Histological Analysis of Urachal Remnants in Surgery
Histological Evaluation of Urachal remnants
Kyoko Baba 1,2*, Risako Ito 1, Yuki Ando 1, Ami Kuwabara 1, Yuta Nimi 1, Junpei Wato 1, Akira Takeda 1
- Department of Plastic Surgery, Kitasato University Medical Center; 6-100 Arai, Kitamoto, Saitama 364-8501, Japan
- Department of Plastic Surgery and Aesthetic Surgery, Kitasato University School of Medicine; 1-15-1 Kitasato, Minami ku, Sagamihara, Kanagawa 252-0374, Japan
*Correspondence: [email protected]
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Baba, K., Ito, R., et al., 2024. Histological Evaluation of Urachal remnants. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.6044
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6044
ISSN 2375-1924
ABSTRACT
Introduction: Urachal remnant disease occurs when the fetal urachus fails to close after birth. We perform surgical excision of umbilical sinus-type urachal remnants, aiming for complete resection to prevent residual urachal epithelium, which may become a source for urachal carcinoma. This study reports immunohistochemical findings from surgical specimens to assess epithelial involvement and inform resection strategies.
Materials and Methods: We reviewed cases of urachal remnant excision performed over one year at the Department of Plastic Surgery, Kitasato University Medical Center. Patient characteristics, including age, sex, symptoms, MRI findings, length of resected cord-like structures, and histopathological results, were examined. Histological evaluation included hematoxylin and eosin (H&E) staining and immunohistochemistry. Markers used were keratin AE1/AE3 and epithelial membrane antigen (EMA) for epithelial cells, factor VIII for vascular endothelium, α-smooth muscle actin (αSMA) for smooth muscle cells, and S-100 protein for nerve fibers.
Results: Five patients (3 males, 2 females; age range 16–69 years) underwent surgery for urachal remnant disease. All presented with peri-umbilical inflammation. Preoperative MRI identified tubular structures ranging from 1.2 to 6.4 cm, while the resected cord-like structures measured 6 to 11 cm. The resected specimens were 3 to 5.2 cm longer than the tubular structures seen on MRI, and no tubular structures were observed at the surgical margins. Three cases showed clear tubular structures on H&E staining, all of which were positive for keratin AE1/AE3 and EMA, with keratin AE1/AE3 showing stronger staining. The remaining two cases, without clear tubular structures, were negative for keratin AE1/AE3 and EMA. αSMA was positive in all cases, while no significant findings were noted with factor VIII or S-100 protein.
Discussion: The ideal resection range for urachal remnants remains unclear. In this study, resecting at least 2 cm beyond inflammatory signs during surgery, combined with intraoperative confirmation of the absence of tubular structures, resulted in complete excision. Immunohistochemical staining, particularly keratin AE1/AE3, was valuable in identifying residual epithelial components, while αSMA was useful for assessing remnant structures.
Keywords:
Urachal remnant, urachal epithelium, histological evaluation, keratin AE1/AE3, resection strategies.
INTRODUCTION
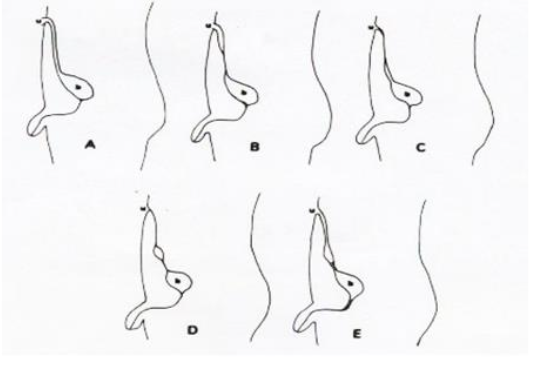
Urachal remnants are characterized by the persistence of the urachus, a fetal structure that fails to close postnatally. In our department, we perform surgical interventions on patients diagnosed with urachal sinus, classified as type B according to the Blichert-Toft classification (Figure 1), where the lesion is confined to the area between the umbilical fossa and the abdominal wall, without concomitant urological complications. Although rare, there have been reports of urachal carcinoma originating from the urachal epithelium, underscoring the necessity for complete excision of the urachal epithelium during surgery to prevent malignant transformation.
