Telemedicine in Huntington’s Disease: Feasibility and Value
TeleHD: Establishing feasibility, validity, and value of telemedicine in Huntington’s disease
Juan Deliz1, Nicola Lancki2, Danny Bega1, Danielle Larson1
- Department of Neurology, Northwestern University Feinberg School of Medicine, Chicago, Illinois USA
- Department of Preventive Medicine-Biostatistics, Northwestern University Feinberg School of Medicine, Chicago, Illinois USA
OPEN ACCESS
PUBLISHED 31 October 2024
CITATION Deliz, J., Lancki, N., et al., 2024. TeleHD: Establishing feasibility, validity, and value of telemedicine in Huntington’s disease. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5960
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5960
ISSN 2375-1924
ABSTRACT
Objective: To demonstrate the feasibility of conducting telemedicine visits, establish the validity and reliability of tele-administered motor/non-motor assessments, and determine the satisfaction of virtual visits for individuals with Huntington disease (HD).
Background: Telemedicine has become a prevalent method for patients to interact with their healthcare providers. Patients with HD often cite time and travel burden as significant barriers to receive care in HDSA centers of excellence. TeleHD studied the applicability of telemedicine in a specialized HD clinic at a large urban center.
Methods: Individuals aged 18 to 70 with genetically confirmed HD, MoCA score >26, and a smartphone or computer were enrolled. Participants completed 2 pairs of in-person clinic and televisits 1 month apart, completing all 4 visits within a 9-month time frame. Validity of tele-administered composited Unified Huntington’s Disease Rating Scale (UHDRS) component of Total Motor Score (TMS), Total Functional Capacity (TFC) Score, Symbol Digit Modalities Test (SMT), and Stroop Color and Word Test (SCWT) was assessed by measuring the strength of the association with clinic-administered scales using Pearson correlation coefficients. Intraclass correlation coefficients (ICC) were calculated to assess cross-sectional reliability and agreement between in-person and tele-administered scales. The value of televisits was assessed with a Satisfaction Survey and Resource Utilization Survey.
Results: Thirty-one participants were enrolled. 55% completed all study visits. The cross-sectional correlations for TMS and mTMS at visits 1 and 2 were very high (r=0.95 and r= 0.98), respectively with p<0.01. Similarly high correlations were observed for cognitive tests including TFC, SWRT Word and Color and SDMT (r >0.8, p<0.01), but were lower for Color-Word and Interference. ICCs showed excellent agreement for TMS and mTMS, TFC, SCWT-Color, SCWT-Word, and SDMT. Ten participants completed the satisfaction and utilization surveys.
Conclusions: The results failed to meet the pre-defined measure of feasibility (80% completion of both telehealth visits). Nonetheless, there was evidence of strong validity and reliability of tele-administration of multiple assessment scales, including administered modified TMS, TFC scale, and cognitive assessments SWCT and SDMT. Respondents to the satisfaction and utilization surveys (N=10) were either satisfied or very satisfied with televisit convenience, comfort, and care.
Keywords: Telemedicine, Huntington’s disease, Ambulatory clinic, Movement disorders
INTRODUCTION
Telemedicine, including real-time videoconferencing between healthcare providers, offers increased patient access to specialized care, overcoming barriers of location, distance, and accessibility of academic centers. This advantage is integral in the neurologic sub-specialty of Movement Disorders which includes caring for those with limited mobility and/or cognitive impairment, and often rare neurologic conditions. Within this field, telemedicine has been deemed feasible for assessments of individuals with Parkinson’s disease (PD), with established validity and reliability of the tele-administered Unified Parkinson’s Disease Rating Scale (UPDRS). Furthermore, telemedicine for PD enables multidisciplinary care provides patient satisfaction, uniquely providing the “four C pillars for PD patients: care, convenience, comfort, and confidentiality. The utility of telemedicine in Movement Disorders was realized, and accelerated by necessity during the COVID-19 pandemic.
