Three Major Infectious Diseases in Africa: HIV, TB, Malaria
Controversies and Questions Regarding the Three Great Killers in Africa
J. Clarke McIntosh¹, China Mayol Kuot²
- Director Medical Ward, Mary Immaculate Hospital, Mapuordit, South Sudan
- Doctor Medical Ward, Mary Immaculate Hospital, Mapuordit, South Sudan
OPEN ACCESS
PUBLISHED 31 May 2025
CITATION McIntosh, JC., and Kuot, CM., 2025. Controversies and Questions Regarding the Three Great Killers in Africa. Medical Research Archives, [online] 13(5). https://doi.org/10.18103/mra.v13i5.6538
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i5.6538
ISSN 2375-1924
ABSTRACT
Introduction: If we speak of the three great killers in the first world (those countries with stable governments and relative affluence in comparison to the developing world), cancer, heart disease, and respiratory diseases (particularly if we include lung cancer) come to mind. In the developing world (third world), the three great killers are infectious diseases. The differentiation between first and third world have been obscured in many nations, where there is a mixture of mixture of problems, both the diseases of the affluent societies and those that have less developed medical systems. This review will focus on the three great killers afflicting sub-Saharan Africa: HIV, TB, and malaria.
Methodology: This manuscript is primarily a literature review of HIV, TB, and malaria using publicly available sources such as Google Search, PubMed and NIH reviews. When using personal experience, it is limited to East Africa, particularly Sudan and South Sudan, where the authors have their primary experience with these diseases.
Keywords
- HIV
- TB
- Malaria
- Africa
Results and Discussion: HIV (Human immunodeficiency virus)
Far too much has been written about HIV to attempt anything close to a comprehensive review of this killer who emerged in the 1980’s. Most people know that 75% of the cases and 90% of the deaths from HIV come from Africa. Many people fail to give sufficient credit to Condoleeza Rice and President George W Bush for their work in the early 21st century to provide testing and medications for HIV in Africa through PEPFAR (President’s Emergency Plan for AIDS Relief), but estimates of the effectiveness of the program suggest that over 25,000,000 lives have been saved through the program. The call for transfer to more local ownership and direction has to be balanced with a realistic appraisal of the likelihood of abuse of funds. The Health Pooled Funds (HPF) attempted to address the dire state of medicine in South Sudan, but much of its limited success could be attributed to by-passing the Government of South Sudan (GoSS) and ministry of health (MoH) and directing funds through carefully vetted NGO’s (non-government organizations). A similar attempt through UNICEF has been disastrous because the funds were given to GoSS and MoH.
In South Sudan, we still run into significant problems with supplying testing kits and medications, but the major issue in South Sudan is overcoming the stigma and denial concerning the disease. ICAP (International Center for AIDS Care and Treatment Programs) is the NGO designated to administer PEPFAR at Mary Immaculate Hospital (MIH) in Mapuordit, South Sudan. I have tried to engage ICAP representatives regarding their outreach methods. Their procedure is to counsel a patient once and then give him six months of medications and a return appointment half a year later. Their rationale is that the patients do not want to face the stigma of going into the VCT (voluntary counseling and testing) to get their drugs, so they attempt to reduce the stigma by making them come only twice a year. Though I acknowledge some validity to their concerns, I think their approach is destined for failure in a large percentage of the patients for numerous reasons. First, we are giving medications for a condition that can be controlled, not cured. As our experience with treating hypertension and other chronic diseases shows, patients are prone to noncompliance when they must continually take medications that do not cure or make them feel any different. With our hypertension patients, we need constant reinforcement to obtain compliance, and the situation with HIV is far more prone to noncompliance. Second, the patients are returning to an environment that is in denial of the disease and has great sway over the patient. These are his family and friends who have played such a vital role in his life, and they are pushing him not to comply with treatments that seem foreign to them. Third, we know that any message we deliver is retained only in part. My understanding of physiology came after repeated efforts to master the material. Patients told in one session that they have a lifelong disease that can be controlled but not cured will retain little. I think that multiple visits shortly after making the diagnosis and repeated counseling to ensure basic understanding is essential to make any significant progress in long term compliance. The need for multiple counseling encounters should be balanced with the desire for privacy, but I am convinced that one such encounter is insufficient.
