SIBS and PRESERFLO MicroShunt: Advanced Glaucoma Treatment
Development of SIBS and the PRESERFLOTM MicroShunt to Treat Advanced Glaucoma: A Review
Leonard Pinchuk, Ph.D., D.Sc. (h.c.), NAE, FNAI, F-AIMBE, ICFBSE, 2019 Russ Prize Laureate1
- Founder and Senior Vice President and Executive Principal Scientist, Polymer Engineer, InnFocus, Inc., a Santen Company; Distinguished Research Professor, Biomedical Engineering, University of Miami, Miami, FL.
OPEN ACCESS
PUBLISHED: 28 Febuary 2025
CITATION:PINCHUK, Leonard. The Development of SIBS and the PRESERFLO TM MicroShunt to Treat Advanced Glaucoma. Medical Research Archives,Available at: <https://esmed.org/MRA/mra/article/view/6169>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6169
ISSN 2375-1924
ABSTRACT
Polyisobutylene-based biomaterials are elastomeric polymers specifically designed for long-term implant applications. These polymers, which are comprised of only carbon and hydrogen atoms, contain no cleavable groups and are highly purified. Their intrinsic methyl groups interact with tissue, minimizing foreign body reactions. The present review describes the development of a glaucoma micro-shunt made from one of these polymers, poly(styrene-block-isobutylene-block-styrene) (SIBS). This review also reports recent findings in patients implanted with these PRESERFLOTM MicroShunts.
Keywords
- Poly(styrene-block-isobutylene-block-styrene)
- SIBS
- Glaucoma
- MicroShunt
- Preserflo
- MIGS
Introduction
Polyisobutylene-based biomaterials are both extremely inert and highly flexible and have been applied commercially in medicine for over 25 years. The first commercial application of a polyisobutylene-based material in medicine was the thermoplastic polymer poly(styrene-block-isobutylene-block-styrene) (SIBS), used as the drug carrier in paclitaxel-eluting coronary stents (Taxus® from Boston Scientific, Natick, MA, USA). SIBS and the processes used to fabricate these stents have been described1-3. The second commercial product based on a polyisobutylene-based biomaterial is the PRESERFLO MicroShunt (MicroShunt) (InnFocus/Santen, Miami, FL, USA), a drainage device for the treatment of glaucoma. Other possible applications of polyisobutylene-based polymers are under development.
This review first discusses the development of SIBS, the premise behind its bio-inertness and biocompatibility, and its use in the PRESERFLO MicroShunt. The article then reviews early studies of the clinical development of the MicroShunt, followed by a description of continued improvements in its performance through its implantation in approximately 100,000 eyes world-wide. The article concludes with a discussion on why this well-recognized treatment for advanced glaucoma, i.e., the PRESERFLO MicroShunt, has not yet been approved for sale by the United States Food and Drug Administration (FDA). A goal of the article is to elucidate the nuances of confidence intervals, the criteria of non-inferiority relative to an existing procedure, the learning curve on new devices when compared with frequently-practiced procedures, and the restrictions of performing a clinical study under strict limitations.
Polyisobutylene Chemistry and its Advantages as a Biomaterial
Certain polyisobutylene-based biomaterials have been classified as elastomers, generally defined as materials capable of stretching more than 20% and returning on their own to their original shapes. Work on this specific class of elastomers for long-term medical implants began in the early 1990s, when conventional polyether urethane implantable biomaterials, such as those comprising the insulators on pacemaker leads (Pellethane 2363 80A) and synthetic vascular grafts attracted granulocytes as a consequence of their unintended biodegradation in the body4. Macrophages, polymorphonuclear leukocytes (PMNs), and foreign body giant cells migrated towards the device to either wall-off the degraded material by forming thick capsules around it or to remove degraded fragments by phagocytosis. Many pacer lead insulators were, and still are, made from these industrial-grade polyether urethanes that were never designed for long-term applications in the body. Consequently, there remained a need to develop a biostable polymer that met four major criteria. The first was that the polymer be devoid of sites for degradation, including the absence of urethane (carbamate), ether, ester, carbonate, and amide linkages, on its backbone or side-groups. The second was that the backbone, in the event of oxidation by granulocytes, would be unable to form double bonds that could embrittle and degrade the polymer, leading to cracking or deterioration of the backbone when subsequently stretched or bent. The third criterion was that the polymer be composed only of carbon and hydrogen atoms, preferably with tissue interfacing methyl groups, which is a common tissue interface in the body. More specifically, the polymer should not contain silicon or fluorine atoms, which are not found in the human body and could, if introduced, provoke a foreign body reaction. The fourth criterion was that the polymer be purifiable; that is, it must be soluble or extractable in specific solvents to remove oligomers, initiators and other contaminants that can otherwise leach out and cause inflammation.
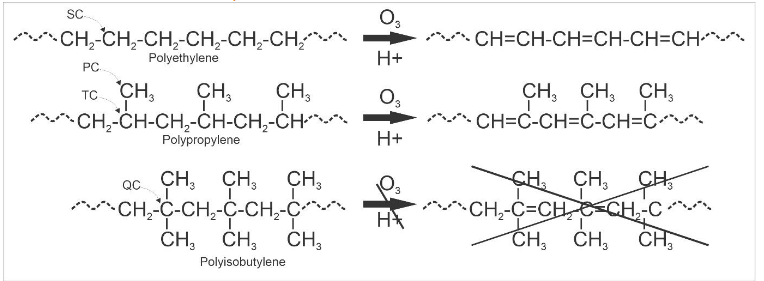
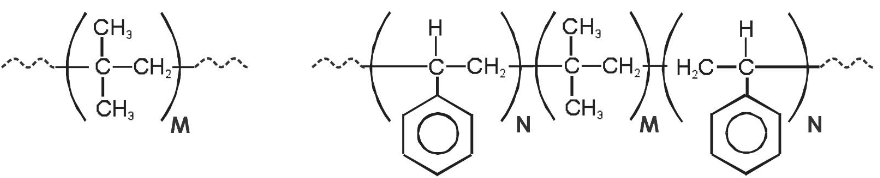
Polyisobutylene is a gum, with many types of chewing gum comprised of naturally-derived polyisobutylene from natural sources (Figure 2, left). Conversion to a processable elastomer (e.g., for injection molding or extrusion) requires its crosslinking with meltable or solvent-dissolvable crosslinks. In SIBS, the polyisobutylene center block is capped on both sides with polystyrene (Figure 2, right). During melting or in solution, the polystyrenes combine and segment due to hydrophobic interactions, and, when cooled or removed from solution, the polystyrene end-groups crystallize into glassy segments that hold the polyisobutylene ends together, with the product being a meltable crosslinked elastomer. The type of chemistry used to synthesize polyisobutylene and SIBS (Figure 2) was developed by Dr. Joseph P. Kennedy and his team at the University of Akron (Akron, OH, USA)5,6, with the products being refined, purified and patented for specific implantable use by the present author7.
