Neuro, Cardio, and Hepato Protection by Namodenoson
The Neuro- Cardio- and Hepato- Protective Effects of Namodenoson are Mediated by Adiponectin
Ohad Etzion ¹, Avital Bareket-Samish ², David Yardeni ¹, Pnina Fishman ³*
¹ Department of Gastroenterology and Liver Diseases, Soroka University Medical Center, Beer Sheva, Israel.
² BioInsight, Ltd., Binyamina, Israel.
³ Can-Fite BioPharma, Ramat Gan, Israel.
Email: [email protected]
OPEN ACCESS
PUBLISHED 28 February 2025
CITATION Etzion, O., Bareket-Samish, A., et al., 2025. The Neuro- Cardio- and Hepato- Protective Effects of Namodenoson are Mediated by Adiponectin. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6343
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6343
ISSN 2375-1924
ABSTRACT
Namodenoson (CF102, Cl-IB-MECA) is a small, water-insoluble, orally bioavailable highly selective A3 adenosine receptor agonist. Namodenoson exerts its clinical activity by binding to the A3 adenosine receptor, which has a role in both inflammation and cancer and was shown to be highly expressed on cancer and inflammatory cells. Namodenoson is currently under clinical development as a treatment for hepatocellular carcinoma (phase 3 trial is ongoing) and metabolic dysfunction-associated steatohepatitis (phase 2b trial is ongoing). Beyond the liver, namodenoson has also been shown to provide protective effects in the central nervous system and the cardiovascular system. The protective effects of namodenoson include anti-ischemic, anti-inflammatory, anti-toxicity, and anti-fibrotic effects. Adiponectin is a hormone produced by adipocytes. It is a polypeptide containing 244 amino acids and can be found in 3 forms (low, moderate, and high molecular weight). It binds to 3 receptors: adiponectin receptor 1, adiponectin receptor 2, and T-cadherin. Adiponectin plays protective roles in a variety of physiological functions across many organs, including the central nervous system, cardiovascular system, and the liver, as well as the kidneys, muscles, and bones. Similar to namodenoson, adiponectin provides anti-ischemic, anti-inflammatory, as well as anti-fibrotic protection, but it is also involved in energy regulation, protection against oxidative stress, promotion of insulin sensitivity, and more. The current review discusses the protective effects of namodenoson in the central nervous system, cardiovascular system, and liver focusing on the available preclinical evidence supporting such protective effects in each system, and presents evidence from preclinical models and translational data from the phase 2 trial in metabolic dysfunction-associated steatohepatitis protective effects are mediated via pathways that involve adiponectin.
Keywords: A3AR, adiponectin, cardioprotection, hepatoprotection, namodenoson, neuroprotection.
Introduction
Namodenoson (CF102, Cl-IB-MECA) is a small, water-insoluble, orally bioavailable highly selective agonist of A3 adenosine receptor (A3AR), one of 4 known adenosine receptors (A1, A2A, A2B, and A3). It is a synthetic ribose-based purine nucleoside leading to its A3AR specificity. Its molecular formula is C18H18CIN6O4, and its molecular weight is 544.73 Da.
A3AR is expressed primarily in the liver, kidney, heart, and lung, whereas its expression in the skeletal muscles and the brain is very limited. In tumor and inflammatory cells it is highly expressed, whereas the expression in the adjacent healthy tissue remains low. Interestingly, a recent study focusing on the nervous system demonstrated an expression of A3AR not only on the plasma membrane but also on the mitochondrial outer membrane. Binding of namodenoson to the A3AR leads to inhibition of phosphoinositide 3 kinase (PI3K) and subsequent deregulation of the Wnt/β-catenin and nuclear factor kappa B (NF-kB) pathways, ultimately causing apoptosis of these cells. Accordingly, namodenoson is currently under clinical development as a treatment for both hepatocellular carcinoma (HCC) where a phase 3 trial is ongoing, and for metabolic dysfunction-associated steatohepatitis (MASH) where phase 2b trial is ongoing.
Interestingly, namodenoson has been shown by us and others to have anti-ischemic, anti-inflammatory, anti-toxicity, and anti-fibrotic effects that provide protective benefits across organ systems. The first three effects were reported in the central nervous system (CNS) and the cardiovascular system. All four effects were reported in the liver. These protective effects suggest that namodenoson may offer advantages in terms of safety and efficacy as a treatment for oncological or inflammatory diseases, and specifically in liver diseases.
Adiponectin is a hormone produced by adipocytes. It is a polypeptide containing 244 amino acids and can be found in 3 forms. These include a low molecular weight (LMW) form (a trimer, approximately 90 kDa), a moderate molecular weight (MMW) form (a hexamer, approximately 180 kDa), and a high molecular weight (HMW) form, which includes a variety of multimers with molecular weights over 400 kDa. In circulation, adiponectin can be found in multimeric forms or as globular adiponectin (gAd) formed by cleaving the full-length form by leukocyte esterase. Each of the adiponectin forms mediates distinct functions.
