Enhancing Skin Cell Aging Resistance with Polyoxometalates
Enhancing effect of polyoxometalates on the aging stress responses of skin cells
Katsuyuki Fujinami ¹ ⁻ ², Katsuaki Dan ³ ⁻ ⁴, Nanami Tominaga ¹, Toshiko Tanaka-Kagawa ², Ikuo Kawamura ²
¹ FSX, Inc., 1-12-3, Izumi, Kunitachi, Tokyo 186-0012, Japan.
² Department of Health Medicine, Yokohama University of Pharmacy, 601, Matano-cho, Totsuka-ku, Yokohama 245-0066, Japan.
³ Division of Research and Development, Research Organization of Biological Activity, Ebisu-nishi 5F, 2-8-4, Ebisu-nishi, Shibuya-ku, Tokyo 150-0021, Japan.
⁴ Department of Pathophysiology, Yokohama University of Pharmacy, 601, Matano-cho, Totsuka-ku, Yokohama 245-0066, Japan.
*Corresponding author: [email protected]
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION Fujinami, K., Dan, K., et al., 2025. Enhancing effect of polyoxometalates on the aging stress responses of skin cells. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6726
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i8.6726
ISSN 2375-1924
ABSTRACT
The accumulation of senescent cells has attracted attention as a cause of aging. Various environmental factors have a harmful effect on the skin, causing aging stress in skin cells, which in turn promotes cellular aging through the accumulation of reactive oxygen species. Stem cell-secreted exosomes also play important roles in skin regeneration and stress relief. Among the polyoxometalates with various biological activities, VB1: vanadyl sulfate (VOSO4), VB2: K11H[(VO)3(SbW9O33)2]・27H2O and VB3: Na2[SbW9O34]・19H2O have been developed as cosmetic ingredients that can be applied to the skin. We evaluated whether these VBs could enhance the resistance of cells to skin aging stress and examined the underlying mechanisms. In this experiment, skin fibroblasts were subjected to glycation (advanced glycosylated end products), oxidation (hydrogen peroxide), and photoaging (ultraviolet irradiation for 5 or 25 min), and VBs were applied before or after treatment. Whether VBs enhance the cellular stress response was determined by changes in the mRNA levels of three parameters (collagen, elastin, and hyaluronic acid synthase). We also investigated whether treating stem cells with VBs results in the secretion of exosomes with properties different from those normally secreted. In addition, because intracellular reactive oxygen species accumulation was suppressed in human dermal fibroblasts treated with VBs, as shown here and in our previous study, the mitochondrial oxygen consumption rate and uptake of cystine, a raw material for the synthesis of the intracellular antioxidant glutathione, were also examined. According to the results of those experiments, we concluded that VBs could enhance stress responses to all the aging stress examined. Vanadyl sulfate likely showed direct effects on dermal fibroblasts, while VB3 was supposed to exhibit the effect via modification of exosomes secreted from mesenchymal stem cells. In addition, VB2 might be involved in the stress response through both direct action on the cells and by inducing the production of exosomes suitable for the stress response. It has been suggested that a combination of several types of VB may exert multifaceted anti-aging effects.
Keywords
polyoxometalates, skin aging, oxidative stress, fibroblasts, exosomes
Abbreviations
- PMs: polyoxometalates
- VB1: vanadyl sulfate (VOSO4)
- VB2: K11H[(VO)3(SbW9O33)2] 27H2O
- VB3: Na2[SbW9O34] 19H2O
- SASP: senescence-associated secretory phenomenon
- BSA: bovine serum albumin
- AGEs: advanced glycation end products
- ROS: reactive oxygen species
- UV: ultraviolet
- MSCs-Exo: mesenchymal stem cells-derived exosomes
- H2O2: hydrogen peroxide
- OCR: oxygen consumption rate
- HDFs: human dermal fibroblasts
- qRT-PCR: quantitative reverse transcription polymerase chain reaction
- DCF-DA: dichlorofluorescein diacetate
- FCCP: carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone
- DAPI: 4’,6-diamidino-2-phenylindole.
Introduction
Various hypotheses have been proposed as to the cause of aging. In recent years, it has been revealed that telomere shortening due to cell division is the main cause of aging in human fibroblasts, while oxidative stress is the main cause in mice. It has also been suggested in recent years that the accumulation of senescent cells in the body causes various age-related diseases. Cellular senescence is a phenomenon in which somatic cells stop dividing in response to stress. For many years, it has been considered a cancer-suppression mechanism. Senescent cells are accompanied by a senescence-associated secretory phenomenon (SASP), and it is assumed that SASP has various effects on the surrounding cells. Furthermore, the elimination of senescent cells has been reported to extend a healthy lifespan. Consequently, senolysis, which targets and removes these cells, has garnered significant attention.
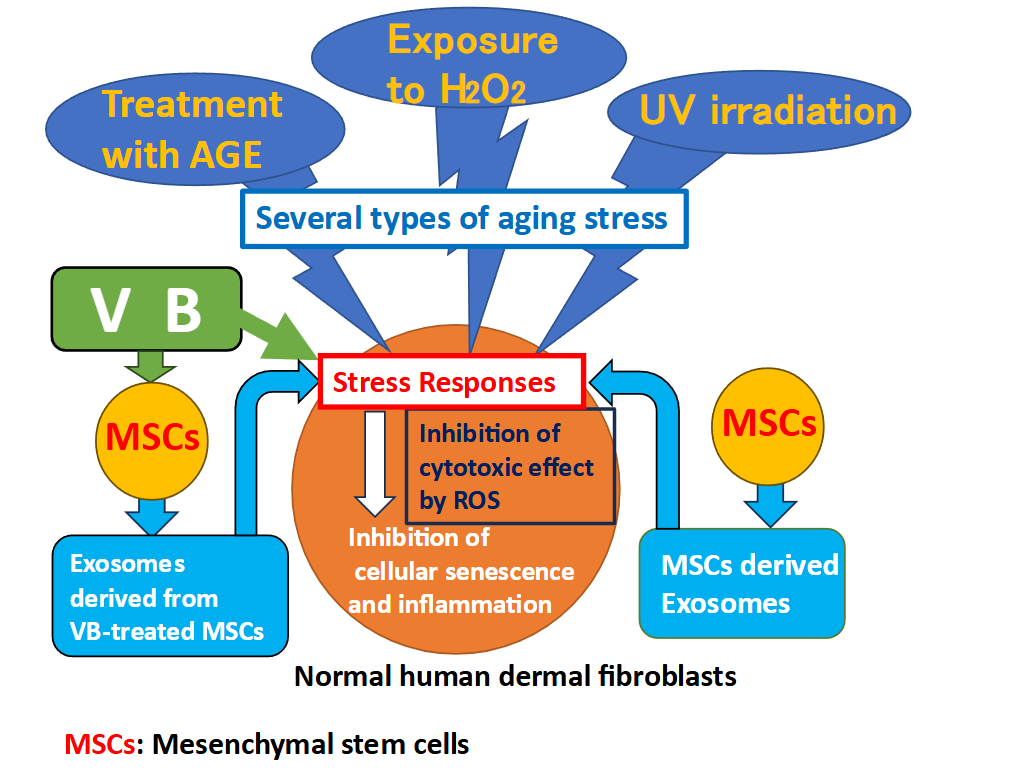
The main causes of skin aging include glycation (accumulation of advanced glycation end products [AGEs]), oxidation (irritation by ROS), and photoaging (exposure to UV rays), among many other environmental factors. These stressors put cells in a state of aging stress, which results in the accumulation of intracellular ROS, leading to cellular aging. Therefore, increasing the resistance to such stress that negatively affects cells is one way to fight skin aging, since suppressing the accumulation of intracellular ROS is important. It is believed that increasing mitochondrial energy metabolism and oxygen consumption, or producing antioxidants within cells, are effective in suppressing the accumulation of ROS. Cystine is the raw material for the synthesis of the intracellular antioxidant glutathione, which is taken up from outside the cell by the cystine transporter xCT and plays an important role in controlling the intracellular redox balance. Recently, in regenerative medicine, in vitro cultured stem cells have been incorporated into skin anti-aging research, and the skin anti-aging effects of extracellular vesicles, such as human mesenchymal stem cells-derived exosomes (MSCs-Exo), have been examined. Stem cells have been recognized as having a homing effect. When one receives rescue signals from damaged tissues or cells, it rushes to the affected area and regenerates the cells. Stem cell-derived exosomes transmit these signals and contribute to wound healing in the skin. However, it takes 3–4 months for stem cells to regenerate into tissues. Therefore, to apply this process to skin anti-aging, it is important to enhance the role of stem cell-derived exosomes so that they can act earlier.
