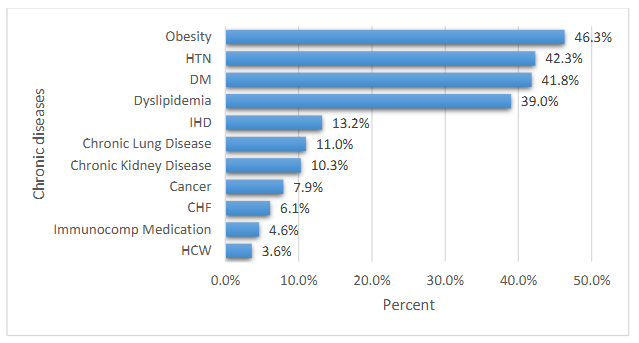
We found that hospitalized patients have had predetermined clinical risk factors associated with their condition at admission. These factors were obesity (46.3%), HTN (42.3%), DM (41.8%), dyslipidemia (39%), IHD (13.2%), chronic lung disease (11%), chronic kidney disease (10.3%), cancer (7.9%), CHF (6.1%), and immunocompromised patients (4.6%). Moreover, only (18%) of the patients were in the normal range of Body mass index (BMI), and (6.6%) were underweight, making overweight and obese patients an obvious risk factor accounting for (76%) of the patients. Another risk factor associated with hospitalization upon admission was the nature of their job as healthcare workers being a front-liner against the pandemic (3.6%) (
The vast majority of patients were hospitalized because they presented symptoms (76%) and abnormal x-ray findings upon diagnosis (56.2%); however, most of these patients were stable upon admission (82.1%). Symptoms were mostly mild (47.5%) and moderate (42.9%). Severe and critical symptoms accounted for (9.7%) of the cases combined. Moreover, 133 cases deteriorated during hospitalization; 65 of them (48.9%) deteriorated from asymptomatic or mild to moderate, while 43 (32.3%) from moderate to severe, and 25 (18.8%) from severe to critical. In addition, 106 (20.3%) of the study patients had required Intensive Care Unit (ICU) admission, and only 24 (4.7%) required intubation. By the end of the study, 34 (5.3%) patients died during hospitalization. Table 1 describes both clinical and non-clinical characteristics in detail.
Table 1: Demographical and clinical characteristics of hospitalized patients with COVID-19
| Variable | Category | N | % |
|---|---|---|---|
| Gender | Male | 340 | 53.2 |
| Female | 299 | 46.8 | |
| Age (years) | 18 -30 | 78 | 12.6 |
| 31 – 40 | 72 | 11.7 | |
| 41 – 50 | 95 | 15.4 | |
| 51 – 60 | 195 | 31.6 | |
| 61 – 70 | 101 | 16.3 | |
| 71+ | 77 | 12.5 | |
| Nationality | Saudi | 558 | 87.3 |
| Non-Saudi | 81 | 12.7 | |
| Blood Group | A | 143 | 28.6 |
| AB | 21 | 4.2 | |
| B | 108 | 21.6 | |
| O | 228 | 45.6 | |
| Smoking | No | 554 | 92.0 |
| Yes | 48 | 8.0 | |
| Body Mass Index Groups | Underweight | 39 | 6.0 |
| Normal weight | 118 | 18.1 | |
| Overweight | 194 | 29.8 | |
| Obese I | 187 | 28.7 | |
| Obese II | 71 | 10.9 | |
| Obese III | 43 | 6.6 | |
| Pregnancy | No | 531 | 94.8 |
| Yes | 29 | 5.2 | |
| Mode of Exposure | Domestic Travel | 12 | 2.0 |
| International Travel | 18 | 2.9 | |
| Contact | 222 | 36.2 | |
| Unknown | 356 | 58.0 | |
| Hospital | 6 | 1.0 | |
| Flu Shot 2019 | No | 491 | 80.2 |
| Yes | 94 | 15.4 | |
| Unknown | 27 | 4.4 | |
| Travel History | No | 520 | 93.9 |
| Yes | 34 | 6.1 | |
| CXR Finding (upon diagnosis) | Normal | 244 | 39.7 |
| Abnormal | 345 | 56.2 | |
| Not Done | 25 | 4.1 | |
| Symptoms (upon admission) | Asymptomatic | 147 | 24.0 |
| Symptomatic | 465 | 76.0 | |
| Severity (upon admission) | Mild | 289 | 47.5 |
| Moderate | 261 | 42.9 | |
| Severe | 57 | 9.4 | |
| Critical | 2 | 0.3 |
Table 2 exhibits the clinical and non-clinical risk factors associated with deterioration severity. The risk factors that had a statistically significant impact on patients’ condition are age, presentation with symptoms upon admission, severity upon admission, and instability during patients’ course of hospitalization, all with p-value <0.05. Table 3 indicates that medical history has a statistically significant impact on the severity of deterioration with a p-value <0.05. Specifically, 27.3% of patients with diabetes, followed by 28.4% of hypertension patients, 29.6% of chronic lung disease patients, 31.3% of cancer patients, and 36.5% of chronic kidney disease patients had their condition deteriorate during the course of hospitalization. Moreover, intubation had a statistically significant (p<0.001) impact on the deterioration severity amongst 19 patients, including the deterioration of 1 patient to moderate, 4 to severe, and 14 to critical cases. ICU admission had a statistically significant (p<0.001) impact as 13 patients have deteriorated to moderate, 40 to severe, and 24 to critical cases. It has also been determined that the longer the patient stays in the hospital, the more likely their condition will deteriorate significantly. Furthermore, it was found that it is most likely (68%) that the patient will die if their condition is critical (Table 2). In addition, it has been found that patients with higher BMI (overweight and Obese) are significantly at a higher risk of condition severity deterioration when associated with older age, diabetes mellitus, hypertension, and dyslipidemia (Table 3).
