Prostate vs. Seminal Vesicle Cancer: Insights and Comparisons
Prostate Cancer and Seminal Vesicles Cancer – Lessons Learned from the Commonest and the Rarest Malignancy Types in Men
Ofer N. Gofrit
Department of Urology, Hadassah Hebrew University Hospital, Jerusalem.
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION Gofrit O.N., 2025. Prostate Cancer and Seminal Vesicles Cancer – Lessons Learned from the Commonest and the Rarest Malignancy Types in Men. Medical Research Archives, [online] 13(5). https://doi.org/10.18103/mra.v13i5.6566
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i5.6566
ISSN 2375-1924
Abstract
Purpose: The prostate and the seminal vesicles (SVs) are analogous in terms of pelvic location, function, and response to hormones; however, prostate cancer (PC) is the most common cancer in men, whereas adenocarcinoma of the SVs is one of the rarest cancer types. This manuscript studies the similarities and dissimilarities between these organs and through them attempts to provide insights into the pathogenesis of PC.
Views: The number of prostatic luminal (the origins cells of PC) and SV cells is similar (1.5 × 109 and 1.9 × 109 cells), and so is the number of daily mitotic cycles in these organs (3 × 106 in the prostate and 3.8 × 106 in the SVs). Thus, numerical differences cannot account for the difference in cancer incidence. Nevertheless, remarkable anatomical and physiological differences can be noted. The SVs are surrounded by loose connective tissue, whereas the prostate is surrounded by a non-expandable collagenous-muscular capsule. The SVs excrete into two thin and long ducts, whereas the prostate into multiple short ducts. The prostate is subjected to the high pressure generated by the contractions of the levator ani muscle on which it lies. By the end of the contraction, the intra-prostatic pressure drops, and urine is sucked from the urethra to the acini, exposing them to high concentrations of pro-inflammatory substances, such as monosodium-urate. Repeated exposures to high pressure and to urine initiate prostatic inflammation, commonly observed in prostate biopsies, but exceedingly rare in SVs. Inflammation promotes proliferative inflammatory atrophy which can degenerate into prostatic intraepithelial neoplasia and to PC. Neutralizing the deleterious components of urine could possibly prevent PC.
Conclusions: The prostate and SVs are similar in number of cells and in daily mitotic cycles. The prostate with its non-expandable capsule and short excretory ducts is repeatedly exposed to urine at high pressures. This offence initiates a chain of events that starts with inflammation and continues with proliferative inflammatory atrophy and PC. The SVs are not exposed to these insults.
Keywords
Prostate cancer, Adenocarcinoma of the seminal vesicles, Urine reflux, Intraprostatic pressure, Proliferative inflammatory atrophy
Introduction
The seminal vesicles (SVs) and prostate are the most important male accessory sex glands. Their secretions constitute most of the seminal fluid volume, with the SVs accounting for 70–80% and the prostate to 20–30% of the seminal fluid volume. The prostate develops from epithelial buds originating in the urogenital sinus (Endoderm) during the 10th week of gestation, whereas the SVs develop from the mesonephric (Wolffian) duct (Mesoderm) around the 12th week of gestation. Both glands respond to testosterone and exhibit 5-alpha reductase activity.
Prostate cancer (PC) is the most common type of cancer affecting men. A total of 313,780 men are expected to be diagnosed with prostate cancer in the US in 2025, with 35,770 patients to succumb to this disease 1. In contrast, malignancies of the SVs are extremely rare. In fact, the commonest type of SVs cancer is invasion by PC, a condition that is more common in base-dominant tumors and has a variable effect of patient’s prognosis 2,3. There are approximately 60 recorded cases of primary adenocarcinoma of the SVs in the medical literature 4. The risk factors of PC are well known and include age, race, family history and mutations like in the BRCA1/2 genes and Lynch syndromes, the pathogenesis of PC is mostly obscure. This manuscript theorizes that study of the similarities and dissimilarities between the prostate and SVs in terms of anatomy, histology, physiology, mechanical pressures and exposures to body fluids can provide the missing links in the pathogenesis of PC and hopefully suggests ways for its prevention.
The following issues will be addressed:
- The number of normal cells in the prostate vs. those in the SVs (assuming that cancer originates from previously normal luminal cells).
- The number of daily mitoses in the prostate vs. those in the SVs (assuming that mitosis is the cell-cycle phase most vulnerable to mutations).
- The anatomical differences between the prostate and SVs.
- The pressures applied to the prostate vs. those applied to the SVs.
- The degree of exposure of the prostate and SVs to urine and sperm.
- The potential effects of exposure to urine.
- The degree and role of inflammation in the prostate and SVs.
- Age-related changes in the prostate and SVs.
