Iodine and Thyroid Health: Myths and Clinical Insights
Iodine and Thyroid Health: Clarifying Myths, Mechanisms, and Clinical Application
Angela D. Mazza1
- Metabolic Center for Wellness
OPEN ACCESS
PUBLISHED: 30 April 2026
CITATION: Author1, X., Author2, Z., et al., 2026. Iodine and Thyroid Health: Clarifying Myths, Mechanisms, and Clinical Application. Medical Research Archives, [online] 14(4).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Background: Iodine is essential for thyroid hormone synthesis and has historically played a central role in global efforts to prevent iodine deficiency disorders. Despite widespread iodization programs and general sufficiency in many developed nations, iodine remains one of the most debated nutrients in thyroid medicine. Conflicting messaging regarding supplementation, autoimmunity, seaweed consumption, urinary iodine testing, nodular disease, and iodinated contrast exposure has contributed to persistent clinical confusion.
Objective: To clarify common myths and misconceptions surrounding iodine and thyroid health by integrating physiologic mechanisms, epidemiologic data, and evidence-based clinical guidance relevant to contemporary iodine-sufficient populations.
Methods: A narrative review of the literature was conducted, focusing on thyroid physiology, iodine autoregulation, epidemiologic transitions from deficiency to sufficiency, and clinical studies evaluating iodine excess, autoimmune thyroid disease, nodular pathology, and iodinated contrast exposure. Emphasis was placed on distinguishing population-level recommendations from individualized patient management.
Results: Thyroid function demonstrates a nonlinear, U-shaped relationship with iodine intake, in which both deficiency and excess may precipitate dysfunction. In iodine-sufficient regions, autoimmune thyroid disease—not iodine deficiency—is the predominant cause of hypothyroidism. Evidence does not support routine supraphysiologic iodine supplementation in iodine-replete individuals. Excess iodine exposure may increase risk of thyroid dysfunction, particularly in those with autoimmune or nodular thyroid disease. Spot urinary iodine testing is most reliable for population assessment and should be interpreted cautiously at the individual level. Iodinated contrast typically causes transient physiologic changes rather than permanent thyroid injury in most patients.
Conclusions: Optimal iodine management requires a balanced, context-specific approach. Maintaining intake within established physiologic ranges—while avoiding indiscriminate supplementation—supports thyroid stability in most individuals. Precision thyroid care should integrate dietary assessment, underlying thyroid pathology, and risk stratification rather than rely on generalized assumptions regarding deficiency or excess.
Keywords
Iodine; Thyroid dysfunction; Autoimmune thyroid disease; Hashimoto’s thyroiditis; Wolff–Chaikoff effect; Iodine excess; Iodine deficiency; Thyroid nodules; Urinary iodine; Precision endocrinology
1. Introduction
Iodine is an essential micronutrient required for thyroid hormone synthesis and normal metabolic regulation. Over the past century, public health initiatives—most notably universal salt iodization—have dramatically reduced the global burden of iodine deficiency disorders. Yet despite this success, iodine remains one of the most debated nutrients in thyroid medicine. In clinical practice, patients are often exposed to conflicting messages: some are encouraged to take high-dose iodine supplements to “support” thyroid function, while others are advised to avoid iodine altogether, particularly in the setting of autoimmune thyroid disease.
At the core of this tension is a fundamental misunderstanding of iodine physiology. Thyroid hormone production depends on adequate iodine availability, but more is not necessarily better. Both deficiency and excess can disrupt normal thyroid function. The relationship between iodine intake and thyroid health is best understood as a nonlinear, U-shaped curve: insufficient intake may contribute to goiter and hypothyroidism, whereas excessive exposure can precipitate dysfunction, including iodine-induced hypo- or hyperthyroidism and autoimmune thyroiditis—especially in genetically susceptible individuals. Acute exposure to high iodine loads can transiently suppress hormone synthesis through the Wolff–Chaikoff effect, followed by an “escape” phenomenon that restores normal function in most individuals. When this adaptive response fails, however, or when excess exposure is sustained, clinically significant dysfunction may emerge.
The increasing prevalence of autoimmune thyroid disease in iodine-sufficient and iodine-replete regions has renewed attention to the complex relationship between iodine exposure, immune regulation, and thyroid antigenicity. Epidemiologic studies of populations transitioning from iodine deficiency to sufficiency suggest that rising iodine intake may influence both the incidence and clinical expression of thyroid disorders, including autoimmune thyroiditis. These observations underscore an important distinction: what is beneficial at the population level does not always translate directly to individual patient care.
In day-to-day practice, clinical decision-making is further complicated by the challenges of assessing iodine status, variability in dietary and supplemental intake, and differences related to life stage, pregnancy, and underlying thyroid pathology. As a result, clinicians are often left navigating practical questions—when to supplement, how to interpret testing, and how to balance potential risks and benefits across diverse patient populations.
This review aims to clarify common misconceptions surrounding iodine and thyroid health by integrating physiologic mechanisms, epidemiologic data, and clinical evidence. Particular emphasis is placed on bridging the gap between population-level recommendations and individualized care, with the goal of supporting more precise, clinically relevant thyroid management.