Polymers comprised of crosslinked collagen, such as those used in XEN Gel-Stents (AbbVie, Dublin, Ireland), contain both amides and many unprotected carbons, with no alternating quaternary carbons8. These properties are thought to be responsible for the slow embrittlement and degradation of XEN Gel-Stents.8
Design of the PRESERFLO MicroShunt
The development began on a glaucoma shunt in 2003, when SIBS disks 3-mm in diameter and 1-mm thick were implanted in the corneal stroma, as well as under the conjunctiva and Tenon’s capsule of New Zealand white rabbits’ eyes9. For comparison, disks made of silicone rubber (polydimethylsiloxane [PDMS]) were implanted alongside the SIBS disks. After 2 months, there were no myofibroblasts or angiogenesis in the vicinity of the SIBS disks or integral capsules surrounding these disks10. In contrast angiogenesis, myofibroblasts, and significant capsules were observed in the vicinity of the PDMS control disks. These findings suggested that SIBS was very inert and innocuous in rabbit eyes. Moreover, the absence of clinically significant fibrous capsule formation suggested that SIBS could theoretically be used for a plateless glaucoma shunt to lower intraocular pressure (IOP) and thwart vision loss due to glaucoma9.
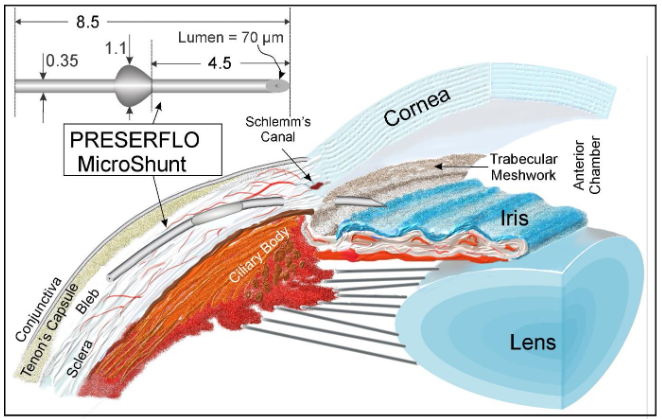
Anatomical features require that SIBS tubes be at least 8 mm long (8.5 mm was selected) to shunt from the anterior chamber to the mid-posterior eye. The lumen serves as a flow restrictor to prevent the pressure in the eye from dropping to below approximately 6 mmHg for a prolonged period of time, as this deflation of the eye (hypotony) could lead to retinal detachment and other serious pathologies. Based on a normal aqueous humor flow rate of 2.5 µL/min and a desired IOP drop of 12 mmHg, the Hagen-Poiseuille equation suggests a lumen diameter of approximately 55 µm. Due to the high surface tension of SIBS, the desired flow was difficult to obtain at this lumen diameter; therefore, the lumen was increased to approximately 70 µm to allow flow at physiological pressures. The flow characteristics of the PRESERFLO MicroShunt have been comprehensively analyzed in vitro11. Implantations into rabbit eyes12,13 confirmed that these requirements were satisfied by a nominal lumen diameter of 70 µm. To prevent kinking and to allow the tube to be pushed through a needle tract formed by a 25-G or 27-G needle inserted under the limbus, the outer diameter of the tube was set at 350 µm, providing a tough tube with a wall thickness of 140 µm.
First Clinical Implants of the PRESERFLO MicroShunt
The MicroShunt was first implanted in humans in January 2006, predominantly in French patients who failed incisional glaucoma surgery. The lack of the intraoperative antifibrotic mitomycin C (MMC) resulted in a surgical success rate of only 42%, whereas a subsequent study, which included 0.2 mg/mL, MMC, improved the surgical success rate to 67%9. A study in the Dominican Republic of 23 patients in a pre-dominant Afro-Caribbean population with primary open-angle glaucoma (POAG) and no previous conjunctival surgery who had failed maximum tolerated glaucoma medication found that MicroShunt implantation, accompanied by a 3-minute intraoperative application of 0.4-mg/mL MMC, resulted in a qualified success rate of 100%, with a 55% drop in IOP from baseline at 1 year. The results of these four independent clinical trials in France and the Dominican Republic, which led to the final design and method of implantation of the MicroShunt, have been summarized14. Follow-up of the patients in the Dominican Republic showed prolonged success at 3 and 5 years15,16.
The MicroShunt was marked as Conformité Européenne (CE) on January 9, 2012, in Europe and is available in 35 countries world-wide. It was initially used in European, Middle Eastern and African countries in 2018, followed by Canada and Australia in 2021 and Japan in 2022. To date, however, the MicroShunt has not been approved for sale in the United States.