Comparison of the prostate and the seminal vesicles
COMPARISON BETWEEN THE NUMBER OF NORMAL CELLS IN THE PROSTATE AND SEMINAL VESICLES
The prostate is composed of transitional zone- the source of benign prostatic hyperplasia, anterior fibromuscular zone, central zone and peripheral zone- the source of prostate cancer. Two layers of cells, basal and luminal, cover the normal prostatic acini. Prostate cancer is believed to arise from the luminal cells 5. Thus, determining the number of luminal cells in the peripheral zone of the prostate is imperative. Prostatic volume is highly variable and tends to increase with age. Notably, most of the increase occurs in the transition zone. Changes in the volume of the peripheral zone are much smaller. The “normal” prostatic volume is approximately 25 cc 6. This volume will be used to calculate the number of susceptible cells in the prostate. The peripheral volume of the normal prostate accounts for about 50% of total gland volume and is therefore, approximately 12 cc 7. Prostatic glands occupy approximately half of this volume (6 cc); the remaining is stroma.
A typical adult prostatic acinus is an ellipsoid, with dimensions of 500 × 250 × 250 µm 8. The volume of a single prostatic acinus (V1) can be determined using the ellipsoid volume formula (v = 4/3π × a axis × b axis × c axis): 4/3π × 500 µ × 250 µ × 250 µ = 1.3 × 108 µm3. The diameter of the cellular lining of the acinus is approximately 60 µm. Accordingly, the volume of prostatic lumen (V2) is: 4/3π × 440 µm × 190 µm × 190 µm = 6.6 × 107 µm3. The volume of the acinar cells (V3) is V1-V2: 1.3 × 108 µm3 – 6.6 × 107 = 6.4 × 107 µm3. Since the volume of the luminal cells (V4) is half that of the glandular cells: V3/2 = 3 cc or 3 × 1012 µm3. The volume of a single luminal prostatic cell ranges from 1000 µm3 to 3000 µm3. Considering an average volume of 2000 µm3, the number of epithelial luminal cells in the peripheral zone is: 3 × 1012 µm3/ 2000 µm3 = 1.5 × 109 cells.
The SVs volume is highly variable. The mean volume of each gland is 7.1 cc (SD 5.2); however, organs as small as 1.9 cc and as large as 12.6 cc have been reported 9,10. An average volume of 14.2 cc (both glands) will be considered in further calculations. Three-dimensional images of seminal vesicles revealed that the luminal structure of SVs is similar to that of their external morphology. Each SV comprises an average of 9.2 curls and 11.8 diverticula. Most of the SV volume is occupied by diverticula. A typical diverticulum measures 5 mm in diameter, comprising an outer smooth muscle layer of 2 mm diameter, a lumen of approximately 1.5 mm diameter, and an inner folded glandular cell layer covering approximately 1.5 mm of the lumen. The volume of a single SV diverticulum with (V1) and without (V2) its muscular coating is 523 mm3 (4/3π × 53) and 113 mm3 (4/3π × 33), respectively. The volume of a single SV diverticulum lumen (V3) is 14 mm3 (v = 4/3π × 1.53), and the volume of a single SV diverticulum glandular cells (V4) is 99 mm3 (V4 = V2-V3). The ratio of the glandular cell volume to the total glandular volume (V4/V1) is 0.19. Thus, the volume of the SV glandular cell, at 2.84 cc, accounts for approximately 20% of all gland volume. The volume of a single SV glandular cell ranges from 1000 µm3 to 2000 µm3. Considering an average of 1500 µm3, the number of SV cells can be estimated at 1.9 × 109 (2.84 cc/ 1500 µm3), which is remarkably similar to the number of prostatic luminal cells. To the best of our knowledge, this is the first attempt to calculate the number of prostatic luminal cells and number of glandular cells in the SVs.
COMPARISON BETWEEN THE NUMBER OF DAILY MITOSES IN THE PROSTATE AND SEMINAL VESICLES
S phase is the most vulnerable cell cycle to mutations. The number of S phases is equal to the number of mitoses which is reflected in the mitotic index. The basal cells of the prostate proliferate. Their number is roughly equal to that of the luminal cells. The mitotic index of these cells is 0.2% 11. Accurate rates remain to be established for SVs, but experts estimate a similar rate of 0.2%. Thus, there are 3 × 106 daily mitotic cycles in the prostate and 3.8 × 106 in the SVs. Differences in the number of daily mitotic cycles cannot explain the variations in cancer incidence. To the best of our knowledge, this is the first attempt to calculate the number of daily mitoses in the prostate and in the SVs.