Adiponectin binds to 3 receptors: adiponectin receptor 1 (AdipoR1), adiponectin receptor 2 (AdipoR2), and T-cadherin. AdipoR1 and AdipoR2 are highly expressed in the brain, muscle, liver, and adipose tissue in humans. T cadherin is widely expressed in the cardiovascular system. The receptors differ in their affinity to the different forms of adiponectin (e.g., AdipoR1 is a high-affinity receptor for gAd, whereas T-cadherin binds to the HMW forms of adiponectin). Interestingly, in alignment with the findings of adiponectin receptors in the brain, adiponectin was demonstrated to cross the blood-brain barrier. Adiponectin was identified in the cerebral spinal fluid (CSF) in humans, but only in its trimeric and hexameric forms. The CSF concentrations of adiponectin were approximately 0.1% of those found in the serum.
The current review discusses the neuro-, cardio-, and hepato- protective effects of namodenoson and the evidence supporting the hypothesis that these protective effects are mediated via adiponectin.
in the Central Nervous Systems
Namodenoson has numerous protective effects on the nervous system, including anti-ischemic, anti-inflammatory, and anti-toxicity (attenuating chemotherapy-induced neuropathic pain) effects. The protective anti-ischemic effects of namodenoson in the brain were investigated in several in-vitro and in-vivo models. These studies included in-vitro examination utilizing primary cortical cultures derived from rat embryos which demonstrated that pretreatment with namodenoson protected against hypoxia-induced decrease in cell viability.
Furthermore, in an in-vivo model of focal cerebral ischemia induced by transient middle cerebral artery (MCA) ligation, namodenoson administration was associated with significantly decreased infarction volume and higher locomotor activity compared to control. The effect of namodenoson on the infarction volume was mediated through A3AR, as repeating the procedures in A3AR knockout vs wild-type (WT) mice revealed that without namodenoson pretreatment, the infarction volume was larger in the knockout mice compared to WT, and that the namodenoson pretreatment had a protective effect only in the WT but not in the knockout mice. In another study evaluating oxygen and glucose deprivation (OGD) in hippocampal slices obtained from rats by field excitatory postsynaptic potentials (fEPSPs), which were recorded extracellularly at the dendritic level of the CA1 pyramidal region, long administration of namodenoson (before, during, and after the OGD) was found to protect the hippocampal slices from irreversible fEPSP depression induced by seven minutes of OGD. Namondeson treatment also prevented/delayed anoxic depolarization appearance. Notably, no namodenoson effect was observed under normoxic conditions.
In addition to the anti-ischemic effects, namodenoson was also shown to have protective anti-inflammatory effects in the CNS. For example, in a rat model of experimental subarachnoid hemorrhage (SAH), namodenoson attenuated neurological impairment and brain edema, as well as inhibited activation of microglia, the innate immune cells of the CNS, and reduced the expression of pro-inflammatory cytokines. In a subsequent study investigating the effect of namodenoson in an in-vivo SAH model in aging rats, treating the rats with namodenoson before SAH significantly improved neurological functions, and this effect was mediated via A3AR, as this improvement was impaired upon treating the rats with the selective A3AR antagonist MRS1523. Moreover, neuronal apoptosis observed in the SAH group was dramatically attenuated by namodenoson treatment, and this effect was also blocked by MRS1523. Analyses of mRNA and protein levels after SAH revealed that namodenoson treatment was associated with considerably decreased levels of pro-inflammatory cytokines interleukin (IL)-1β and tumor necrosis factor (TNF)-α that was partially reversed by MRS1523, whereas the levels of the anti-inflammatory cytokines IL-4 and IL-10 increased compared to the SAH group. The increased levels of IL-4 and IL-10 were also shown in an in-vitro system of microglia cells. Interestingly, findings from a recent in-vitro murine study are not aligned with the aforementioned results. This study, in which total RNA was isolated from activated primary microglia cells with or without namodenoson treatment followed by mRNA sequencing, demonstrated that treatment with namodenoson led to downregulation of certain pro-inflammatory genes (most notably, CCL19), but also to upregulation of other pro-inflammatory genes (most notably, CXCL11), suggesting that additional studies are required to better elucidate the effect of namodenoson (dosages, timing, microglia activation approach used, etc). Another recent study focused on the neuroprotection effects of namodenoson in murine primary microglia cells and the HMC3 cell line (human primary microglial cells) related to mitochondrial function and oxidative stress. In activated primary microglial cells, namodenoson treatment was associated with upregulation of nuclear genes that are involved (directly or indirectly) in redox homeostasis, and downregulation of nuclear genes that are relevant to the immunological function of microglia. Analysis of mitochondrial respiration using activated HMC3 cells as a model demonstrated that namodenoson in combination with the A2AR antagonist SCH582561 but not by itself protected the mitochondria from the maximal and compensatory respiratory pro-inflammatory effects, pointing to a potential role of dual targeting of the A3 and A2A adenosine receptor.