Several studies by investigators in the Dominican Republic, Europe and Canada helped define the advantages and limitations of the PRESERFLO MicroShunt14,17-20. A thorough overview of the MicroShunt has been provided21, and a consensus on the use of the PRESERFLO MicroShunt for the treatment of glaucoma has been summarized by a Delphi Panel of expert users of this device22. In brief, the Delphi Panel sought consensus on 25 statements, including: the MicroShunt is suited to those with a failed trabeculectomy, hypermetropia, high pre-operative IOP or with contact lenses. In addition, the MicroShunt allows for rapid post-operative visual recovery, the posterior position of the bleb can minimize device erosion. Blebs may potentially be revived by needling or removing scar tissue, often described by surgeons as a loosely adherent fibrous sock surrounding the MicroShunt. The incidence of flare is lower with the PRESERFLO MicroShunt than with other devices and treatments used in the eye23. A recent meta-analysis found that the magnitude of IOP decrease is lower with the MicroShunt than with trabeculectomy, but that the two had a similar safety profile and the MicroShunt had a lower reintervention rate24.
Delayed Approval of the PRESERFLO MicroShunt in the USA
InnFocus was granted an Investigational Device Exception (IDE) by the U.S. Food and Drug Administration (FDA) in May 2013 to begin clinical testing of the MicroShunt in the U.S. (herein called the “IDE” study). The FDA required a two-year, prospective, randomized, multicenter, non-inferiority study (NCT01881425) comparing the MicroShunt with primary trabeculectomy in the United States and Europe. The study population consisted of 527 patients, uncontrolled on maximum tolerated glaucoma medication at 29-sites, with 395 eyes in the MicroShunt group and 132 in the control group, who underwent primary trabeculectomy. The study was completed in 201925.
The one-year results of this IDE trial have been reported20. The data obtained on patients who underwent MicroShunt implantation with intraoperative 0.2 mg/mL MMC were consistent with previous findings (Table 1)16-18. From baseline to 1-year, non-washout IOP decreased from 21.1 ± 4.9 mmHg to 14.3 ± 4.3 mmHg (32%) and the number of glaucoma medications decreased from 3.1 to 0.6 in the MicroShunt group20. In the trabeculectomy group, non-washout IOP decreased from 21.1± 5.0 mmHg to 11.1± 4.3 mmHg (47%) and the number of glaucoma medications decreased from 3.0 to 0.3 from baseline to 1 year. Rates of post-operative interventions, cataract progression and hypotony were lower in the MicroShunt than in the trabeculectomy group. Of importance, the PRESERFLO MicroShunt did not fail due to bad MicroShunt data. Rather, the trabeculectomy control arm performed better than in any previously published randomized control trial (RCT) of primary trabeculectomy. The FDA has guidelines for “Non-Inferiority Studies”26, including a “Constancy Assumption”, which states that results in the control group must be statistically similar to previously reported results to allow their use in determining non-inferiority. The Primary Tube versus Trabeculectomy (PTVT) study27 was the only study of primary trabeculectomy available prior to the IDE study and differed from the IDE study in regard to inclusion criteria, baseline IOP and mitomycin C dose. At 1 year, the mean ± SD IOP for trabeculectomy in the PTVT study was 12.4 ± 4.4 mmHg (n=105), which was significantly higher (p = 0.023) than the mean IOP (11.1 ± 4.3 mmHg) in the trabeculectomy group of the IDE study (n=132). Thus, for all the above reasons, the control group in the IDE study failed the Constancy Assumption, casting doubt on the overall appropriateness of this RCT.
Table 1
| Responder Rate | MicroShunt | Trabeculectomy |
|---|---|---|
| 53.9% | 333 | 72.7% |
The IDE trial had two endpoints. Endpoint #1 was defined as a responder (non-failure) rate of the MicroShunt group being within 15% of the responder rate of the trabeculectomy group, whereas Endpoint #2, was defined as an IOP of the MicroShunt group at 1 year being within 2.5 mmHg of the IOP of the trabeculectomy group. Meeting both endpoints would therefore suggest that the MicroShunt was non-inferior to trabeculectomy.
It is important to understand the implications of the non-inferior margins in regard to a CI of 95%. For example, if the responder rates of the MicroShunt and trabeculectomy were 65.1% and 73.5%, respectively, with the difference being -8.4%, then this difference would be within a margin of 15%, indicating that Endpoint #1 would be met. Unfortunately, this was not the case. The term “non-inferior”, as defined by the FDA, required that the 15% difference between groups be within a 95% confidence interval (CI), indicating that with a sample size of 527 eyes, the difference would have to be within 6%, not 15%. To state it statistically, to be 95% confident that the difference between the MicroShunt and trabeculectomy is within 15%, the difference in population responder rate, for the number of patients enrolled, would have to be less than 6%.
At 1 year, the success rates in the MicroShunt and trabeculectomy groups were 53.9% (n=333) and 72.7% (n=113), respectively, a difference of -18.8%, indicating a failure to meet the primary endpoint20. The Farrington-Manning test using the above data showed that the 95% CI was -28.6% to -9.0% (see Figure 4). To meet the 95% CI, the -15% value would have to be outside the range of -28.6% to -9.0%, a function of the null hypothesis, which is why the primary endpoint was not met.
However, the definition of “responder rate” in the comparison of MicroShunt and trabeculectomy20 in the IDE study differed from that provided by the World Glaucoma Association (WGA) guidelines28. If defined according to WGA guidelines, the difference would have been significantly lower. The failure rate for the primary endpoint, as defined in the IDE study20, was IOP <5 mmHg or <20% reduction in IOP compared with baseline, with no increase in glaucoma medications, reoperation for glaucoma, or loss of light perception vision at 1 year. In contrast, the WGA guidelines defined failure rate as an IOP >21 mmHg or a less than 20% reduction below baseline at two consecutive follow-up visits after 3 months, an IOP ≤5 mmHg at two consecutive follow-up visits after 3 months, reoperation for glaucoma, or loss of light perception vision28. That is, the standard WGA guidelines allow patients to be placed back on medication, with a single >20% responder rate drop not considered a failure. This study did not allow for the patients being placed back on medications in order to be considered a success. Moreover, if IOP was not at target in patients who underwent trabeculectomy, suture lyses could be performed to further reduce IOP, indicating a bias in favor of the trabeculectomy arm.