In our clinical experience, evaluating the presence of urachal epithelium at the resection margin using conventional hematoxylin and eosin (H&E) staining has proven difficult in certain cases. We have encountered specimens where the characteristic tubular structure of the urachal lumen was either unclear or absent. Even in cases where the tubular structure was visible, H&E staining alone was sometimes insufficient for definitive assessment of residual epithelial cells at the margin.
To address these challenges, we aimed to improve the precision of resection margin evaluation by incorporating immunohistochemical staining. To our knowledge, few studies have applied immunohistochemical techniques to evaluate the margins of urachal remnants. Our case report findings suggested that immunohistochemical staining provides a more reliable method for determining the presence of residual urachal epithelium at the resection margin. There have been several reports in pediatric urachal remnants where, despite clear clinical symptoms, the tubular structure could not be definitively identified using H&E staining. The exact histological mechanisms underlying urachal closure remain poorly understood. Furthermore, few studies have investigated urachal remnants using immunohistochemical techniques, leaving gaps in knowledge about other histological features that may indicate the disappearance of the urachus.
We conducted immunohistochemical staining not only for urachal epithelium but also for vascular endothelial cells, nerves, and smooth muscle, considering the embryological tissues that compose the urachus. This report presents our findings on the immunohistochemical analysis of resection margins in urachal remnants, representing one of the first studies to investigate adult urachal remnants using this method. Our results contribute to a more comprehensive understanding of the histopathological evaluation of urachal remnants and may help to enhance surgical management strategies.
MATERIALS AND METHODS
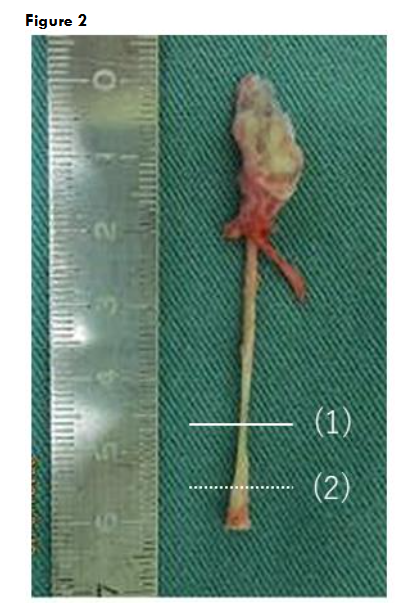
This study was conducted with the approval of our institutional ethics committee (Approval No. 2019012). We retrospectively reviewed cases that underwent surgery in our department of plastic surgery over a one-year period starting in March 20XX, using patient medical records. The parameters analyzed included patient age, sex, the presence or absence of periomphalitis, the distance between the umbilical fossa and the distal end of the tubular structure as observed on preoperative magnetic resonance imaging (MRI), the length of the excised specimen, and histopathological findings. Histological specimens were prepared from transverse sections of the tubular structure in each excised specimen.
In addition to hematoxylin and eosin (H&E) staining, immunohistochemical staining was performed. Immunostaining for keratin AE1/AE3 and epithelial membrane antigen (EMA) was used for epithelial cells, factor VIII for vascular endothelial cells, α-smooth muscle actin (αSMA) for smooth muscle cells, and S-100 protein (S100) for nerve fibers.
RESULTS
A total of five cases were analyzed, aged 16 to 69 years (3 males, 2 females). All cases had preoperative periomphalitis. The length from the umbilical fossa to the distal end of the tubular structure as observed on preoperative MRI ranged from 1.2 to 6.4 cm, while the length of the excised specimens ranged from 6 to 11 cm. The excised specimens were 3 to 5.2 cm longer than those visible on MRI. None of the excised specimens showed evidence of tubular structure at the resection margin. Details are presented in Table 1.
