Musculoskeletal Ultrasound in Diagnosing Arthritis Types
The role of musculoskeletal ultrasonography in the early diagnosis and differentiation of rheumatoid arthritis, psoriatic arthritis and cutaneous psoriasis
Eugeniu Russu 1,2, Alesea Nistor 1, Mircea Betiu 1, Victor Cazac 3 Marinela Homițchi 1,2, Liliana Groppa 1
- Nicolae Testemitanu” State University of Medicine and Pharmacy, Chisinau, Republic of Moldova,
- Timofei Mosneaga” Republican Clinical Hospital, Chisinau, Republic of Moldova
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Russu, E., Nistor, A., et al., 2025. The role of musculoskeletal ultrasonography in the early diagnosis and differentiation of rheumatoid arthritis, psoriatic arthritis and cutaneous psoriasis. Medical Research Archives, [online] 13(4). https://doi.org/10.18103/mra.v13i4.6488
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i4.6488
ISSN: 2375-1924
Abstract
Introduction. Inflammatory arthritis, including rheumatoid arthritis (RA) and psoriatic arthritis (PsA), presents with overlapping clinical manifestations, making early differentiation crucial for effective treatment. Musculoskeletal ultrasonography has emerged as a sensitive imaging tool for detecting early inflammatory and structural changes, aiding in precise diagnosis.
Objective. This study aims to identify specific musculoskeletal ultrasonography parameters distinguishing RA, PsA, and psoriasis (PsO), facilitating early diagnosis and optimizing therapeutic strategies.
Methods. A cross-sectional study was conducted on 274 patients diagnosed with inflammatory musculoskeletal diseases, categorized into three groups: 127 PsA, 91 RA, and 56 PsO patients. All participants underwent clinical and ultrasonographic assessment, evaluating synovial hypertrophy, joint effusions, tenosynovitis, enthesitis, and bone erosions using Gray Scale and Power Doppler techniques. Statistical analysis was performed using SPSS 26.0.
Results. Musculoskeletal ultrasonography revealed significant differences between PsA, RA, and PsO. Synovial hypertrophy and bone erosions were predominant in RA (75.67%) and PsA (48.48%), with tenosynovitis affecting flexor tendons in PsA and extensor tendons in RA. Subclinical enthesitis was present in 72.72% of PsA patients and 45% of PsO patients, while radiocarpal joint effusions were more common in RA (59%). PsO patients exhibited mild, subclinical changes, with 40% showing early synovio-entheseal inflammation.
Conclusions. Musculoskeletal ultrasonography is a valuable tool for the early differentiation of inflammatory arthritis types, allowing detection of subclinical enthesitis and tenosynovitis in PsA, as well as progressive synovial hypertrophy and erosions in RA. Periodic musculoskeletal ultrasonography monitoring in PsO patients may help predict PsA onset, improving early intervention strategies.
Keywords: musculoskeletal ultrasonography, rheumatoid arthritis, psoriatic arthritis, enthesitis, early diagnosis
Introduction
Despite advancements in immunological and serological diagnostics, the clinical features of inflammatory arthritis often overlap, especially in early disease stages. Differentiation is further complicated by extra-articular manifestations, comorbidities such as obesity, and patient-specific anatomical variations. For instance, rheumatoid arthritis (RA) and psoriatic arthritis (PsA) may initially exhibit similar joint involvement patterns, while patients with cutaneous psoriasis (PsO) may present with subclinical joint inflammation that progresses silently before overt arthritis develops.
Musculoskeletal ultrasonography (MSUS) has emerged as a pivotal imaging modality in rheumatology. Its high resolution and sensitivity allow for real-time, non-invasive evaluation of synovial and periarticular structures. Unlike conventional radiography, MSUS can detect subtle inflammatory changes such as synovitis, tenosynovitis, enthesitis, and bone erosions at a subclinical level – prior to irreversible joint damage. It also enables dynamic comparison of multiple joints in the same session, which is critical in polyarticular presentations. Studies have demonstrated that MSUS findings correlate well with disease activity and are more sensitive than clinical examination alone in detecting early synovial and entheseal inflammation.