COMPARISON BETWEEN THE ANATOMICAL CHARACTERISTICS OF THE PROSTATE AND THE SEMINAL VESICLES
Major anatomical differences have been observed between the prostate and SVs in terms of their excretory ducts and capsule. The SVs excrete into ducts that are joined by the vasa deferencia to form the ejaculatory ducts. A long tube, 2–4 cm in length with lumen diameter of 1–3 mm. It runs through the prostatic parenchyma and enters the prostatic urethra at an acute angle. The prostate secrets through multiple short ducts that enter the urethra at a right angle. The SVs have no capsules; they are surrounded by loose connective tissue that blends with the retroperitoneal fat and can accommodate pressure. The prostate is surrounded by a collagenous and muscular capsule, which limits its expansion upon an increase in pressure. The impact of these unique anatomical differences on organ physiology and potentially on carcinogenesis will be discussed in later sections.
COMPARISON BETWEEN PRESSURES APPLIED TO THE PROSTATE AND SEMINAL VESICLES
Owing to their anatomy, the intraluminal pressure of the SVs is always low and equals that of the retroperitoneum. Conversely, the prostatic ductal and acinar cells are exposed to intra- and periacinar pressures. The acinar pressure theory proposes that during micturition, the bladder neck is open and the pressure within the prostatic urethra equals that of the bladder. Between micturitions, when the bladder neck is closed, contractions of the bulbourethral portion of the levator ani control the prostatic urethra pressure. This striated muscle generates forceful contractions during emergent switching off urination, defecation, and ejaculation. This pressure is transmitted directly to the acini by the short prostatic ducts. On the other hand, the distance between the SVs and the levator ani, the long and thin ejaculatory ducts, their compression by prostatic parenchyma upon pressure rises and the absence of a capsule, all protect the SVs from pressure rises.
Wadhera 12 suggested that the peripheral zone, which is sited right on the levator ani, is exposed to the highest pressure, whereas the distant central zone to the lowest. He also suggested that the apex of the prostate, which essentially rests on the muscle, is subjected to the utmost pressure levels and that prostatic pressure distribution parallels the distribution of cancer.
Cyclic stretching of cells is obnoxious by itself. In-vitro studies showed that cyclic stretching conferred resistance to etoposide-induced apoptosis in prostatic stromal and epithelial cells. Notably, an increase in the production of the anti-apoptotic factor Bcl-2 and the proliferation inducer platelet-derived growth factor (PDGF) was observed in this model 13. Atrophic changes in the epithelium, which are commonly observed in prostate biopsies, and proliferative inflammatory atrophy (PIA) have been observed in prostatic acini repeatedly exposed to high pressure. The significance of this finding is discussed in subsequent sections of this paper.
COMPARISON BETWEEN THE DEGREE OF EXPOSURE OF THE PROSTATE AND SEMINAL VESICLES TO URINE AND SPERM
Prostatic acinar pressure increases during the contractions of the levator ani. By the end of the contraction, pressure drops, and the distended acini suck urine and sperm from the urethra. Kirby et al. demonstrated this by injecting carbon-particle suspension into the bladder prior to prostatectomy and detecting it intraductally in the peripheral zone 15. Reflux of contrast media into the prostate can also be seen in urethrography but is extremely rare in SVs 16, 17.
Reflux of sperm on the other hand, into both the prostate and SVs is common. In a study of 69 radical prostatectomies sperm was found in the SVs only and in the SVs and prostate in 31.9% and 26.1% of the cases, respectively 18.
THE POTENTIAL EFFECTS OF EXPOSURE TO URINE
Only 84,870 Americans develop bladder cancer annually despite the bladder being constantly exposed to urine 1. The normal urothelium can cope with exposure to urine owing to the presence of multi-layered, terminally differentiated, non-dividing apical (umbrella) cells covered with uroplakins that provide resistance to chemicals and microbes. However, non-urothelial tissues exposed to urine do not possess this ability. The risk of developing adenocarcinoma increases by 7000-fold after ureterosigmoidostomy 19,20. This extremely high risk is attributed to high levels of carcinogenic N-nitroso compounds. These molecules are not found in significant amounts in urine or feces. They originate from reduction of nitrates from urine by gut bacterial nitrate reductase. The risk of developing cancer upon exposure to urine is high even in the absence of stools. The inclusion of any bowel segment into the urinary system, including small bowel and stomach, has been associated with an increased incidence of cancer. Most tumors are adenocarcinomas, and the latency between bladder augmentation and cancer diagnosis is usually >10 years 21,22. The proposed mechanism for carcinogenesis includes inflammatory changes and papillary hyperplasia at the enterovesical junction. In-vivo studies showed these changes in half of the animals one year after ileocystoplasty, gastrocystoplasty, or sigmoid cystoplasty 23.