Huntington’s disease (HD) is a severe genetic neurodegenerative that affects mobility, cognition and mental health, ultimately leading to disability, dependence and mortality. As such, HD care is complex and requires expert multidisciplinary care, best offered at Huntington’s disease Society of America Centers of Excellence (HDSA COEs), of which there are 56 in the U.S. HDSA COEs are congregated around urban areas which leave large areas of the U.S. without convenient access to experts in the field. Telemedicine provides an opportunity to nullify the distance and time burden to patients and care-partners to receive this specialized care. However, standard motor, cognitive and functional HD assessments have not been deemed valid and reliable for virtual administration. A small pilot study of tele-administration of a modified Unified Huntington’s Disease Rating Scale Total Motor Score (UHDRS-TMS) examination (excluding rigidity and balance assessments) showed good reliability with intraclass coefficients (ICC) = 0.78 virtual home visits were found to be reliable for conducting motor assessments in HD. However, additional measures within the composite UHDRS (cUHDRS) have not been validated, including the Total Functional Capacity (TFC) Score, and cognitive assessments Symbol Digit Modalities Test (SDMT), and Stroop Color and Word Test (SCWT) for cognitive assessments. It has previously been demonstrated that the Montreal Cognitive Assessment (MoCA) can be conducted remotely in PD and HD, suggesting the feasibility of conducting the SDMT and SWRT via telemedicine. Our study, TeleHD, builds on the limited work on telemedicine in the HD population to determine the feasibility, validity, and value of televisits to assess motor and non-motor features of HD individuals.
METHODS
The TeleHD pilot study included genetically-confirmed HD patients, between the ages of 18 and 70, HD Stage I-IV, recruited at Northwestern University (NU) Parkinson’s Disease and Movement disorder Center’s HDSA COE. It was supported by a Huntington’s Disease Society of America Human Biology Fellowship grant, and approved by NU’s IRB and assigned protocol number STU00211872. Participants were primarily English-speaking, with a personal computer, laptop, or tablet, equipped with high-speed internet, camera, and microphone, and allowance for caregiver assistance in visit facilitation and assessment completion. Individuals with cognitive impairment (defined as MoCA score < 21) were excluded.
Participants were asked to complete a total of two in-clinic visits and two televisits over a 9-month period. The first televisit was conducted within 30 days of the first in-person clinic visit. At enrollment, participants were randomized on a 1:1 basis to participate in their second televisit 30 days before (“Group A”) or 30 days after (“Group B”) their second in-person clinic visit. Outcomes assessed at each in-person and televisit included the components of the cUHDRS, as described in further detail below. A trained clinical research coordinator or clinician completed all assessments for in-person clinic visits and televisits. The same clinician rater completed a participant’s paired in-person TMS and televisit mTMS to eliminate inter-rater variability. The SDMT and SCWT were provided in clinic or mailed in sealed envelopes to participants ahead of their televisit to facilitate completion during the televisit. To assess value, patients and caregivers completed two surveys during in-person visits (i.e. the “Satisfaction Survey” and “Resource Use Survey”), which were used to assess ease of technology use, preference for televisits, and impact on time and cost burden.
TMS and mTMS: The UHDRS‐TMS is formed of 15 items assessing for oculomotor dysfunction, chorea, dystonia, parkinsonism, and balance. Each item is rated from 0 (normal) to 4 (severe impairment), with a total maximum score of 124, with higher scores indicating more severe motor impairment. Complete TMS assessments were performed at in-person clinic visits, and a mTMS was completed during televisits. The mTMS included 13 of the 15 motor assessments, omitting rigidity and balance.
TFC: This scale contains 5 items assessing independence with occupation, finances, domestic chores, activities of daily living and living environment. Each item has a 4-point ordinal scale ranging from 0 to 3 for a total score of 0-13, with higher scores indicating higher functional capacity.
SDMT: This test assesses visual scanning, processing speed, and attention. The subject has 90 seconds to pair numbers with geometric symbols provided in a coding key. The score, from 0 to 110, is the number of correct pairs recorded in 90 seconds.
SCWT: This test involves three trials: WORD and COLOR are measures of processing speed, and COLOR-WORD is a measure of inhibition and executive functions. The score for each trial is the number read correctly in 45 seconds. Higher scores indicate better cognitive performance. Raw scores were standardized to a reference population (general population) to generate T scores, controlling for age and years of education.
STATISTICAL ANALYSIS
Descriptive statistics were used to summarize the study population demographics, clinical baseline characteristics and motor and cognitive scores. Means and standard deviations or medians and ranges for continuous variables and frequency percent for categorical variables were reported.