We have been studying the biological activities related to the anti-aging effects of polyoxometalates (PMs). In particular, we investigated the effect of PMs VB1: VOSO4, VB2: K11H[(VO)3(SbW9O33)2] 27H2O, and VB3: Na2[SbW9O34]・19H2O, which have proven the antibacterial and antiviral activities. These have been incorporated into hand towels and other products, but we are incorporating them into cosmetics so that they can be used not only for hand hygiene, but also on the whole body. We are currently investigating the mechanism of action of PMs on skin cells, and have already found that PMs increase the expression of AGE receptors (present on the surface of skin fibroblasts), which participate in cytoprotection against glycation (AGE) stress, and suppress ROS that accumulate in cells due to oxidative (H2O2) stress.
In this study, we examined whether three types of VB (VB1, VB2, and VB3) enhanced the resistance of cultured human dermal fibroblasts (HDFs) to aging stress, including glycation (AGE loading), oxidation (H2O2 stimulation), and photoaging (UV irradiation) by measuring the expression levels of collagen, elastin and hyaluronic acid synthase, which were used as indicators of stress response in skin cells. We also examined the effect of exosomes secreted by human mesenchymal stem cells (MSCs) treated with VBs (VBs-MSCs-Exo). Furthermore, to verify the suppression mechanism of intracellular ROS levels by VBs and the exosomes, we measured their effects on mitochondrial oxygen consumption rate (OCR) and cystine uptake, which is involved in glutathione synthesis. The purpose of this study was to clarify whether VBs can enhance the initial resistance of human skin to aging stress. Furthermore, we aimed to demonstrate whether the addition of VBs to MSCs would induce the secretion of exosomes with different properties compared to those normally secreted, and whether the effects of preconditioning would enhance resistance to aging-related stresses.
Materials and methods
POLYOXOMETALATES
Vanadyl sulfate (VB1) was purchased from Fujifilm Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Bulk powders of VB2: K11H[(VO)3(SbW9O33)2] 27H2O and VB3: Na2[SbW9O34] 19H2O were synthesized using a patented method (International Patent Publication Number: WO2019/230210). Each bulk powder was ground in a mortar, dissolved in ultrapure water, and the solution was filtered through a 0.45 μm filter before use in the experiment. The concentration of VB added was examined in the range of 10 to 300 μg/ml, but here only the results obtained from stimulation with 100 μg/ml were shown.
REAGENTS
DL-Glyceraldehyde and H2O2 were purchased from FujiFilm Wako Pure Chemical Industries, Ltd. Bovine serum albumin (BSA) (fraction V) was purchased from SIGMA (St. Louis, MO, USA).
GLYCATION OF BOVINE SERUM ALBUMIN
Glycation of BSA was performed according to the method of Maeda et al. Briefly, BSA (25 mg/mL) was dissolved in 0.2 M phosphate buffer (pH 7.4) and reacted with 0.1 M DL-glyceraldehyde for seven days. The glyceraldehyde that did not bind to BSA was removed using a PD-10 gel filtration column equilibrated with phosphate-buffered saline. Glycosylated BSA in the obtained samples was quantified using a glyceraldehyde-derived AGE ELISA kit.
OXIDATIVE STRESS REAGENT
Hydrogen peroxide (FUJIFILM Wako Pure Chemical Corporation) was added to the culture medium by diluting the stock solution with purified water to a final concentration of 0.2 mM.
CELL CULTURE
Human dermal fibroblasts (HDFs) derived from juvenile foreskin (C-12300; PromoCell, Heidelberg, Germany) were cultured in a dedicated fibroblast growth medium (C-23010, PromoCell). Mesenchymal stem cells were purchased from PromoCell and cultured in a serum-free medium (opti-MEM). In the experiment in which exosomes were added, HDFs were cultured in advance in the same serum-free medium (opti-MEM) as MSCs and then used in the experiment.
PREPARATION OF MESENCHYMAL STEM CELLS-DERIVED EXOSOMES
Mesenchymal stem cells were cultured with or without 100 μg/ml VBs for 18 h in a serum-free medium (opti-MEM). The culture supernatants were collected and used as MSCs-derived exosomes (MSCs-Exo) in the experiments shown in Fig. 7 and 8. In the experiment investigating the effect of exosomes on mitochondrial OCR (Fig. 9), the culture supernatants were subjected to the miRCURY exosome isolation kit (product#: 300102; Exiqon, Hovedstaden, Denmark), and the purified exosomes were used.