2. Thyroid Physiology Refresher
Iodide Transport and Sodium–Iodide Symporter Activity
Thyroid hormone synthesis begins with active transport of iodide across the basolateral membrane of thyroid follicular cells via the sodium–iodide symporter (NIS), a transmembrane glycoprotein regulated primarily by thyroid-stimulating hormone (TSH). In physiologic states, this system is remarkably efficient—concentrating iodide intracellularly to levels far exceeding those in circulation and allowing stable hormone production despite moderate variation in dietary intake. Once transported into the thyrocyte, iodide is translocated across the apical membrane into the follicular lumen, where it undergoes oxidation and incorporation into thyroglobulin.
Sodium-iodide symporter expression is dynamic and responsive to iodine availability. In iodine deficiency, NIS expression increases under TSH stimulation to enhance iodide trapping. Conversely, excess iodine exposure can downregulate NIS transcription, contributing to adaptive autoregulatory responses. This bidirectional regulation underscores the thyroid gland’s capacity to buffer fluctuations in iodine intake within a physiologic range.
Organification and Thyroid Hormone Synthesis
Within the follicular lumen, iodide is oxidized by thyroid peroxidase (TPO) in the presence of hydrogen peroxide and incorporated into tyrosyl residues of thyroglobulin, forming monoiodotyrosine (MIT) and diiodotyrosine (DIT). Coupling reactions then generate triiodothyronine (T3) and thyroxine (T4), which remain stored in colloid until proteolytic release. This process is tightly regulated. Adequate iodine availability is essential for hormone synthesis; however, excessive iodine may alter thyroglobulin iodination patterns and potentially increase the immunogenicity of thyroid antigens. These molecular changes are thought to contribute to the association between high iodine intake and autoimmune thyroid disease in susceptible individuals.
The Wolff–Chaikoff Effect
Acute exposure to high iodine loads triggers a transient suppression of thyroid hormone synthesis known as the Wolff–Chaikoff effect. This autoregulatory phenomenon occurs when elevated intrathyroidal iodine concentrations inhibit organification, temporarily reducing hormone production. The mechanism is believed to involve inhibition of hydrogen peroxide generation and TPO-mediated iodination.
In most individuals, this response is protective and self-limited. It prevents excessive thyroid hormone synthesis in the setting of sudden iodine excess, such as iodinated contrast exposure or high-dose supplementation.
Escape Phenomenon
Normal thyroid function is restored within several days through an “escape” from the Wolff–Chaikoff effect. This escape is mediated primarily by downregulation of NIS expression, which reduces intrathyroidal iodine concentration to levels below the inhibitory threshold, allowing organification to resume. Failure of this escape mechanism may result in iodine-induced hypothyroidism, particularly in individuals with underlying thyroid autoimmunity, prior thyroid injury, or impaired adaptive capacity. Conversely, in iodine-deficient individuals with autonomous nodular tissue, iodine repletion may precipitate hyperthyroidism (Jod-Basedow phenomenon).
The U-Shaped Dose–Response Relationship
The relationship between iodine intake and thyroid function is nonlinear and is best understood as a U-shaped curve (Figure 1). Both insufficient and excessive iodine exposure are associated with an increased risk of thyroid dysfunction. At one end of the spectrum, deficiency limits substrate availability for hormone synthesis, leading to goiter and hypothyroidism. At the other extreme, chronic iodine excess may increase the risk of autoimmune thyroiditis, subclinical hypothyroidism, or iodine-induced hyperthyroidism—particularly in individuals with underlying susceptibility.
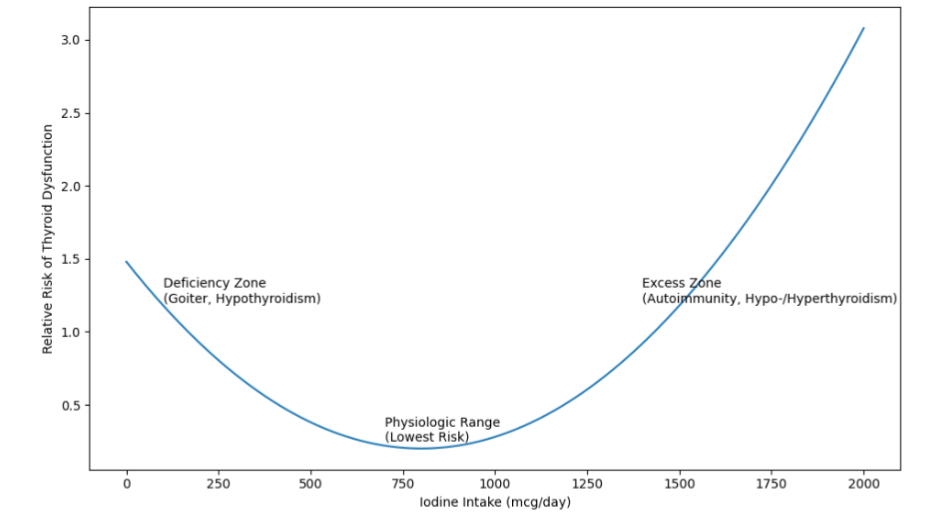
Table 1: Common Myths About Iodine and Thyroid Health: Evidence-Based Clarifications
| Myth | Evidence-Based Clarification | Clinical Implication |
|---|---|---|
| 1. Most thyroid dysfunction is caused by iodine deficiency. | In iodine-sufficient regions, autoimmune thyroid disease is the predominant cause of hypothyroidism. True iodine deficiency remains a global issue but is uncommon in many developed nations. | Evaluate geographic and dietary context before recommending supplementation. Do not assume deficiency as default cause. |
| 2. More iodine improves thyroid function. | Thyroid physiology follows a U-shaped dose–response curve. Once adequate intake is achieved, additional iodine does not enhance hormone production and may increase dysfunction risk. | Avoid supraphysiologic iodine supplementation in iodine-replete individuals. |
| 3. Patients with Hashimoto’s must avoid iodine entirely. | Physiologic iodine intake remains necessary for hormone synthesis. Excess—not normal intake—is associated with increased autoimmune risk in susceptible individuals. | Maintain intake within RDA range; avoid high-dose supplementation. |
| 4. Seaweed and “natural” iodine sources are inherently safe. | Iodine content in seaweed varies widely and may exceed tolerable upper intake levels. Excess intake can trigger thyroid dysfunction in susceptible individuals. | Review kelp/seaweed supplements and counsel on variability and dosing risk. |