Another support for the importance of urine in carcinogenesis is found in the pathogenesis of Skene’s gland cancer. These glands are the female homolog of the prostate. They empty through small ducts near the urethral orifice and are not normally exposed to urine. Malignant tumors of Skene’s gland are rare. They resemble PC in histologically, age distribution (median age 70 years), are associated with elevated serum PSA that drops with treatment and can be classified according to the Gleason score 24. More than a quarter of the reported cases were associated with urethral diverticula, and constant exposure of the glands to urine.
Thus, anatomical and physiological differences between the prostate and SVs results in exposure of the prostatic luminal cells to urine while SVs cells are protected. Inflammation could be the missing link between exposure to urine and cancer.
COMPARISON BETWEEN THE DEGREE AND ROLE OF INFLAMMATION IN THE PROSTATE AND SEMINAL VESICLES
Chronic inflammation, especially in the context of proliferation, has been identified as a risk factor for carcinomas of the colon, urinary bladder, stomach, and liver. The proposed mechanism involves tissue damage and regeneration in a reactive oxygen and nitrogen species-rich environment (H2O2, NO, etc.). These substances are mutagenic 25.
Inflammation of the SVs is uncommon. It usually presents as an acute condition accompanying acute prostatitis. Asymptomatic seminal vesiculitis is extremely rare. Asymptomatic (type IV) prostatitis on the other hand, is found in approximately 40% of prostate biopsies. When accompanying PC, the aggressiveness of the inflammation correlates with PC grade 26,27. High-grade inflammation surrounding malignant glands is a prognostic factor for biochemical failure after radical prostatectomy 28. Follow-up of participants in the prostate cancer prevention trial (PCPT) and Selenium and Vitamin E Cancer Prevention Trial (SELECT) cohorts with a negative end-of-study biopsy found that the odds of being diagnosed with PC six years after study completion correlated with an increase in the mean percentage of inflamed tissue area (OR 3.31, p-trend 0.047) 29.
Epidemiological, experimental and therapeutic clues suggest that inflammation contributes to cancer initiation and progression, especially in aging people 30. The oxidative stress associated with inflammation directly induces double-strand DNA breaks, indirectly promotes carcinogenesis by suppression of anti-tumor immune response and recruits of epigenetically regulated proteins, such as DNA methyltransferases and chromatin modulators which alter genetic transcription patterns. If key protective proteins are silenced, previously repressed genes can become active resulting in genomic instability, disturbance in cell cycle regulation and impairment of differentiation. Glutathione-S-transferase P1 (GSTP1) that codes for the enzyme detoxification electrophilic substances is such a gene. It is the most altered gene in PC, epigenetically hypermethylated in >90% of cases 28.
The inflammatory response of the prostate to insults (exposure to high pressure and to urinary components) is subjected to inherited variations. This could be the inherited risk component of PC, which is strong in this disease. Variations in many immune-related genes such as RNASEL; MSR1; TLR1; IL1B; IL-2, 4, 6, 8, and 10; and others have been reported 25. The frequent findings of asymptomatic prostatic inflammation and its rarity in the SVs suggest that it has a role in the pathogenesis of PC.
COMPARISON BETWEEN AGE-RELATED CHANGES IN THE PROSTATE AND SEMINAL VESICLES
With aging, the SVs undergo mild age-related atrophy and fibrosis resulting in minor decrease in size and volume of fluid production. The prostate on the other hand, exhibits dramatic age-related changes. The most significant changes occur in the transition zone – benign prostatic hyperplasia (BPH), but age-related changes appear also in the peripheral zone including inflammation, focal atrophy, proliferation, and PIA. PIA could be the link between inflammation and cancer. PIA often accompanies inflammation, PIN, and PC. It typically exhibits increased expression of the proliferative marker Ki-67, downregulation of P27Kip1 (a cyclin-dependent kinase inhibitor), increased expression of the anti-apoptotic Bcl-2 protein, and increased expression GSTP1, suggesting stress response. PIA may develop into carcinoma directly or indirectly through PIN 32,33.
SUMMARY OF THE COMPARISONS
The similarities and dissimilarities between the prostate and SVs are summarized in Table 1 and Fig. 1. Calculations of the number of cells and number of daily mitoses in the prostate and SVs, while far from being accurate, showed no significant differences. Thus, numerical variations cannot explain the difference in cancer incidence. There are, however, significant anatomical differences between the organs in the excretory ducts, in the capsule, and in proximity to the levator ani muscle. The results of these differences are repeated exposures of the prostate but not the SVs to pressure and to urine. Since high incidence of cancer is observed following the exposure of any non-urothelial tissue to urine, this could also be the case in PC. Inflammation is probably the bridge between exposure to urine and cancer. It plays a role in carcinogenesis in the colon, urinary bladder, stomach, and liver. Asymptomatic inflammation (type IV prostatitis) is found in 40% of the prostatic biopsies, and the presence of inflammation in prostatic biopsy with no malignancy is a risk factor of later diagnosis of PC 26,27,29 The inflammatory response of the prostate is genetically determined. Asymptomatic inflammation of the SVs is extremely rare.