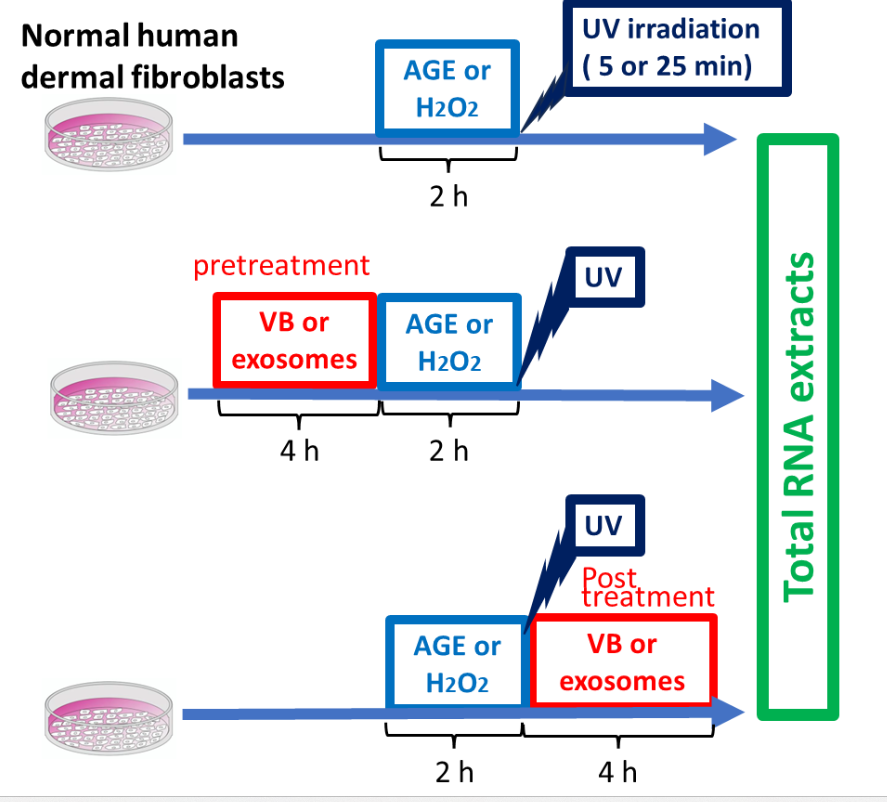
MEASUREMENT OF STRESS-RESPONSIVE MESSENGER RNA LEVELS IN HUMAN DERMAL FIBROBLASTS SUBJECTED TO GLYCATION, OXIDATION, AND UV IRRADIATION
(1) Glycation stress: Human dermal fibroblasts were exposed to 100 mg/ml AGE for 2 h. The cells were treated with VBs for 4 h before or after stress loading, and total RNA was extracted.
(2) Oxidative stress: Human dermal fibroblasts were exposed to 0.2 mM H2O2 for 2 h. The cells were treated with VBs for 4 h before or after stress loading, and total RNA was extracted.
(3) Photoaging (UV) stress: Human dermal fibroblast culture plates were irradiated from the UV lamp for 5 or 25 min, and the plates were then quickly returned to a 37°C CO2 incubator to continue culturing. The UV irradiation dose was set to 2 J/cm2 (for 5 min) or 10 J/cm2 (for 25 min) using a UV intensity meter. The cells were treated with VBs for 4 h before or after UV irradiation, and total RNA was extracted.
Total RNA samples obtained from the three types of experiments were used as templates to measure the mRNA levels of three stress response parameters (collagen, elastin, and hyaluronic acid synthase) using qRT-PCR.
TOTAL RNA EXTRACTION
Total RNA was extracted using TRIzol reagent (Ambion, Austin, TX, USA) according to the manufacturer’s instructions.
QUANTITATIVE REVERSE TRANSCRIPTION POLYMERASE CHAIN REACTION
The mRNA expression levels of collagen, elastin, and hyaluronic acid synthase were determined using one-step quantitative reverse transcription polymerase chain reaction (qRT-PCR) with specific primers. Specifically, one-step PCR was performed in the same tube using the Luna Universal One-Step qRT-PCR Kit (New England Biolabs, Ipswich, MA, USA) and Thermal Cycler Dice Real Time System II (Takara Bio, Shiga, Japan). Reactions were performed according to the manufacturer’s instructions for the reagent kit. Delta Ct was calculated with an internal standard using the constantly expressed gene glyceraldehyde-3-phosphate dehydrogenase. Differences from experimental controls were determined using the delta delta Ct method and expressed as fold changes in mRNA expression.
Table 1. Primers for qRT-PCR.
| Gene | Primer | References |
|---|---|---|
| Collagen1A2 | Forward: CTGGACCTCCAGGTGTAAGC Reverse: TGGCTGAGTCTCAAGTCACG |
[26] |
| Elastin | Forward: GGCCATTCCTGGTGGAGTTCC Reverse: AACTGGCTTAAGAGGTTTGCCTCCA |
[27] |
| Hyaluronic acid synthase | Forward: CACGTAACGCAATTGGTCTTGTCC Reverse: CCAGTGCTCTGAAGGCTGTGTAC |
[26] |
| GAPDH | Forward: GACATGCCGCCTGGAGAAAC Reverse: AGCCCAGGATGCCCTTTAGT |
[26] |
EVALUATION OF INTRACELLULAR REACTIVE OXYGEN SPECIES LEVELS
Human dermal fibroblasts were cultured for 4 h with 100 μg/ml VB2, MSCs-Exo or VB2-MSCs-Exo, followed by treatment with 0.2 mM H2O2 for 2 h. The intracellular ROS levels were measured using the fluorometric method with dichlorofluorescein diacetate (DCF-DA; Invitrogen, Carlsbad, CA, USA). After labeling the cells of the experimental groups with 10 mM DCF-DA for 20 min at 37°C, we visualized the fluorescence emission of the cells using a fluorescence microscope and the levels of intracellular ROS were evaluated by measuring the fluorescence intensity at the excitation wavelength (525 nm) using an Agilent microplate reader (Agilent Technologies, Santa Clara, CA, USA). Furthermore, the positions of individual cells were identified by staining the nuclei with 4’,6-diamidino-2-phenylindole (DAPI).
EVALUATION OF MITOCHONDRIAL OXYGEN CONSUMPTION RATE
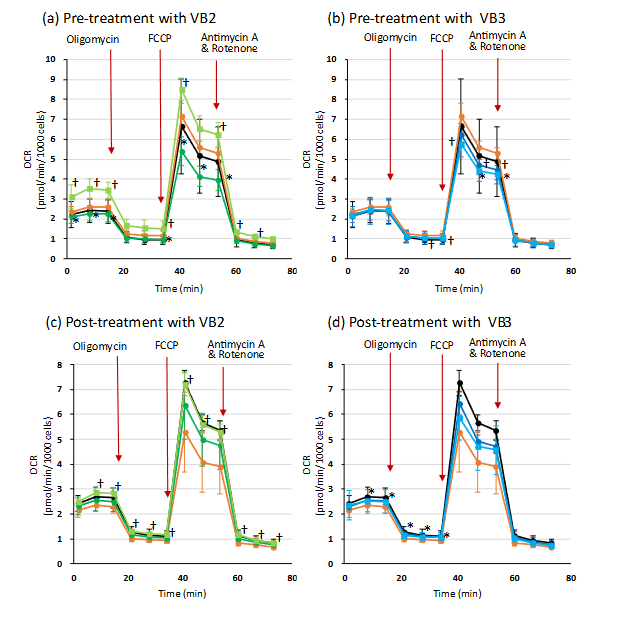
After stimulating with VB2 or VB3, with or without exosomes, before and after treating HDFs with H2O2, HDFs were suspended in phenol red-free XF RPMI medium (Agilent Technologies) containing 10 mM glucose, 1 mM pyruvate, and 2 mM glutamine and seeded at 1.2 × 104 cells/well in 8-well plastic plates (Agilent Technologies). The cells were incubated for 1 h at 37°C without CO2. Then, the oxygen consumption rate (OCR) was measured with an XF HS mini analyzer (Agilent Technologies) over time while sequentially adding 20 μM oligomycin (final concentration 2 μM), 20 μM Carbonyl Cyanide 4-(Trifluoromethoxy) phenylhydrazone (FCCP) (final concentration 2 μM), and 10 μM antimycin A/rotenone (final concentration 1 μM) according to the standard protocol of the Mito Stress Test kit.