| 5. Urinary iodine testing reliably reflects individual thyroid sufficiency. | Spot urinary iodine is best used for population monitoring. Individual values are highly variable and reflect recent intake rather than long-term thyroid iodine status. | Avoid overinterpreting single UIC measurements; prioritize dietary history and clinical context. |
| 6. Iodine treats thyroid nodules. | In iodine-sufficient settings, most nodules are not caused by deficiency. Supplementation does not reliably reduce nodular disease and may precipitate dysfunction in autonomous tissue. | Do not use iodine as primary nodule therapy; assess for autonomy before iodine exposure. |
| 7. Iodinated contrast permanently damages the thyroid in everyone. | Most individuals experience transient autoregulatory changes. Clinically significant dysfunction occurs primarily in high-risk populations. | Monitor high-risk patients after exposure; routine avoidance is unnecessary. |
Summary of common misconceptions regarding iodine and thyroid health with corresponding evidence-based clarifications and clinical implications. The nonlinear (U-shaped) relationship between iodine intake and thyroid function underlies many of these misconceptions.
3. Breaking Down the Myths
Common misconceptions regarding iodine and thyroid health, along with their evidence-based clarifications and clinical implications, are summarized in Table 1.
Myth #1: Most Thyroid Dysfunction Is Caused by Iodine Deficiency
Iodine deficiency is historically recognized as a leading cause of goiter and hypothyroidism worldwide, and it remains an important public health issue in certain regions. However, in everyday clinical practice within iodine-sufficient countries—including the United States and much of Europe—this is no longer the dominant driver of thyroid dysfunction.
In contemporary clinical practice within iodine-replete regions, the most common cause of hypothyroidism is autoimmune thyroid disease, particularly Hashimoto’s thyroiditis. National survey data from the United States indicate that the majority of individuals maintain urinary iodine concentrations within adequate ranges, although mild deficiency may occur in specific subgroups such as pregnant women and women of reproductive age.
Importantly, population-level sufficiency does not equate to universal adequacy, but neither does it support the assumption that most thyroid dysfunction stems from iodine deficiency.
The persistence of this myth may reflect historical teaching, global epidemiologic data that do not apply uniformly to developed nations and increasing consumer exposure to messaging that frames iodine as a universal thyroid “support” nutrient. Yet evidence suggests that indiscriminate iodine supplementation in iodine-sufficient populations does not improve thyroid function and may, in some cases, exacerbate underlying pathology.
Furthermore, autoimmune thyroid disease demonstrates complex interactions with iodine exposure. While adequate iodine intake is necessary for hormone synthesis, excessive intake may increase thyroglobulin iodination, alter antigen presentation, and promote autoimmune responses in genetically susceptible individuals. Population studies have documented increased rates of autoimmune thyroiditis following shifts from iodine deficiency to sufficiency, underscoring that higher intake does not necessarily equate to improved thyroid health.
Clinically, this distinction is critical. In iodine-deficient individuals, targeted repletion restores hormone synthesis and reduces goiter risk. In contrast, in patients with autoimmune hypothyroidism in iodine-sufficient settings, the underlying pathology reflects immune-mediated glandular destruction rather than substrate deficiency. Routine high-dose iodine supplementation in this context lacks supportive evidence and may contribute to thyroid instability.
Recognizing the geographic, epidemiologic, and pathophysiologic context of thyroid disease is essential to avoiding both under-treatment of true deficiency and over-supplementation in iodine-replete populations.
Myth #2: “More Iodine Improves Thyroid Function”
The assumption that increasing iodine intake enhances thyroid performance is intuitively appealing: because iodine is required for thyroid hormone synthesis, greater intake is often presumed to support greater hormone production. However, thyroid physiology is not linear—and this is where the misconception begins. Rather, iodine exposure follows a tightly regulated, nonlinear response curve in which both deficiency and excess may impair function.
In iodine-sufficient individuals, additional iodine does not stimulate increased thyroid hormone synthesis under normal physiologic conditions. Thyroid hormone production is regulated primarily by hypothalamic–pituitary feedback, not by substrate abundance once adequate intake is achieved. Excess iodine does not “boost” thyroid output; instead, acute high exposure may transiently suppress organification via the Wolff–Chaikoff effect. While most individuals escape this inhibitory response within several days, repeated or sustained high intake may increase the risk of thyroid dysfunction in susceptible populations.