Table 2: Presentation of clinical and non-clinical risk factors associated with deterioration severity
| Deterioration Severity | No Deterioration | Moderate | Severe | Critical | P-value | |
|---|---|---|---|---|---|---|
| Gender | Male | 266 (78.2) | 35 (10.3) | 23 (6.8) | 16 (4.7) | 0.738 |
| Female | 240 (80.3) | 30 (10.0) | 20 (6.7) | 9 (3.0) | ||
| Age (years) | 18 -30 | 69 (88.5) | 6 (7.7) | 3 (3.8) | 0.021 | |
| 31 – 40 | 64 (88.9) | 7 (9.7) | 1 (1.4) | 0.021 | ||
| 41 – 50 | 83 (87.4) | 9 (9.5) | 3 (3.2) | 0.021 | ||
| 51 – 60 | 154 (79.0) | 18 (9.2) | 18 (9.2) | 5 (2.6) | ||
| 61 – 70 | 74 (73.3) | 12 (11.9) | 12 (11.9) | 3 (3.0) | ||
| 71+ | 55 (71.4) | 12 (15.6) | 6 (7.8) | 4 (5.2) | ||
| Nationality | Saudi | 440 (78.9) | 55 (9.9) | 40 (7.2) | 23 (4.1) | 0.523 |
| Non-Saudi | 66 (81.5) | 10 (12.3) | 3 (3.7) | 2 (2.5) | ||
| Smoking | No | 435 (78.5) | 58 (10.5) | 38 (6.9) | 23 (4.2) | 0.959 |
| Yes | 37 (77.1) | 5 (10.4) | 4 (8.3) | 2 (4.2) | ||
| Body Mass Index Groups | Underweight | 26 (100.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0.347 |
| Normal weight | 95 (81.9) | 11 (9.5) | 8 (6.9) | 2 (1.7) | ||
| Overweight | 148 (77.1) | 19 (9.9) | 13 (6.8) | 12 (6.3) | ||
| Obese I | 142 (75.9) | 25 (13.4) | 14 (7.5) | 6 (3.2) | ||
| Obese II | 60 (84.5) | 5 (7.0) | 3 (4.2) | 3 (4.2) | ||
| Obese III | 31 (72.1) | 5 (11.6) | 5 (11.6) | 2 (4.7) | ||
| Pregnancy | No | 409 (77.0) | 58 (10.9) | 41 (7.7) | 23 (4.3) | 0.159 |
| Yes | 20 (69.0) | 7 (24.1) | 2 (6.9) | 0 (0.0) | ||
| Mode of Exposure | Domestic Travel | 10 (83.3) | 1 (8.3) | 0 (0.0) | 1 (8.3) | 0.813 |
| International Travel | 18 (100.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||
| Contact | 172 (77.5) | 25 (11.3) | 16 (7.2) | 9 (4.1) | ||
| Unknown | 276 (77.5) | 39 (11.0) | 26 (7.3) | 15 (4.2) | ||
| Hospital | 5 (83.3) | 0 (0.0) | 1 (16.7) | 0 (0.0) | ||
| Flu Shot 2019 | No | 382 (77.8) | 54 (11.0) | 34 (6.9) | 21 (4.3) | 0.744 |
| Yes | 73 (77.7) | 11 (11.7) | 8 (8.5) | 2 (2.1) | ||
| Travel History | No | 394 (75.8) | 61 (11.7) | 43 (8.3) | 22 (4.2) | 0.077 |
| Yes | 31 (91.2) | 1 (2.9) | 0 (0.0) | 2 (5.9) | ||
| Symptoms (upon admission) | Asymptomatic | 135 (91.8) | 5 (3.4) | 4 (2.7) | 3 (2.0) | <0.001 |
| Symptomatic | 344 (74.0) | 60 (12.9) | 39 (8.4) | 22 (4.7) | ||
| Severity (upon admission) | Mild | 246 (85.1) | 31 (10.7) | 8 (2.8) | 4 (1.4) | <0.001 |
| Moderate | 192 (73.6) | 29 (11.1) | 27 (10.3) | 13 (5.0) | ||
| Severe | 38 (66.7) | 5 (8.8) | 8 (14.0) | 6 (10.5) | ||
| Critical | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (100.0) |
Table 3 presents the significant association between BMI and the risk factors for severity deterioration. Obesity, age (years), and other factors were analyzed for their impact on deterioration severity.