The protective anti-toxic effect of namodenoson in the nervous system involves its mitigation of chemotherapy-induced peripheral neuropathy (CIPN), which occurs in up to 85% and more of patients receiving chemotherapy with severity depending on the chemotherapeutic agents used and the duration of treatment. In a murine model of neuropathic pain induced by chronic constriction injury (CCI) of the sciatic nerve, intraperitoneal administration of namodenoson was shown to reverse the development of established mechanoallodynia (abnormal pain response to mechanical stimuli), as measured by probing the paws of the mouse (ipsilaterally, and contralaterally as a control) with increasing pressure and determining the paw withdrawal threshold. The observed protective effect of namodenoson was mediated by A3AR, as it was blocked by MRS1523.
in the Cardiovascular System
Namodenoson has been shown to render multiple protective effects in the cardiovascular system, which were first demonstrated over 25 years ago. Since then, the protective roles of namodenoson in preclinical models of ischemia, myocardial infarction, and ischemia/reperfusion (I/R) injury have been well-established. The studies consistently demonstrated that the effect of namodenoson is mediated via A3AR, by either using specific A3AR antagonists or by employing A3AR knockout animal models.
| Author, year | Models | Main findings |
|---|---|---|
| Stambaugh et al., 1997 | In vitro: Primary cardiac ventricular myocytes (cultured from chick embryos), under prolonged hypoxic conditions. | Reduction in hypoxia-induced injury demonstrated by increased cell viability and a decrease in the amount of CK released. The reduction was dose-dependent and blocked by MRS1191 and MRS1097 (selective A3AR antagonists). |
| Thourani et al., 1999 | Ex vivo: Cardioplegia model using isolated perfused rat hearts. | Administering namodenoson before the cardioplegic procedure protected against post-ischemic left ventricular dysfunction, myocardial edema, and CK release. The protective effects were not observed when namodenoson was administered during the cardioplegic procedure. |
| Shneyvays et al., 2002 | In vitro: Primary cardiac myocyte culture derived from newborn rat hearts and treated in a hypoxic chamber in glucose-free media. | Namodenoson treatment during the hypoxic treatment protected against myocyte injury. It attenuated hypoxia-induced LDH release, as well as hypoxia-induced decrease in ATP levels. The protective effect (measured by LDH release) was mediated by A3AR since it was blocked by MRS1523 (a selective A3AR antagonist) and was dose-dependent. |
| Harrison et al., 2002 | Ex vivo: I/R injury in isolated perfused hearts from mice. | Namodenoson significantly enhanced post-ischemic contractile recovery (left ventricular function) and did not impact coronary re-flow. The effect was mediated by A3AR since it was blocked by MRS1220 (a selective A3AR antagonist). |
| Maddock et al., 2002 | In vitro: Hypoxia/reoxygenation in primary cardiac myocyte culture derived from adult rats. Ex vivo: I/R injury in perfused hearts from rats. | In vitro: The presence of namodenoson during the reoxygenation phase attenuated hypoxia-induced necrosis and apoptosis. Ex vivo: The presence of namodenoson during the reperfusion phase limited the infarct size. In both in-vitro and ex-vivo models, the effect was dose-dependent (at least up to a certain concentration) and mediated by A3AR since it was blocked by MRS1191 (a selective A3AR antagonist). |
| Hochhauser et al., 2004 | Ex vivo: I/R injury in isolated perfused hearts from rats. | Isolated hearts were either perfused with namodenoson or namodenoson was injected prior to the I/R procedure. The beneficial effects were mainly in the injected group. In the injected group, namodenoson attenuated left ventricular dysfunction and coronary flow, and the effect was mediated via A3AR, as it was blocked by MRS1523 (a selective A3AR antagonist). Namodenoson also reduced infarct size and attenuated the following I/R-induced effects: CK release, decrease in ATP levels, and mitochondrial damage. |
| Zatta et al., 2006 | Ex vivo: I/R injury in isolated perfused hearts from mice. | Namodenoson treatment (pre-ischemia and for the first 10 min of the reperfusion) protected against impaired sensitivity to 2-chloroadenosine and ADP, limited post-ischemic coronary dysfunction and improved peak flow in post-ischemic hearts. |
| Ge et al., 2006 | In vivo: I/R injury in WT and A3AR knockout mice. Ex vivo: I/R injury in isolated perfused hearts from WT and A3AR knockout mice. | In vivo: Pretreatment with namodenoson reduced in a dose-dependent manner, the size of myocardial infarct induced by I/R in WT but not in A3AR knockout mice. The effect in WT was blocked by MRS1523 (a selective A3AR antagonist). Ex vivo: Namodenoson protected against left ventricular dysfunction in isolated hearts from WT but not from A3AR knockout mice. |
| Hochhauser et al., 2007 | Ex vivo: I/R injury in isolated perfused hearts from normotensive and hypertensive strains of rats. | The isolated hearts were perfused with namodenoson or namodenoson was injected before the I/R procedure. The beneficial effects were seen more in the injected group. Namodenoson was beneficial in the recovery of left ventricular function, coronary flow and ATP levels in hearts from both strains. The recovery of left ventricular recovery dysfunction was better in the normotensive strain. |