Psoriatic arthritis represents a chronic and heterogeneous form of seronegative spondyloarthropathy. It typically involves peripheral and axial joints, as well as entheses and tendons, and is frequently accompanied by PsO, nail changes, and other systemic features such as uveitis or inflammatory bowel disease. Notably, musculoskeletal symptoms may precede skin involvement, complicating the diagnostic process. The Classification Criteria for Psoriatic Arthritis (CASPAR) are currently used for diagnosis, although the lack of specific biomarkers continues to pose a challenge. As a result, diagnosis relies on a combination of clinical manifestations, imaging findings, and nonspecific inflammatory markers, which become particularly relevant in advanced disease stages. The absence of PsO in the presence of active arthritis can lead to an undifferentiated arthritis diagnosis, delaying the initiation of targeted treatment. Musculoskeletal ultrasonography studies have proven valuable in detecting joint involvement even in the absence of cutaneous signs of PsO.
Among the most frequently encountered inflammatory arthritides, RA and PsA are the most clinically relevant. Differentiation between them is based on the anatomical structures involved, with diagnosis established according to the EULAR/ACR criteria for RA and the CASPAR criteria for PsA. Since RA is associated with specific serological markers, diagnosis can be established more quickly. In contrast, PsA may present with joint manifestations before the onset of cutaneous psoriasis, complicating the diagnostic process. Identifying PsO is crucial, given its association with degenerative bone pathologies. These patients require periodic monitoring via MSUS, as they have an increased risk of developing PsA. Patients with psoriasis, even in the absence of articular symptoms, are at increased risk of developing PsA. Approximately 30–40% of these individuals may exhibit sonographic evidence of synovio-entheseal inflammation before clinical arthritis manifests. As such, periodic MSUS evaluation of PsO patients may enable earlier identification of at-risk individuals and prompt preemptive intervention.
Standardized ultrasonographic scoring systems – such as the Glasgow Ultrasound Enthesitis Scoring System (GUESS) and the Madrid Sonographic Enthesitis Index (MASEI) – have shown promise in quantifying entheseal involvement and enhancing diagnostic accuracy. MASEI, in particular, has demonstrated high specificity (up to 85%) in identifying PsA features when enthesitis is present. Taken together, the integration of MSUS into clinical evaluation provides a powerful tool for early detection and classification of inflammatory arthritis. By recognizing distinctive ultrasonographic patterns associated with RA, PsA, and PsO, clinicians can make more informed diagnostic and therapeutic decisions.
Study objective
To identify specific musculoskeletal ultrasonographic parameters for psoriatic arthritis, cutaneous psoriasis, and rheumatoid arthritis.
Material and Methods
This cross-sectional study included 274 patients diagnosed with inflammatory musculoskeletal diseases, who were hospitalized and evaluated in the Rheumatology and Arthrology Departments of the Republican Clinical Hospital “Timofei Moșneaga” between 2022–2024. The study population was divided into three distinct groups:
| Group | No. of patients | % | No. of women | % women | No. of men | % men |
|---|---|---|---|---|---|---|
| PsA | 127 | 46.35 | 69 | 54.33 | 58 | 45.67 |
| RA | 91 | 33.21 | 57 | 62.64 | 34 | 37.36 |
| PsO | 56 | 20.44 | 39 | 69.64 | 17 | 30.36 |
| Total | 274 | 100.00 | 165 | 60.22 | 109 | 39.78 |
Sex distribution revealed a predominance of female patients in all subgroups, with 69 women (54.33%) in the PsA group, 57 (62.64%) in the RA group, and 39 (69.64%) in the PsO group, compared to 58 men (45.67%), 34 (37.36%), and 17 (30.36%) in the respective groups. The mean age of participants was 47 ± 12.1 years. All patients underwent comprehensive clinical and imaging evaluations performed by board-certified rheumatologists following standardized international guidelines. The study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of USMF “Nicolae Testemițanu” (21.12.2019, No. 21).
DIAGNOSTIC AND INCLUSION CRITERIA
Patients were classified based on internationally validated criteria: the CASPAR criteria for PsA, the EULAR/ACR 2010 criteria for RA, and the clinical confirmation of psoriasis vulgaris by a dermatologist. To ensure diagnostic precision and cohort homogeneity, the following exclusion criteria were applied: age under 18 years; disease duration exceeding five years; pregnancy; history of biological treatment; and presence of overlapping autoimmune or degenerative joint disorders.