High-dose iodine supplementation—often in milligram quantities far exceeding the Recommended Dietary Allowance (RDA) of 150 µg/day for adults—has gained popularity in certain integrative and consumer health communities. Yet evidence supporting supraphysiologic iodine dosing in iodine-replete populations is limited. In contrast, excess iodine intake has been associated with iodine-induced hypothyroidism, subclinical thyroid dysfunction, and autoimmune thyroiditis, particularly in individuals with preexisting thyroid autoimmunity or structural thyroid disease.
Population-based studies provide further insight into this phenomenon. Regions transitioning from iodine deficiency to sufficiency have demonstrated shifts in the pattern of thyroid disease, including increased rates of autoimmune thyroiditis and subclinical hypothyroidism. These findings suggest that while correcting deficiency is essential, exceeding physiologic requirements does not confer additional benefit and may alter immune–thyroid interactions.
Importantly, certain clinical scenarios illustrate iodine’s bidirectional risk profile. In iodine-deficient individuals with autonomous nodular tissue, repletion may precipitate hyperthyroidism (Jod-Basedow phenomenon). Conversely, in patients with autoimmune thyroid disease, chronic high iodine exposure may exacerbate thyroid dysfunction by increasing antigenicity of highly iodinated thyroglobulin.
Taken together, available evidence does not support the premise that more iodine enhances thyroid function in iodine-sufficient individuals. Instead, optimal thyroid health appears to depend on maintaining iodine intake within a physiologic range—adequate to support hormone synthesis, yet not excessive enough to disrupt autoregulatory and immune mechanisms.
Myth #3: “Patients With Hashimoto’s Must Avoid Iodine Entirely”
Autoimmune thyroid disease, particularly Hashimoto’s thyroiditis, is the leading cause of hypothyroidism in iodine-sufficient regions. Because excessive iodine intake has been associated with increased thyroid autoimmunity in susceptible populations, a common clinical belief has emerged that patients with Hashimoto’s should eliminate iodine exposure altogether. However, this perspective oversimplifies a complex physiologic and immunologic relationship.
Iodine is essential for thyroid hormone synthesis, even in individuals with autoimmune thyroid disease. Complete avoidance is neither physiologically appropriate nor realistically achievable, given the widespread presence of iodine in common dietary sources, as iodine is present in common dietary sources including dairy, seafood, iodized salt, and many multivitamins. The Recommended Dietary Allowance (RDA) for iodine in nonpregnant adults remains 150 µg/day, with increased requirements during pregnancy and lactation. These requirements do not disappear in the presence of autoimmunity.
The concern arises primarily with excess iodine exposure. Experimental and epidemiologic data suggest that high iodine intake may increase thyroglobulin iodination, alter antigen presentation, and enhance oxidative stress within the thyroid gland, potentially amplifying autoimmune processes in genetically predisposed individuals. Population studies have observed increases in autoimmune thyroiditis following iodine fortification in previously deficient regions, particularly when intake shifts rapidly from low to more-than-adequate levels.
However, it is important to distinguish physiologic intake from supraphysiologic supplementation. Evidence does not support routine iodine restriction below recommended levels in patients with Hashimoto’s thyroiditis. Rather, available data suggest that maintaining intake within a normal physiologic range—avoiding both deficiency and excess—is associated with the most stable thyroid function. Severe restriction may risk inadequate substrate for hormone synthesis, while high-dose supplementation (often in milligram quantities) may increase the likelihood of thyroid dysfunction or antibody elevation.
Clinical management therefore requires contextual assessment. In iodine-sufficient regions, most patients with Hashimoto’s do not require additional iodine supplementation unless deficiency is documented or strongly suspected. Conversely, patients with restrictive diets, limited dairy or seafood intake, or those who avoid iodized salt entirely may be at risk for mild insufficiency and should be evaluated accordingly. Pregnancy represents a special scenario in which adequate—but not excessive—iodine intake is essential for fetal neurodevelopment, even in women with autoimmune thyroid disease.
Thus, the appropriate clinical message is not universal avoidance, but physiologic balance. For patients with Hashimoto’s thyroiditis, maintaining iodine intake within recommended ranges while avoiding high-dose supplementation represents a prudent, evidence-aligned approach.
Myth #4: “Seaweed and ‘Natural’ Iodine Sources Are Inherently Safe”
Seaweed and kelp-based products are frequently promoted as “natural” sources of iodine and are often perceived as safer alternatives to synthetic supplements. While sea vegetables can provide dietary iodine, the assumption that natural sources are inherently safe overlooks the substantial variability in iodine content and the potential for excessive exposure.
The iodine concentration of seaweed varies dramatically depending on species, geographic origin, processing methods, and preparation. Brown seaweeds such as kelp (Laminaria species) and kombu may contain iodine concentrations ranging from several hundred micrograms to several thousand micrograms per gram of dry weight. In some cases, a single serving of certain kelp products may exceed the tolerable upper intake level (UL) for adults, which is set at 1,100 µg/day in the United States.
Unlike regulated pharmaceutical preparations, over-the-counter kelp supplements may exhibit inconsistent iodine labeling and batch variability. Case reports and observational data have documented thyroid dysfunction—including both hypothyroidism and hyperthyroidism—associated with high seaweed consumption or kelp supplementation, particularly in individuals with underlying thyroid susceptibility.