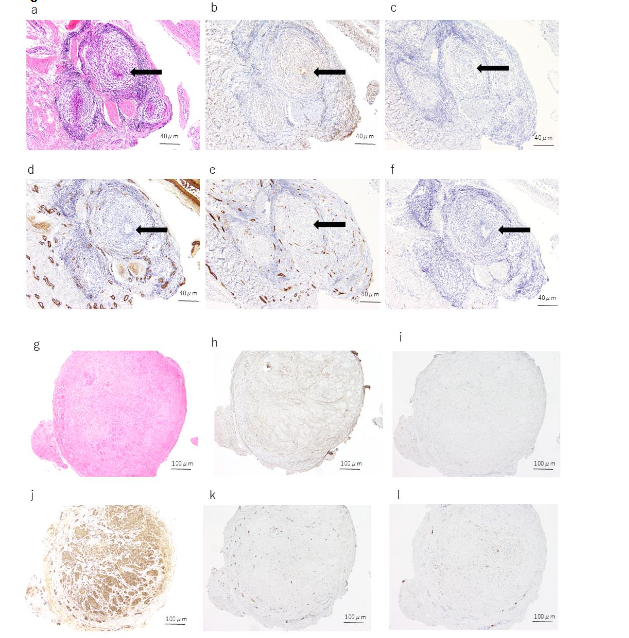
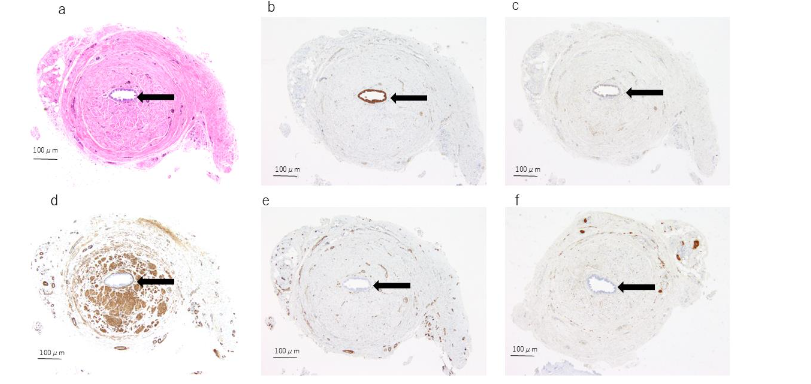
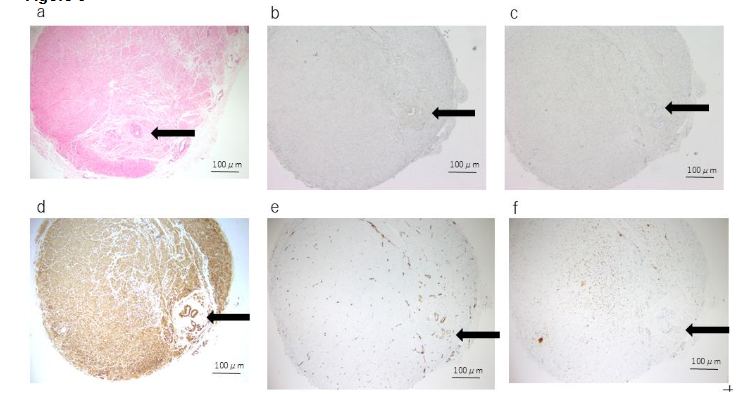
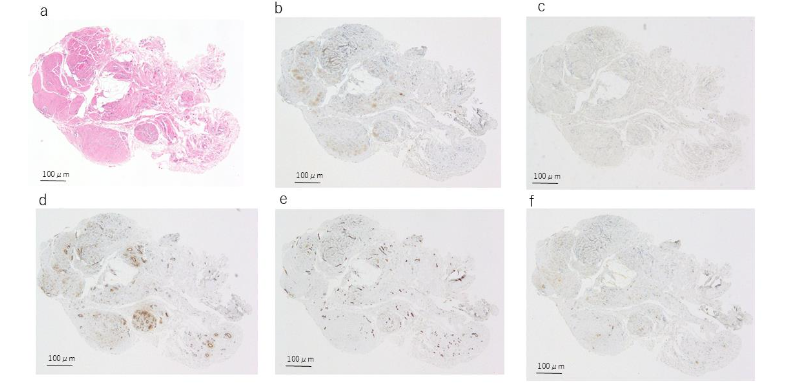
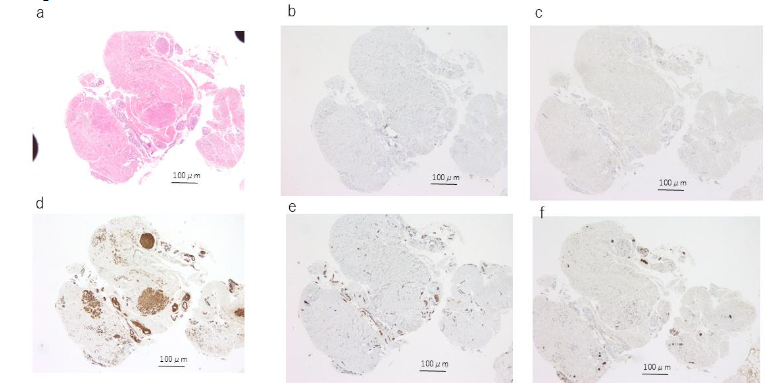
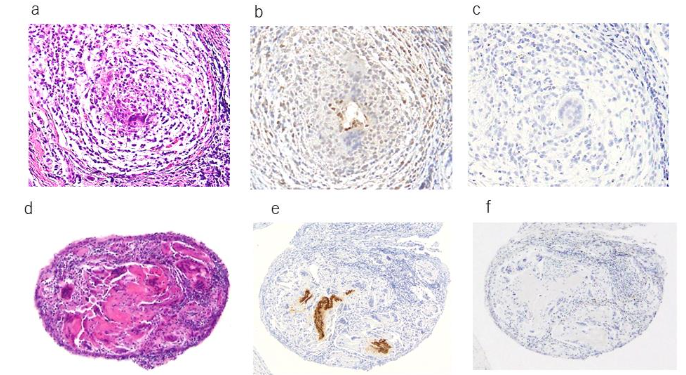
Histopathologically, three cases exhibited a distinct tubular structure on H&E staining, while no tubular structure was observed in the remaining two cases. In all three cases where a tubular structure was identified, the urachal epithelium was positive for both keratin AE1/AE3 and EMA. Among these three cases, two slices from two specimens contained cells that were stained with keratin AE1/AE3, although no distinct tubular structure was observed on H&E staining; however, no EMA-positive cells were noted in these slices. In all cases, no tubular structure or urachal epithelium was identified at the resection margin using H&E, keratin AE1/AE3, or EMA staining. αSMA staining was positive in all cases, with focal staining observed in cases without a tubular structure, while more extensive staining was seen in cases with a tubular structure. The staining patterns around the tubular structures varied; some specimens exhibited no staining around the tubular structure, while others showed staining extending close to the tubular structure. Factor VIII was positive in all cases but did not show any distinctive staining pattern. S-100 protein was positive in three cases, but no notable characteristics were observed.
DISCUSSION
DEVELOPMENT OF THE URACHUS
The urachus appears by approximately the 16th day of embryonic development as a diverticulum originating from the caudal end of the yolk sac, later extending into the umbilical stalk, eventually forming the umbilical cord. By the second month of embryonic development, the extraembryonic portion of the urachus regresses, while the intraembryonic portion continues to extend anteriorly and merges with the cloaca. As the cloaca differentiates into the bladder and descends into the pelvic cavity, the urachus elongates as a tubular structure extending from the anterior wall of the bladder. The epithelial lining of the urachus consists predominantly of transitional epithelium, with connective tissue in the middle layer and muscle tissue in the outer layer. The caudal portion of the urachus extends into the muscular layer of the bladder wall, while its cranial end connects with the umbilical ring, which contributes to the formation of the umbilical cord. The urachus is believed to regress once the bladder has matured and the ureters have fully formed, eventually becoming a narrow tube by the fourth or fifth month of fetal development and resulting in the formation of the median umbilical ligament.