Table 3: Present the significant association between BMI and the risk factors for severity deterioration.
| Obesity | Chi-square | df | Sig. |
|---|---|---|---|
| Age (years) | 27.971 | 5 | .000* |
| HCW | .130 | 1 | .719 |
| IHD | .515 | 1 | .473 |
| CHF | .180 | 1 | .672 |
| DM | 8.281 | 1 | .004* |
| HTN | 10.715 | 1 | .001* |
| Dyslipidemia | 16.157 | 1 | .000* |
| Cancer | .484 | 1 | .487 |
| Chronic Kidney Disease | .119 | 1 | .730 |
| Chronic Lung Disease | .757 | 1 | .384 |
| Immunocomp Medication | .042 | 1 | .837 |
The severity of deterioration is also affected by the patient’s medical history. Preexisting chronic health conditions such as diabetes mellitus, hypertension, cancer, chronic kidney disease, and chronic lung disease had a statistically significant (p<0.005) association with severity of deterioration (Table 4). Ordinal regression analysis was performed at a 95% confidence interval (CI) and revealed that age, chest x-ray (CXR) finding upon diagnosis, and symptoms upon admission significantly impacted the deterioration pattern. We found that younger patients, especially those 31-40 years old, were nearly 1:3 less likely to deteriorate when compared to older patients (71+ years old). Upon diagnosis, patients with abnormal CXR findings were three times more likely to deteriorate than those with normal CXR. Furthermore, patients who presented with no symptoms upon admission were 61% less likely to deteriorate when compared to symptomatic patients (Table 5).
Table 4: Medical history and association with deterioration severity during hospitalization of COVID-19 patients
| Deterioration Severity | No Deterioration | Moderate | Severe | Critical | P-value | |
|---|---|---|---|---|---|---|
| HCW | No | 465 (78.4) | 61 (10.3) | 42 (7.1) | 25 (4.2) | 0.624 |
| Yes | 17 (77.3) | 4 (18.2) | 1 (4.5) | 0 (0.0) | ||
| IHD | No | 421 (79.1) | 55 (10.3) | 36 (6.8) | 20 (3.8) | 0.577 |
| Yes | 59 (72.8) | 10 (12.3) | 7 (8.6) | 5 (6.2) | ||
| CHF | No | 451 (79.0) | 61 (10.7) | 39 (6.8) | 20 (3.5) | 0.092 |
| Yes | 25 (67.6) | 4 (10.8) | 4 (10.8) | 4 (10.8) | ||
| DM | No | 295 (82.6) | 37 (10.4) | 19 (5.3) | 6 (1.7) | <0.001 |
| Yes | 186 (72.7) | 27 (10.5) | 24 (9.4) | 19 (7.4) | ||
| HTN | No | 295 (83.3) | 35 (9.9) | 20 (5.6) | 4 (1.1) | <0.001 |
| Yes | 186 (71.5) | 30 (11.5) | 23 (8.8) | 21 (8.1) | ||
| Dyslipidemia | No | 297 (79.4) | 39 (10.4) | 21 (5.6) | 17 (4.5) | 0.338 |
| Yes | 183 (76.6) | 26 (10.9) | 22 (9.2) | 8 (3.3) | ||
| Obesity | No | 263 (79.9) | 33 (10.0) | 20 (6.1) | 13 (4.0) | 0.711 |
| Yes | 217 (76.4) | 32 (11.3) | 23 (8.1) | 12 (4.2) | ||
| Cancer | No | 445 (79.3) | 56 (10.0) | 41 (7.3) | 19 (3.4) | 0.022 |
| Yes | 33 (68.8) | 7 (14.6) | 2 (4.2) | 6 (12.5) | ||
| Chronic Kidney Disease | No | 441 (80.0) | 58 (10.5) | 33 (6.0) | 19 (3.4) | 0.003 |
| Yes | 40 (63.5) | 7 (11.1) | 10 (15.9) | 6 (9.5) | ||
| Chronic Lung Disease | No | 414 (80.2) | 56 (10.9) | 26 (5.0) | 20 (3.9) | 0.023 |
| Yes | 45 (70.3) | 7 (10.9) | 10 (15.6) | 2 (3.1) | ||
| Immunocomp Medication | No | 439 (79.1) | 60 (10.8) | 36 (6.5) | 20 (3.6) | 0.689 |
| Yes | 21 (77.8) | 3 (11.1) | 1 (3.7) | 2 (7.4) |
Table 5: presents the ordinal logistic regression for deterioration severity with the baseline characteristics.