When I came to southern Sudan in 2009 (prior to their independence), the incidence of HIV was low. The probable reason was inaccessibility. The civil war with Khartoum ended in 2005 with the signing of the Comprehensive Peace Agreement (CPA). Until that time, no trucks would venture into southern Sudan; the economic risks were too great. With peace came the lories; with the lories came the truck stops. With the truck stops came the prostitutes and HIV. War and inaccessibility made South Sudan different. During my years in the Nuba Mountains (2016-20) the incidence of HIV remained low because it was an active war zone and the threat of war still looms in that region, though officially under cease-fire.
In the 1990’s Uganda had the highest incidence of HIV in the world, estimated to be between 18-30%. When we consider those populations of low incidence, such as the elderly, that number suggests that close to 50% of “reproductive age” adults had HIV. But when effective medications became available through PEPFAR, the stigma changed. Instead of despising all patients with HIV, the shame was placed on those who refused to get tested and those who had the disease and refused to take their medication. That was particularly true for pregnant women who have a 70% likelihood of transmitting the disease to their child if they refuse to take their medications, but less than 1% chance if they take their anti-retroviral therapy (ART). With that kind of attitude and having effective medications available, Uganda became the only country in sub-Saharan Africa with a declining HIV rate, now estimated to be around 6.5%.
The greatest task for clinicians in South Sudan, and probably much of sub-Saharan Africa, with respect to HIV is changing the stigmata, as the people of Uganda were able to do. Condoms have been promoted by first world interventionists, but they carry their own stigma. Most couples in South Sudan refuse to use them, even as a contraceptive, because they associate condoms with HIV; condom use signaled mistrust. Though the mistrust is probably appropriate, the association makes condoms unfeasible. PEPFAR must be secure and supply lines for medicines and testing need to improve, but what is really needed in South Sudan is to change the stigma. The best way for us achieve the goal of stopping the spread of the disease is to change the attitude toward testing and taking the medications. That is particularly true for pregnant women. We know from experience in encouraging women to have their babies in the hospital that little things carry weight. Perhaps we should start giving out shirts saying, “I’ve been tested…and proven” and another saying, “I’m intelligent…and compliant.” South Sudanese love clothing from the west. If we can change the shame factor and somehow change compliance with medications into a virtue, we will greatly slow the spread of the disease and extend the lives of those who have HIV.
Mycobacterium Tuberculosis (MTB)—the consumptive killer
Anyone who has read Jane Austen or other writers from the 18th and 19th century is familiar with consumption, the old name for MTB. MTB dominated the respiratory problems until the mid-20th century. The American Thoracic Society (ATS), the largest society of pulmonologists/respirologists in the world, was formerly called the American Tuberculosis Society. Only as MTB has receded that we discovered COPD (chronic obstructive pulmonary disease) and CF (cystic fibrosis). In the US, MTB is virtually confined to immigrants and patients with HIV, but in Africa, MTB remains a dangerous and frequent killer.
The relationship between HIV and MTB is synergistic. In South Sudan and in much of sub-Saharan Africa, primary TB is ubiquitous. In virtually every CXR I have seen, perihilar nodes are prominent. The BCG vaccine is given widely, but the noted benefit of BCG is to prevent TB meningitis, miliary TB or other overwhelming forms disease in the first years of life. It does not reduce the incidence of primary (latent) TB, or at least it has not been demonstrated to reduce the risk for active TB. In primary TB, the tubercles enter the body, either through inhalation or consumption of contaminated milk. Pasteurization is non-existent, as is testing of cattle. The illness in primary TB is short-lived and indistinguishable from a viral illness. A few hundred of the organisms are deposited and encased, virtually incarcerated. The body’s immune system keeps them at bay. The cells most clearly involved in that process are CD4 lymphocytes, which are the targets of HIV. As immunity wanes, from HIV or other causes, the dormant organisms revive and proliferate.
When their numbers are in the 10’s of billions, we have reactivation disease, or what we call TB. People living with HIV are 16 (uncertainty interval 14–18) times more likely to fall ill with TB disease than people without HIV. TB is the leading cause of death among people with HIV. HIV and TB form a lethal combination, each speeding the other’s progress. In 2023, about 161,000 people died of HIV-associated TB. The percentage of notified TB patients who had a documented HIV test result in 2023 was 80%, this was the same level as in 2022, but up from 76% in 2021. The WHO African Region has the highest burden of HIV-associated TB. Overall, in 2023, only 56% of TB patients known to be living with HIV were on antiretroviral therapy (ART).