The feasibility of conducting telemedicine visits was computed as the proportion of individuals completing all telehealth assessments, and the 95% confidence interval was constructed. Feasibility was defined as at least 80% of participants completing all televisits as scheduled, including completion of all cUHDRS components. To assess validity of tele-administered scales compared to in-person administration, scatter plots of scores from the clinic visit on the x-axis and televisit on the y-axis were plotted, and Pearson’s correlation coefficients were calculated to measure the strength of association, with r > 0.80 demonstrating strong validity. These were assessed cross-sectionally and reported separately at each time point for all available pairs of assessments. To assess reliability between scores on in-person and tele-administered scales, intraclass correlation coefficients (ICCs) were calculated for each visit pair using a two-way random-effects model with absolute agreement. ICCs and 95% confidence intervals were reported. An ICC>0.7 indicates excellent agreement. Descriptive statistics were used to summarize responses to the Satisfaction Survey and Resource Use Survey. Response rates were low, therefore the characteristics of respondents were also assessed and reported. All analyses were conducted using R version 4.1.2.
RESULTS
A total of 31 participants with HD consented to the study with a mean (SD) age of 45 (13) years (Table 1). Fifty-five percent of participants were female, 16% identified as Hispanic or Latino, and 87% were white. The mean (SD) MoCA score at the first assessment was 25.5 (3.2).
| Characteristic | Group A n = 16 | Group B n = 15 | Overall n = 31 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Age (years), Mean (SD) | 44 (12) | 47 (15) | 45 (13) | ||||||
| Gender, n (%) | Male 7 (44) | Male 7 (47) | Male 14 (45) | Female 9 (56) | Female 8 (53) | Female 17 (55) | |||
| Ethnicity, n (%) | Not Hispanic or Latino 13 (81) | Not Hispanic or Latino 13 (87) | Not Hispanic or Latino 26 (84) | Hispanic or Latino 3 (19) | Hispanic or Latino 2 (13) | Hispanic or Latino 5 (16) | |||
| Race, n (%) | Caucasian 14 (88) | Caucasian 13 (87) | Caucasian 27 (87) | African American 1 (6.3) | African American 0 (0) | African American 1 (3.2) | Other 1 (6.3) | Other 2 (13) | Other 3 (9.7) |
| MoCA score, Mean (SD) | 26.0 (3.2) | 25.0 (3.3) | 25.5 (3.2) | ||||||
| Unknown | 0 | 1 | 1 | ||||||
| Pairs of assessments, n (%) | 0 2 (13) | 0 1 (6.7) | 0 3 (9.7) | 1 3 (19) | 1 8 (53) | 1 11 (35) | 2 11 (69) | 2 6 (40) | 2 17 (55) |
| Satisfaction Survey, n (%) | Incomplete 10 (63) | Incomplete 11 (73) | Incomplete 21 (68) | Complete 6 (38) | Complete 4 (27) | Complete 10 (32) |
Seventeen participants (55%) completed both pairs of assessments. At the first visit, 28 participants completed both clinic and televisit TMS assessments and 16 completed both clinic and televisit TMS assessments and could be included in analyses of reliability and validity. One pair of visits was >60 days between each other and excluded from analyses. Among the 31 participants enrolled, 17 participants (55%) completed all televisits (95% CI: 36%, 72%).