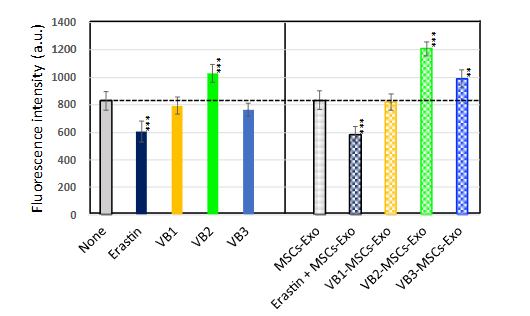
CYSTINE UPTAKE INTO CELLS
After treating HDFs with VBs or exosomes for 4 h, the relative amount of intracellular cystine was measured using a cystine uptake assay kit (Dojindo Chemical Industries, Ltd., Mashiki, Japan), according to the manufacturer’s instructions. Like cystine, the cystine analog (CA) is taken up into cells via xCT, a cystine transporter. After cell lysis, a reducing agent was added to reduce CA, which reacted specifically with the detection dye fluorescein O, O’-diacrylate (FOdA), generating a green fluorescent emission proportional to the amount of CA taken up by the cells. The fluorescence intensity was measured (Ex=485 nm, Em=535 nm) using a microplate reader (SYNERGY/HT; BioTek, Tokyo, Japan). Additionally, to clarify that the results obtained from this experiment depend on the cystine transporter, the effect of erastin, a transporter inhibitor, was also analyzed.
STATISTICAL ANALYSIS
In the qRT-PCR analysis, the difference from the control or stress stimulation alone was shown as delta delta Ct, and a difference of 2 or more (a difference of 4-fold or more at the mRNA level) was considered significant. All experiments, except for qRT-PCR analysis, were performed in triplicate. The data are presented as mean ± SD. Student’s t-test was performed, and statistical significance was set at p < 0.05.
Results
CHANGES IN MESSENGER RNA EXPRESSION OF THREE STRESS PARAMETERS IN HUMAN DERMAL FIBROBLASTS STIMULATED WITH VBS OR EXOSOMES DERIVED FROM MESENCHYMAL STEM CELLS THAT WERE TREATED WITH OR WITHOUT VBS
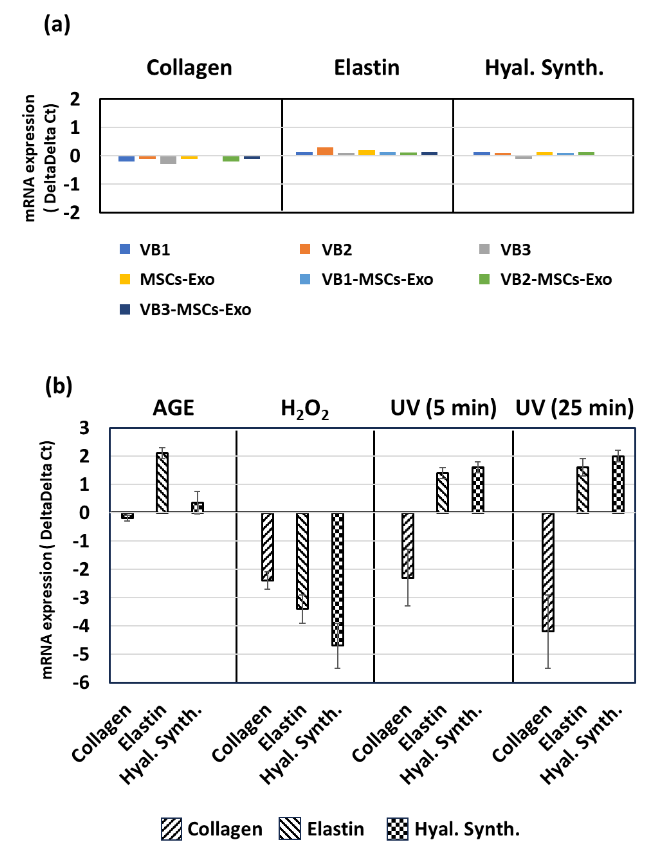
Four hours after VB1, 2, 3 and MSCs-Exo were added to HDFs, the mRNA expression levels of collagen, elastin, and hyaluronic acid synthase were all within ±1 compared to the untreated control level set to zero. Neither VBs nor MSCs-Exo, regardless of treatment of MSCs with VBs, affected the expression of stress parameters in HDFs.
REACTION OF HUMAN DERMAL FIBROBLASTS TO FOUR TYPES OF AGING STRESS (advanced glycation end products, hydrogen peroxide, or ultraviolet irradiation)
Human dermal fibroblasts were subjected to four types of aging stress, and the mRNA expression levels of the three stress parameters were evaluated. With AGE loading, elastin levels increased; however, no other significant changes were observed. After H2O2 treatment, all three parameters decreased. Upon UV irradiation, the expression of collagen mRNA decreased at both 5 and 25 min, while the expression of elastin and hyaluronic acid synthase tended to increase. The range of fluctuation was greater at 25 min.
RESPONSE OF HUMAN DERMAL FIBROBLASTS SUBJECTED TO GLYCATION STRESS (ADVANCED GLYCATION END PRODUCTS)
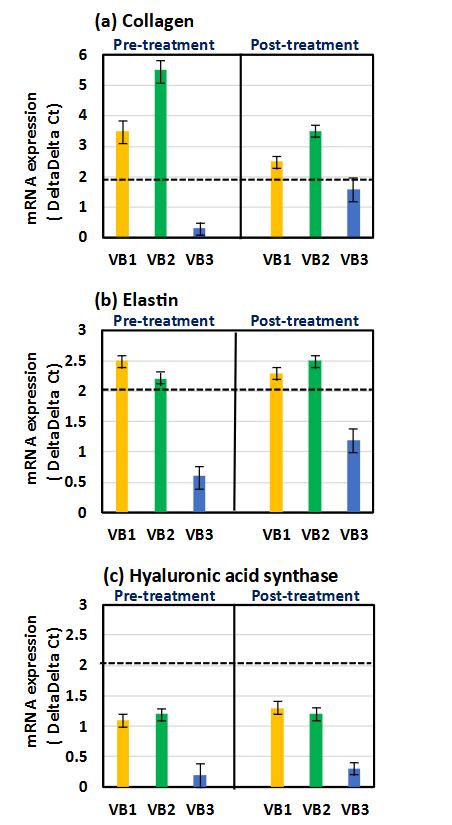
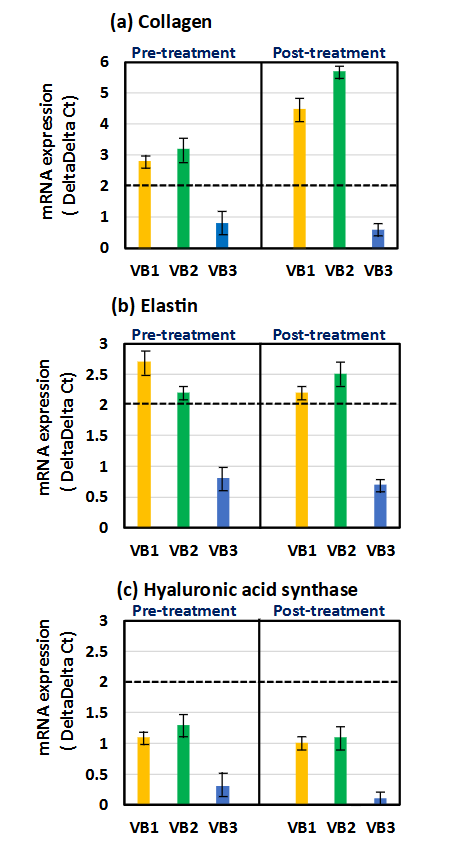
The levels of the three parameters in HDFs treated with the three types of VB for 4 h before or after the treatment with AGE for 2 h were compared with those in the group treated with AGE alone. Collagen levels were significantly increased by VB1 and VB2, both pre- and post-treatment. The strongest enhancement in expression of collagen mRNA was observed when stimulated with VB2, and this activity was similarly observed whether applied before or after AGE treatment. On the other hand, treatment with AGE alone increased elastin level. Both VB1 and VB2 had an ability to further enhance their expression before and after AGE treatment. The activity of VB3 was weaker compared to VB1 and VB2. Furthermore, it seemed that the expression of hyaluronic acid synthase was slightly increased by VB1 and VB2 than by VB3; however, the difference was considered not significant because the delta delta Ct value did not exceed 2.