Why has TB been virtually eliminated from the first world, but remains rampant in Africa? To a large degree, MTB is a disease of poverty and overcrowding. Streptomycin, discovered in the 1947, and Isoniazid (INH), discovered in the 1950’s, impacted the world as greatly as penicillin, but the incidence of MTB had been declining steadily before the discovery of INH because of improved living conditions. The currently accepted international definition of poverty is per capita income of less than $2/day; upwards of 80% of South Sudanese would qualify by that definition, including many of the workers in our hospitals. Poverty is widespread throughout sub-Saharan Africa, and development is desperately needed, though hampered by the widespread political upheaval, war, and random destruction.
But the medical community must share the blame. Part of the problem is the lack of sufficient drugs. In MIH, we often have a diagnosis and prescribed medications, but the patients who are willing to comply are often thwarted by a lack of drugs either at our center or the TB center closer to their home. Often the supply chain is the victim of bureaucratic incompetence and results in partially treated patients who then require a longer course of therapy and raise the risk for resistance.
Clinicians are also to blame. We regularly receive patients with prolonged cough and a MUAC (mid-upper arm circumference) of < 24 cm (indicating malnutrition) who are eventually referred to us after being treated multiple times with various antibiotics, most commonly Amoxicillin and Ceftriaxone. How a clinician in South Sudan could fail to consider TB in that setting is beyond me, but for many, there is little support in making the diagnosis. The majority of TB administrators will only treat if MTB is confirmed by sputum analysis, but we know that AFB (acid-fast bacillus) is only found in sputum about 40% of the pulmonary TB. Confirmation is rare in extra-pulmonary TB, which accounts for 30-40% of all TB. Many facilities in South Sudan do not have x-ray capability. The ability to culture MTB is widely available in the first world, where MTB is rare, but is not available at all in South Sudan and in most of sub-Saharan Africa, where the predominant burden of the disease lies.
In MIH, we have the ability to take CXR, a major boon for diagnosing pulmonary TB. But much of our success has been a willingness to look for and diagnose the disease. If someone has never been treated for TB, but has a prolonged cough, a low MUAC, an elevated ESR (erythrocyte sedimentation rate), and multiple suspicious findings on CXR, we treat for MTB even if the sputum is negative. If the above patient has localized accentuated breath sounds and crackles, we do not even wait for the CXR, which can only be obtained on Tuesdays and Thursdays. Have we made some overcalls? I am sure that we have, but false negative patients (with the disease, but not diagnosed) are a far greater risk to our country.
MTB can infect all areas of the body except for nails, teeth, and hair, but extra-pulmonary TB (ETB) is much more difficult to diagnose. Our experience was that a quarter of our patients with MTB had ETB. One of the areas that has surprised me its incidence is TB pericarditis. Even though MTB is virtually eliminated from the first world, MTB is still recognized as the leading cause of pericarditis in sub-Saharan Africa. In a recent study in consecutive patients with pulmonary TB, 47% of the patients had signs of pericardial involvement at enrollment, and that dropped to 16% after 6 months of anti-TB therapy. Again, a high index of suspicion is needed to make the diagnosis, because there is little chance of direct confirmation. The signs are non-specific. Chest pain that increases as the patient lies down suggests the disease, but chest pain is such a common problem worldwide, and much of it is from relatively benign diseases such as GERD (gastro-esophageal reflux disease). Congestive cardiac failure (CCF) is also common in South Sudan, but coronary heart disease is not. In my experience in South Sudan, there are a few patients with CCF because of uncontrolled hypertension (HTN).
Rheumatic carditis is common, but typically has a mitral insufficiency murmur (blowing murmur at the apex of the heart), sometimes with accompanying mitral stenosis (a change in the murmur at the end, early diastole), or it has Aortic valvular disease with the harsh murmur at the left sternal border in Aortic stenosis or the extremely low diastolic pressures from Aortic insufficiency. In the absence of HTN and valvular murmurs, the patient with a large, congestive liver, particularly when accompanied by a low MUAC and high ESR, TB pericarditis should be suspected.
It is my contention that the surprisingly high incidence of TB pericarditis is because we have better diagnostic tools today than when MTB was a serious concern in the first world. Modern ultrasound can probably detect physiologic pericardial fluid, but a large effusion strongly suggests TB. A CXR showing a “water bottle heart” is about as strongly confirmatory as is possible, but a large heart otherwise not explained with a significant effusion by ultrasound is strongly suggestive. A high ESR also adds to the confidence of the diagnosis.