In particular, patients with clinical or radiographic features suggestive of osteoarthritis, gout, fibromyalgia, or other systemic rheumatic diseases (e.g., lupus, scleroderma) were excluded. Differential diagnosis was confirmed through correlation of clinical, serological, and ultrasonographic data. For PsO patients, a detailed musculoskeletal assessment using MSUS was performed to exclude concomitant inflammatory or degenerative arthropathies and ensure accurate classification as non-articular psoriasis. Synovial hypertrophy, erosions, and Doppler signal were considered markers of inflammatory arthritis, while the presence of isolated osteophytes or joint space narrowing without inflammatory signs were suggestive of degenerative disease and led to exclusion.
ULTRASOUND ASSESSMENT
High-resolution MSUS was performed using both Grey Scale (GS) and Power Doppler (PD) techniques to detect inflammatory and structural changes in articular and periarticular tissues. A standardized scanning protocol included small and large joints bilaterally, with emphasis on synovial thickening, joint effusion, osteophyte formation, and bone erosions. Tendons and entheses were assessed in detail, including the flexor and extensor tendons of the hands and feet, the Achilles tendon, the plantar aponeurosis, and the quadriceps tendon, in order to identify subclinical inflammation or degenerative changes.
The degree of synovial inflammation was graded on a semi-quantitative PD scale (0–3): grade 0 indicating no signal; grade 1 – ≤3 discrete Doppler signals; grade 2 – moderate signal involving <50% of the synovial area; and grade 3 – intense signal involving >50% of the synovial area.
STATISTICAL ANALYSIS
Data were analyzed using SPSS version 26.0 (IBM, Armonk, NY). The Kolmogorov-Smirnov test was used to assess normality of continuous variables. Normally distributed data were presented as mean ± standard deviation (SD), and non-normally distributed data as median (25th–75th percentiles). Group comparisons were performed using the Student’s t-test or Mann-Whitney U test, as appropriate. Categorical variables were expressed as absolute and relative frequencies and analyzed using chi-square or Fisher’s exact test. Correlations were assessed using Pearson’s or Spearman’s coefficient depending on variable distribution. A p-value < 0.05 was considered statistically significant.
Results
The study included 33 patients (36.6%) with PsA, 37 patients (41.1%) with RA, and 20 patients (22.2%) with PsO, with a mean age of 47±12.1 years. Among them, 55 were women and 35 were men. Patients diagnosed with PsA and PsO exhibited cutaneous manifestations specific to psoriasis, while RA patients were selected exclusively from those with the seropositive form and no prior biological treatment. Ultrasound investigations revealed the presence of joint and periarticular changes in all three groups, with a higher incidence in patients with PsA and RA. The results showed ultrasonographic changes in 85% of clinically affected joints and in 20% of apparently healthy joints. Thus, in patients with the oligoarticular form of PsA, the diagnosis was reassessed and reclassified as the polyarticular form, leading to an adjustment in therapeutic strategy.
In patients with PsA, asymmetric joint involvement was predominantly observed, most commonly affecting the knees, followed by the first metatarsophalangeal (MTP) joint, the radiocarpal joint, the talocrural joint, and the small joints of the hands. Among these, the radiocarpal joint was affected in 20% of cases, followed by the third metacarpophalangeal (MCP) joint (13%) and the third distal interphalangeal (DIP III) joint (11%).
| Affected joint structures | PsA (%) | RA (%) |
|---|---|---|
| Knee | 57 | – |
| MTP I joint | 38 | – |
| Talocrural joint | 23 | – |
| Radiocarpal joint | 20 | 59 |
| MCP III joint | 13 | 19 |
| DIP III joint | 11 | – |
| MCP II joint | 3 | 17 |
In RA patients, ultrasonographic changes were more frequently observed in MCP II, III joints, radiocarpal joints, and MTP II, III joints, in both GS and PD modes. Among RA patients, the radiocarpal joints were the most affected (59%), followed by MCP III (19%) and MCP II (17%). There was a more pronounced progression of joint involvement in RA patients compared to PsA patients.