RESPONSE OF HUMAN DERMAL FIBROBLASTS SUBJECTED TO OXIDATIVE STRESS (HYDROGEN PEROXIDE)
The levels of these three parameters were compared to those in the group treated with H2O2 alone. Both VB1 and VB2 nearly restored the reduced expressions of collagen and elastin mRNAs in HDFs under H2O2-induced stress conditions. Similarly, VB1 and VB2 increased the expression of hyaluronic acid synthase more strongly than VB3, but it seems that the expression level could not be restored to the original level.
RESPONSE OF HUMAN DERMAL FIBROBLASTS SUBJECTED TO PHOTOAGING STRESS (ULTRAVIOLET IRRADIATION)
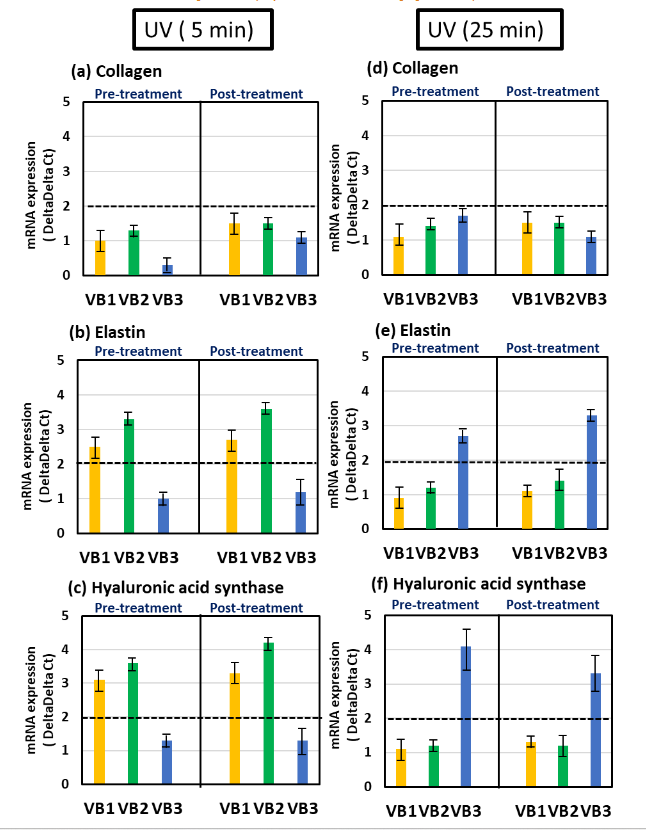
The levels of these three parameters were compared with those of the UV irradiation-only group at 5 and 25 min. In the stress condition of 5 min of UV irradiation, pre- and post-treatment with VB1 and VB2 tended to increase the expression of three parameters, with significant differences observed in the elastin and hyaluronic acid synthase. It appeared that all VBs had the ability to increase collagen mRNA expression levels, albeit weakly, but they were not able to restore the reduced expression caused by UV irradiation. In 25 min of UV irradiation, VB3 tended to increase the expression of these parameters, regardless of the timing of VB treatment, and elastin and hyaluronic acid synthase showed significant increases.
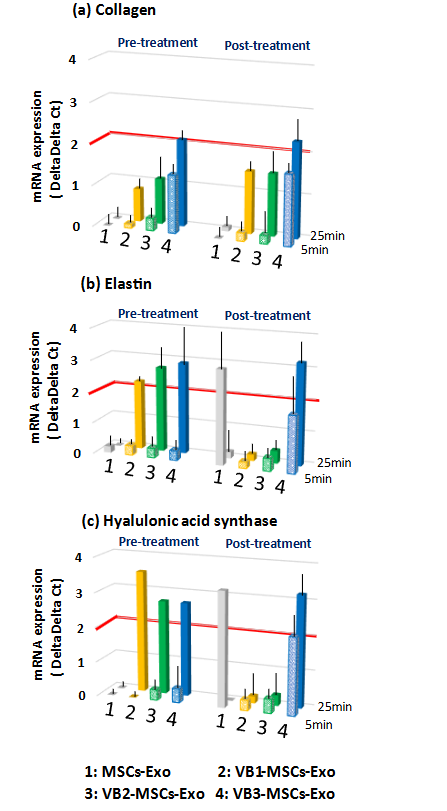
Furthermore, before and after 5 or 25 min of UV exposure, the levels of the three parameters were measured when HDFs were treated with exosomes — obtained by stimulating MSCs with VBs, and the results were compared with those of the UV stress alone group, 5 min, or 25 min. Compared to exosomes derived from unstimulated MSCs (MSCs-Exo), exosomes obtained through stimulation with VB1-3 tended to mitigate the decrease in collagen expression induced by 25 min UV irradiation, as opposed to 5 min. Especially, stronger activity was observed in the exosomes obtained by VB3 stimulation, regardless of whether it was pre-treatment or post-treatment. As shown in Fig. 3b, UV irradiation alone increased the expression of elastin and hyaluronic acid synthase, but VBs appear to further enhance these expressions. Slight increases in the expression of elastin and hyaluronic acid synthase were observed in 5 min UV irradiation when pre-treating HDFs with exosomes obtained from VB1-3 stimulation whereas post-treatment with VB3-MSCs-Exo showed a rather strong increase in these transcriptions. On the other hand, in 25 min UV irradiation, pre-treatment with exosomes obtained from VB1-3 stimulation strongly enhanced the expression of elastin and hyaluronic acid synthase, whereas the activities of VB1 and VB2 were not detected in the post-treatment. Post-treatment with exosomes obtained from VB3 stimulation similarly showed an enhancement in their expressions. Post-treatment with untreated MSCs-derived exosomes (MSCs-Exo) showed a strong increase in elastin and hyaluronic acid synthase in 5 min UV irradiation, but the effect disappeared in 25 min UV irradiation.