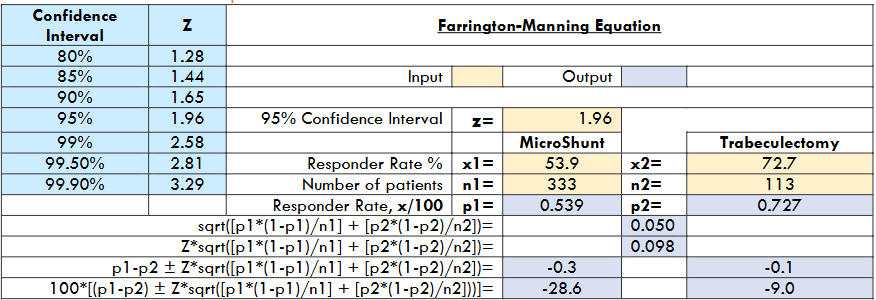
Post hoc reanalysis of success according to WGA guidelines showed that the responder rates in the MicroShunt and trabeculectomy groups at Year-1 were 65.1% and 73.5%, respectively, a difference of -8.4%. According to the Farrington-Manning test (Figure 4) for 95% CI, recalculated using the 65.1% and 73.5% responder rates, the range was -17.3% to 0.5%, again indicating an endpoint failure because -15% was within this range. If, however, the CI was 85% (Z=1.44), the range would be -14.9% to -1.9%, which would meet the criteria as -15% is outside this range. Further, if the patient population was doubled, the 95% CI would be -14.7 to 2.1, thereby meeting the primary endpoint.
The primary reason for the large discrepancy in responder rate in comparisons of the MicroShunt and trabeculectomy groups for both endpoints was largely due to many trabeculectomy patients having IOPs <6 mmHg at 1 year. If all <6 mmHg data from both arms of the study were removed, the responder rates in the MicroShunt and trabeculectomy groups using the WGA definition would be 82.4% (n=329) and 86.8% (n=102), respectively, a difference of -4.4% with a 95% CI of -12.2% to 3.4% thereby meeting the primary Endpoint.
The large number of patients in the trabeculectomy group with IOP < 6 mmHg at 1 year may have been due to the recruitment of these patients by surgeons with extensive experience in performing trabeculectomy. These patients were deemed by the surgeons as able to undergo the risk of hypotony. In contrast, the PRESERFLO MicroShunt was a new device, with surgeons having little knowledge of the device or methods to optimize its performance. For example, the first approximately 100 procedures were performed with the MicroShunt implanted at 12 o’clock, which was reported to be suboptimal by several surgeons. Rather, implanting the device in the superior quadrant of the eye at 11 or 1 o’clock to avoid the rectus muscles is now preferred. In addition, a higher MMC dose of 0.4 mg/mL is preferred, by glaucoma surgeons for filtering surgeries, over the 0.2 mg/mL dose previously approved for commercial use by the FDA. According to the Advanced Glaucoma Intervention Study (AGIS)29, the control of IOP to < 14 mmHg in the majority of patients suggests that glaucomatous progression of vision loss would be unlikely. There are no definitive data suggesting that control of IOP to < 6 mmHg provides further benefit; rather, it places patients at higher risk of pathological hypotony.
Table 1
| Study | No. of Eyes (n) | Pre-op IOP (mmHg) (A) | Pre-op Drugs | Follow-Up Final (yrs) | IOP (mmHg) | Post-op Drugs |
|---|---|---|---|---|---|---|
| Riss et al. (2015) [30] | 23 | 23.8 | 2.6 | 1 | 10.7 | 0.3 |
| Schlenker et al. (2015) [17] | 164 | 21.4 | 3.4 | 1 | 13.3 | 0.5 |
| Beckers et al. (2017) [31] | 91 | 24.3 | 2.4 | 1 | 13.3 | 0.4 |
| Batlle et al. (2021) [16] | 23 | 23.8 | 2.4 | 5 | 12.4 | 0.4 |
| Martinez-de-la-Casa (2021) [32] | 58 | 21.5 | 2.3 | 1 | 14.6 | 0.2 |
| Beckers et al. (2022) [19] | 81 | 21.7 | 2.5 | 2 | 14.1 | 0.5 |
| Fili et al. (2022) [33] | 150 | 23.5 | 2.5 | 1 | 12.9 | 0.4 |
| Van Lancker et al. (2023) [34] | 64 | 24.3 | 3.4 | 1 | 14.0 | 0.5 |
| Tanner et al. (2023) [37] | 104 | 23.4 | 3.4 | 1 | 14.7 | 0.7 |
| Gubser et al. (2023) [38] | 70 | 21.7 | 3.5 | 2 | 15.5 | 1.1 |
Cumulative Averages: 828, 22.8, 2.9, 1.6, 13.7, 0.5
The PRESERFLO MicroShunt Learning Curve
Once the PRESERFLO MicroShunt was approved for use, many surgeons optimized their own procedural techniques. The reader should be aware that InnFocus/Santen cannot condone or encourage the use of these off-label procedures.
- Use of 0.4 mg/mL MMC: Compared with 0.2 mg/mL MMC, 0.4 mg/mL MMC was found to result in a lower IOP and generally better clinical outcomes16,19,35,36. The MicroShunt drains to the mid-posterior Tenon’s capsule, which is thicker than the anterior Tenon’s capsule, the site of trabeculectomy. The mid-posterior Tenon’s capsule contains larger numbers of fibroblasts and smooth muscle cells than the anterior Tenon’s capsule37.