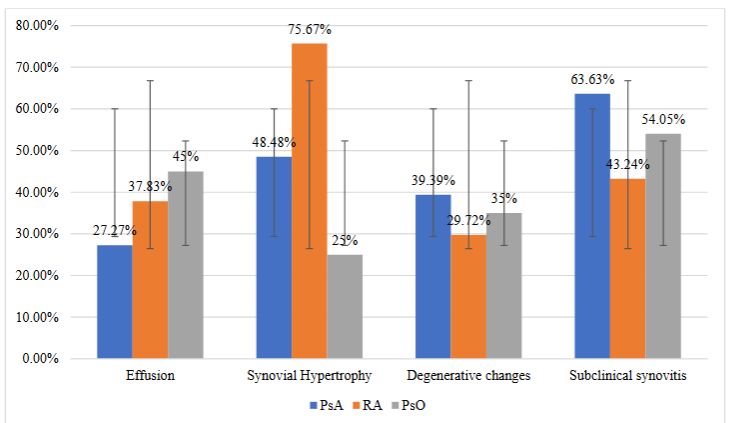
The ultrasonographic features of PsA are comparable to those observed in RA, being characterized by synovial hypertrophy, intra-articular effusion, the presence of PD signal, and bone erosions. Among the analyzed patients, joint effusions without ultrasonographic signs of synovitis were more frequently observed in PsO patients (45%), compared to PsA (27.27%) and RA (37.83%) patients.
| Ultrasonographic findings | PsA (%) | RA (%) | PsO (%) |
|---|---|---|---|
| Joint effusions | 27.27 | 37.83 | 45 |
| Synovial hypertrophy | 48.48 | 75.67 | 25 |
| Degenerative changes | 39.39 | 29.72 | 35 |
| Subclinical synovitis | 63.63 | 43.24 | 54.05 |
Degenerative changes, including osteophytes, were similarly present in all three patient groups, with a slightly higher prevalence in PsO and PsA patients.
Ultrasonographic examination of the MCP joints revealed the presence of articular synovitis without periarticular involvement in RA patients, while in 48.48% of PsA patients, peritendinous inflammation of the extensor tendons at the MCP level was observed. Tenosynovitis of the radiocarpal joints was detected in both RA patients (43.24%) and PsA patients (27.27%). A higher specificity for flexor tenosynovitis was observed in PsA, whereas RA showed a predominance of extensor tendon involvement.
Approximately one-third of PsA patients exhibited flexor tenosynovitis, accompanied by soft tissue edema and PD signal. The most frequently affected tendons were the flexor pollicis longus (46%) and the flexor digitorum profundus (49%). In contrast, RA patients predominantly showed extensor tendon involvement (63%), particularly in compartments 1 and 4. In patients without radiological erosions, compartment 6 involvement, including the extensor carpi ulnaris tendon, was observed. Tenosynovitis of this tendon correlated with the appearance of erosions at the ulnar styloid process, a specific feature of RA.
In addition to tenosynovitis, tendon involvement without a synovial sheath was also observed in PsA and PsO patients. On ultrasound, these tendons appeared hypoechoic and thickened, with or without PD signal. The most frequently affected structures were: Achilles tendon (60.6% in PsA and 55% in PsO), quadriceps tendon (42.42% in PsA and 35% in PsO), plantar aponeurosis (36.36% in PsA and 25% in PsO).
Discussion
The early differentiation of inflammatory arthritis subtypes remains a clinical challenge due to significant overlap in signs and symptoms. The current study reinforces the diagnostic value of MSUS as a non-invasive, sensitive, and dynamic imaging modality capable of identifying both articular and periarticular inflammatory changes before the onset of irreversible damage.
In patients with PsA, the study revealed a predominance of asymmetric joint involvement, particularly affecting the knees, MTP I, and small joints of the hands, such as MCP II and III. These findings are consistent with existing literature, which reports peripheral asymmetric arthritis as the most common phenotype in PsA, especially in early disease stages. Musculoskeletal ultrasonography enabled not only the confirmation of clinically suspected synovitis but also the detection of structural lesions in joints considered asymptomatic, facilitating reclassification of oligoarticular forms into polyarticular patterns in certain cases. This highlights the pivotal role of MSUS in treatment planning and monitoring.