| P-value | OR | 95% CI for OR | Lower | Upper | |
|---|---|---|---|---|---|
| Gender | Male | 0.336 | 1.27 | 0.78 | 2.09 |
| Female* | . | 1 | . | . | |
| Age (years) | 18 -30 | 0.166 | 0.50 | 0.19 | 1.34 |
| 31 – 40 | 0.044 | 0.37 | 0.14 | 0.98 | |
| 41 – 50 | 0.057 | 0.42 | 0.17 | 1.03 | |
| 51 – 60 | 0.604 | 0.83 | 0.42 | 1.66 | |
| 61 – 70 | 0.87 | 1.07 | 0.48 | 2.38 | |
| 71+ | . | 1 | . | . | |
| Nationality | Saudi | 0.776 | 0.90 | 0.44 | 1.84 |
| Non-Saudi* | . | 1 | . | . | |
| Blood Group | A | 0.929 | 0.98 | 0.56 | 1.70 |
| AB | 0.700 | 1.24 | 0.41 | 3.75 | |
| B | 0.659 | 1.14 | 0.64 | 2.05 | |
| O* | . | 1 | . | . | |
| Smoking | No | 0.562 | 0.78 | 0.33 | 1.83 |
| Yes* | . | 1 | . | . | |
| Body mass index groups | Overweight | 0.170 | 0.52 | 0.21 | 1.32 |
| Obese I | 0.398 | 0.67 | 0.26 | 1.71 | |
| Obese II | 0.200 | 0.49 | 0.16 | 1.47 | |
| Obese III* | . | 1 | . | . | |
| Pregnancy | No | 0.326 | 0.64 | 0.26 | 1.57 |
| Yes* | . | 1 | . | . | |
| Mode of Exposure | Domestic/International Travel | 0.787 | 1.28 | 0.22 | 7.48 |
| Contact | 0.332 | 1.25 | 0.80 | 1.97 | |
| Unknown* | . | 1 | . | . | |
| Flu Shot 2019 | No | 0.994 | 1.00 | 0.56 | 1.78 |
| Yes* | . | 1 | . | . | |
| Travel History | No | 0.421 | 2.27 | 0.31 | 16.81 |
| Yes* | . | 1 | . | . | |
| CXR Finding (Upon diagnosis) | Normal | 0.005 | 0.36 | 0.18 | 0.74 |
| Abnormal* | . | 1 | . | . | |
| Symptoms (Upon admission) | Asymptomatic | 0.007 | 0.39 | 0.19 | 0.77 |
| Symptomatic* | . | 1 | . | . | |
| Severity (Upon admission) | Mild | 0.619 | 0.81 | 0.35 | 1.86 |
| Moderate | 0.327 | 0.73 | 0.38 | 1.38 | |
| Severe/Critical* | . | 1 | . | . |
Discussion
PATIENT CHARACTERISTICS
The COVID-19 pandemic has introduced many uncertainties regarding factors influencing infection severity, complicating clinical management. The widespread COVID-19 infection, due to the high infectivity of the virus, allowed many healthcare providers to observe and experience different behavior of COVID-19 severity among the different patient populations. Those observations raised the clinical question of what factors can determine COVID-19 disease severity?
Several studies suggest that factors like age, gender, and underlying comorbidities largely determine COVID-19 severity. Recent studies have found that older adults are at higher risk for severe COVID-19-associated illness; hence, they are most vulnerable to the disease and have the most severe form of the illness and the worst outcome. In this study, we could not observe a significant difference between males and females among confirmed cases. However, much data in the literature suggest that males are more prone to severe COVID-19 infection that may need intensive intervention. Although it is not statistically significant, we found that male patients are (27%) more likely to deteriorate and have severe illness than females.
Observational studies demonstrate a higher risk of contracting COVID-19 infection and disease severity among healthcare providers. Such findings did not appear to be significant among our studied population, which could be because healthcare workers are mostly of a younger age group, have fewer comorbidities, and are obliged to follow institutional strict infection control measures. Other studies have also linked the disease severity to certain ethnicities. This factor was not evaluated in this study since most of the cohort are Saudi nationals (87.3%).
Moreover, we found that comorbidities such as diabetes mellitus, chronic obstructive lung disease, obesity, and cardiovascular diseases were associated with severe COVID-19, similar to what was reported in the literature.
BEHAVIORS AND PRACTICES
Research studying the link between tobacco smoking and COVID-19 infection is still limited. We found that smoking was not strongly associated with deterioration severity of COVID-19 infection and severe outcomes. This finding is contrary to what Patanavanich & Glantz have reported in their meta-analysis published in 2020, that smoking is a significant risk factor for severe progression of COVID-19 infection compared to non-smokers. There may have been missed cases in the literature because of mislabeling of smoking status, clarity of smoking methods, the number of cigarettes smoked in general, smoking-status duration, and whether or not smoking cessation has occurred and for how long. In our study, we believe that some smokers might not have disclosed their smoking habits, which might be due to cultural barriers and fear of being judged. Besides, some patients who smoke electronic cigarettes classify themselves as non-smokers.
A study in Brazil found that getting the seasonal Flu vaccine decreased the severity and mortality of COVID-19 illness. On the contrary, our data suggest that the flu vaccine did not affect the severity outcome. Among our study cohort of 639 individuals, only 73 reported receiving the previous year’s flu vaccine. Additionally, there is a scarcity in the literature that examines the impact of the Flu vaccine on COVID-19 illness. Hence, this study cannot entirely determine the effect of the Flu vaccine on the severity of COVID-19 disease.