| Ge et al., 2010 | In vivo: I/R injury in WT, A3AR knockout mice, and a chimeric mice without A3AR expression in the bone marrow. | In WT mice, namodenoson administered during the reperfusion phase reduced infarct size, and limited leukocyte infiltration into the myocardium. The protective effect was mediated by A3AR expressed in the bone marrow, as it was not seen in A3AR knockout mice, or in chimeric mice without A3AR expression in the bone marrow. |
| Chanysheva et al., 2012 | In vitro: Primary cardiac myocyte culture derived from newborn rat hearts. | Namodenoson treatment during the hypoxic treatment attenuated hypoxia-induced LDH release. |
| Hussain et al., 2014 | Ex vivo: I/R injury in isolated perfused hearts from rats. In vitro: Hypoxia/reoxygenation in primary myocyte culture derived from adult rats. | Ex vivo: Namodenoson administered during the reperfusion phase significantly reduced infarct size. This effect was abolished by either a PI3K or MEK1/2 inhibitor. In vitro: Namodenoson presence during reoxygenation reduced apoptosis, necrosis, cleaved-caspase 3 activity and increased phosphorylation of BAD at Ser-112 and at Ser-136. All these effects were abolished by either a PI3K or MEK1/2 inhibitor. |
The anti-inflammatory properties of namodenoson were specifically demonstrated in an in-vivo murine model of I/R injury. Consistent with the other I/R studies, this in-vivo study showed that receiving namodenoson during the reperfusion phase was associated with significantly reduced infarction size. It also showed that namodenoson administration led to a significantly lower number of leukocytes infiltrating into the ischemic-reperfused myocardium. This effect was A3AR-mediated as it was not observed in A3AR knockout mice or chimeric mice with no A3AR expression in bone-marrow-derived cells. The researchers attributed this anti-inflammatory effect to the inhibition of neutrophil migration through activation of the A3AR in the bone marrow.
An additional protective effect observed preclinically with namodenoson treatment is the mitigation of chemotherapy-induced cardiotoxicity and specifically that of doxorubicin-induced cardiotoxicity, which occurs in up to 9% of patients with cancer receiving this drug. This protective effect was observed preclinically both in vitro and in vivo. In studies using primary cardiac myocyte culture derived from newborn rats, incubation of the cells with doxorubicin for 18 h, was shown to result in cell damage. This damage was attenuated by namodenoson introduced prior to and during the treatment with doxorubicin. The protective effect was mediated via A3AR, as it was considerably reduced by MRS1523. This protection reduced cell loss and attenuated several doxorubicin-induced dysfunctions such as diminished sarcoplasmic reticulum calcium uptake, mitochondrial calcium overload, generation of free radicals, peroxidation of lipids, mitochondrial damage, and decreased ATP production. Consistent with the in-vitro studies, in-vivo studies in rats demonstrated that namodenoson administration protected against doxorubicin-induced left ventricular dysfunction, bradycardia, ST-segment elevation, increased levels of creatine kinase (CK)-MB, and myocardial degeneration. This cardioprotective effect was A3AR-mediated, as it was blocked by MRS1523.
in the Liver
The protective effects of namodenoson in the liver include anti-ischemic, anti-inflammatory, anti-fibrotic, and anti-toxicity effects. The anti-ischemic effects were evaluated in a study using a surgical procedure to induce I/R injury in rats by first clamping the blood flow to the liver and then were administered by subcutaneous injection during the reperfusion phase and orally thrice daily thereafter. Serum levels of alanine transaminase (ALT) and aspartate aminotransferase (AST) were used as proxy for the I/R injury-induced liver damage. Namodenoson attenuated the I/R liver injury as serum ALT and AST levels were significantly reduced compared to control 48 h after the I/R procedure. Furthermore, histopathological analyses 48 h after the I/R procedure revealed that unlike the control group which exhibited large necrotic areas surrounded by inflammatory cells, in the namodenoson-treated rats, no necrotic areas were observed and only a few inflammatory cells were observed. A similar protective effect was observed in rats that underwent liver hepatectomy on 70% of their liver, whilst being exposed to ischemia for 10 min. Administration of namodenoson during the injection and orally thrice daily thereafter attenuated the increase in ALT and AST observed in the control as well as increased the regeneration of the remnant liver.
The anti-toxic effects of namodenoson were evaluated in the context of its anti-inflammatory effects in concanavalin A (Con. A)-induced liver injury. Namodenoson treatment attenuated the Con. A-induced increases in serum ALT and AST levels compared to control. Furthermore, pathological analyses revealed that the livers of the control mice were characterized by an extensive area of necrosis at the parenchyma whereas those of the namodenoson-treated mice exhibited no signs of necrosis. This pathologic finding was consistent with decreased expression of pro-apoptotic proteins (FasR, Bax, and Bad) in the namodenoson-treated mice vs controls. Another study using a murine STAM model demonstrated that treatment with namodenoson significantly less inflammation as was evident from histopathological analysis of H&E-stained liver sections from namodenoson-treated animals vs controls.