| Characteristic | Clinic VI, N = 31 | Televist VI, N = 29 | Clinic VII, N = 18 | Televisit VII, N = 18 | ||||
|---|---|---|---|---|---|---|---|---|
| TMS | Mean (SD) 16 (13) | NA | 19 (12) | NA | Median (Range) 13 (0, 50) | 18 (0, 40) | Missing 0 | 0 |
| mTMS | Mean (SD) NA | 15 (12) | NA | 18 (15) | Median (Range) 14 (0, 42) | 13 (0, 45) | Missing 1 | 1 |
| TFC Total | Mean (SD) 10.5 (3.1) | 10.3 (3.1) | 9.8 (3.4) | 10.2 (3.4) | Median (Range) 12 (5, 13) | 12 (5, 13) | Missing 0 | 1 |
| Stroop Word | Mean (SD) 28 (15) | 26 (14) | 28 (14) | 25 (14) | Median (Range) 25 (6, 56) | 23 (4, 55) | Missing 3 | 1 |
| Stroop Color | Mean (SD) 30 (17) | 31 (17) | 30 (18) | 28 (16) | Median (Range) 27 (7, 67) | 27 (6, 61) | Missing 3 | 1 |
| Stroop Word-Color | Mean (SD) 44 (14) | 44 (14) | 46 (13) | 44 (12) | Median (Range) 42 (18, 71) | 41 (23, 77) | Missing 3 | 1 |
| Stroop Interference | Mean (SD) 55 (10) | 56 (9) | 57 (7) | 58 (7) | Median (Range) 56 (40, 80) | 56 (41, 77) | Missing 3 | 1 |
| SDMT | Mean (SD) 40 (16) | 37 (16) | 35 (16) | 36 (12) | Median (Range) 40 (16, 74) | 34 (13, 67) | Missing 0 | 1 |
In assessing validity, the cross-sectional correlations for TMS and mTMS at visits 1 and 2 were very high (r=0.95 and r= 0.98), respectively with p<0.01 (Figure 1). Similar high correlations were seen for the TFC, and cognitive tests SDMT and SCWT-WORD and COLOR and SDMT (r >0.8, p<0.01). Correlations were lower for SCWT-COLOR-WORD and Interference sub-scores.
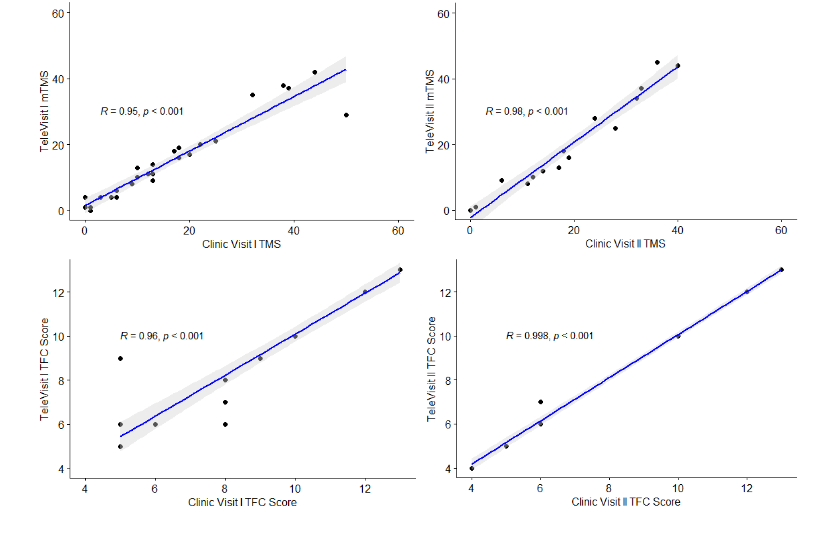
In assessing agreement between in-person and tele-administered scales, ICCs showed excellent agreement (ICCs and 95% CIs >0.7) for TMS, TFC, SDMT, and most components of the SCWT (Table 3).
| ICC Model | Outcome | ICC (95% CI) |
|---|---|---|
| Cross Sectional Visit 1 (n=28) | TMS | 0.94 (0.87,0.97) |
| Cross Sectional Visit 2 (n=15) | TMS | 0.96 (0.9,0.99) |
| Cross Sectional Visit 1 (n=28) | TFC | 0.96 (0.92,0.98) |
| Cross Sectional Visit 2 (n=16) | TFC | 1.00 (0.99,1.00) |
| Cross Sectional Visit 1 (n=28) | SCWT- WORD | 0.96 (0.92,0.98) |
| Cross Sectional Visit 2 (n=15) | SCWT- WORD | 0.87 (0.67,0.95) |
| Cross Sectional Visit 1 (n=28) | SCWT- COLOR | 0.97 (0.94,0.99) |
| Cross Sectional Visit 2 (n=15) | SCWT- COLOR | 0.90 (0.74,0.96) |
| Cross Sectional Visit 1 (n=28) | SCWT-WORD-COLOR | 0.76 (0.56,0.88) |
| Cross Sectional Visit 2 (n=15) | SCWT-WORD-COLOR | 0.90 (0.75,0.96) |
| Cross Sectional Visit 1 (n=28) | SCWT-Interference | 0.51 (0.19,0.74) |
| Cross Sectional Visit 2 (n=15) | SCWT-Interference | 0.76 (0.44,0.91) |
| Cross Sectional Visit 1 (n=28) | SDMT | 0.89 (0.78,0.95) |
| Cross Sectional Visit 2 (n=15) | SDMT | 0.93 (0.82,0.98) |
About a third of participants completed satisfaction and resource utilization surveys. Satisfaction was high, with most very satisfied for convenience, comfort, care and overall. A slightly lower percentage were satisfied with technical quality. Most reported no difference or better than in-person visits for convenience, time, comfort, connection, quality of care. Among those who responded to the resource utilization survey, median travel time was 120 minutes (range 20-180 minutes) and all used a car their transportation method.