My experience suggests that Rheumatic carditis and TB are seen together more often than would be anticipated by their relative frequency of the diseases. The link between MTB and HIV is obvious and can be explained by basic science. The decline in CD4 lymphocytes allows for reactivation of dormant MTB. There is no such basic science support for the link between Rheumatic fever and TB, but epidemiology helps. Streptococcal pharyngitis is still a common disease in the US, perhaps more so than previously, but we have never identified the “rheumatogenic strains” of Strept. Although ARF was the leading cause of mortality in 5- to 20-year-olds in the United States during the 1920s, the annual incidence of ARF in the United States today is low (<2 cases per 100 000 school-aged children compared with up to 150 cases per 100 000 worldwide). Finally, the complexity of the immune response to TB suggests a possible mechanism for the association. After 150 years or so of seeking for a vaccine for TB, we have the relatively ineffective BCG and little else to show for our efforts.
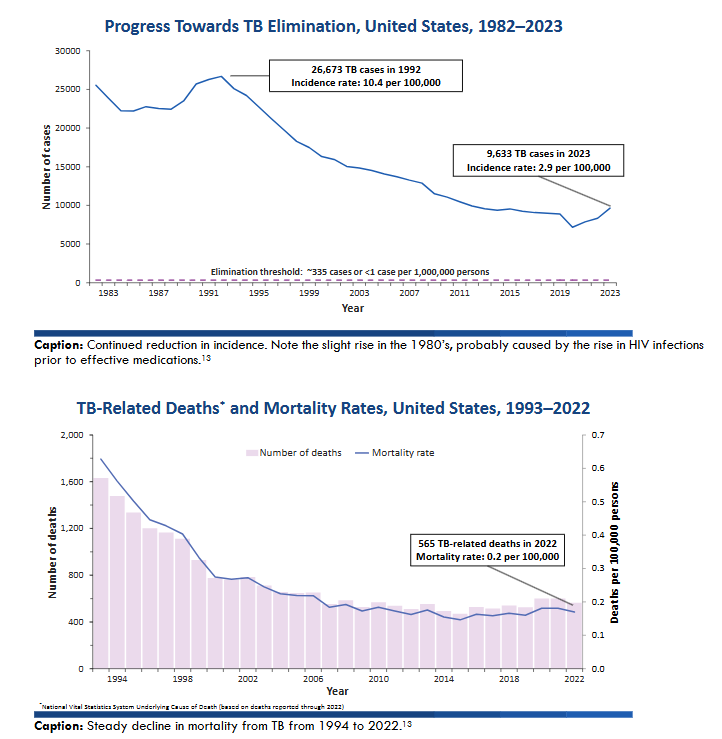
Progress Towards TB Elimination, United States, 1982–2023
| Year | Number of cases |
|---|---|
| 1983 | 25000 |
| 1987 | 20000 |
| 1991 | 15000 |
| 1995 | 10000 |
| 1999 | 5000 |
| 2003 | 3000 |
| 2007 | 2000 |
| 2011 | 1500 |
| 2015 | 1000 |
| 2019 | 500 |
| 2023 | 300 |
Though there are a number of controversies that could be covered, I want to address the use of Streptomycin. As it sounds, Streptomycin is an aminoglycoside and the first effective drug against TB. Though Streptomycin and INH made an immediate impact, it quickly became apparent that MTB has a strong propensity for resistance. It has been estimated that in completely naïve MTB, there exist a resistant organism for certain of the TB drugs when there is more than 1010 organisms. The holy grail of TB therapy has been the use of multiple effective drugs simultaneously. In South Sudan, we have RHZE (Rifampin, Isoniazid, Pyrazinamide, and Ethambutol) in a single pill. We give multiple pills based on weight. We have recently had introduced GenXprobe. It is a machine capable of carrying out PCR (polymerase chain reaction) on numerous samples looking for either HIV or TB. It was also useful during the Covid-19 pandemic. It can also detect a certain gene leading to resistance to Rifampin, and that gene is associated with MDRTB (multiple drug resistant TB). MDRTB patients are typically resistant to Streptomycin, at least in first world countries. This led to the ill-advised ban on Streptomycin in South Sudan. In the various hospitals where I have worked, we used Streptomycin for two months and RHZE for three months followed by RH for 5 months for relapse of TB and treatment failure. We had a high degree of success. It appears that the ban has finally (perhaps not officially) been lifted and we have used it effectively multiple times in the last three months. MDRTB is primarily a first world problem. It was driven by patients who came to receive free medications, quit them when they felt better, and then returned when they fell sick again. In Mapuordit, we have identified perhaps five patients with MDRTB, but there have been scores of patients who relapsed or failed therapy and then benefited from Streptomycin.