The anti-fibrotic effects of namodenoson in the liver were assessed in two studies, both of which used a murine model of carbon tetrachloride (CCL4)-induced liver fibrosis. Both studies demonstrated that in this model, not only did namodenoson (intraperitoneal administration twice daily) show its liver protective characteristics by significantly attenuating the CCL4-induced increase in ALT and AST, it also significantly reduced the CCL4-induced fibrosis, as evident from histopathological analyses of liver sections stained with Sirius Red.
Adiponectin Effects in the Central Nervous System, Cardiovascular System, and the Liver
Adiponectin plays protective roles in a variety of physiological functions across many organs, including the CNS, cardiovascular system, and the liver, as well as the kidneys, muscles, and bones. Overall, it provides anti-ischemic, anti-inflammatory, as well as anti-fibrotic protection, and in addition, was shown to be involved in energy regulation, protection against oxidative stress, promotion of insulin sensitivity, and more. In the CNS, adiponectin has protective effects in numerous anxiety/depression. The anti-ischemic and anti-inflammatory effects of adiponectin were evident in preclinical studies in mice, where adiponectin-knockout mice exhibited larger infarction size and greater neurological function deficits after an ischemic injury compared to WT. Supplementing mice with adiponectin (via adenoviral vector) reduced the infarct size in both the knockout and WT mice. In the cardiovascular system, adiponectin has also been linked to several pathologic states including cardiac hypertrophy, ischemic heart disease, and chronic heart failure. A recent meta-analysis of preclinical animal studies examined the cardioprotective effects of adiponectin against myocardial I/R injury. The meta-analysis, which included 20 studies and 426 animals, demonstrated that adiponectin significantly reduced the infarct size, improved left ventricular function, as well as reduced inflammatory factor (TNFα), oxidative stress indicators (superoxide production), and apoptosis indicators (caspase 3), supporting a multi-faceted cardioprotective role for adiponectin. In the liver, adiponectin has been linked to metabolic dysfunction-associated steatotic liver disease (MASLD)/MASH and HCC. Specifically, adiponectin was shown to prevent hepatic steatosis, inflammation, and fibrosis, promote apoptosis and antagonize pathways related to cell proliferation, migration, and invasion. For example, in a mouse model where MASH was induced by a choline-deficient L-amino acid defined diet, after a week on this diet, hepatic steatosis was greater in an adiponectin knockout mice compared to WT, and after 24 weeks, approximately half of the knockout mice developed liver cirrhosis and hepatic tumors vs none of the 15 WT mice. Furthermore, in a mouse model where MASH was induced by high-fat diet with low methionine and no added choline, administration of an adiponectin replacement therapy (an adiponectin-derived peptide), significantly ameliorated hepatic steatosis, inflammation, and fibrosis, thus preventing the development of MASH.
The Protective Effects of Namodenoson are Adiponectin-mediated: Preclinical and Clinical Evidence
Several lines of evidence demonstrate that namodenoson exerts its protective effects through adiponectin. In an in-vitro study examining the impact of namodenoson on human bone marrow mesenchymal stem cells (hBM-MSCs), in which adipocyte differentiation was induced, treatment with namodenoson resulted in a significant increase in adiponectin levels as determined by enzyme-linked immunosorbent assay (ELISA). Namodenoson resulted in 2.2- and 6.0-fold increase in adiponectin levels, respectively, compared to control. Similarly, treating murine adipocytes with 10 nM namodenoson was also associated with increased levels of adiponectin expression compared to control.
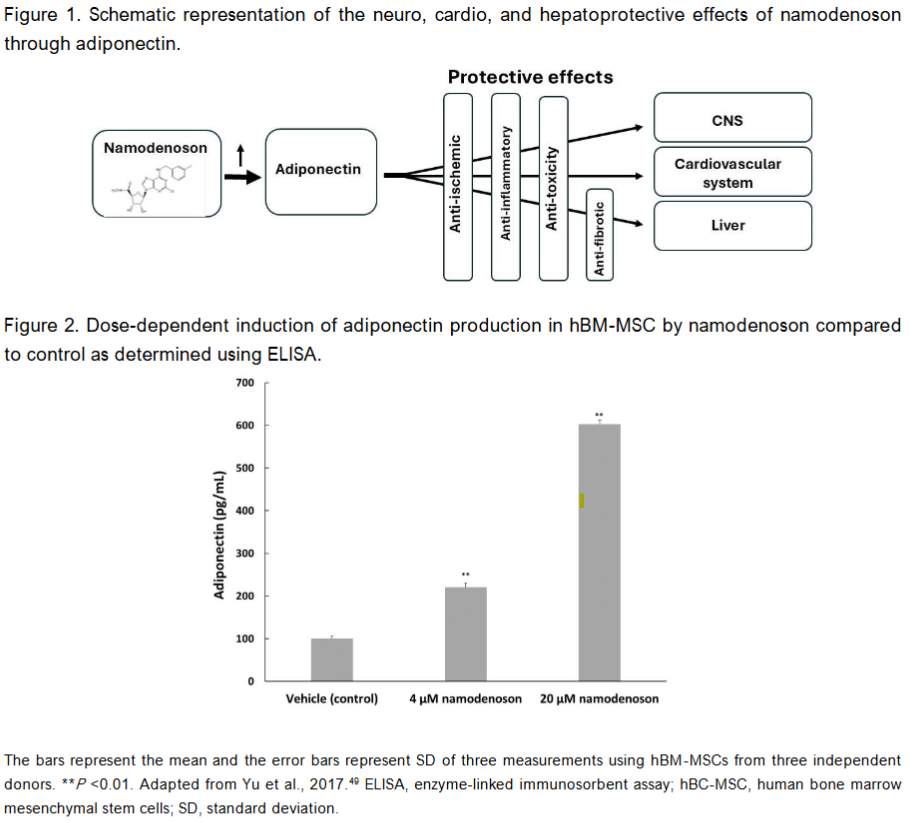
The findings of these in-vitro analyses are supported by in-vivo studies utilizing the murine STAM model which mimics the progression of human MASH and HCC by using male C57BL/6 mice with a background of late type 2 diabetes (T2D) to which a single dose of streptozotocin is administered followed by a high-fat diet. The study showed that daily) for 4 weeks was associated with significantly increased adiponectin levels in the liver compared to vehicle, as determined by immunohistochemistry staining of liver sections.