Enthesitis, a cardinal feature of PsA, was frequently observed in both symptomatic and asymptomatic patients. MSUS revealed entheseal changes—thickening, loss of fibrillar pattern, and Doppler signal – in 72.72% of PsA cases and in 45% of PsO patients without clinical arthritis. These subclinical changes may precede the onset of arthritis, supporting the concept of a “prodromal PsA phase” in which enthesitis acts as a primary pathogenic driver. These observations align with the findings of Gutierrez et al., who demonstrated that 35–50% of PsO patients show MSUS-detectable enthesitis or tenosynovitis prior to clinical PsA onset. Therefore, early identification and close monitoring of PsO patients with subclinical inflammatory lesions could allow for early intervention and potentially disease-modifying strategies.
In rheumatoid arthritis patients, MSUS confirmed a classical pattern of synovitis with symmetric distribution, particularly affecting the radiocarpal and MCP II/III joints. Synovial hypertrophy and active Doppler signal were predominant and correlated with disease activity. Bone erosions, which are hallmark features of RA, were detected in 64.86% of RA patients, in line with previous reports. The involvement of the extensor tendon compartments – especially compartments 1, 4, and 6 – was strongly associated with RA. Compartment 6 tenosynovitis, involving the extensor carpi ulnaris, was noted as an early feature and often preceded erosive changes at the ulnar styloid, consistent with prior sonographic studies.
Interestingly, peritendinous inflammation and tenosynovitis were also observed in PsA and PsO, but with different distribution. PsA was associated with predominant flexor tenosynovitis (flexor pollicis longus, flexor digitorum profundus), often accompanied by thickening of the tendon sheath and positive Doppler signal. These features, although also detectable in PsO, were significantly more prevalent and severe in PsA, suggesting that flexor tenosynovitis may be an early biomarker of psoriatic arthropathy.
The importance of identifying subclinical synovitis is increasingly recognized. In our study, subclinical synovitis was found in 63.63% of PsA patients, 43.24% of RA patients, and 54.05% of PsO patients. Several longitudinal studies have demonstrated that PsO patients with sonographic evidence of synovitis or enthesitis are at significantly higher risk of progressing to PsA within 12–24 months. These findings support MSUS as a screening tool for PsO patients, enabling early risk stratification and tailored follow-up.
Degenerative changes (e.g., osteophytes, cartilage loss) were noted in all patient groups, but were not considered discriminatory. However, when accompanied by inflammatory markers such as Doppler signal or synovial thickening, these features became suggestive of mixed pathologies or inflammatory exacerbation on pre-existing osteoarthritis. The simultaneous presence of bone erosions, enthesitis, and Doppler signal in PsA patients proved to be a highly specific ultrasonographic signature for PsA, in line with the findings from Gutierrez et al. and Naredo et al. This combination may serve as a practical diagnostic algorithm in ambiguous cases, particularly in seronegative patients or those without cutaneous lesions.
CLINICAL IMPLICATIONS
These findings reinforce the clinical utility of MSUS not only in diagnosis, but also in disease monitoring and prognosis assessment. The ability to assess multiple joints and soft tissue structures during a single session makes MSUS especially valuable in real-world rheumatology practice. Moreover, its sensitivity in detecting preclinical inflammation highlights its potential role in preventive rheumatology – particularly for high-risk PsO patients or those with suspected early PsA.
Limitations
This study has several limitations. The sample size was moderate, and the analysis was cross-sectional, limiting assessment of long-term disease evolution and response to treatment. Patients with long-standing disease or prior biological therapy were excluded, which may restrict generalizability to broader populations. Additionally, interobserver variability in MSUS interpretation was not formally assessed, although all scans were performed by experienced rheumatologists following a standardized protocol.
Conclusions
Musculoskeletal ultrasonography has proven to be an essential diagnostic tool for the early detection, characterization, and differentiation of inflammatory arthritis subtypes. Its sensitivity in identifying both articular and periarticular changes – particularly in subclinical disease – positions it as a frontline imaging modality in rheumatologic practice. The ability of MSUS to capture subtle synovial and entheseal inflammation allows for a more accurate stratification of patients, especially in cases where clinical and serological findings are inconclusive.
In rheumatoid arthritis, MSUS confirms the classical intra-articular inflammatory pattern, with dominant synovial hypertrophy in the MCP II/III and radiocarpal joints, accompanied by extensor tenosynovitis (notably in compartments 1, 4, and 6). Bone erosions are more extensive and appear earlier compared to PsA, reinforcing the importance of early ultrasonographic surveillance in RA. In PsA, distinctive periarticular findings such as peritenon extensor inflammation, flexor tenosynovitis, and entheseal changes – including digital enthesitis and palmar fascia involvement – provide ultrasonographic signatures that distinguish it from RA and may precede clinical onset. Similar but less pronounced structural alterations were observed in PsO, where MSUS-detected inflammation may serve as a predictor for future PsA development.