INHIBITORY EFFECT OF VB2 AND EXOSOMES ON INTRACELLULAR REACTIVE OXYGEN SPECIES ACCUMULATION IN HUMAN DERMAL FIBROBLASTS SUBJECTED TO OXIDATIVE STRESS
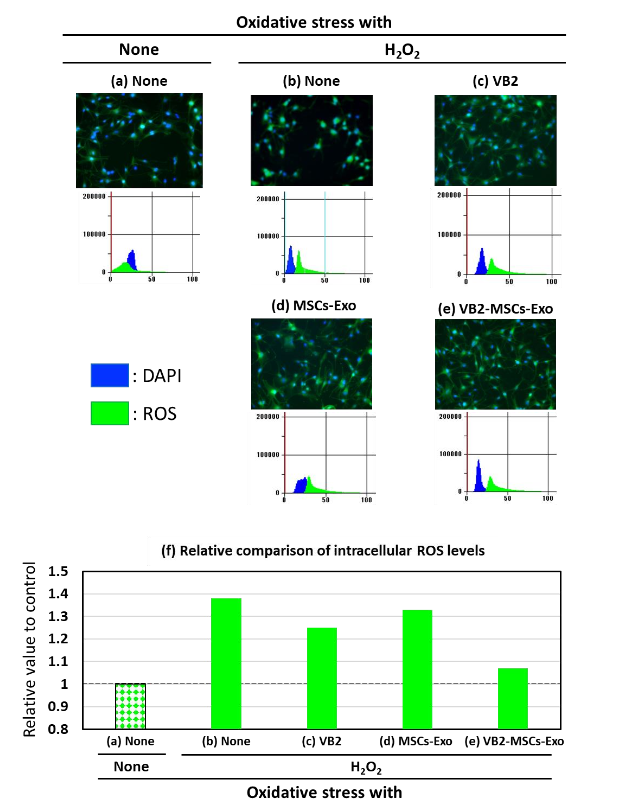
It is important to examine the regulation of collagen, elastin and hyaluronic acid synthase expressions when analyzing the anti-aging effects of VBs on the skin. Therefore, in this experiment, we investigated the impact of VB2 pre-treatment on intracellular ROS levels, which are considered crucial for regulating the expression of those three parameters. Indeed, it has been shown that under stress conditions, ROS accumulation in cells increased by up to 370% compared to the control, which was set at 100%. In addition, VB2 was used in this experiment because, as shown in Fig. 5, it has strong activity in mitigating the decrease in the expression of three types of parameters. In oxidative stress condition caused by H2O2, a relative increase in intracellular ROS levels was observed. However, VB2 suppressed this increase.
EFFECTS ON MITOCHONDRIA OXYGEN CONSUMPTION RATE
The results shown in Fig. 8 indicate that intracellular ROS levels increase in oxidative stress conditions, and that VB2 has the ability to suppress this increase. Therefore, to investigate whether this inhibitory effect was due to the suppression of ROS production by VB2, we first measured mitochondrial OCR in cells pre-treated or post-treated with VB2 alone, or with VB2 plus MSCs-Exo, in oxidative stress condition using a flux analyzer. In this experiment, we also examined the activity of VB3, which showed lower inducing ability for the expression of three types of parameters in the stress condition, as shown in Fig. 5. By following the experimental protocol and adding FCCP reagent, the maximum OCR of mitochondria was induced. The patterns of OCR changes in cells stimulated with VBs and MSCs-Exo in oxidative stress conditions were similar to those of the control group. There were no distinctive differences that explained the variations in intracellular ROS levels observed when stimulated with VBs or MSCs-Exo. Furthermore, the data observed before the addition of oligomycin, up to 20 minutes after the start of the experiment, reflects the OCR values induced by stimulation with VBs and MSCs-Exo. A slight increase in data was observed in the pre-treatment group with VB2 and MSCs-Exo, but otherwise, there were almost no differences. Based on these results, it was considered that VBs have little direct impact on mitochondrial OCR.
EFFECTS ON CYSTINE UPTAKE IN HUMAN DERMAL FIBROBLASTS
To further examine the mechanism of antioxidant action of VBs, which suppressed accumulation of intracellular ROS, we measured the amount of cystine taken up by the cells in non-stress conditions as a raw material for glutathione, an intracellular antioxidant. The amount of cystine was significantly decreased by treatment with erastin, an inhibitor of cystine transporter xCT. In contrast, VB2 and exosomes derived from MSCs stimulated with both VB2 and VB3 (VB2-MSCs-Exo and VB3-MSCs-Exo) exhibited a significant ability to increase intracellular cystine levels, whereas VB1 or VB1-MSCs-Exo did not exhibit such activity. These results suggest that the reduction of intracellular ROS by VB2 or exosomes may be due to the antioxidant effect of increased glutathione production, which results from the enhanced uptake of cystine.
Discussion
We investigated the effect of VBs on the cellular responses of HDFs exposed to aging-inducing stress, and the effectiveness of adding VBs to human MSCs. We hypothesized that the resulting exosomes (VBs-MSCs-Exo) would have different and more beneficial properties than those normally secreted. Glycation (AGE), oxidation (H2O2), and UV irradiation (5 or 25 min) were administered to HDFs to induce aging-related stress responses. Several types of VB or exosomes were added before and after treatment. The resulting effects were verified by measuring the mRNA levels of extracellular matrix components (collagen, elastin, and hyaluronic acid synthase), which are important for maintaining skin homeostasis.
When VBs and various types of VBs-MSCs-Exo were added to HDFs that had not been subjected to stress, the number of parameters did not change, indicating that VBs and VBs-MSCs-Exo did not affect normal cellular functions. On the other hand, depending on the parameter, the damage caused by various stresses may increase with a decrease in mRNA levels. Conversely, in other cases, resistance may be enhanced by an increase in mRNA levels. Therefore, changes in these three parameters in various aging-inducing stresses were examined. Collagen mRNA levels decreased in response to these stressors. Additionally, since all three parameters showed a decrease in response to H2O2, it is thought that the damage caused by the 0.2 mM H2O2 used in this study was strong or caused rapid damage. Furthermore, the increases in certain parameters in response to stress (elastin vs. AGE, elastin and hyaluronic acid synthase vs. UV irradiation) are thought to be due to a defense response or repair action. Therefore, using the changes in the expression levels of these extracellular matrix components as an indicator, we examined whether VB and exosomes have the ability to enhance the stress response involving skin fibroblasts.