Data from the phase 2 clinical trial of oral namodenoson in MASLD/MASH further supports this mechanistic link. This study randomized 60 MASLD patients with or without MASH to 12 weeks of namodenoson 12.5 mg BID (n = 21), namodenoson 25 mg BID (n = 19), or placebo (n = 20). The main endpoints of the study included serum ALT levels at 12 weeks and safety. One of the secondary endpoints was the change from baseline (CFB) in serum adiponectin levels at 12 weeks. The CFB was statistically significantly higher in the 12.5 mg BID arm compared to placebo (mean, 539 ng/mL vs -78 ng/mL, P = 0.032), and was numerically but not statistically significantly higher in the 25 mg BID arm compared to placebo (mean, 220 ng/mL vs -78 ng/mL, P = 0.216).
Conclusions
The preclinical evidence supporting the protective characteristics of namodenoson and those of adiponectin in the CNS, cardiovascular system, and the liver is extensive. The mirroring of the effects rendered by namodenoson and adiponectin combined with the available preclinical and clinical evidence showing that administering namodenoson is associated with an increase in adiponectin, suggests that the protective effects of namodenoson are mediated by pathways that involve adiponectin.
The protective effects of namodenoson could explain its excellent safety profile observed in the clinical trials evaluating namodenoson for MASLD/MASH and HCC. Furthermore, these effects suggest that the role of namodenoson could potentially be expanded beyond liver diseases to include conditions in the CNS or cardiovascular system, or to be an add-on drug to address chemotherapy-induced toxicities.
Conflict of Interest:
P. Fishman is employed by and owns stock in Can-Fite BioPharma Ltd.; O. Etzion and D. Yardeni received research funds from Can-Fite BioPharma, Ltd. A. Bareket-Samish has a consultant role in Can-Fite BioPharma; Ltd.
Funding Statement:
Not applicable.
Acknowledgements:
None.
Author Contributions:
Writing original draft preparation, P. Fishman and A. Bareket-Samish. Writing review and editing, all authors. All authors have read and agreed to the published version of the manuscript.
References:
- Fishman P, Stemmer SM, Bareket-Samish A, et al. Targeting the A3 adenosine receptor to treat hepatocellular carcinoma: anti-cancer and hepatoprotective effects. Purinergic Signal. 2023; 19(3):513-22.
- Fishman P. Drugs targeting the A3 adenosine receptor: Human clinical study data. Molecules. 2022; 27(12).
- Etzion O, Bareket-Samish A, Yardeni D, et al. Namodenoson at the crossroad of metabolic dysfunction-associated steatohepatitis and hepatocellular carcinoma. Biomedicines. 2024; 12(4).
- Jacobson KA, Gao ZG. Adenosine receptors as therapeutic targets. Nat Rev Drug Discov. 2006; 5(3):247-64.
- Doyle TM, Janes K, Xiao WH, et al. Mitochondrial A(3) adenosine receptor as a mechanism for the protective effects of A(3)AR agonists on chemotherapy-induced neuropathic pain. J Neurosci. 2024. Online ahead of print.
- Clinicaltrials.gov. Description of the “Namodenoson in the treatment of advanced hepatocellular carcinoma in patients with Child-Pugh Class B7 cirrhosis (LIVERATION)” study (NCT05201404). Available at: https://www.clinicaltrials.gov/study/NCT05201404. Accessed Jan 2, 2025.
- Clinicaltrials.gov. Description of the “Namodenoson in the treatment of non-alcoholic steatohepatitis (NASH)” study (NCT04697810). Available at: https://clinicaltrials.gov/search?term=NCT04697810. Accessed Jan 2, 2025.
- Khoramipour K, Chamari K, Hekmatikar AA, et al. Adiponectin: Structure, physiological functions, role in diseases, and effects of nutrition. Nutrients. 2021; 13(4).