Excess iodine intake from seaweed has been implicated in iodine-induced hypothyroidism via sustained Wolff–Chaikoff inhibition, as well as iodine-induced hyperthyroidism in those with autonomous nodular tissue. Importantly, the issue is not that seaweed is intrinsically harmful. In populations with traditionally high seaweed consumption, such as Japan, thyroid physiology appears adapted to chronically higher iodine intake, although even in these settings, excessive intake may be associated with increased rates of subclinical hypothyroidism. The key distinction lies in habitual, culturally integrated dietary exposure versus intermittent high-dose supplementation in iodine-sufficient individuals without physiologic adaptation.
Moreover, the “natural” designation does not mitigate iodine’s pharmacologic effects. Iodine remains biologically active regardless of source, and excessive intake from whole foods can exert the same autoregulatory and immunologic effects as synthetic preparations.
Clinically, patients using seaweed products for perceived thyroid “support” should be counseled regarding variability in iodine content and the absence of evidence supporting supraphysiologic intake in iodine-replete populations. For most individuals in iodine-sufficient regions, routine consumption of iodized salt or a standard multivitamin provides adequate intake without the risk of large, unpredictable iodine loads.
Thus, while sea vegetables can be part of a balanced diet, they should not be assumed to be inherently safe or universally beneficial for thyroid health. As with all iodine sources, dose and context determine risk.
Myth #5: “Urinary Iodine Testing Reliably Reflects Individual Thyroid Sufficiency”
Urinary iodine concentration (UIC) is widely used to assess iodine nutrition because more than 90% of ingested iodine is excreted in urine. However, a key misconception is that a single urinary iodine measurement can reliably determine iodine sufficiency (or excess) for an individual patient. In reality, UIC is primarily a population-level biomarker, not a definitive individual diagnostic test.
UIC reflects recent iodine intake, often over the prior 24–48 hours, and is highly influenced by day-to-day variation in diet, hydration status, and supplement use. A single spot urine sample can therefore vary substantially within the same individual across different days. For this reason, public health agencies recommend interpreting iodine status using median UIC values across a population, rather than relying on single measurements for individual diagnosis.
While 24-hour urinary iodine excretion better approximates recent intake than a spot sample, it remains logistically challenging and still reflects short-term exposure rather than long-term thyroid iodine economy. Creatinine-adjusted spot UIC can reduce some variability but introduces new sources of error related to muscle mass, age, sex, and nutritional status.
In practice, over-reliance on spot UIC testing can lead to two common clinical missteps:
- False reassurance: A normal spot UIC does not exclude intermittent deficiency (or excess), particularly if intake fluctuates.
- Overcorrection: A low spot UIC may prompt unnecessary high-dose supplementation, even in iodine-sufficient regions where autoimmune thyroid disease—not deficiency—is the dominant driver of hypothyroidism.
This matters because iodine exhibits a U-shaped relationship with thyroid function: both deficiency and excess can contribute to dysfunction. Excess iodine exposure, especially from supplements or seaweed products, may precipitate thyroid dysfunction and may exacerbate autoimmune thyroid disease in susceptible individuals.
When (and How) Urinary Iodine Concentration Testing Can Be Useful
Urinary iodine concentration testing is most useful when applied thoughtfully in the right context:
- Population assessment: Monitoring iodine sufficiency within communities, regions, or study cohorts (its intended use).
- High-risk clinical scenarios: Pregnancy (where requirements increase), restrictive diets, avoidance of iodized salt, or unusual dietary patterns—particularly when paired with careful dietary/supplement history.
- Research or repeated-measure protocols: Individual assessment becomes more reliable when multiple samples are collected over time, allowing estimation of usual intake.
In routine thyroid practice, a structured clinical approach—dietary review (iodized salt, dairy/seafood intake), supplement audit (including kelp), and assessment of thyroid autoimmunity and function—often provides greater actionable clarity than a single UIC result.
Bottom line: Urinary iodine concentration is an excellent tool for public health and research, but it is commonly overinterpreted at the individual level. Precision thyroid care requires integrating iodine exposure history with thyroid physiology and underlying disease context rather than relying on isolated urinary iodine values.
Myth #6: “Iodine Treats Thyroid Nodules”
Thyroid nodules are exceedingly common, particularly in women and with advancing age, and most are benign. Because iodine deficiency is historically associated with goiter and nodular thyroid enlargement in affected populations, a persistent misconception is that iodine supplementation can “shrink nodules” or reverse nodular disease broadly. While iodine repletion can reduce diffuse goiter in iodine-deficient settings, most thyroid nodules in iodine-sufficient regions are not caused by iodine deficiency, and routine iodine supplementation is not an evidence-based nodule treatment.
When Iodine Repletion Helps—and When It Doesn’t
In areas of true iodine deficiency, chronic TSH stimulation contributes to thyroid hypertrophy, multinodular goiter, and functional autonomy; correcting deficiency can reduce goiter prevalence over time. However, in iodine-sufficient countries, nodular thyroid disease is influenced by multiple factors—including age, genetics, local growth factors, and the natural history of follicular hyperplasia—rather than simple substrate lack.
Importantly, increasing iodine intake in patients with nodular thyroid disease can sometimes carry risk. In individuals with autonomous nodules or multinodular goiter (particularly those with low/low-normal TSH), iodine exposure—through supplements, seaweed products, or iodinated contrast—may precipitate iodine-induced hyperthyroidism (Jod-Basedow phenomenon).