- Surgical implant site: Initially, in the first 100 implants in the IDE clinical study, the MicroShunt was implanted at the 12 o’clock position. Implantation superiorly at the 11 or 1 o’clock position was later found by the surgeons in this study to be optimal, as it avoided the superior rectus muscle.
- Single versus double MicroShunts: A recent retrospective analysis compared outcomes in glaucoma patients who underwent single (n=29) or double (n=28) MicroShunt implantation41. Although mean preoperative IOP was significantly higher in patients who underwent double than single implantation (29.4 ± 10.0 vs. 21.7 ± 8.2 mmHg, P = 0.003), mean postoperative IOP was significantly lower in the double implantation group at 1 day, 1 week and 3 and 6 months (all P <0.021). In the 17 patients who underwent a two-stage procedure, mean IOP lowering was similar after both procedures, with a longer sustainable effect observed after the second procedure. These findings suggest that double MicroShunt implantation was safe and effective in lowering IOP in glaucoma patients, although the risk of hypotony was higher when two devices were implanted in the same eye.
- Stenting the MicroShunt: Beginning in 2022, the MicroShunt has been stented by inserting a suture, generally polypropylene or Nylon, 9.0 or 10.0, at the time of surgery, 2 to 3 mm into the posterior lumen of the MicroShunt, inhibiting short-term flow to prevent hypotony42-46. The free end of the suture has often been placed close to the limbus to ensure later access to the “ripcord,” with the suture-ripcord pulled 2 to 6 weeks after MicroShunt implantation45. The outflow pathways in the eye and through several MIGS devices, including the MicroShunt, have been modeled47, and studies are assessing the effects of stenting.
- Maintenance of the bleb: Several methods have been described for maintenance of the bleb, including filling the bleb with Ologen containing MMC. Ologen is a porous collagen–glycosaminoglycan copolymer matrix used to modulate wound healing in connective and epithelial tissues48,49. At 6 months, however, IOP reductions in groups with and without Ologen implantation did not differ significantly. Beta radiation provides another non-invasive method of modulating fibrosis50-53. Trabeculectomy with a single adjunctive dose of 1000 rad β radiation in Chinese eyes with POAG was found to achieve a qualified success rate of 88.4% at 7 years50. Annual prophylactic exposure of the bleb area to β radiation for 20 seconds may prevent long-term bleb fibrosis51,52. Administration of a biodegradable wafer infused with MMC to rabbit eyes yielded results similar to a controlled dose of MMC applied intraoperatively after one month53. These rabbit eyes, however, were not glaucomatous, preventing assessment of the true efficacy of the biodegradable wafer. In addition, many practitioners inject MMC and 5-FU periodically after surgery, as well as performing needling and bleb revisions21.
- MicroShunt implantation in conjunction with cataract surgery: The PRESERFLO MicroShunt has been implanted into eyes undergoing cataract surgery. An evaluation of 35 eyes that underwent MicroShunt implantation alone and 23 eyes that underwent MicroShunt implantation in combination with phacoemulsification found no between-group differences in reduction of IOP and glaucoma medications at 12 months32. Overall mean IOP dropped from 21.5 ± 3.3 mmHg at baseline to 14.6 ± 3.5 mmHg at 12 months, and the average number of glaucoma medicines dropped from 2.3 at baseline to 0.2 at 12 months. The complete success rates in these two groups were 68.6% (24/35 eyes) and 52.2% (12/23 eyes), respectively. Similarly, an evaluation of 51 eyes that underwent MicroShunt implantation alone and 13 that underwent MicroShunt implantation with phacoemulsification found no significant between-group differences in the number of IOP-lowering medication (0.2 ± 0.08 vs. 0.1 ± 0.1; P =0.2)54. In the overall study population, mean IOP decreased significantly from 22.03 ± 0.7 mm Hg at baseline to 12.7 ± 0.4 mm Hg at the final visit (mean follow-up: 11 ± 1.4 months) (P <0.0001), and the mean number of ocular hypotensive medication was reduced significantly from 2.7 ± 0.7 to 0.2 ± 0.5 (P <0.0001).
- Pseudoexfoliation glaucoma: A retrospective, single-center study compared the safety and efficacy of the PRESERFLO MicroShunt with trabeculectomy in patients diagnosed with pseudoexfoliation glaucoma55. Thirty-one eyes underwent MicroShunt implantation, and 29 underwent trabeculectomy. Surgical success was defined as an IOP between 5 mmHg and 17 mmHg at one-year, no need for surgical revisions or secondary glaucoma surgery, and no loss of light perception. Mean IOP decreased from 20.8 ± 5.9 mmHg at baseline to 12.4 ± 2.8 mmHg (p < 0.0001) in the MicroShunt group and from 22.3 ± 6.5 mmHg to 11.1 ± 3.7 mmHg (p < 0.0001) in the trabeculectomy group. The mean number of glaucoma medications showed similar reductions in the two groups, from 2.7 ± 1.2 to 0.2 ± 0.7 (p < 0.0001) in the MicroShunt group and from 2.9 ± 1.2 to 0.3 ± 0.9 (p < 0.0001) in the trabeculectomy group. Complete and qualified success rates were 83.9% and 90.3%, respectively, in the MicroShunt group and 82.8% and 93.1%, respectively, in the trabeculectomy group. Postoperative complications were comparable in the two groups. These findings indicated that MicroShunt implantation had a non-inferior efficacy and safety profile compared with trabeculectomy at 1 year in patients with pseudoexfoliation glaucoma.