In addition, the prostate undergoes marked age-related changes while the SVs undergo only minor atrophy and fibrosis. In addition to inflammation, age-related prostatic changes include focal atrophy and PIA, which probably link inflammation and cancer 32,33.
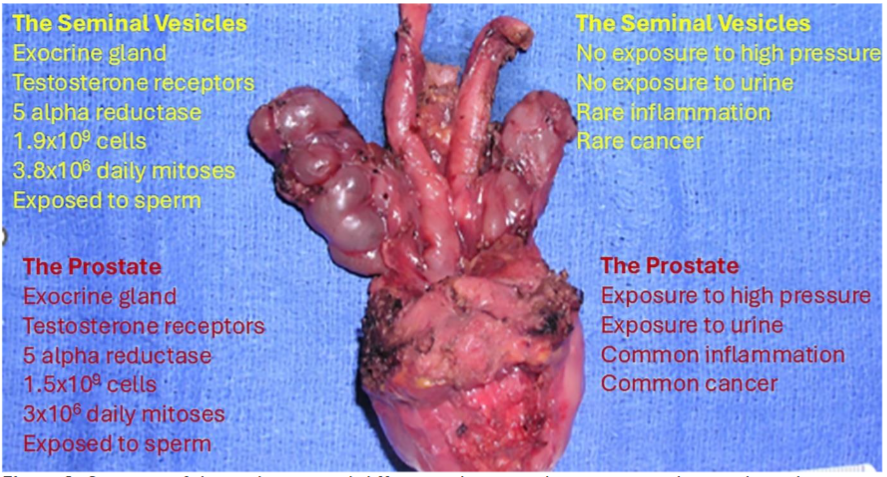
| Seminal Vesicles | Prostate |
|---|---|
| Location | Posterior inferior to the bladder |
| Function | Exocrine gland |
| Excretion to | Prostatic urethra |
| % of seminal fluid | 70-80% |
| Embryonal origin of epithelium | Mesonephric (Wolffian) duct |
| Testosterone receptors | Yes |
| 5-alpha reductase activity | Yes |
| Estimated number of Epithelial cells | 1.9 × 109 |
| Estimated number of daily mitoses | 3.8 × 106 |
| Exposure to high pressures | No |
| Reflux of urine | Rare |
| Reflux of sperm cells | Common |
| Clinical Inflammation | Extremely rare (Seminal vesiculitis) |
| Pathological inflammation | Extremely rare |
| Age related changes | Almost none |
| Malignant tumors | Extremely rare |
| Malignant Tumor type | Adenocarcinoma |
| Response of cancer to anti-androgens | Probably yes |
| Benign tumors prevalence | Extremely rare |
| Benign tumor types | papillary adenomas and cystadenomas |
| Benign Prostatic Hyperplasia | Yes |
Discussion
In the attempts to prevent PC, the next analytical step should be allocating the offensive component of urine and neutralizing it. The primary suspect is monosodium urate (MSU). MSU is a strong proinflammatory substance that activates the NALP3 inflammasome, stimulates the release of inflammatory mediators such as TNF-α, IL-1 α and β and many others and recruits inflammatory cells 34,35. Its levels are in the range of 30 to 60 mg% in the urine, compared to 3.4–7 mg% in the serum and 0.2–0.4 mg% in prostatic secretions. Exposure of prostate cells to MSU at levels two-fold higher than their “working range” may have deleterious effects. Other characteristic features of urine, such as acidic pH, the presence of calcium oxalate, proinflammatory cytokines, and multiple other factors may also play a role in carcinogenesis.
If exposure of luminal cells to MSU has a role in PC pathogenesis, its incidence should be higher in patients with gout, and indeed this is so 36. Treatment with allopurinol, xanthine oxidase blocker, should therefore decrease the incidence of PC. In a cohort study from Taiwan, significant reduction in PC incidence was found among allopurinol treated patients. (HR=0.64, CI 0.45-0.9) 37. These findings were not verified in Finish population (HR 1.03, CI 0.92-1.16) and in meta-analysis of seven studies (risk ratio 1.13, CI 0.96-1.34) 38,39. All these studies were retrospective and are subjected to confounding and selection biases. Obviously, patients treated with allopurinol have gout, and their prostates were exposed to high levels of MSU for years before treatment. Only prospective trial randomizing patients without PC (preferably at high risk for developing PC) to receive allopurinol or placebo in a methodology similar to that of the PCPT trial can answer the question 40.