Thus, recommending iodine for “nodule shrinkage” is not only unsupported in most iodine-replete populations, but may also be counterproductive in selected patients.
What the Evidence Supports for Nodules
For benign nodules, evidence-based management includes ultrasound risk stratification, biopsy when indicated, surveillance, and—when symptomatic or growing—interventional therapies such as surgery or minimally invasive approaches (e.g., thermal ablation in appropriate candidates). While thyroid hormone suppression therapy has been studied historically, routine suppressive dosing is generally not recommended due to limited efficacy and potential adverse effects, particularly in older adults.
In contrast, iodine supplementation is not a standard therapy for benign nodules in iodine-sufficient settings. Any discussion of iodine in nodule management should focus on: (1) ensuring physiologic intake (avoid deficiency), (2) avoiding excess exposure, and (3) identifying patients at risk for iodine-induced hyperthyroidism or hypothyroidism based on underlying thyroid pathology.
Clinical Takeaway
The appropriate clinical message is that iodine is essential for thyroid hormone synthesis, but it is not a primary therapeutic tool for thyroid nodules in iodine-sufficient populations. For patients with nodules, the more relevant iodine-related intervention is often risk reduction—avoiding unpredictable high-dose iodine sources (kelp/seaweed supplements) and recognizing scenarios where iodine loads may destabilize thyroid function.
Myth #7: “Iodinated Contrast Permanently Damages the Thyroid in Everyone”
Iodinated contrast media used in radiographic imaging can contain iodine loads several thousand times greater than the recommended daily intake. This has led to a common concern that exposure to contrast agents inevitably causes lasting thyroid dysfunction. While iodinated contrast can transiently affect thyroid physiology, it does not lead to permanent thyroid damage in the vast majority of individuals.
Acute Iodine Load and the Wolff–Chaikoff Effect
Following iodinated contrast administration, serum iodine levels rise substantially, and the thyroid gland is exposed to a large iodine load. In response, most individuals experience a transient inhibition of thyroid hormone synthesis via the acute Wolff–Chaikoff effect. This autoregulatory mechanism protects against excessive hormone production in the setting of sudden iodine excess.
In individuals with normal thyroid function, escape from the Wolff–Chaikoff effect typically occurs within several days through downregulation of the sodium–iodide symporter (NIS), allowing thyroid hormone synthesis to normalize. Consequently, clinically significant thyroid dysfunction is uncommon in patients without underlying thyroid disease.
Who Is at Risk?
While most patients tolerate iodinated contrast without long-term consequences, certain populations are more vulnerable to iodine-induced thyroid dysfunction:
- Individuals with autonomous nodular goiter or latent Graves disease (risk of iodine-induced hyperthyroidism, or Jod-Basedow phenomenon)
- Patients with underlying Hashimoto’s thyroiditis (risk of iodine-induced hypothyroidism if escape mechanisms fail)
- Elderly individuals with nodular thyroid disease
- Neonates, particularly premature infants
- Individuals residing in areas of chronic iodine deficiency
Even in these groups, dysfunction is often transient, although some cases may require temporary or permanent treatment depending on severity and baseline thyroid reserve.
Hyperthyroidism vs Hypothyroidism After Contrast
Iodine-induced hyperthyroidism tends to occur in patients with autonomous thyroid tissue, where iodine availability fuels unregulated hormone synthesis. Conversely, iodine-induced hypothyroidism more commonly results from failure to escape from the Wolff–Chaikoff effect, particularly in autoimmune or previously injured glands.
The reported incidence of clinically overt thyroid dysfunction following iodinated contrast varies depending on baseline population risk, but it remains relatively low in iodine-sufficient regions. Routine prophylactic treatment is not recommended for the general population.
Practical Clinical Approach
For most patients undergoing contrast-enhanced imaging:
- Routine thyroid testing before contrast is not necessary in the absence of risk factors.
- In high-risk individuals (e.g., known nodular goiter, suppressed TSH, Graves disease, or significant autoimmune thyroid disease), clinicians may consider baseline TSH assessment and post-exposure monitoring.
- Patients should be counseled that transient thyroid changes are possible but permanent damage is uncommon.
Importantly, iodinated contrast is not contraindicated in the majority of patients with stable thyroid disease. The clinical need for diagnostic imaging should not be avoided solely out of concern for universal thyroid harm.
Bottom line: Iodinated contrast represents a substantial but usually temporary iodine load. In most individuals, thyroid autoregulatory mechanisms restore homeostasis. Risk is concentrated in specific vulnerable populations, not the general public.
Recognizing iodine as a critical modulator of thyroid physiology supports a more integrated and individualized approach to care. Rather than applying uniform recommendations, clinicians are better served by considering context—baseline thyroid status, dietary exposure, life stage, and susceptibility to both deficiency and excess.