- Refractory glaucoma: Patients with refractory glaucoma have been defined as those with POAG who have failed incisional glaucoma surgery or presented with severe forms of secondary glaucoma (e.g., after penetrating keratoplasty or globe penetrating injury). The efficacy of MicroShunt implantation has been assessed in 40 eyes of 38 patients with refractory glaucoma, with complete success defined as reaching the target IOP (>6 mm Hg and <14 mm Hg) without additional IOP-lowering medication and qualified success defined as reaching the same target IOP regardless of changes in medication56. The complete success rate after 12 months was 85.7%, with an average IOP of 10.5 ± 2.0 mm Hg, without the use of glaucoma eye drops. The average IOP reduction from baseline was 58.4%. Five (12.5%) eyes experienced failure, with these eyes requiring revision surgery. Another study evaluated the effects of MicroShunt implantation in 47 eyes with uncontrolled IOP despite maximally tolerated medical therapy and at least one previous failed glaucoma surgery57. At baseline, the mean preoperative IOP was 30.1 ± 7.1 mmHg, the mean number of glaucoma medications was 3.4 ± 1, and the mean number of previous surgeries was 2.3 ± 1.3. One year after MicroShunt implantation, the mean IOP was significantly reduced to 18.8 ± 4.6 mmHg, with the mean number of medications significantly reduced to 1.4 ± 1.2. Complete success was achieved in 35% of eyes, and qualified success in 60%. Moreover, 55% of eyes showed a ≥30% reduction in IOP. Needling or bleb repair was performed in 49% of eyes. Complications were minimal and transient, except for one eye that presented with tube extrusion, and another eye with a transected tube. Repeat glaucoma surgery was performed in 17% of eyes. These findings indicated that the PRESERFLO MicroShunt provided moderate success but a significant reduction in IOP, with a good safety profile after 1 year of follow-up, in eyes at high risk for failure of filtering surgery.
- Treatment of refractory childhood glaucoma: MicroShunt implantation into 12 eyes of 12 children, aged 15 months to 14 years, with mean preoperative IOP of 22.72 ± 4.8 mmHg and taking a mean 3.3 ± 0.65 glaucoma medications, showed that none of these eyes experienced any intraoperative complications59. Of the eyes followed-up for ≥1 year (range 12-23 months), nine were successfully controlled, with IOP decreasing from 21.6 ± 4.9 mm Hg preoperatively to 11.9 ± 3.8 mmHg at 1 year, a decrease of 45%. Seven patients were off glaucoma medications at 12 months, and two required two medications (fixed-combination dorzolamide-timolol). Three eyes failed, requiring additional surgery. These results, in which patients were administered 0.4 mg/mL MMC, suggest that the device is safe and effective in patients with refractory childhood glaucoma. A prospective, multicenter trial is planned.
Conclusion
The development of the PRESERFLO MicroShunt was an educated iterative process that occurred over 10 years. The process required formulating a long-term indwelling thermoplastic glaucoma shunt using chemistry and engineering principles. The chemistry and design of the SIBS MicroShunt yielded an atraumatic flow-restrictor tube that minimized foreign body reactions, with few reports of erosion or expulsion of the tube from the eye. Draining to under the conjunctiva and Tenon’s capsule is important because the shunt bypasses areas of high resistance in the drainage path for aqueous humor, including the trabecular meshwork, Schlemm’s canal, the collector channels, the aqueous veins, and the episcleral veins. In addition, the bleb formed in the posterior eye is thicker-walled and potentially less prone to adverse events such as device erosion than blebs formed in the anterior part of the eye. However, the posterior drainage site also contains more fibroblasts and smooth muscle cells, which often necessitate a higher dose of MMC. The MicroShunt is suited to eyes with a failed trabeculectomy, hypermetropia, high pre-operative IOP or contact lenses. Results also suggest that the MicroShunt may be useful in the treatment of childhood glaucoma, pseudoexfoliation glaucoma and other types of refractory glaucoma. In addition, innovations in the field, such as increased MMC dosing, bleb management, stenting the MicroShunt or insertion of a second MicroShunt, may further improve MicroShunt performance. Finally, the MicroShunt allows for rapid post-surgery visual recovery with little risk of corneal decompensation.
Acknowledgments
Editorial assistance with referencing and formatting was provided by BelMed Professional Resources supported by Santen Inc. according to Good Publication Practice guidelines (link).
Author Disclosure Statement
No competing financial interests exist other than being an employee of Santen. No funding was received for this article.
References
- Pinchuk L, Wilson GJ, Barry JJ, Schoephoerster RT, Parel JM, Kennedy JP. Medical applications of poly(styrene-block-isobutylene-block-styrene) (“SIBS”). Biomaterials 2008;29(4):448-460. doi:10.1016/j.biomaterials.2007.09.041
- Pinchuk L, Boden M, Bluestein D. In: SIBS in Medicine. Macromolecular Engineering: Design, Synthesis and Application of Polymers, edited by Lubnin and Erdodi, 2021. 978-0-12-821998-0, Chapter 11, Section V, Disruptive Medical Applications.
- Boden M, Richard R, Schwarz MC, Kangas S, Huibregtse B, Barry JJ. In vitro and in vivo evaluation of the safety and stability of the TAXUS paclitaxel-eluting coronary stent. J Mater Sci Mater Med 2009;20(7):1553–1562. doi:10.1007/s10856-009-3705-5
- Pinchuk L. A review of the biostability and carcinogenicity of polyurethanes in medicine and the new generation of “biostable” polyurethanes. J Biomater Sci Polym Ed 1994;6(3):225-267. doi:10.1163/156856294X00347
- Kennedy JP, Puskas JE, Kaszas G, Hager WG. Patent. Thermoplastic Elastomers of Isobutylene and Process of Preparation. US Patent No. 4946899. 7 August 1990.; 1990.
- Storey RF, Chisholm BJ, Masse MA. Morphology and physical properties of poly(styrene-b-isobutylene-b-styrene) block copolymers. Polymer 1996;37(14):2925-2938.
- Pinchuk L. Patent. Biostable Elastomeric Polymers Having Quaternary Carbons. US Patent No. 5741331. 21 April 1998.