The integration of MSUS into the clinical assessment of patients with psoriasis and inflammatory arthritis enhances diagnostic precision and informs earlier therapeutic intervention. When applied longitudinally and in combination with clinical and laboratory parameters, MSUS can support personalized treatment strategies, improve disease classification, and ultimately contribute to better long-term outcomes.
Conflicts of Interest Statement
The authors have no conflicts of interest to declare.
Funding Statement
No specific funding was received for the work discussed in this manuscript.
Acknowledgments
The authors express their sincere gratitude to the Republican Clinical Hospital “Timofei Moșneaga” for their technological and methodological support in conducting this study. This research was carried out within the framework of the institutional project (2024–2027) titled “Optimization of the Diagnosis of Clinical Forms of Psoriatic Arthritis” (DOPA 15-01-01).
References
- Ahmed A.R., Tharwat S., Abd el-Khalek A.M., et al. Ultrasound findings in hand joints involvement: a comparative study between psoriatic and rheumatoid arthritis. Annals of the Rheumatic Diseases, 2020; 79:1857.
- Tom Stephanie, Zhong Yujie, Cook Richard, Zehra Aydin Sibel, Kaeley Gurjit, Eder Lihi. Development of a Preliminary Ultrasonographic Enthesitis Score in Psoriatic Arthritis — GRAPPA Ultrasound Working Group. The Journal of Rheumatology, Apr 2019, 46 (4) 384-390; doi:10.3899/jrheum.171465.
- Di Matteo A., Mankia K., Azukizawa M., Wakefield R.J. The Role of Musculoskeletal Ultrasound in the Rheumatoid Arthritis Continuum. Curr Rheumatol Rep. 2020 Jun 19;22(8):41. doi:10.1007/s11926-020-00911-w. PMID: 32562012; PMCID: PMC7305070.
- Dubash S.R., De Marco G., Wakefield R.J., Tan A.L., McGonagle D., Marzo-Ortega H. Ultrasound Imaging in Psoriatic Arthritis: What Have We Learnt in the Last Five Years? Front Med (Lausanne). 2020 Aug 25; 7:487. doi:10.3389/fmed.2020.00487. PMID: 32984374; PMCID: PMC7477070.
- Filer A., Cader M., Abhishek A., et al. Ultrasound defined tenosynovitis improves the prediction of rheumatoid arthritis and persistent disease in patients with very early synovitis. Annals of the Rheumatic Diseases, 2013; 71:506-507.
- Gudu T., Ng B., Jethwa H., Graham C., Kudva V., Rammanohar J., Zhang C., Sapsford M., Jadon D.R. Improving Imaging Modalities in Early Psoriatic Arthritis: The Role of Ultrasound in Early Diagnosis of Psoriatic Arthritis. Front Med (Lausanne). 2022 Jan 7;8:804695. doi: 10.3389/fmed.2021.804695. PMID: 35071281; PMCID: PMC8776996.
- Gutierrez M., Filippucci E., Salaffi F., Di Geso L., Grassi W. Differential diagnosis between rheumatoid arthritis and psoriatic arthritis: the value of ultrasound findings at metacarpophalangeal joints level. Ann Rheum Dis. 2011 Jun;70(6):1111-4. doi:10.1136/ard.2010.147272. Epub 2011 Mar 15. PMID: 21406459.
- Kaeley, G.S., Bakewell, C. & Deodhar, A. The importance of ultrasound in identifying and differentiating patients with early inflammatory arthritis: a narrative review. Arthritis Res Ther 22, 1, 2020. doi.org/10.1186/s13075-019-2050-4.
- Kamp Felbo S., Wiell C., Ǿstergaard M., et al. Do tender joints in psoriatic arthritis reflect inflammation on ultrasound? Annals of the Rheumatic Diseases, 2020;79:1842-1843.
- Merola J.F., Espinoza L.R., Fleischmann R. Distinguishing rheumatoid arthritis from psoriatic arthritis. RMD Open 2018;4:e000656. doi:10.1136/rmdopen-2018-000656.