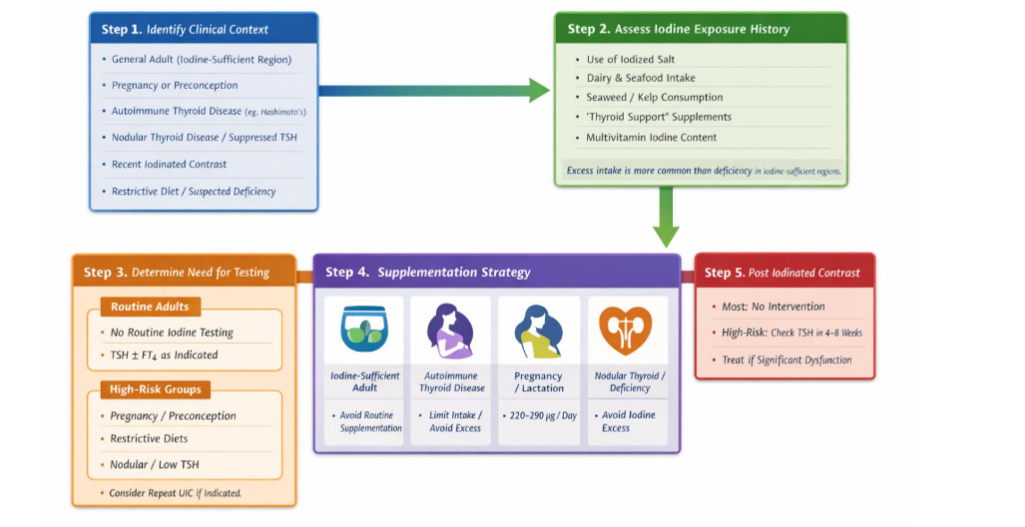
A practical, stepwise approach to iodine assessment and management in patients with thyroid-related conditions is outlined in Figure 2.
Key Clinical Takeaways
- Iodine is essential for thyroid hormone synthesis, but both deficiency and excess can disrupt thyroid function.
- The relationship between iodine intake and thyroid health follows a nonlinear, U-shaped curve, with risk at both low and high levels of exposure.
- In iodine-sufficient regions, autoimmune thyroid disease—not iodine deficiency—is the most common cause of hypothyroidism.
- Indiscriminate iodine supplementation in iodine-replete individuals offers no clear benefit and may exacerbate thyroid dysfunction, particularly in those with underlying autoimmunity or nodular disease.
- The Wolff–Chaikoff effect provides short-term protection against iodine excess; however, failure to escape this effect may lead to hypothyroidism in susceptible individuals.
- Common clinical misconceptions—regarding seaweed consumption, urinary iodine testing, contrast exposure, and thyroid nodules—often arise from applying population-level data to individual patients.
- Assessment of iodine status in clinical practice is challenging; careful dietary and supplement history remains essential for identifying both deficiency and excess.
- A balanced, individualized approach—maintaining iodine intake within physiologic ranges and targeting supplementation to specific populations such as pregnancy—represents the most effective strategy for optimizing thyroid health.
Conclusion
Iodine remains both essential and frequently misunderstood in modern thyroid practice. While historical iodine deficiency shaped global thyroid epidemiology, the landscape in iodine-sufficient regions has shifted. Today, autoimmune thyroid disease—not substrate deficiency—is the dominant driver of hypothyroidism, and routine or indiscriminate iodine supplementation offers little clinical benefit. In fact, more is not necessarily better, and in some cases, it may be counterproductive.
The relationship between iodine intake and thyroid function is nonlinear and best conceptualized as a U-shaped curve. Both insufficient and excessive exposure can disrupt thyroid homeostasis, particularly in susceptible individuals. Protective autoregulatory mechanisms, including the Wolff–Chaikoff effect, help buffer acute iodine excess, but this response is not universal and may fail in the setting of autoimmunity or nodular disease. In clinical practice, many of the most persistent misconceptions—whether related to seaweed intake, urinary iodine testing, contrast exposure, or thyroid nodules—stem from applying population-level assumptions to individual patients.
Effective thyroid management, therefore, requires context rather than extremes. Attention to dietary and supplemental iodine exposure, recognition of higher-risk groups, and maintaining intake within physiologic ranges are central to care. Supplementation should be reserved for clearly defined situations, such as pregnancy or documented deficiency, while avoiding unnecessary high-dose use in iodine-replete individuals. Ultimately, iodine is neither universally deficient nor inherently harmful—its impact is shaped by dose, duration, baseline thyroid status, and individual susceptibility. A balanced, physiology-based, and patient-centered approach offers the most reliable path forward in contemporary thyroid care.
References
- Zimmermann MB, Boelaert K. Iodine deficiency and thyroid disorders. Lancet Diabetes Endocrinol. 2015;3(4):286–295. DOI: 10.1016/S2213-8587(14)70225-6
- Laurberg P, Pedersen KM, Vestergaard H, Sigurdsson G. Iodine intake as a determinant of thyroid disorders in populations. Best Pract Res Clin Endocrinol Metab. 2010;24(1):13–27. DOI: 10.1016/j.beem.2009.08.013
- Teng W, Shan Z, Teng X, et al. Effect of iodine intake on thyroid diseases in China. N Engl J Med. 2006;354(26):2783–2793. DOI: 10.1056/NEJMoa054022
- Leung AM, Braverman LE. Consequences of excess iodine. Nat Rev Endocrinol. 2014;10(3):136–142. DOI: 10.1038/nrendo.2013.251
- Wang B, He W, Li Q, Jia X, Yao Q, Song R, Qin Q, Zhang JA. U-shaped relationship between iodine status and thyroid autoimmunity risk in adults. Eur J Endocrinol. 2019 Sep;181(3):255-266. doi: 10.1530/EJE-19-0212. PMID: 31252413.