- Widder RA, Kühnrich P, Hild M, Rennings C, Szumniak A, Rossler GF. Intraocular degradation of XEN45 Gel Stent 3 years after its implantation. J Glaucoma 2019;28(12):e171–e173. Doi:10.1097/IJG.0000000000001364
- Pinchuk L, Riss I, Batlle JF, et al. The development of a micro-shunt made from poly(styrene-block-isobutylene-block-styrene) to treat glaucoma. J Biomed Mater Res B Appl Biomater 2017;105(1):211-221. doi:10.1002/jbm.b.33525.
- Acosta AC, Espana EM, Yamamoto H, et al. A newly designed glaucoma drainage implant made of poly(styrene-b-isobutylene-b-styrene): biocompatibility and function in normal rabbit eyes. Arch Ophthalmol 2006;124(12):1742-1749. doi:10.1001/archopht.124.12.1742
- Ibarz Barberá M, Hernandez-Verdejo JL, Bragard J, et al. Evaluation of the ultrastructural and in vitro flow properties of the PRESERFLO MicroShunt. Transl Vis Sci Technol 2021;10(13):26. Doi:10.1167/tvst.10.13.26.
- Fantes F, Acosta AC, Carraway J, et al. An independent GLP evaluation of a new glaucoma drain, the Midi. Invest Ophthalmol Vis Sci. 2006;47(13):3547.
- Arrieta EA, Aly M, Parrish R, et al. Clinicopathologic correlations of poly-(styrene-b-isobutylene-b-styrene) glaucoma drainage devices of different internal diameters in rabbits. Ophthalmic Surg Lasers Imaging 2011;42(4):338-345. doi:10.3928/15428877-20110603-01
- Pinchuk L, Riss I, Batlle JF, et al. The development of SIBS in the InnFocus MicroShunt®. In: Samples JR, Knepper PA, eds. Glaucoma Research and Clinical Advances 2018–2020. Amsterdam, The Netherlands: Kugler Publications; 2018.
- Batlle JF, Fantes F, Riss I, et al. Three-year follow-up of a novel aqueous humor microshunt. J Glaucoma 2016;25(2):e58-e65. doi:10.1097/IJG.0000000000000368
- Batlle JF, Corona A, Albuquerque R. Long-term results of the PRESERFLO MicroShunt in patients with primary open-angle glaucoma from a single-center nonrandomized study. J Glaucoma 2021;30(3):281–286. doi:10.1097/IJG.0000000000001734
- Schlenker MB, Durr GM, Michaelov E, Ahmed IIK. Intermediate outcomes of a novel standalone ab externo SIBS Microshunt with mitomycin C. Am J Ophthalmol 2020;215:141–153. Doi:10.1016/j.ajo.2020.02.020
- Pillunat KR, Herber R, Haase MA, Jamke M, Jasper CS, Pillunat LE. PRESERFLO™ MicroShunt versus trabeculectomy: first results on efficacy and safety. Acta Ophthalmol 2021;100(3):e779–e790. Doi:10.1111/aos.14968
- Beckers HJM, Aptel F, Webers CAB, et al. Safety and effectiveness of the PRESERFLO MicroShunt in primary open-angle glaucoma: results from a 2-year multicenter study. Ophthalmol Glaucoma 2022;5(2):195–209. Doi:10.1016/j.ogla.2021.07.008
- Baker ND, Barnebey HS, Moster MR, et al. Ab-externo MicroShunt versus trabeculectomy in primary open-angle glaucoma: one-year results from a 2-year randomized, multicenter study. Ophthalmology 2021;128(12):1710–1721. Doi:10.1016/j.ophtha.2021.05.023
- Gambini G, Carla MM, Giannuzzi F, et al. PreserFlo® MicroShunt: an overview of this minimally invasive device for open-angle glaucoma. Vision (Basel) 2022;6(1):12. Doi:10.3390/vision6010012
- Khawaja AP, Stalmans I, Aptel F, et al. Expert consensus on the use of the PRESERFLO MicroShunt device in the treatment of glaucoma: a modified delphi panel. Ophthalmol Ther 2022;11(5):1743–1766. Doi:10.1007/s40123-022-00529-4
- Ranjit A, Patel B, Sherman TEJ, et al. Evaluating post-operative flare in glaucoma surgery: Miniject, Microshunt, Baerveldt tube, cataract & ECP and trabeculectomy. Invest Ophthalmol Vis Sci 2024;65(7):3522 (abstract).
- Khan A, Khan AU. Comparing the safety and efficacy of Preserflo Microshunt implantation and trabeculectomy for glaucoma: A systematic review and meta‐analysis. Acta Ophthalmol 2024;102(4):e443–e451. Doi:10.1111/aos.16658
- Panarelli JF, Moster MR, Garcia-Feijoo J, et al. Ab-externo MicroShunt versus trabeculectomy in primary open-angle glaucoma: two-year results from a randomized, multicenter study. Ophthalmology 2024;131(3):266–276. Doi:10.1016/j.ophtha.2023.09.023
- Non-Inferiority Clinical Trials to Establish Effectiveness Guidance for Industry, U.S. Department of Health and Human Services Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER), November 2016, Clinical/Medical
- Gedde SJ, Chen PP, Heuer DK, et al. The Primary Tube Versus Trabeculectomy Study: Methodology of a multicenter randomized clinical trial comparing tube shunt surgery and trabeculectomy with mitomycin C. Ophthalmology 2018;125(5):774–781. Doi:10.1016/j.ophtha.2017.10.037.
- World Glaucoma Association. In: Shaarawy T, ed. Guidelines on Design and Reporting of Glaucoma Surgical Trials. Amsterdam: Kugler; 2009. 22.