Compared with the expression of their parameters in HDFs in AGE-induced stress condition, pre- and post-treatment with VB1 and VB2 increased the expression of collagen and elastin and slightly increased the expression of hyaluronic acid synthase. It seemed that VB3’s performance was slightly inferior compared to VB1 and VB2. In addition, in stress conditions induced by H2O2, the expression levels of collagen and elastin were largely suppressed. However, VB1 and VB2 mostly restored these mRNA levels in both pre- and post-treatment, whereas the expression level of hyaluronic acid synthase was not fully restored. The expression of elastin and hyaluronic acid synthase increased only with UV irradiation in HDFs. These expressions were also enhanced by treatment with VBs; however, the degree of increase differed depending on the irradiation time (5 and 25 min). At 5 min, VB1 and VB2 significantly increased the expression of elastin and hyaluronic acid synthase, but at 25 min, VB3, but not VB1 or VB2, increased the expression of those parameters. On the other hand, VB1-3 could induce but not fully recover the expression of collagen in UV-exposed HDFs. Furthermore, the expression of elastin and hyaluronic acid synthase was especially increased in HDFs treated with VBs-MSCs derived exosomes in 25 min UV irradiation. Particularly, VB1- and VB2-MSCs derived exosomes exhibited their activities only when HDFs were pre-treated. On the other hand, VB3-MSCs derived exosomes showed a significant increase regardless of pre-treatment or post-treatment.
As a mechanism of aging, it has been reported that cells are stressed by exposure to stressors such as glycation, oxidation, UV irradiation, and chemicals. These stressors cause ROS to accumulate in the cells, leading to the aging phenomenon. On the other hand, we have already found that VB2 and VB3 have the ability to inhibit ROS accumulation that was induced by H2O2. In this study, we also demonstrated that pretreating cells with VB2 or exosomes derived from mesenchymal stem cells stimulated with VB2 reduced the accumulation of intracellular ROS in HDFs cultured in H2O2-induced stress conditions. Since the ROS detection kit allows for the identification of relative cell numbers within the measurement area through DAPI staining, it is possible to accurately evaluate the increase and decrease of ROS levels per cell. The DAPI staining images of each experimental group showed similar patterns, demonstrating that there were comparable numbers of cells within the measurement area. Based on these findings, it is suggested that VB2 and the exosomes obtained after VB2 stimulation have the ability to inhibit the accumulation of ROS and exert anti-aging effects on cells. To investigate whether VBs and MSCs derived exosomes confer resistance to the aging process by suppressing the production of ROS, we measured the effect of VBs and exosomes on mitochondrial OCR. As a result, no clear inhibitory effect on mitochondrial OCR was observed, suggesting that VBs and exosomes do not exhibit stress responses by suppressing ROS production.
There are several reports indicating that H2O2 treatment and UV irradiation lead to the accumulation of ROS, with stress-induced damage being repaired by antioxidants. As a mechanism for VB-induced resistance to aging stress and suppression of ROS, we focused on the production of intracellular antioxidants. We showed that VB2 itself, as well as exosomes obtained by VB2 or VB3 stimulation, increase cystine uptake and may suppress the accumulation of ROS through glutathione synthesis.
Bannai et al. first reported a cystine transport system in cultured cells. This transport system, called the xc-system, is strongly induced by various stimuli, including ROS, and has been shown to increase intracellular glutathione levels. Glutathione is a major endogenous antioxidant composed of glutamate, cysteine, and glycine. If the activity of the xc-system is inhibited or extracellular cystine is removed, intracellular glutathione levels drop rapidly, and cells die. In this study, to demonstrate that the increased uptake of cystine by VBs and exosomes is due to their action on the cystine transporter, we aimed to analyze cystine analogues taken up into cells via the cystine transporter. As the results indicate, the increase in cystine uptake via the xCT transporter by VB2 alone or exosomes derived from VB2- and VB3-treated MSCs is strongly related to their antioxidant effects and the suppression of ROS.
Some cancer cells exhibit strong xCT expression, resulting in high intracellular glutathione levels. It is expected that this will become one of the treatments for brain tumors in the future.
Results indicated that VB1 and VB2 showed direct effects on HDFs to resist aging stresses. Furthermore, VB2, in addition to VB3, might exert its effect through the production of exosomes from MSCs. On the other hand, VBs-MSCs-exosomes are contained in the culture supernatant obtained by stimulating MSCs with VBs. However, it is not clear whether the VBs used for cell stimulation have been removed from the preparations containing exosomes. Therefore, further analysis is needed to determine whether the activity observed in VB-MSCs-Exo is due to changes in the content of exosomes induced by VB stimulation, or simply the result of simultaneous stimulation by VB and normally produced exosomes.
This study revealed that VB enhances the ability of cells to resist various early aging stresses and may have a preventive effect. In the future, we plan to investigate the ability to improve or eliminate established aging cells, which is a desirable effect. In recent years, it has been suggested that senescent cells in skin tissues may be eliminated by signals (JAG1) sent from surrounding cells, after which a request (epidermal growth factor) is sent to surrounding stem cells to regenerate and replenish skin cells. Phagocytic ability is increased by activating macrophages, which leads to an increase in the expression of the scavenger receptor STAB1. As a result, senescent skin cells may be phagocytosed, and activated macrophages may secrete fibroblast growth factor.
Conclusions
VB was shown to have no specific effect on normal HDFs, but it enhanced the resistance of cells subjected to aging stress. It is likely that this effect was stronger in VB2 and VB1; however, VB3 appeared to act on MSCs to promote stress resistance through the exosomes secreted. Therefore, it has been suggested that a combination of these VB types may exert a multifaceted anti-aging effect. It is probable that one of the mechanisms involves enhancing the synthesis of the antioxidant glutathione in cells by increasing cystine uptake.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding Statement
This study received no external funding.
Acknowledgments
The authors express their sincere gratitude to Dr. Keita Takanashi (Yokohama Pharmaceutical University) for technical guidance regarding the XF assay. They are also grateful to the FSX Company and those engaged in VB-compounded hygienic products for their generous cooperation.