CHRONIC ILLNESSES
In this study, the data strongly suggest that patients with preexisting diabetes mellitus were at an increased risk for severe COVID-19. This finding is supported by several published studies, including a meta-analysis by Kumar et al. published in 2020 showed an increase in the severity of Covid-19 infection and mortality by two folds in diabetic patients compared to non-diabetic. Another meta-analysis published in 2020 by Huang, Lim, and Pranata showed that diabetes was associated with adverse outcomes in COVID-19 patients in terms of severity of the infection, rapid disease progression, and acute respiratory distress syndrome (ARDS); and mortality. However, the publication was limited by confounding factors such as preexisting risk factors, including older age and hypertension. Diabetes mellitus as a single risk factor was more prominent in studies that included younger diabetic patients without any other comorbidity.
Furthermore, cardiovascular diseases, such as ischemic heart disease (IHD) and congestive heart failure (CHF), on COVID-19 illness were startling and unforeseen. Our data showed no significant correlation between cardiovascular diseases (e.g., IHD and CHF) and deterioration severity in Covid-19 patients. A meta-analysis showed that preexisting chronic diseases, including coronary artery disease (CAD) and cerebrovascular diseases (CVD), increased the risk of developing severe COVID-19 disease and ICU admission. Another meta-analysis indicated that CHF is an independent risk factor that increases the risk for hospitalization, deterioration severity, and mortality in patients infected by COVID-19; however, more data about the stage of CHF was needed.
Regarding hypertension, our results strongly correlate with hospitalized patients’ deterioration severity of COVID-19 illness. This observation is consistent with previous studies reporting that hypertension is significantly associated with the increased risk of illness progression in patients with COVID-19. The fact that hypertension is the most common comorbidity among COVID-19 patients may explain this association. A plausible theory is that pathogenic severe acute respiratory syndrome coronavirus family (e.g., SARS-CoV, MERS-CoV, and SARS-CoV-2) bind to their target cells through angiotensin-converting enzyme 2 (ACE2). Subsequently, it is expressed by epithelial cells of the lung, intestine, kidney, and blood vessels. However, it remains unclear whether or not poorly controlled blood pressure is a risk factor for getting infected with COVID-19 and developing the severe disease.
To our surprise, the analysis revealed that dyslipidemia was not significantly correlated with COVID-19 illness severity or progression, but other studies showed otherwise. Dyslipidemia was strongly associated with more severe COVID-19 illness. In this study, different cholesterol levels were not measured; however, it is believed that elevated low-density lipoprotein (LDL) and low high-density lipoprotein (HDL) levels potentiate the inflammatory process caused by the COVID-19 infection.
Moreover, our study showed that obesity strongly correlates with hospital course deterioration of COVID-19 infection, especially when associated with age, diabetes mellitus, hypertension, and dyslipidemia. This finding is consistent with previous studies showing that obesity was significantly associated with the increased risk of illness progression in patients with coronavirus disease 2019 (COVID-19). Additionally, obesity increases the risk for hospitalization, ICU admission, need for invasive ventilation, and death among patients with COVID-19. About 47.3 % of our population is obese. Out of them, patients aged between 51-60 years (53.6%), patients with DM (53%), patients with HTN (53.9%), and patients with dyslipidemia (56.3%). Obesity is a significant health threat due to its associated metabolic diseases, such as type 2 diabetes, heart diseases, stroke, and certain cancers. Obesity has appeared as a strong risk factor for disease severity in the current COVID-19 pandemic. Moreover, several studies have demonstrated that obese patients with COVID-19 have a higher risk of severe disease, hospitalization, and an increased probability of death.
Recent studies have shown the negative impact on the immune system, making the host vulnerable to infectious diseases.
Amongst our study cohort, cancer patients had a worse clinical course than non-cancer patients, ranging from moderate to critical. Our result was consistent with multiple studies published globally. A multi-center study has shown that cancer patients infected with COVID-19 were more susceptible to severe deterioration of illness and outcomes, with hematological and lung cancers being the highest in adverse events. Another study showed that cancer patients were intubated more frequently than others, which could be attributed to cytokine-associated lung injury in cancer patients with weakened immune systems. Another study found that cancer patients have elevated levels of “Neutrophil, NLR, IL-6, LDH and PCT”.
Additionally, our data revealed that patients with chronic kidney disease (CKD) and those infected with COVID-19 were more susceptible to severe illnesses. Previous studies have shown that patients with CKD were at a higher risk for deterioration and poor prognosis. A study by Chan et al. in 2021 documented a higher incidence of neutrophilia in such patients. Another study concluded that CKD significantly predicted COVID-19 severity among hospitalized patients. The exact mechanism is not precise. Nonetheless, concomitant high blood pressure may play a role due to the decrease in ACE2 by SARS-CoV-2.