- The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol 2000;130(4):429-440. doi:10.1016/S0002-9394(00)00538-9
- Riss I, Batlle J, Pinchuk L, Kato YP, Weber BA, Parel JM. One-year results on the safety and efficacy of the InnFocus MicroShunt™ depending on placement and concentration of mitomycin C. J Fr Ophtalmol 2015;38(9):855–860. doi:10.1016/j.jfo.2015.05.005
- Beckers HJM, Kujovic-Aleksov S, Webers CAB, Riss I, Batlle J, Parel JM. One-year results of a three-site study of the MicroShunt®. Acta Ophthalmol 2017;95(S258):28-29. doi:10.1111/aos.13419
- Martínez-de-la-Casa JM, Saenz-Frances F, Morales-Fernandez L, et al. Clinical outcomes of combined Preserflo Microshunt implantation and cataract surgery in open-angle glaucoma patients. Sci Rep 2021;11(1):15600. doi:10.1038/s41598-021-95217-x
- Fili S, Kontopoulou K, Vastardis I, Perdikakis G, Kohlhaas M. PreserFlo MicroShunt versus trabeculectomy in patients with moderate to advanced open-angle glaucoma: 12-month follow-up of a single-center prospective study. Curēus 2022;14(8) e28288. doi:10.7759/cureus.28288.
- Van Lancker L, Saravanen A, Abu-Bakra M, et al. Clinical outcomes and cost analysis of PreserFlo versus trabeculectomy for glaucoma management in the United Kingdom. Ophthalmol Glaucoma 2023;6(4):342–357. Doi:10.1016/j.ogla.2022.11.006
- Tanner A, Haddad F, Fajardo-Sanchez J, et al. One-year surgical outcomes of the PreserFlo MicroShunt in glaucoma: a multicentre analysis. Br J Ophthalmol 2023;107(8):1104–1111. doi:10.1136/bjophthalmol-2021-320631
- Abegao Pinto L, Sunaric Megevand G, Stalmans I, et al. European Glaucoma Society – A guide on surgical innovation for glaucoma. Br J Ophthalmol 2023;107(Suppl 1):1–114. doi:10.1136/bjophthalmol-2023-egsguidelines
- Kakizaki H, Takahashi Y, Nakano T, et al. Anatomy of Tenons capsule. Clin Exp Ophthalmol 2012;40(6):611–616. doi:10.1111/j.1442-9071.2011.02745.x
- Gubser PA, Pfeiffer V, Hug S, et al. PRESERFLO MicroShunt implantation versus trabeculectomy for primary open-angle glaucoma: a two-year follow-up study. Eye Vis (Lond) 2023;10(1):50. doi:10.1186/s40662-023-00369-8.
- Gedde SJ, Feuer WJ, Lim KS, et al. Treatment outcomes in the primary tube versus trabeculectomy study after 3 years of follow-up. Ophthalmology 2020;127(3):333–345. doi:10.1016/j.ophtha.2019.10.002
- De Francesco T, Armstrong JJ, Hussein IM, Costa MP, Ahmed IIK. Mitomycin 0.2 mg/ml versus mitomycin 0.4 mg/ml during the implantation of an ab externo polystyrene-isobutylene-styrene microshunt: a mega-analysis. Ophthalmol Glaucoma 2024;7(5):454-465. doi:10.1016/j.ogla.2024.06.001
- Dervos T, Gugleta K, Scholl HPN, Gatzioufas Z, Enz TJ. Single versus double PreserFlo MicroShunt implantation in glaucoma patients: a retrospective cohort study. Ophthalmic Res 2023;66(1):1362-1375. doi:10.1159/000535276
- Lupardi E, Laffi GL, Moramarco A, Barboni P, Fontana AL. Systematic Preserflo MicroShunt intraluminal stenting for hypotony prevention in highly myopic patients: a comparative study. J Clin Med 2023;12(4):1677. doi:10.3390/jcm12041677
- Ophthalmology Unit Researchers Provide New Data on Clinical Medicine (Systematic Preserflo MicroShunt Intraluminal Stenting for Hypotony Prevention in Highly Myopic Patients: A Comparative Study). NewsRX LLC, 2023. Print.
- Aguilar-Munoa S, Tham YH, Barton K. A simple surgical solution for the treatment of persistent postoperative hypotony after PRESERFLO MicroShunt implantation. Eye (Lond) 2023;37(10):2126–2129. doi:10.1038/s41433-022-02301-1.
- Verma-Fuehring R, Dakroub M, Bamousa A, Kann G, Hillenkamp J, Kampik D. The use of intraluminal PRESERFLO stenting in avoiding early postoperative hypotony. Graefes Arch Clin Exp Ophthalmol 2024, in press. doi:10.1007/s00417-024-06567-x
- Lupardi E, Laffi GL, Ciardella A, Bartoni P, Fontana L. Ab-externo intraluminal stent for prolonged hypotony and choroidal detachment after Preserflo implantation. Eur J Ophthalmol 2023;33(5):NP63–NP66. doi:10.1177/11206721221137166
- Farrar N, Yan DB, Johnson M. Modeling the effects of glaucoma surgery on intraocular pressure. Exp Eye Res 2021;209:108620. doi:10.1016/j.exer.2021.108620.
- Lüke JN, Enders P, Lappa A, Dietlein TS. Revision of the PRESERFLO MicroShunt with ologen and mitomycin C. Ophthalmologie 2023;120(4):440-442. doi:10.1007/s00347-023-01816-6
- Vastardis I, Fili S, Perdikakis G, et al. Preliminary results of Preserflo Microshunt versus Preserflo Microshunt and ologen implantation. Eye Vis (Lond) 2021;8(1):33. doi:10.1186/s40662-021-00253-3
- Lai JSM, Poon ASY, Tham CCY, Lam DSC. Trabeculectomy with beta radiation: long-term follow-up. Ophthalmology 2003;110 (9):1822–1826. doi:10.1016/S0161-6420(03)00561-X
- Constable PH, Crowston JG, Occleston NL, Cordeiro MF, Khaw PT. Long term growth arrest of human Tenon’s fibroblasts following single applications of beta radiation. Br J Ophthalmol 1998;82(4):448–452. doi:10.1136/bjo.82.4.448