Other mechanisms that could be involved in carcinogenesis and were not considered here include the potential different effects of hormones on the SVs and prostate, the (unlikely) possibility of infectious agent involvement, the role of ingested carcinogens (which probably play a minor role in PC), the importance of Zinc, present in high concentration in prostatic secretion and almost absent in SVs sections, and potential significance of physical trauma caused by corpora amylacea 41.
Conclusion
Comparisons between the anatomy and physiology of the SVs can potentially reveal noteworthy hints to the pathogenesis of PC. These differences result in the exposure of prostatic acini and ducts to high pressure and to urinary reflux, containing MSU and other potentially toxic constituents. The SVs are not exposed to these insults. The damage induced by these offenses, as well as age-related changes in the prostate, promotes chronic asymptomatic inflammation, which is common in the prostate and extremely rare in SVs. Inflammation can progress to PIA, PIN, and PC. Neutralizing the effects of MSU and possibly of other offending agents could potentially decrease the risk of PC.
Conflict of interest:
None
Financial support:
None
References
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17-48. doi: 10.3322/caac.21763. PMID: 36633525.
- Lee YI, Lee HM, Jo JK, Lee S, Hong SK, Byun SS, Lee SE, Oh JJ. Association between Seminal Vesicle Invasion and Prostate Cancer Detection Location after Transrectal Systemic Biopsy among Men Who Underwent Radical Prostatectomy. PLoS One. 2016;11:e0148690. doi: 10.1371/journal.pone.0148690. PMID: 26848747; PMCID: PMC4743841.
- Galosi AB, Milanese G, Montesi L, Cimadamore A, Franzese C, Palagonia E, Chiacchio G, Castellani D. The pathway of isolated seminal vesicle invasion has a different impact on biochemical recurrence after radical prostatectomy and pelvic lymphadenectomy. Urol Oncol. 2023;41:293.e9-293.e14. doi: 10.1016/j.urolonc.2023.03.011. Epub 2023 May 2. PMID: 37142452.
- Bhat A, Banerjee I, Kryvenko ON, Satyanarayana R. Primary seminal vesicle adenocarcinoma: a lethal yet cryptic malignancy with review of literature. BMJ Case Rep. 2019;12:e232994. doi: 10.1136/bcr-2019-232994. PMID: 31852695; PMCID: PMC6936479.
- Okada H, Tsubura A, Okamura A, Senzaki H, Naka Y, Komatz Y, Morii S. Keratin profiles in normal/hyperplastic prostates and prostate carcinoma. Virchows Arch A Pathol Anat Histopathol. 1992;42:157-61. doi: 10.1007/BF01607049. PMID: 1381129.
- Mitterberger M, Horninger W, Aigner F, Pinggera GM, Steppan I, Rehder P, Frauscher F. Ultrasound of the prostate. Cancer Imaging. 2010;10:40-8. doi: 10.1102/1470-7330.2010.0004. PMID: 20199941; PMCID: PMC2842183.
- Brawer MK. The influence of prostate volume on prostate cancer detection. Eur Urol Supp 2002;1:35-39.
- Cahill LC, Fujimoto JG, Giacomelli MG, Yoshitake T, Wu Y, Lin DI, Ye H, Carrasco-Zevallos OM, Wagner AA, Rosen S. Comparing histologic evaluation of prostate tissue using nonlinear microscopy and paraffin H&E: a pilot study. Mod Pathol. 2019;32:1158-1167. doi: 10.1038/s41379-019-0250-8. Epub 2019 Mar 26. PMID: 30914763; PMCID: PMC6663612.
- Gofrit ON, Zorn KC, Taxy JB, Zagaja GP, Steinberg GD, Shalhav AL. The dimensions and symmetry of the seminal vesicles. J Robot Surg. 2009;3:29-33. doi: 10.1007/s11701-009-0134-x. Epub 2009 Feb 27. PMID: 27628450.
- Hernandez AD, Urry RL, Smith JA Jr. Ultrasonographic characteristics of the seminal vesicles after ejaculation. J Urol. 1990;144:1380-2. doi: 10.1016/s0022-5347(17)39747-1. PMID: 2231933.
- Berges RR, Vukanovic J, Epstein JI, CarMichel M, Cisek L, Johnson DE, Veltri RW, Walsh PC, Isaacs JT. Implication of cell kinetic changes during the progression of human prostatic cancer. Clin Cancer Res. 1995;1:473-80. PMID: 9816006; PMCID: PMC4086477.