| Characteristic | N = 10 | ||
|---|---|---|---|
| Technical quality, n (%) | Neutral 2 (20) | Satisfied 2 (20) | Very Satisfied 6 (60) |
| Convenience, n (%) | Satisfied 2 (20) | Very Satisfied 8 (80) | |
| Comfort, n (%) | Neutral 1 (10) | Satisfied 2 (20) | Very Satisfied 7 (70) |
| Care, n (%) | Neutral 1 (10) | Satisfied 1 (10) | Very Satisfied 8 (80) |
| Overall, n (%) | Satisfied 3 (30) | Very Satisfied 7 (70) | |
| Compare: Convenience, n (%) | No difference from in-person clinic visits 2 (20) |
In assessing validity, the cross-sectional correlations for TMS and mTMS at visits 1 and 2 were very high (r=0.95 and r= 0.98), respectively with p<0.01 (Figure 1). Similar high correlations were seen for the TFC, and cognitive tests SDMT and SCWT-WORD and COLOR and SDMT (r >0.8, p<0.01). Correlations were lower for SCWT-COLOR-WORD and Interference sub-scores.
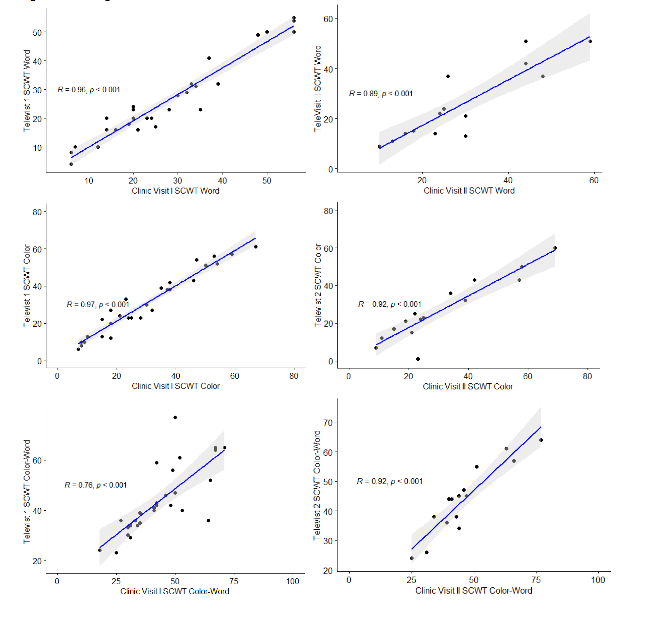
The TeleHD pilot study demonstrates strong validity and reliability of virtually administered cUHDRS components, including a modified TMS, the TFC scale, and cognitive assessments (e.g. SCWT and SDMT). These results have important implications for HD clinical care and research, as the ability to complete these assessments virtually can decrease the time and travel burden for patients and their care partners and research participants. Virtual use of such scales can extend the scope of care at HDSA COEs for HD care, as there are limited COEs in the U.S., leaving HD patients outside of specialized care networks with decreased access to clinical trial participation. Additionally, demonstration of validity and reliability has implications for clinical trial design and implementation, as the cUHDRS, and specific sub-scales of the measure are used as both an endpoint for clinical research. Specifically, this study adds valuable demonstration of reliability and validity of cognitive scales SDMT and SCWT which is commonly used in HD and other neurologic conditions. Previously, the MoCA has been the only cognitive scale analyzed for virtual use. Nonetheless, the virtual UHDRS study sponsored by the Huntington Study Group (HSG) is currently underway to formally determine the reliability of administering all sections of the UHDRS virtually compared to in-person. Utilization of tele-administered scales for remote assessments has significant potential to reduce clinical trial time and cost burden, thus increasing participant access to trial participation.