References
- Bodnar AG, Ouellette M, Frolkis M, et al. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279(5349):349-352. doi:10.1126/science.279.5349.349
- Parrinello S, Samper E, Krtolica A, Goldstein J, Melov S, Campisi J. Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat Cell Biol. 2003;5(8):741-747. doi:10.1038/ncb1024
- Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585-621. doi:10.1016/0014-4827(61)90192-6
- Watanabe S, Kawamoto S, Ohtani N, Hara E. Impact of senescence-associated secretory phenotype and its potential as a therapeutic target for senescence-associated diseases. Cancer Sci. 2017;108(4):563-569. doi:10.1111/cas.13184
- Baker DJ, Childs BG, Durik M, et al. Naturally occurring p16(Ink4a)-positive cells shorten healthy lifespan. Nature. 2016;530(7589):184-189. doi:10.1038/nature16932
- Chaib S, Tchkonia T, Kirkland JL. Cellular senescence and senolytics: the path to the clinic. Nat Med. 2022;28(8):1556-1568. doi:10.1038/s41591-022-01923-y
- Shin SH, Lee YH, Rho N-K, Park KY. Skin aging from mechanisms to interventions: focusing on dermal aging. Front Physiol. 2023;14:1195272 [review]. doi:10.3389/fphys.2023.1195272
- Wang Y, Branicky R, Noë A, Hekimi S. Superoxide dismutases: dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 2018;217(6):1915-1928. doi:10.1083/jcb.201708007
- Sharma A, Smith HJ, Yao P, Mair WB. Causal roles of mitochondrial dynamics in longevity and healthy aging. EMBO Rep. 2019;20(12):e48395. doi:10.15252/embr.201948395
- Ploumi C, Daskalaki I, Tavernarakis N. Mitochondrial biogenesis and clearance: a balancing act. FEBS Journal. 2017;284(2):183-195. doi:10.1111/febs.13820
- Takahashi A, Ohtani N, Yamakoshi K, et al. Mitogenic signalling and the p16INK4a-Rb pathway cooperate to enforce irreversible cellular senescence. Nat Cell Biol. 2006;8(11):1291-1297. doi:10.1038/ncb1491
- Bannai S, Kitamura E. Transport interaction of L-cystine and L-glutamate in human diploid fibroblasts in culture. J Biol Chem. 1980;255(6):2372-2376. doi:10.1016/S0021-9258(19)85901-X
- Sato H, Tamba M, Ishii T, Bannai S. Cloning and expression of a plasma membrane cystine/glutamate exchange transporter composed of two distinct proteins. J Biol Chem. 1999;274(17):11455-11458. doi:10.1074/jbc.274.17.11455
- Lv J, Yang S, Lv M, Lv J, Sui Y, Guo S. Protective roles of mesenchymal stem cells on skin photoaging: a narrative review. Tissue Cell. 2022;76:101746. doi:10.1016/j.tice.2022.101746
- Chen B, Sun Y, Zhang J, et al. Human embryonic stem cell-derived exosomes promote pressure ulcer healing in aged mice by rejuvenating senescent endothelial cells. Stem Cell Res Ther. 2019;10(1):142. doi:10.1186/s13287-019-1253-6
- Weiliang Z, Lili G. Research advances in the application of Adipose-derived stem cells derived exosomes in cutaneous wound healing. Ann Dermatol. 2021;33(4):309-317. doi:10.5021/ad.2021.33.4.309
- An Y, Lin S, Tan X, et al. Exosomes from adipose-derived stem cells and application to skin wound healing. Cell Prolif. 2021;54(3):E12993. doi:10.1111/cpr.12993
- Dan K, Yeh H-L. Biological activity of polyoxometalates and their applications in anti-aging. Med Res Arch. 2024;12(12). doi:10.18103/mra.v12i12.6108
- Dan K, Katoh N, Matsuoka T, Fujinami K. In vitro antimicrobial effects of virus block, which contains multiple polyoxometalate compounds, and hygienic effects of virus block-supplemented moist hand towels. Pharmacology. 2019;104(1-2):1-15. doi:10.1159/000500897
- Dan K, Fujinami K, Sumitomo H, et al. Application of antiviral polyoxometalates to living environments—antiviral moist hand towels and stationery items. Appl Sci. 2020;10(22):8246. doi:10.3390/app10228246
- Fujinami K, Dan K, Tanaka-Kagawa T, Kawamura I. Anti-aging effects of polyoxometalates on skin. Applied Sciences. 2021;11(24):11948. doi:10.3390/app112411948
- Eckes B, Mauch C, Hüppe G, Krieg T. Differential regulation of transcription and transcript stability of pro-alpha 1 (I) collagen and fibronectin in activated fibroblasts derived from patients with systemic scleroderma. Biochem J. 1996;315(2):549-554. doi:10.1042/bj3150549
- Hwang K-A, Yi B-R, Choi K-C. Molecular mechanisms and in vivo mouse models of skin aging associated with dermal matrix alterations. Lab Anim Res. 2011;27(1):1-8. doi:10.5625/lar.2011.27.1.1
- Maeda S, Matsui T, Ojima A, Takeuchi M, Yamagishi S-I. Sulforaphane inhibits advanced glycation end product-induced pericyte damage by reducing expression of receptor for advanced glycation end products. Nutr Res. 2014;34(9):807-813. doi:10.1016/j.nutres.2014.08.010
- Kartal B, Akçay A, Palabiyik B. Oxidative stress upregulates the transcription of genes involved in thiamine metabolism. Turk J Biol. 2018;42(5):447-452. doi:10.3906/biy-1801-51
- Liu W, Ma C, Li H-Y, Chen L, Yuan S-S, Li K-J. MicroRNA-146a downregulates the production of hyaluronic acid and collagen I in Graves’ ophthalmopathy orbital fibroblasts. Exp Ther Med. 2020;20(5):38. doi:10.3892/etm.2020.9165
- Deslee G, Woods JC, Moore CM, et al. Elastin expression in very severe human COPD. Eur Respir J. 2009;34(2):324-331. doi:10.1183/09031936.00123008
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods. 2001;25(4):402-408. doi:10.1006/meth.2001.1262
- Lyons SA, Chung WJ, Weaver AK, Ogunrinu T, Sontheimer H. Autocrine glutamate signaling promotes glioma cell invasion. Cancer Res. 2007;67(19):9463-9471. doi:10.1158/0008-5472.CAN-07-2034
- N. E. Savaskan, Heckel A, Hahnen E, et al. Small interfering RNA-mediated xCT silencing in gliomas inhibits neurodegeneration and alleviates brain edema. Nature Med. 2008;14(6):629-632. doi:10.1038/nm1772
- Zhang X, Lan L, Niu L, et al. Oxidative stress regulates cellular bioenergetics in esophageal squamous cell carcinoma cell. Biosci Rep. 2017;37(6):BSR20171006. doi:10.1042/BSR20171006
- Yu J-X, Lin M, Zhang W-X, Lao F-X, Huang H-C. Astaxanthin prevents oxidative damage and cell apoptosis under oxidative stress involving the restoration of mitochondrial function. Cell Biochem Funct. 2024;42(8):e70027. doi:10.1002/cbf.70027
- McNish H, Mathapathi MS, Figlak K, Damodaran A, Birch-Machin MA. The effect of blue light on mitochondria in human dermal fibroblasts and the potential aging implications. FASEB J. 2025;39(11):e70675. doi:10.1096/fj.202500746R
- Hudson L, Rashdan E, Bonn CA, Chavan B, Rawlings D, Birch‐Machin MA. Individual and combined effects of the infrared, visible, and ultraviolet light components of solar radiation on damage biomarkers in human skin cells. FASEB J. 2020;34(3):3874-3883. doi:10.1096/fj.201902351RR
- Birch‐Machin MA, Russell EV, Latimer JA. Mitochondrial DNA damage as a biomarker for ultraviolet radiation exposure and oxidative stress. Br J Dermatol. 2013;169(Suppl 2):9-14. doi:10.1111/bjd.12207
- Yoshioka H, Yamada T, Hasegawa S, et al. Senescent cell removal via JAG1-NOTCH1 signalling in the epidermis. Exp Dermatol. 2021;30(9):1268-1278. doi:10.1111/exd.14361
- Ogata Y, Yamada T, Hasegawa S, et al. SASP-induced macrophage dysfunction may contribute to accelerated senescent fibroblast accumulation in the dermis. Exp Dermatol. 2021;30(1):84-91. doi:10.1111/exd.14205