That brings up another clinical dilemma facing South Sudanese clinicians—deciding whether the patient has a relapse of his TB, or is suffering from bronchiectasis, a common complication of TB, particularly if there was a delay in seeking therapy. Bronchiectasis is when an airway is damaged to the degree that it loses its structural integrity. Bronchoscopically, it looks like airway collapse, but at autopsy, if paraffin is poured into the airways, the bronchiectatic airways are dilated, almost sack like in severe cases. MTB and measles are two of the leading causes of bronchiectasis. In a TB patient who has completed therapy and symptoms return, deciding the culprit is difficult. If there is wheezing and course crackles, bronchiectasis is more likely. A normal to modestly high ESR helps. A trial of bronchodilators, steroids, and co-trimoxazole may also be helpful. Cachexia, high ESR, and hemoptysis point to a relapse. Positive sputum makes the diagnosis easy, but CXR is often not helpful, since old films are virtually never available, and deciding what is from previous infection versus a new TB reactivation is difficult.
Another difficult questions to answer concerning a patient with a second TB infection is whether this was a relapse (the old organism was not completely eliminated) versus re-infection (picked up from another patient). Is this the same organism that made her sick before, or is this newly acquired infection? That brings up a final controversy (for this article) regarding the length of therapy. In the late 1980’s, when I was a pulmonary fellow, there was a series of articles detailing the depth of care and investigation that went in to the decision to change the recommended length of treatment for MTB from 12 to 9 months. The conclusion of those investigations was that 9 months of therapy was as efficacious as 12 months. Of course, at that point, TB was still a significant problem in the US with an incidence of 10.4/100,000. It appears to me that the decision to change to 6 months therapy was based upon compromises—the costs versus the efficacy. The expected outcome was 85% cure rate. Perhaps the compromises were needed, but I ask “Would we accept a 15% failure rate for the most deadly single infectious agent in the world if TB was still a major problem in Europe and the US?” I think the obvious answer is no. Why not extend the phase of more intense therapy? In patients who are Hepatitis B surface antigen (+) or evidence of liver dysfunction, there is some danger of Pyrazinamide-induced liver disease, but is that risk sufficient to condemn everyone in the third world to a 15% failure rate? By my observations, there appear to be too many relapses, even in patients compliant for the six month duration. Insufficient killing of the MTB is a setup for MDRTB.
Malaria—vector-borne death
One of the keys to the building of the Panama Canal was the recognition that mosquitoes bore both malaria and yellow fever, and eradication of mosquito breeding areas helped the project immensely. Yellow fever was dangerous and killed many, but malaria was far more problematic to that venture and, indeed, to all of history. One of the great controversies of the latter 19th century was whether malaria was vector-borne or caused by “bad air,” as the name implies. By eradicating the breeding places of mosquitoes, Reed and Goethals eradicated malaria and gave the needed proof that mosquitoes carried the pathogen.
Potomac Fever now means political intrigue in the US capital, but it used to mean malaria. The land around Washington, DC was swampy and the politicians left the capital during summer to move to higher ground and better air. In his book The Mosquito: A Human History of Our Deadliest Predator, Timothy Winegard convincingly shows how much of Western Civilization has been shaped by malaria and the fear of swamp fever. Hannibal marched his troops and elephants over the Alps, but he could not attack Rome because of the mosquito infested swamp around the city, so he dared not set up a siege. Alexander the Great withdrew from his planned attack on India because malaria ravaged his troops. In the battle of Vicksburg in the American Civil War, General Grant was able to purchase all the quinine available, so malaria decimated the southern troops.
The war against malaria in America was won by draining swamps and DDT. By eliminating the Anopheles mosquito, malaria was eradicated in most of the world, but that is not true in Africa. In South Sudan, there are expected to be over 8.3 million cases out of 12 million inhabitants in 2025. Like TB, the immune response to malaria is complex, probably because the Plasmodia responsible have the ability to change the antigens on their surface. If a patient has Streptococcus pneumoniae infection, six weeks later, the IgM is just beginning to wane and the IgG is increasing, so another attempted attack is easily thwarted. Not so in malaria. In my first few years in South Sudan, I was amazed to see children come in with 40C fever, large spleens, and verified Plasmodium falciparum (PF) infection six weeks after we had successfully treated them for the same problem. It is one of the aspects of this disease that made it so difficult to come up with a vaccine, though there appears to be an effective vaccine on the horizon. WHO maintains that the two vaccines are equally efficacious, though the initial studies suggest that the R21 vaccine is more efficacious. A new vaccine, PfSPZ-LARC, is estimated to have over 90% efficacy, though that is yet to be proven in clinical trials.