The COVID-19 pandemic dramatically showcased the value of telemedicine, especially for patients with chronic neurological conditions and particularly those with movement disorders. Most research on telemedicine within the field has focused on Parkinson’s disease, with a growing body of evidence supports its feasibility and effectiveness for remote patient management. Nonetheless, the use of telemedicine for hyperkinetic movement disorders (such as HD chorea, tremor, and myoclonus) remains relatively unexplored. However, many experts continue to advocate for expanding virtual services to improve access and care in underserved areas.
While this study failed to demonstrate a pre-defined measure of feasibility based on the percentage of participants who completed all study visits, this failure was in a large part due to a limited sample size. The target for enrollment was 40 participants and only 31 participants were enrolled. A secondary main limitation was the drop-out of study participants, such that only 48% of participants completed both the first and second pair of in-person and clinic visits. Unfortunately, this was driven by the Covid-19 pandemic and staff turnover having a negative impact on participant recruitment and retention. As these extenuating circumstances prevented participants from completing all visits, this is unlikely to be a true representation of televisit feasibility in HD. Though limited by small sample size, value of televisits was demonstrated by the outcomes of the satisfaction survey, with the majority of respondents either satisfied or very satisfied with televisit convenience, comfort, and care. Results of the resource utilization survey reiterate what has been previously reported for HD patients, which is a significant time and travel burden for clinical care.
CONCLUSION
The TeleHD pilot study demonstrates strong validity and reliability of virtually administered cUHDRS components, including a modified TMS, the TFC scale, and cognitive assessments (i.e. SCWT and SDMT). Survey respondents reported satisfaction with televisit convenience, comfort and care compared to in-person visits. These results have important implications for HD clinical care and research trial design and implementation, as they suggest telemedicine may reduce financial and geographical barriers for a vulnerable population of patients. Utilization of virtually administered scales for remote assessments may therefore improve access to HDSA COEs and increase participation in clinical trials.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
FUNDING STATEMENT
This study was funded by the Huntington’s Disease Society of America Human Biology Fellowship Award, awarded to Dr. Danielle Larson in 2019.
ACKNOWLEDGMENTS
The authors would like to thank the efforts of the clinical research team of the Movement Disorders Division at Northwestern University and all the clinical staff helping to take care of HD patients on a daily basis.
REFERENCES
- Barbosa W, Zhou K, Waddell E, Myers T, Dorsey ER. Improving Access to Care: Telemedicine Across Medical Domains. Annu Rev Public Health. 2021;42:463-481.
- Houston E, Kennedy AG, O’Malley D, Rabinowitz T, Rose GL, Boyd J. Telemedicine in Neurology: A Scoping Review of Key Outcomes in Movement Disorders. Telemed J E Health. 2022;28(3):295-308.
- Abdolahi A, Scoglio N, Killoran A, Dorsey A, Biglan K. Potential reliability and validity of a modified version of the Unified Parkinson’s disease rating scale that could be administered remotely. Parkinsonism Related Disord. 2013 February. 19(2): 218-221.
- Dorsey R, Deuel L, Voss T, et al. M. Increasing access to specialty care: a pilot, randomized controlled trial of telemedicine for Parkinson’s disease. Movement Disorder. 2010: 25(11), 1652-1659.
- Ben-Pazi H, Browne P, Chan P, et al. The promise of telemedicine for movement disorders: an interdisciplinary approach. Current neurology and neuroscience reports. 2018; 18(5): 1-10.
- Wilkinson J, Spindler M, Wood S, et al. High patient satisfaction with telehealth in Parkinson disease: a randomized controlled study. Neurology: Clinical Practice. 2016; 6(3): 241-251.
- Dorsey ER, Okun MS, Bloem BR (2020) Care, Convenience, Comfort, Confidentiality, and Contagion: The 5 C’s that Will Shape the Future of Telemedicine. J Parkinsons Dis 10(3):893-897.