Moreover, a significant correlation between chronic lung disease (CLD) and deterioration of COVID-19 illness was found. This finding is supported by a meta-analysis of 19 studies presenting the negative impact of preexisting COPD and tobacco smoking on the severity of COVID-19 infection. Patients with COPD were more likely to have severe COVID infection by four-folds than those without COPD. However, a significant correlation between the use of immunosuppressive medications and deterioration severity for COVID-19 infection was not found in this study. A cohort study reported a similar finding involving more than 5,000 patients with inflammatory bowel disease (IBD). Systemic immunosuppressive therapy in those patients did not increase the risk for COVID-19 infection. While this observation is encouraging, the study had a few limitations. Patients with mild symptoms may not have been tested for COVID-19; the patients who self-discontinued their medications were not classified as non-users. The exact dose of the immunosuppressive medications was not factored in.
CLINICAL PRESENTATION
Early in the COVID-19 pandemic, the health authority of Saudi Arabia mandated all COVID-19 positive patients to be admitted to hospitals, regardless of the presence of symptoms. It is worth mentioning that in Saudi Arabia, the COVID-19 confirmed cases peaked in mid-June 2020, and the highest record of critical cases reached more than 2000 cases in mid-April 2020. Consequently, our study showed a significant correlation between the presence of symptoms upon admission and the deterioration severity. Among those admitted to the hospital with no symptoms, (91.8%) showed no deterioration, (3.4%) showed moderate deterioration, (2.7%) showed severe deterioration, and only (2%) showed critical deterioration. On the other hand, patients who presented with symptoms upon admission were slightly more likely to deteriorate, leaving us with (74%) of symptomatic patients showing no deterioration.
Our study included both asymptomatic and symptomatic patients, which gives our study more strength in its findings. Chin et al. study showed that patients with severe symptoms and unstable hospital courses were more likely to deteriorate. In comparison, the remaining (26%) of patients showed moderate (12.9%), severe (8.4%), and critical deterioration (4.7%). Similar findings were reported by Chin et al. in their study on patients admitted to the Zhongnan Hospital of Wuhan University. One of the limitations of their study, they only included symptomatic patients since the study was conducted in a center only for those with severe symptoms.
Previous studies showed an association between specific symptoms upon admission and deterioration severity of the COVID-19 illness course. A cohort study by Zhou et al. concluded that of 191 hospitalized patients, the most common presentations were fever (94%) and cough (79%) upon admission, which showed an increase in mortality with tachypnea. Another systematic review and meta-analysis by Rodriguez-Morales et al. described the most common symptoms were fever (88.7%), cough (57.6%), dyspnea (45.6%), myalgia or fatigue (29.4%), sore throat (11.0%), headache (8.0%), and diarrhea (6.1%). Anosmia was associated with a mild clinical course.
We are not aware of prior work that studied the correlation between the severity of the symptoms upon admission and the deterioration severity of the COVID-19 illness course. In our study, we looked at the presence and the severity of symptoms upon admission. Among those who presented with mild symptoms majority (85.1%) showed no clinical deterioration in their hospital course, and only (5.4%) of this group showed severe or critical deterioration. In contrast, for those with moderate symptoms upon admission, (73.6%) showed no deterioration, and up to (15.3%) showed severe to critical deterioration. Whereas if they had severe symptoms upon admission, they had an (8.8%) chance for moderate deterioration and (24.5%) severe or critical deterioration. This significant finding showed a strong correlation between the severity of the symptoms upon admission and the likelihood of deterioration during the hospital stay.
HOSPITAL COURSE AND CARE COMPLEXITY
Hospital course and care complexity among hospitalized patients with COVID-19 varied considerably. We evaluated specific indicators of care complexity such as invasive ventilation, admission or transfer to the intensive care unit (ICU) or step down (SD), and the hospital length of stay (LOS) to explore their association with deterioration severity of COVID-19 illness. Expectedly, these indicators correlated positively with the deterioration severity of COVID-19 illness. For instance, patients who required invasive ventilation and admission to the ICU or SD were more likely to have severe to critical deterioration of their illness. Among our study cohort, about 10% of patients with severe deterioration and two-thirds (63.6%) of those with the critical clinical course were intubated.
On the other hand, a third of the patients with moderate deterioration had to stay in the ICU for some time, while more than 90% of those with severe to critical course ended up in the ICU. Consistent with prior work, patients undergoing moderate to severe deterioration of their illness (e.g., respiratory distress) required invasive ventilation and admission to the ICU. Moreover, those patients had additional risk factors such as old age and obesity, which have led to a more extended hospital stay.
According to a meta-analysis published in 2020, the hospital length of stay (LOS) of patients admitted with COVID-19 varied from less than seven days to almost two months. Patients who spent more time (days) in the hospital were more likely to have gone through more severe deterioration of COVID-19 illness and subsequently unfavorable outcomes (death). A hospital LOS of more than seven days was significantly associated with our study’s severe or critical clinical course. While more extended hospital stays (more than seven days) tend to be in intensive care (ICU) or step-down (SD) units, we did not further characterize our LOS data based on the location of admission. This limitation was also reported in the meta-analysis by Rees et al.