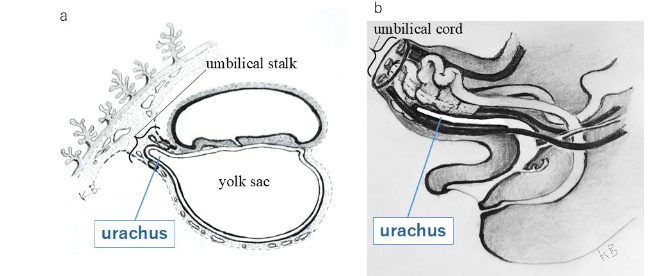
URACHAL REMNANT
Urachal remnants occur when the urachus, which normally closes after birth but remains patent, causing symptoms such as urinary leakage from the umbilicus, peri-umbilical inflammation, and abdominal pain. These symptoms may be present not only during infancy or childhood but can also first appear during adolescence or adulthood. Urachal remnants are reported to occur in 50% of fetuses immediately before birth and in 2% of adults. MRI is considered to be a useful diagnostic tool for identifying urachal remnants, with Blichert-Toft et al. proposing a classification system that categorizes urachal remnants into five types: (A) congenital patent urachus, (B) umbilical urachal sinus, (C) vesicourachal diverticulum, (D) urachal cyst, and (E) the alternating sinus, which develops as a result of infection of a urachal cyst. Reported incidence rates for these types are 15% for patent urachus, 49% for urachal sinus, and 36% for urachal cyst, though the clinical classification of these conditions can be difficult. At our department, the urachal sinus type (Blichert-Toft type B), which manifests with lesions in the abdominal wall and typically presents after adolescence, is the primary target of our treatment.
Although urachal carcinoma is rare, accounting for approximately 0.4% of all urological malignancies, the risk of developing urachal carcinoma must be considered. Most reported cases of urachal carcinoma involve either adenocarcinoma or transitional cell carcinoma, underscoring the importance of completely excising epithelial components to mitigate the risk of cancer development. As such, surgical excision remains the cornerstone of treatment. Preoperative assessments, including cystoscopy and cytological evaluation of urine, are conducted in collaboration with the urology department.
EVALUATION OF THE EXTENT OF RESECTION IN THIS STUDY
The appropriate extent of resection for urachal remnants has not been definitively established. In our practice, to minimize the risk of urachal carcinoma, we aim to achieve complete resection of the urachal epithelium. Our surgical focus is on cases of umbilical urachal sinus (type B), which often presents with inflammation in the umbilical area. During surgery, we prioritize two key points: (1) resecting at least 2 cm caudally from any adhesions or inflammatory signs (such as capillary dilation) of the peritoneum, and (2) confirming the absence of any tubular structures at the resection margin. However, we did not find any literature providing preoperative guidance for determining the extent of resection in our review. It remains unclear whether the urachus narrows conically and gradually disappears or forms a bottleneck before abruptly terminating. As such, it is difficult to determine the appropriate distance from the point of luminal disappearance at which resection should occur to ensure the complete removal of urachal epithelium. In this study, resection was performed at least 2 cm caudally from the site of peritoneal inflammation, and the urachus was excised at least 3 cm caudally from any tubular structures observed on MRI. Pathological examination confirmed the absence of residual urachal epithelium at the surgical margin. While our results suggest that these guidelines may serve as a reference for determining resection sites in cases of umbilical urachal sinus, the actual distance between the site of luminal disappearance and the resection site was not assessed in this study. Given that pathological specimens were prepared at approximately 5 mm intervals, we could not obtain statistically significant data. Furthermore, in cases of urachal remnants, the luminal structure may not always be clearly identifiable on MRI. Therefore, postoperative histopathological evaluation of the resection margin for residual urachal epithelium is recommended.