- Wadhera P. An introduction to acinar pressures in BPH and prostate cancer. Nat Rev Urol. 2013;10:358-66. doi: 10.1038/nrurol.2013.86. Epub 2013 May 14. PMID: 23670181.
- Hegarty PK, Watson RW, Coffey RN, Webber MM, Fitzpatrick JM. Effects of cyclic stretch on prostatic cells in culture. J Urol. 2002;168:2291-5. doi: 10.1016/S0022-5347(05)64373-X. PMID: 12394777.
- Nelson G, Culberson DE, Gardner WA Jr. Intraprostatic spermatozoa. Hum Pathol. 1988 May;19(5):541-4. doi: 10.1016/s0046-8177(88)80201-6. PMID: 3371978.
- Kirby RS, Lowe D, Bultitude MI, Shuttleworth KE. Intra-prostatic urinary reflux: an aetiological factor in abacterial prostatitis. Br J Urol. 1982;54:729-31. doi: 10.1111/j.1464-410x.1982.tb13635.x. PMID: 7150931.
- Mitty HA. Roentgen features of reflux into the prostate, seminal vesicles and vasa deferentia. Am J Roentgenol Radium Ther Nucl Med. 1971;112:603-6. doi: 10.2214/ajr.112.3.603. PMID: 5570373.
- Hekimsoy T, Gorur GD, Isgoren S, Uslu H, Demir H. Urinary Reflux Into the Prostate Gland and Seminal Vesicles: A Potential Pitfall in 18F-FDG and 68Ga-PSMA PET/CT. Clin Nucl Med. 2020;45:536-537. doi: 10.1097/RLU.0000000000003069. PMID: 32433177.
- Chen X, Zhao J, Salim S, Garcia FU. Intraprostatic spermatozoa: zonal distribution and association with atrophy. Hum Pathol. 2006;37:345-51. doi: 10.1016/j.humpath.2005.11.011. PMID: 16613330.
- Pettersson L, Tranberg J, Abrahamsson K, Pettersson S, Sillen U, Jonsson O. Half century of followup after ureterosigmoidostomy performed in early childhood. J Urol. 2013;189:1870-5. doi: 10.1016/j.juro.2012.11.179. Epub 2012 Dec 5. PMID: 23220244.
- Husmann DA, Spence HM. Current status of tumor of the bowel following ureterosigmoidostomy: a review. J Urol. 1990;144:607-10. doi: 10.1016/s0022-5347(17)40055-3. PMID: 2179579.
- Filmer RB, Spencer JR. Malignancies in bladder augmentations and intestinal conduits. J Urol. 1990;143:671-8. doi: 10.1016/s0022-5347(17)40055-3. PMID: 2179579.
- Castellan M, Gosalbez R, Perez-Brayfield M, Healey P, McDonald R, Labbie A, Lendvay T. Tumor in bladder reservoir after gastrocystoplasty. J Urol. 2007;178(4 Pt 2):1771-4; discussion 1774. doi: 10.1016/j.juro.2007.05.100. Epub 2007 Aug 17. PMID: 17707009.
- Buson H, Diaz DC, Manivel JC, Jessurun J, Dayanc M, Gonzalez R. The development of tumors in experimental gastroenterocystoplasty. J Urol. 1993;150:730-3. doi: 10.1016/s0022-5347(17)35599-4. PMID: 8326635.
- Li XH, Zhang YT. Skene gland adenocarcinoma of the urethra: A systematic review. Asian J Surg. 2024;47:1587-1588. doi: 10.1016/j.asjsur.2023.12.023. Epub 2023 Dec 14. PMID: 38102007.
- Puhr M, De Marzo A, Isaacs W, Lucia MS, Sfanos K, Yegnasubramanian S, Culig Z. Inflammation, Microbiota, and Prostate Cancer. Eur Urol Focus. 2016;2:374-382. doi: 10.1016/j.euf.2016.08.010. Epub 2016 Aug 28. PMID: 28723469.
- Aglamis E, Tasdemir C, Ceylan C. The role of National Institutes of Health category IV prostatitis in accurately staging the newly diagnosed prostate cancer. Ir J Med Sci. 2013;182:463-7. doi: 10.1007/s11845-013-0914-1. Epub 2013 Feb 1. PMID: 23370973.
- Aglamis E, Ceylan C, Akin MM. Is there a correlation between the aggressiveness of chronic asymptomatic prostatitis National Institutes of Health category IV and the Gleason score in patients with prostate cancer? Can Urol Assoc J. 2020;14:E568-E573. doi: 10.5489/cuaj.6284. PMID: 32520704; PMCID: PMC7673826.