Conclusion
In line with the literature, our study confirms that older patients and patients with chronic medical conditions such as cardiovascular diseases, chronic kidney disease, chronic lung disease, and diabetes are at higher risk for more severe COVID-19 illness than healthy individuals infected with COVID-19. They are at a higher risk of deterioration, ICU admission, ventilation, and death. This population is at higher risk for hospitalization. Patients admitted at an early presentation of their symptoms progressed better than those who came with moderate to severe symptoms. Public health authorities and primary care practitioners are expected to raise awareness about this correlation amongst the population and healthcare providers. COVID-19 threat to this vulnerable population is an alert for the healthcare industry and governments to develop short and long-term plans to reduce the prevalence of modifiable chronic medical conditions. Meanwhile, those at a higher risk of acquiring severe COVID-19 should have strict preventive measures and social distancing to reduce their exposure. We would recommend a post-discharge follow-up study to determine any long-term complications or consequences after their course of illness.
Declarations
ETHICS APPROVAL
The study received the Institutional Review Board’s approval (no. 20-33) in October 2020.
CONSENT TO PARTICIPATE
The patients’ consent form was waived due to the retrospective study design with the definite anonymity of all participants.
AVAILABILITY OF DATA AND MATERIAL
Data is available upon request
COMPETING INTERESTS
The authors declare that there are no conflicts of interest.
FUNDING
This research received no specific grant from the public, commercial, or not-for-profit funding agencies.
AUTHORS’ CONTRIBUTION
All authors testify that all persons designated as authors qualify for authorship and have checked the article for plagiarism. The research idea was conceptualized by MJ and AD. The clinical work was performed by the coauthors MJ, AD, KK, HK, and TZ. Data was collected by YR, NB, WA, and AM. Following that, data validation was performed by MJ and HM to ensure that the data provided was clean and accurate. The main draft of the manuscript was written by HM, MJ, AD, AA, KK, and HK. Finally, TZ, SM, and SQ reviewed and edited the manuscript. Moreover, SQ has approved and supervised all decisions from conceptualization to execution.
ACKNOWLEDGMENTS:
Not Applicable
References
1. Johns Hopkins University. Johns Hopkins Coronavirus Resources Center. Published April 15, 2021. Accessed April 15, 2021. https://coronavirus.jhu.edu/map.html.
2. Liu S, Luo H, Wang Y, et al. Clinical characteristics and risk factors of patients with severe COVID-19 in Jiangsu province, China: a retrospective multicentre cohort study. BMC Infect Dis. 2020;20:584-593.
3. Gao Y, Ding M, Dong X, et al. Risk factors for severe and critically ill COVID-19 patients: A review. Allergy. 2021;76:428-455.
4. Ho F, Celis-Morales C, Gray SR, et al. Modifiable and non-modifiable risk factors for COVID-19, and comparison to risk factors for influenza and pneumonia: results from a UK Biobank prospective cohort study. BMJ Open. 2020;10:e040402.
5. Sanyaolu A, Chuku O, Marinkovic A, et al. Comorbidity and its Impact on Patients with COVID-19. SN Compr Clin Med. 2020;1-8.
6. Pradhan A, Olsson P-E. Sex differences in severity and mortality from COVID-19: are males more vulnerable? Biol Sex Differ. 2020;11(1):53.
7. Mutambudzi M, Niedzwiedz C, Macdonald EB, et al. Occupation and risk of severe COVID-19: prospective cohort study of 120,075 UK Biobank participants. Occup Environ Med. 2021;78:307-314.
8. Gibson DM, Greene J. Risk for Severe COVID-19 Illness Among Health Care Workers Who Work Directly with Patients. J Gen Intern Med. 2020;35(9):2804-2806.
9. Khunti K, Griffiths A, Majeed A, Nagpaul C, Rao M. Assessing risk for healthcare workers during the COVID-19 pandemic. BMJ. 2021;n602.
10. Vila-Córcoles A, Ochoa-Gondar O, Satué-Gracia EM, et al. Influence of prior comorbidities and chronic medications use on the risk of COVID-19 in adults: a population-based cohort study in Tarragona, Spain. BMJ Open. 2020;10(12):e041577.
11. Centers for Disease Control and Prevention. Underlying Medical Conditions Associated with High Risk for Severe COVID-19: Information for Healthcare Providers. Published March 29, 2021. Accessed April 16, 2021.
12. Targher G, Mantovani A, Wang X-B, et al. Patients with diabetes are at higher risk for severe illness from COVID-19. Diabetes Metab. 2020;46(4):335-337.
13. Patanavanich R, Glantz SA. Smoking Is Associated With COVID-19 Progression: A Meta-analysis. Nicotine Tob Res. 2020;22(9):1653-1656.
14. Kumar A, Arora A, Sharma P, et al. Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis. Diabetes Metab Syndr. 2020;14(4):535-545.
15. Huang I, Lim MA, Pranata R. Diabetes mellitus is associated with increased mortality and severity of disease in COVID-19 pneumonia – A systematic review, meta-analysis, and meta-regression. Diabetes Metab Syndr. 2020;14(4):395-403.
16. Liu H, Chen S, Liu M, et al. Comorbid Chronic Diseases are Strongly Correlated with Disease Severity among COVID-19 Patients: A Systematic Review and Meta-Analysis. Aging Dis. 2020;11(3):668-678.