- Eng PHK, Cardona GR, Fang S-L, et al. Escape from the acute Wolff–Chaikoff effect is associated with a decrease in sodium/iodide symporter mRNA and protein. Endocrinology. 1999;140(8):3404–3410. DOI: 10.1210/endo.140.8.6893
- Rose NR, Bonita R, Burek CL. Iodine: an environmental trigger of thyroiditis. Autoimmun Rev. 2002;1(1–2):97–103. DOI: 10.1016/s1568-9972(01)00016-7
- Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017;27(3):315–389. DOI: 10.1089/thy.2016.0457
- Kalarani IB, Veerabathiran R. Impact of iodine intake on the pathogenesis of autoimmune thyroid disease in children and adults. Ann Pediatr Endocrinol Metab. 2022 Dec;27(4):256-264. doi: 10.6065/apem.2244186.093. Epub 2022 Dec 31. PMID: 36567462; PMCID: PMC9816468.
- Dai G, Levy O, Carrasco N. Cloning and characterization of the thyroid iodide transporter. Nature. 1996;379(6564):458–460. DOI: 10.1038/379458a0
- Wolff J, Chaikoff IL. Plasma inorganic iodide as a homeostatic regulator of thyroid function. J Biol Chem. 1948 Jun;174(2):555-64. PMID: 18865621
- Caldwell KL, Pan Y, Mortensen ME, et al. Iodine status in the United States, NHANES 2003–2004. Thyroid. 2011;21(4):419–427.DOI: 10.1089/thy.2010.0077
- Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99:39–51. DOI:10.1093/bmb/ldr030
- Leung AM, Pearce EN, Braverman LE. AAP recommendations on iodine nutrition in pregnancy and lactation. Endocrinol Metab Clin North Am. 2011;40(4):765–777. DOI: 10.1542/peds.2014-2111A
- Hall JE. Thyroid metabolic hormones. In: Guyton and Hall Textbook of Medical Physiology. 13th ed. Elsevier; 2016.
- Teas J, Pino S, Critchley A, Braverman LE. Variability of iodine content in common commercially available edible seaweeds. Thyroid. 2004;14(10):836–841. DOI: 10.1089/thy.2004.14.836
- Institute of Medicine (US) Panel on Micronutrients. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington (DC): National Academies Press (US); 2001. Available from: https://www.ncbi.nlm.nih.gov/books/NBK222310/ doi: 10.17226/10026
- Smyth PPA. Iodine, Seaweed, and the Thyroid. Eur Thyroid J. 2021 Apr;10(2):101-108. doi: 10.1159/000512971. Epub 2021 Jan 27. PMID: 33981614; PMCID: PMC8077470.
- Nishiyama S, Mikeda T, Okada T, et al. Transient hypothyroidism or persistent hyperthyrotropinemia in neonates born to mothers with excessive iodine intake. Thyroid. 2004 Dec;14(12):1077-83. doi: 10.1089/thy.2004.14.1077. PMID: 15650362.
- Clark CD, Bassett B, Burge MR. Effects of kelp supplementation on thyroid function in euthyroid subjects. Endocr Pract. 2003;9(5):363–369. DOI: 10.4158/EP.9.5.363
- Aakre I, Vogt EC, Myrmel LS, et al. Impact of habitual seaweed consumption on iodine nutrition and thyroid function: a non-randomized pre-post clinical study. Eur J Nutr. 2026 Jan 16;65(1):27. doi: 10.1007/s00394-025-03813-8. PMID: 41543576; PMCID: PMC12811364.
- Nagataki S. The average of dietary iodine intake due to the ingestion of seaweeds is 1.2 mg/day in Japan. Thyroid. 2008;18(6):667– 668. DOI: 10.1089/thy.2007.0379
- World Health Organization, UNICEF, International Council for Control of Iodine Deficiency Disorders. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers. 3rd ed. World Health Organization; 2007.
- Pearce EN, Caldwell KL. Urinary iodine, thyroid function, and thyroglobulin as biomarkers of iodine status. Am J Clin Nutr. 2016 Sep;104 Suppl 3(Suppl 3):898S-901S. doi: 10.3945/ajcn.115.110395. Epub 2016 Aug 17. PMID: 27534636; PMCID: PMC5004493.
- Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016 Jan;26(1):1-133. doi: 10.1089/thy.2015.0020. PMID: 26462967; PMCID: PMC4739132.
- Rhee CM, Bhan I, Alexander EK, Brunelli SM. Association between iodinated contrast media exposure and incident hyperthyroidism and hypothyroidism. Arch Intern Med. 2012;172(2):153–159. DOI: 10.1001/archinternmed.2011.677
- Hu Y, Zhong X, Peng D, et al. Iodinated contrast media (ICM)-induced thyroid dysfunction: a review of potential mechanisms and clinical management. Clin Exp Med. 2025 Apr 29;25(1):132. doi: 10.1007/s10238-025-01664-5. PMID: 40299149; PMCID: PMC12040987.
- Leung AM, Braverman LE. Iodine-induced thyroid dysfunction. Curr Opin Endocrinol Diabetes Obes. 2012;19(5):414–419. DOI: 10.1097/MED.0b013e3283565bb2