- Tang Y., Cheng S., Yang Y., Xiang X., Wang L., Zhang L., Qiu L. Ultrasound assessment in psoriatic arthritis (PsA) and psoriasis vulgaris (non-PsA): which sites are most commonly involved and what features are more important in PsA? Quant Imaging Med Surg 2020;10(1):86-95. doi:10.21037/qims.2019.08.09.
- Chiara C. Rizzo. Ultrasound in rheumatoid arthritis. Medical Ultrasonography 15(3):199 Romanian Society of Ultrasonography in Medicine and Biology 2013-9 2066-8643. doi:10.11152/mu.2013.2066.153.cr1fc2.
- Zabotti A., De Marco G., Gossec L., et al. EULAR points to consider for the definition of clinical and imaging features suspicious for progression from psoriasis to psoriatic arthritis. Annals of the Rheumatic Diseases, 2023;82:1162-1170.
- Tang Y., Yang Y., Xiang X., Wang L., Zhang L., Qiu L. Power Doppler Ultrasound Evaluation of Peripheral Joint, Entheses, Tendon, and Bursa Abnormalities in Psoriatic Patients: A Clinical Study. J Rheumatol. 2018 Jun;45(6):811-817. doi:10.3899/jrheum.170765. Epub 2018 Apr 15. PMID: 29657139.
- Boussaid, S., Ben Aissa, R., Rekik, S., Rahmouni, S., Jammali, S., Zouaoui, K., Sahli, H., Elleuch, M. Ultrasonography Enthesitis and Synovitis Screening in Psoriatic Patients: A Case Control Study. Mediterr. J. Rheumatol. 2023, 34, 495–505. doi.org/10.31138/mjr.180923.ues.
- Balulu, G., Furer, V., Wollman, J., Levartovsky, D., Aloush, V., Elalouf, O., Sarbagil-Maman, H., Mendel, L., Borok, S., Paran, D., et al. The association between sonographic enthesitis with sonographic synovitis and tenosynovitis in psoriatic arthritis patients. Rheumatology 2024, 63, 190–197. doi.org/10.1093/rheumatology/kead202.
- Taylor W, Gladman D, Helliwell P, Marchesoni A, Mease P, Mielants H; CASPAR Study Group. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 2006 Aug;54(8):2665-73. doi: 10.1002/art.21972. PMID: 16871531.
- Kay J, Upchurch KS. ACR/EULAR 2010 rheumatoid arthritis classification criteria. Rheumatology (Oxford). 2012 Dec;51 Suppl 6:vi5-9. doi: 10.1093/rheumatology/kes279. PMID: 23221588.
- Ben Abdelghani, K., Boussaa, H., Miladi, S., Zakraoui, L., Fazaa, A., Laatar, A. Value of Hands Ultrasonography in the Differential Diagnosis Between Psoriatic Arthritis and Rheumatoid Arthritis. J. Ultrasound Med. 2023, 42, 1987–1995. doi.org/10.1002/jum.16215.
- Aydin, S.Z., Mathew, A.J., Koppikar, S., Eder, L., Østergaard, M. Imaging in the diagnosis and management of peripheral psoriatic arthritis. Best Pract. Res. Clin. Rheumatol. 2020, 34, 101594. doi.org/10.1016/j.berh.2020.101594.
- Sapundzhieva, T., Sapundzhiev, L., Karalilova, R., Batalov, A. A Seven-Joint Ultrasound Score for Differentiating Between Rheumatoid and Psoriatic Arthritis. Curr. Rheumatol. Rev. 2022, 18, 329–337. doi.org/10.2174/1573397118666220215093323.
- Ramadan, A., Tharwat, S., Abdelkhalek, A., Eltoraby, E. Ultrasound detected synovitis, tenosynovitis and erosions in hand and wrist joints: A comparative study between rheumatoid and psoriatic arthritis. Rheumatology 2021, 59, 313–322. doi.org/10.5114/reum.2021.110550.
- Smerilli, G., Di Matteo, A., Cipolletta, E., Grassi, W., Filippucci, E. Enthesitis in Psoriatic Arthritis, the Sonographic Perspective. Curr. Rheumatol. Rep. 2021, 23, 75. doi.org/10.1007/s11926-021-01039-1.