- Bloem BR, Dorsey RE, Okun M (2020) The coronavirus disease 2019 Crisis as Catalyst for Telemedicine for Chronic Neurological Disorders. JAMA Neurology 77(8), 927-928.
- A, Papa SM, Brundin P, Fung VSC, et al (2020) Impact of the COVID-19 Pandemic on Parkinson’s Disease and Movement Disorders. Mov Disord Clin Pract 7(4):357-360.
- Cubo E, Hassan A, Bloem BR, Mari Z, Group MD-TS (2020) Implementation of Telemedicine for Urgent and Ongoing Healthcare for Patients with Parkinson’s Disease During the COVID-19 Pandemic: New Expectations for the Future. J Parkinsons Dis 10(3), 911-913.
- Grossman et al. Rapid implementation of virtual neurology in response to the COVID-19 pandemic. Neurology 2020; 94:1077-1087.
- Hassan A, Mari Z, Gatto EM, et al. Global Survey on Telemedicine Utilization for Movement Disorders During the COVID-19 Pandemic. Mov Disord. 2020;35(10):1701-1711.
- Larson D, Schneider R and Simuni T. A new era: the growth of video-based visits for remote management of persons with Parkinson’s disease. Journal of Parkinson’s disease 2021; 11(s1):S27-S34.
- Nicholas S Caron, PhD, Galen EB Wright, PhD, and Michael R Hayden. Huntington’s disease. Gene Reviews. October 23, 1998; Last Update: June 11, 2020.
- HDSA Centers of Excellence. https://hdsa.org/find-help/clinical-care-services/hdsa-centers-of-excellence
- Bull MT, Darwin K, Venkataraman V, Wagner J, Beck CA, Dorsey ER, et al. A pilot study of virtual visits in Huntington disease. J Huntingtons Dis. 2014;3:189–95.
- Creighton S, Ho A, McManus B, Hayden MR. Providing predictive testing for Huntington disease via telehealth: results of a pilot study in British Columbia, Canada. Clin Genet. 2013;84:60–4.
- Trundell D, G Palermo, S Schobel, J Long, B Leavitt, S Tabrizi. Defining clinically meaningful change on the composite unified Huntington’s disease rating scale (cUHRS). Neurology April 2019. 92(15) P1.8-043.
- Mestra, MK Forjaz, M Philipp, Cardoso F, Ferreira J, Reilmann R, C Sampaio, C Goetz, E Cubo, P Martinez-Martin, G Stebbins. Rating scales for motor symptoms and signs in Huntington’s disease: Critique and recommendations. Mov Disord Clin Pract. 2018 Mar-Apr; 5(2) 111-117.
- Klempír, Jirí, et al. “Unified Huntington’s disease rating scale: clinical practice and a critical approach.” Functional neurology 21.4 (2006): 217.
- Smith A. Symbol Digit Modalities Test: Manual. Los Angeles: Western Psychological Services; 1982.
- Golden C and Freshwater SM. The stroop color and word test: a manual for clinical and experimental uses. Wood dale, IL stoelting Co, 2002.
- Abdolahi A, Bull M, Darwin K, Venkataramna M, Grana R, Dorsey K, Biglan. A feasibility study of conducting the Montreal Cognitive Assessment remotely in individuals with movement disorders. Health Informatics Journal 2016 22(2) 304-311.
- Frank S. LBA-18: Utility of the Virtual Unified Huntington’s Disease Rating Scale (vUHDRS®) MDS International Congress 2023.
- Mishra S, Dhuna N, Lancki N, Yeh C, Larson DN. Telehealth utilization and patient satisfaction in an ambulatory movement disorders center during the COVID-19 pandemic. J Telemed Telecare. 2024;30(8):1293-1299.
- Cubo E, Delgado-López PD. Telemedicine in the Management of Parkinson’s Disease: Achievements, Challenges, and Future Perspectives. Brain Sci. 2022;12(12):1735.
- Srinivasan R, Ben-Pazi H, Dekker M, et al. Telemedicine for Hyperkinetic Movement Disorders. Tremor Other Hyperkinet Mov (N Y). 2020.
- Shalash A, Spindler M, Cubo E. Global Perspective on Telemedicine for Parkinson’s Disease. J Parkinsons Dis. 2021.