- Bloemer J, Pinky PD, Govindarajulu M, et al. Role of adiponectin in central nervous system disorders. Neural Plast. 2018; 2018:4593530.
- Kusminski CM, McTernan PG, Schraw T, et al. Adiponectin complexes in human cerebrospinal fluid: distinct complex distribution from serum. Diabetologia. 2007; 50(3):634-42.
- Chen Z, Janes K, Chen C, et al. Controlling murine and rat chronic pain through A3 adenosine receptor activation. FASEB J. 2012; 26(5):1855-65.
- Haddad M, Alsalem M, Aldossary SA, et al. The role of adenosine receptor ligands on inflammatory pain: possible modulation of TRPV1 receptor function. Inflammopharmacology. 2023; 31(1):337-47.
- Chen GJ, Harvey BK, Shen H, et al. Activation of adenosine A3 receptors reduces ischemic brain injury in rodents. J Neurosci Res. 2006; 84(8):1848-55.
- Pugliese AM, Coppi E, Volpini R, et al. Role of adenosine A3 receptors on CA1 hippocampal neurotransmission during oxygen-glucose deprivation episodes of different duration. Biochem Pharmacol. 2007; 74(5):768-79.
- Luo C, Yi B, Tao G, et al. Adenosine A3 receptor agonist reduces early brain injury in subarachnoid haemorrhage. Neuroreport. 2010; 21(13):892-6.
- Li P, Li X, Deng P, et al. Activation of adenosine A3 receptor reduces early brain injury by alleviating neuroinflammation after subarachnoid hemorrhage in elderly rats. Aging (Albany NY). 2020; 13(1):694-713.
- Lillo A, Serrano-Marin J, Lillo J, et al. Gene regulation in activated microglia by adenosine A(3) receptor agonists: a transcriptomics study. Purinergic Signal. 2024; 20(3):237-45.
- Serrano-Marin J, Valenzuela R, Delgado C, et al. Neuroprotective compounds alter the expression of genes coding for proteins related to mitochondrial function in activated microglia. Mitochondrion. 2024; 78:101934.
- Mattar M, Umutoni F, Hassan MA, et al. Chemotherapy-induced peripheral neuropathy: A recent update on pathophysiology and treatment. Life (Basel). 2024; 14(8).
- Stambaugh K, Jacobson KA, Jiang JL, et al. A novel cardioprotective function of adenosine A1 and A3 receptors during prolonged simulated ischemia. Am J Physiol. 1997; 273(1 Pt 2):H501-5.
- Thourani VH, Ronson RS, Jordan JE, et al. Adenosine A3 pretreatment before cardioplegic arrest attenuates postischemic cardiac dysfunction. Ann Thorac Surg. 1999; 67(6):1732-7.
- Shneyvays V, Mamedova LK, Leshem D, et al. Insights into the cardioprotective function of adenosine A(1) and A(3) receptors. Exp Clin Cardiol. 2002; 7(2-3):138-45.
- Harrison GJ, Cerniway RJ, Peart J, et al. Effects of A(3) adenosine receptor activation and gene knock-out in ischemic-reperfused mouse heart. Cardiovasc Res. 2002; 53(1):147-55.
- Maddock HL, Mocanu MM, Yellon DM. Adenosine A(3) receptor activation protects the myocardium from reperfusion/reoxygenation injury. Am J Physiol Heart Circ Physiol. 2002; 283(4):H1307-13.
- Hochhauser E, Kaminski O, Shalom H, et al. Role of adenosine receptor activation in antioxidant enzyme regulation during ischemia-reperfusion in the isolated rat heart. Antioxid Redox Signal. 2004; 6(2):335-44.
- Zatta AJ, Matherne GP, Headrick JP. Adenosine receptor-mediated coronary vascular protection in post-ischemic mouse heart. Life Sci. 2006; 78(21):2426-37.
- Ge ZD, Peart JN, Kreckler LM, et al. Cl-IB-MECA [2-chloro-N6-(3-iodobenzyl)adenosine-5′-N-methylcarboxamide] reduces ischemia/reperfusion injury in mice by activating the A3 adenosine receptor. J Pharmacol Exp Ther. 2006; 319(3):1200-10.
- Hochhauser E, Leshem D, Kaminski O, et al. The protective effect of prior ischemia reperfusion adenosine A1 or A3 receptor activation in the normal and hypertrophied heart. Interact Cardiovasc Thorac Surg. 2007; 6(3):363-8.
- Ge ZD, van der Hoeven D, Maas JE, et al. A(3) adenosine receptor activation during reperfusion reduces infarct size through actions on bone marrow-derived cells. J Mol Cell Cardiol. 2010; 49(2):280-6.
- Hussain A, Gharanei AM, Nagra AS, et al. Caspase inhibition via A3 adenosine receptors: a new cardioprotective mechanism against myocardial infarction. Cardiovasc Drugs Ther. 2014; 28(1):19-32.