17. Yonas E, Alwi I, Pranata R, et al. Effect of heart failure on the outcome of COVID-19 – A meta-analysis and systematic review. Am J Emerg Med. 2020;S0735-6757(20)30602-1.
18. Huang S, Wang J, Liu F, et al. COVID-19 patients with hypertension have more severe disease: a multicenter retrospective observational study. Hypertens Res. 2020;43(8):824-831.
19. Liang X, Shi L, Wang Y, et al. The association of hypertension with the severity and mortality of COVID-19 patients: Evidence based on adjusted effect estimates. J Infect. 2020;81(3):e44-e47.
20. Hosseinzadeh R, Goharrizi MA, Bahardoust M, et al. Should all patients with hypertension be worried about developing severe coronavirus disease 2019 (COVID-19)? Clin Hypertens. 2021;27:3.
21. Hariyanto TI, Kurniawan A. Dyslipidemia is associated with severe coronavirus disease 2019 (COVID-19) infection. Diabetes Metab Syndr. 2020;14(5):1463-1465.
22. Ghoi GJ, Kim HM, Kang H. The Potential Role of Dyslipidemia in COVID-19 Severity: an Umbrella Review of Systematic Reviews. J Lipid Atheroscler. 2020;9(3):435-448.
23. Kompaniyets L, Goodman AB, Belay B, et al. Body Mass Index and Risk for COVID-19-Related Hospitalization, Intensive Care Unit Admission, Invasive Mechanical Ventilation, and Death – United States, March-December 2020. MMWR Morb Mortal Wkly Rep. 2021;70(10):355-361.
24. Huang Y, Lu Y, Huang Y, et al. Obesity in patients with COVID-19: a systematic review and meta-analysis. Metabolism. 2020;113:154378.
25. Mehanna O, El Askary A, Ali E, et al. Impact of Obesity and Its Associated Comorbid Conditions on COVID-19 Presentation. Diabetes Metab Syndr Obes Targets Ther. 2021;14:409-415.
26. Mohammad S, Aziz F, Al Mahri S, et al. Obesity and COVID-19: what makes obese host so vulnerable? Immun Ageing. 2021;18(1):1.
27. Dai M, Liu D, Liu M, et al. Patients with Cancer Appear More Vulnerable to SARS-CoV-2: A Multicenter Study during the COVID-19 Outbreak. Cancer Discov. 2020;6:783-791.
28. Miyashita H, Mikami T, Chopra N, et al. Do patients with cancer have a poorer prognosis of COVID-19? An experience in New York City. Ann Oncol. 2020;31:1088-1089.
29. Ma J, Yin J, Qian Y, Wu Y. Clinical characteristics and prognosis in cancer patients with COVID-19: A single center’s retrospective study. J Infect. 2020;81(2):318-356.
30. Yang D, Xiao Y, Chen Y, et al. COVID-19 and chronic renal disease: clinical characteristics and prognosis. QJM. 2020;113(11):799-805.
31. Chan L, Chaudhary K, Saha A, et al. AKI in Hospitalized Patients with COVID-19. J Am Soc Nephrol. 2021;32(1):151-160.
32. Vicenzi M, Di Cosola R, Ruscica M, et al. The liaison between respiratory failure and high blood pressure: evidence from COVID-19 patients. Eur Respir J. 2020;56(1):2001157.
33. Zhao Q, Meng M, Kumar R, et al. The impact of COPD and smoking history on the severity of COVID-19: A systemic review and meta-analysis. J Med Virol. 2020;92(10):1915-1921.
34. Burke KE, Kochar B, Allegretti JR, et al. Immunosuppressive Therapy and Risk of COVID-19 Infection in Patients With Inflammatory Bowel Diseases. Inflamm Bowel Dis. 2021;27(2):155-161.
35. Chen T, Dai Z, Mo P, et al. Clinical Characteristics and Outcomes of Older Patients with Coronavirus Disease 2019 (COVID-19) in Wuhan, China: A Single-Centered, Retrospective Study. J Gerontol A Biol Sci Med Sci. 2020;75(9):1788-1795.
36. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062.
37. Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020;34:101623.
38. Yan C-H, Faraji F, Prajapati DP, Ostrander BT, DeConde AS. Self‐reported olfactory loss associates with outpatient clinical course in COVID‐19. Int Forum Allergy Rhinol. 2020;10(7):821-831.
39. Hur K, Price CP, Gray EL, et al. Factors Associated With Intubation and Prolonged Intubation in Hospitalized Patients With COVID-19. Otolaryngol Head Neck Surg. 2020;163(1):170-178.
40. Rees EM, Nightingale ES, Jafari Y, et al. COVID-19 length of hospital stay: a systematic review and data synthesis. BMC Med. 2020;18(1):270.
41. Fink G, Orlova-Fink N, Schindler T, et al. Inactivated trivalent influenza vaccination is associated with lower mortality among patients with COVID-19 in Brazil. BMJ Evidence-Based Medicine. 2021;26:192-193.