EVALUATION OF IMMUNOHISTOCHEMICAL STAINING IN THIS STUDY
While H&E staining is effective for identifying epithelial components when the tubular structure is clearly defined, detecting epithelial components becomes challenging as the lumen narrows. Therefore, we opted to evaluate immunohistochemical staining. For the detection of epithelial cells, we used keratin AE1/AE3 and epithelial membrane antigen (EMA), which are commonly used for epithelial staining due to their excellent staining capabilities. Keratin AE1/AE3 is an epithelial cocktail antibody, with AE1 reacting with type I cytokeratins and AE3 with type II cytokeratins, making it a highly versatile antibody. EMA is a glycoprotein that serves as a cell membrane antigen found in normal epithelial tissues but not in non-epithelial tissues. To date, no reports have evaluated the immunohistochemical staining of urachal epithelium, and no optimal staining methods have been established. It remains unclear whether staining both antibodies is necessary to confirm the presence of urachal epithelium, as there are no established precedents. Under these limitations, our comparison of the two staining methods suggests that keratin AE1/AE3 provides better visibility in this study. This implies that keratin AE1/AE3 may be more suitable for confirming the presence of epithelial components.
In this study, αSMA was positive in all previous cases, regardless of the presence of a luminal structure. Staining for the smooth muscle marker αSMA typically yields positive results for smooth muscle cells, myoepithelial cells, and myofibroblasts. The perivesical tissue is continuous with the umbilical cord during the fetal period, and mesenchymal stem cells are present in the umbilical cord collected at birth. However, the dynamics of cord-like structures, including the urachus during the perinatal period, and their relationship with the umbilical cord in the formation of the median umbilical ligament remain unresolved. It is intriguing to consider whether smooth muscle-like tissues contribute to the closure process of the urachus and whether these tissues are associated with mesenchymal stem cells. Given the hypothetical presence of vestigial urachal structures, αSMA may be a suitable marker for investigating their existence.
LIMITATIONS
This study was limited by a small sample size of only five cases, and no statistical analyses were performed.
CONCLUSIONS
We conducted a pathological and histological examination of the resection margins in five cases of urachal remnants. In cases of urachal sinus-type urachal excision, our findings suggest that complete resection without residual urachal epithelium may be achievable by excising the structure at least 3 cm caudally from the luminal structure observed on MRI and at least 2 cm caudally from any signs of peritoneal inflammation noted during surgery. Confirming the presence or absence of residual urachal epithelium through pathological examination is essential, and keratin AE1/AE3 staining may serve as a useful tool for this purpose.
Conflict of interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Author contributions
Conceptualization: Kyoko Baba
Methodology: Kyoko Baba
Validation: Yuuta Nimi, Junpei Wato
Investigation: Risako Ito, Yuki Ando, Ami Kuwabara
Resources: Kyoko Baba
Data curation: Kyoko Baba
Writing—original draft preparation: Kyoko Baba
Writing—review and editing: Akira Takeda
Supervision: Akira Takeda
Project administration: Akira Takeda
Funding acquisition: Kyoko Baba
All authors have read and agreed to the published version of the manuscript.
Ethics approval and consent to participate
This study was conducted with the approval of our institutional ethics committee (Approval No. 2019012). Informed consent was obtained from all subjects involved in the study.
References
- Larsen WJ. Human Embryology. Elsevier Health Sciences; 2002.
- Hammond G, Yglesis L, Davis JE. The urachus, its anatomy and associated fascia. Anat Rec. 1941; 80:271.
- Blichert-Toft M, Nielsen OV. Diseases of the urachus simulating intra-abdominal disorders. Am J Surg. 1971;122(2):123-128.