- Chanyshev B, Shainberg A, Isak A, et al. Anti-ischemic effects of multivalent dendrimeric A(3) adenosine receptor agonists in cultured cardiomyocytes and in the isolated rat heart. Pharmacol Res. 2012; 65(3):338-46.
- Duangrat R, Parichatikanond W, Chanmahasathien W, et al. Adenosine A(3) receptor: From molecular signaling to therapeutic strategies for heart diseases. Int J Mol Sci. 2024; 25(11).
- Linders AN, Dias IB, Lopez Fernandez T, et al. A review of the pathophysiological mechanisms of doxorubicin-induced cardiotoxicity and aging. NPJ Aging. 2024; 10(1):9.
- Shneyvays V, Mamedova L, Zinman T, et al. Activation of A(3) adenosine receptor protects against doxorubicin-induced cardiotoxicity. J Mol Cell Cardiol. 2001; 33(6):1249-61.
- Emanuelov AK, Shainberg A, Chepurko Y, et al. Adenosine A3 receptor-mediated cardioprotection against doxorubicin-induced mitochondrial damage. Biochem Pharmacol. 2010; 79(2):180-7.
- Galal A, El-Bakly WM, Al Haleem EN, et al. Selective A3 adenosine receptor agonist protects against doxorubicin-induced cardiotoxicity. Cancer Chemother Pharmacol. 2016; 77(2):309-22.
- Ohana G, Cohen S, Rath-Wolfson L, et al. A3 adenosine receptor agonist, CF102, protects against hepatic ischemia/reperfusion injury following partial hepatectomy. Mol Med Rep. 2016; 14(5):4335-41.
- Cohen S, Stemmer SM, Zozulya G, et al. CF102 an A3 adenosine receptor agonist mediates anti-tumor and anti-inflammatory effects in the liver. J Cell Physiol. 2011; 226(9):2438-47.
- Fishman P, Cohen S, Itzhak I, et al. The A3 adenosine receptor agonist, namodenoson, ameliorates nonalcoholic steatohepatitis in mice. Int J Mol Med. 2019; 44(6):2256-64.
- Yang L, Gao ZW, Wang X, et al. The different effects of four adenosine receptors in liver fibrosis. Front Pharmacol. 2024; 15:1424624.
- Nishimura M, Izumiya Y, Higuchi A, et al. Adiponectin prevents cerebral ischemic injury through endothelial nitric oxide synthase dependent mechanisms. Circulation. 2008; 117(2):216-23.
- Miao J, Shen LH, Tang YH, et al. Overexpression of adiponectin improves neurobehavioral outcomes after focal cerebral ischemia in aged mice. CNS Neurosci Ther. 2013; 19(12):969-77.
- Ouchi N, Shibata R, Walsh K. Cardioprotection by adiponectin. Trends Cardiovasc Med. 2006; 16(5):141-6.
- Yue H, Zhang Q, Chang S, et al. Adiponectin protects against myocardial ischemia-reperfusion injury: a systematic review and meta-analysis of preclinical animal studies. Lipids Health Dis. 2024; 23(1):51.
- Vachliotis ID, Valsamidis I, Polyzos SA. Tumor necrosis factor-alpha and adiponectin in nonalcoholic fatty liver disease-associated hepatocellular carcinoma. Cancers (Basel). 2023; 15(21).
- Polyzos SA, Kountouras J, Zavos C, et al. The role of adiponectin in the pathogenesis and treatment of non-alcoholic fatty liver disease. Diabetes Obes Metab. 2010; 12(5):365-83.
- Kamada Y, Matsumoto H, Tamura S, et al. Hypoadiponectinemia accelerates hepatic tumor formation in a nonalcoholic steatohepatitis mouse model. J Hepatol. 2007; 47(4):556-64.
- Huang Z, Sung HK, Yan X, et al. The adiponectin-derived peptide ALY688 protects against the development of metabolic dysfunction-associated steatohepatitis. Clin Transl Sci. 2024; 17(6):e13760.
- Yu J, Ahn S, Kim HJ, et al. Polypharmacology of N(6)-(3-Iodobenzyl)adenosine-5′-N-methyluronamide (IB-MECA) and related A(3) adenosine receptor ligands: Peroxisome proliferator activated receptor (PPAR) gamma partial agonist and PPARdelta antagonist activity suggests their antidiabetic potential. J Med Chem. 2017; 60(17):7459-75.
- Can-Fite Ltd., data on file. 2025.
- Safadi R, Braun M, Francis A, et al. Randomised clinical trial: A phase 2 double-blind study of namodenoson in non-alcoholic fatty liver disease and steatohepatitis. Aliment Pharmacol Ther. 2021; 54(11-12):1405-15.
- Stemmer SM, Benjaminov O, Medalia G, et al. CF102 for the treatment of hepatocellular carcinoma: a phase I/II, open-label, dose-escalation study. Oncologist. 2013; 18(1):25-6.
- Stemmer SM, Manojlovic NS, Marinca MV, et al. Namodenoson in advanced hepatocellular carcinoma and Child-Pugh B cirrhosis: Randomized placebo-controlled clinical trial. Cancers (Basel). 2021; 13(2).