- Irani J, Goujon JM, Ragni E, Peyrat L, Hubert J, Saint F, Mottet N. High-grade inflammation in prostate cancer as a prognostic factor for biochemical recurrence after radical prostatectomy. Pathologist Multi Center Study Group. Urology. 1999;54:467-72. doi: 10.1016/s0090-4295(99)00152-1. PMID: 10475356.
- Platz EA, Kulac I, Barber JR, Drake CG, Joshu CE, Nelson WG, Lucia MS, Klein EA, Lippman SM, Parnes HL, et al. A Prospective Study of Chronic Inflammation in Benign Prostate Tissue and Risk of Prostate Cancer: Linked PCPT and SELECT Cohorts. Cancer Epidemiol Biomarkers Prev. 2017;26:1549-1557. doi: 10.1158/1055-9965.EPI-17-0503. Epub 2017 Jul 28. PMID: 28754796; PMCID: PMC5626618.
- Guerville F, Bourdel-Marchasson I, Déchanet-Merville J, Pellegrin I, Soubeyran P, Appay V, Lemoine M. Does Inflammation Contribute to Cancer Incidence and Mortality during Aging? A Conceptual Review. Cancers (Basel). 2022;14:1622. doi: 10.3390/cancers14071622. PMID: 35406394; PMCID: PMC8996949.
- Henrique R, Jerónimo C. Molecular detection of prostate cancer: a role for GSTP1 hypermethylation. Eur Urol. 2004;46:660-9; discussion 669. doi: 10.1016/j.eururo.2004.06.014. PMID: 15474280.
- De Marzo AM, Platz EA, Sutcliffe S, Xu J, Grönberg H, Drake CG, Nakai Y, Isaacs WB, Nelson WG. Inflammation in prostate carcinogenesis. Nat Rev Cancer. 2007;7:256-69. doi: 10.1038/nrc2090. PMID: 17384581; PMCID: PMC3552388.
- De Marzo AM, Marchi VL, Epstein JI, Nelson WG. Proliferative inflammatory atrophy of the prostate: implications for prostatic carcinogenesis. Am J Pathol. 1999;155:1985-92. doi: 10.1016/S0002-9440(10)65517-4. PMID: 10595928; PMCID: PMC1866955.
- Dinarello CA. An expanding role for interleukin-1 blockade from gout to cancer. Mol Med. 2014;20 Suppl 1(Suppl 1):S43-58. doi: 10.2119/molmed.2014.00232. PMID: 25549233; PMCID: PMC4374514.
- Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237-41. doi: 10.1038/nature04516. Epub 2006 Jan 11. PMID: 16407889.
- Kuo CF, Luo SF, See LC, Chou IJ, Fang YF, Yu KH. Increased risk of cancer among gout patients: a nationwide population study. Joint Bone Spine. 2012;79:375-8. doi: 10.1016/j.jbspin.2011.09.011. Epub 2011 Nov 15. PMID: 22088929.
- Shih HJ, Kao MC, Tsai PS, Fan YC, Huang CJ. Long-term allopurinol use decreases the risk of prostate cancer in patients with gout: a population-based study. Prostate Cancer Prostatic Dis. 2017;20:328-333. doi: 10.1038/pcan.2017.14. Epub 2017 Apr 11. PMID: 28398294.
- Kukko V, Kaipia A, Talala K, Taari K, Tammela TLJ, Auvinen A, Murtola TJ. Allopurinol and the risk of prostate cancer in a Finnish population-based cohort. Prostate Cancer Prostatic Dis. 2019;22:483-490. doi: 10.1038/s41391-019-0129-2. Epub 2019 Jan 29. PMID: 30696944.
- Lai SW, Hwang BF, Kuo YH, Liu CS, Liao KF. A meta-analysis of allopurinol therapy and the risk of prostate cancer. Medicine (Baltimore). 2022;101:e28998. doi: 10.1097/MD.0000000000028998. PMID: 35356907; PMCID: PMC10684161.
- Thompson IM Jr, Goodman PJ, Tangen CM, Parnes HL, Minasian LM, Godley PA, Lucia MS, Ford LG. Long-term survival of participants in the prostate cancer prevention trial. N Engl J Med. 2013;369:603-10. doi: 10.1056/NEJMoa1215932. PMID: 23944298; PMCID: PMC4141537.
- DuPre NC, Flavin R, Sfanos KS, Unger RH, To S, Gazeeva E, Fiorentino M, De Marzo AM, Rider JR, Mucci LA; Transdisciplinary Prostate Cancer Partnership(ToPCaP). Corpora amylacea in prostatectomy tissue and associations with molecular, histological, and lifestyle factors. Prostate. 2018;78:1172-1180. doi: 10.1002/pros.23692. Epub 2018 Jul 15. PMID: 30009541; PMCID: PMC6501556.