- Pedersen GL, Dahl C, Azawi NH. Non-invasive, low-grade papillary urothelial carcinoma in the urachus. BMJ Case Rep. 2013;2013: bcr2013200635.
- Fujiyama C, Nakashima N, Tokuda Y, Uozumi J. Squamous cell carcinoma of the urachus. Int J Urol. 2007;14(10):966-968.
- Soni HC, Marda S, Goswami KG, Vadvala H, Parekh K, Vadodaria H. Transitional cell carcinoma in urachal cyst. Abdom Imaging. 2010;35(6):764-766.
- Maletic V, Cerovic S, Lazic M, Stojanovic M, Stevanovic P. Synchronous and multiple transitional cell carcinoma of the bladder and urachal cyst. Int J Urol. 2008;15(6):554-556.
- Baba K, Kuwabara A. Efficacy of Keratin AE1/AE3 Staining in Evaluating Resection Margins of a Urachal Remnant and Improving Surgical Precision and Outcomes: A Case Report. Cureus. 2024;16(6):e63197.
- Kishi Y, Kikuchi K, Ogushi K, et al. Indications for Delayed Excision of Urachal Remnants. The Japanese Society of Pediatric Surgeons (JSPS). 2018;54(2):236-241.
- Obatake M, Nishijima E, Takamizawa S, et al. Vitelline Duct Remnants Associated with Omphalitis. The Japanese Society of Pediatric Surgeons (JSPS). 2003;39:22-27.
- Moore KL, Persaud TVN, Mark G. The Developing Human: Clinically Oriented Embryology. 11th ed. Elsevier; 2018.
- Wilson AL, Gandhi J, Seyam O, et al. Urachal anomalies: a review of pathological conditions, diagnosis, and management. Transl Res Anat. 2019; 16:100041.
- Hammond G, Yglesis L, David JE. The urachus, its anatomy and associated fascia. Anat Rec. 1941; 80:271.
- Cilento BG Jr, Bauer SB, Retik AB, et al. Urachal anomalies: defining the best diagnostic modality. Urology. 1998;52(1):120-122.
- Naiditch JA, Radhakrishnan J, Chin AC. Current diagnosis and management of urachal remnants. J Pediatr Surg. 2013;48(10):2148-2152.
- Higashimori T, Sakurai H. Immediate Umbilical Reconstruction for Treatment of Urachal Cyst. Keisei Geka. 2013;56:59-66.
- Patel J, Villegas A. Urachal adenocarcinoma: a rare primary cancer managed with FOLFOX chemotherapy. Cureus. 2023;15:e43849. doi:10.7759/cureus.43849
- Begg RC. The colloid adenocarcinoma of the bladder vault arising from the epithelium of the urachal canal. Br J Surg. 1931; 18:422-466.
- Baba K, Ishiguro M, Takeda A, et al. A case of infected urachal sinus with paralytic ileus. Japanese Soc Occup Med Traumatol. 2014;62:128-132.
- Itoh T. Immunohistochemistry in diagnostic surgical pathology. Kenbikyo. 2013;48:33-38. doi:10.11410/kenbikyo.48.1_33
- Sloane JP, et al. An assessment of the value of epithelial membrane antigen and other epithelial markers in solving diagnostic problems in tumour histopathology. Histochem J. 1983; 15:645-654.
- Baba K, Yamazaki Y, Ikemoto S, et al. Osteogenic potential of human umbilical cord-derived mesenchymal stromal cells cultured with umbilical cord blood-derived autoserum. J Cranio-Maxillofac Surg. 2012; 40:768-772.
- Sarugaser R, Lickorish D, Baksh D, Hosseini MM, Davies JE. Human umbilical cord perivascular (HUCPV) cells: A source of mesenchymal progenitors. Stem Cells. 2005;23:220-229.
- Pivoriūnas A, Bernotiene E, Unguryte A, et al. Isolation and differentiation of mesenchymal stem-like cells from human umbilical cord vein endothelium and subendothelium. Biologija. 2006;2:99-103.