Like all bullies, malaria picks on the vulnerable. Most of the 600,000 deaths attributed to malaria every year in Africa occur in pregnant women and small children. Malaria is the most common cause of abortion (miscarriage) in South Sudan, though PID (pelvic inflammatory disease) gives it good competition. PF is the most dangerous and destructive species of the Plasmodia that cause malaria, but there are others. P ovale, P vivex, and P malaria are more common in western Africa, but as we demonstrated, are not infrequent causes of malaria in South Sudan and East Africa.
As the world experienced during the Covid-19 pandemic, controversies and poor medical practices abound when ignorance reigns. One of the most common misconceptions in South Sudan regarding malaria is an assumed synergistic association with Typhoid fever. South Sudanese frequently speak of having “malaria-Typhoid” as though it was one disease. Much of that confusion arises because of the widespread use of the Widal test. The Widal test is a fairly nonspecific marker of inflammation that, in the context of the typical signs and symptoms of Typhoid fever (infection by Salmonella typhi), added some assurance to the diagnosis. It was never intended to be a standalone test for Typhoid fever, and has been superseded by far better tests. It has been demonstrated to be neither sensitive nor specific. Epidemiology strongly repudiates the association. Typhoid fever is predominantly an Asian disease with about 80% of the documented cases, using blood cultures or IgM measurements, occurring in India. Malaria is predominantly an African disease, with again about 80% of the documented cases occurring in sub-Saharan Africa. If there was a synergistic relationship between malaria and Typhoid, we would expect an endemic area for one would be endemic for the other, as it is with HIV and TB, but in fact, that is not the case.
More specific tests for Salmonella typhi also repudiate the association. In a study looking at the seeming association, Widal tests for patients with documented malaria were often positive, but more specific tests for Salmonella typhi were negative and authors conclude, rightly I think, that the Widal tests in that context were false positives. Looking at blood cultures for S. typhi, the simultaneous infections were exceedingly rare, and the overlap no greater than would be expected randomly. We used IgM antibodies to look for S. typhi infection at the same time that we did blood films for malaria. Whereas over 75% of the blood films of these acutely ill patients were (+) for PF or other species of plasmodia, less than 5% of the patients tested had objective evidence of S. typhi infection. In truth, much of propagation of the “malaria-Typhoid” association has been by unscrupulous medical providers in South Sudan who want to sell Ceftriaxone when they treat for malaria, but the practice has no basis in science and needs to end.
A second controversy involves the best test for malaria diagnosis. Thick and thin blood smears have been the gold standard for the diagnosis of malaria, and it has the added advantage of being able to identify the various species of plasmodia. The RDT (rapid diagnostic test) for malaria has, to a substantial degree, replaced the blood film. It is a simple western blot, with antibodies to expected antigens embedded in the strip and shows up (+) if that antigen is present. It is fast and easily taught, requiring no special equipment and no particular methodological expertise. It also has the advantage (or disadvantage) of remaining positive for about 2 weeks after anti-malarial medications have been given. It is inexpensive by first world standards, costing around $1/test, but in MIH in Mapuordit, that can easily add up to over $1000/week during malaria season, and that is a substantial burden for South Sudanese hospitals. Blood films require both a microscope and some technical expertise, and are more labor intensive, but they are much cheaper, more sensitive, and in the case of recently treated patients, more selective—if the plasmodia have been eliminated by effective treatment, the blood film will be negative. If a treatment failure is suspected, positive blood film confirms the suspicion, whereas RDT may still be (+) after successful eradication of the illness.
There are two major problems with RDT for malaria. One is that many of the tests are specific for PF only. If the malaria is caused by other species, those RDT will miss it. The data from the Yei study suggests about 15% of cases of malaria in South Sudan are caused by plasmodia other than falciparum. The other concern has emerged recently and is probably due to the plasmodia’s ability to alter its surface antigens. In our study from Yei, South Sudan, during the peak season for malaria, 84% of the blood films were (+), whereas only 60% of the RDT’s were positive during the same period. In a study out of Ethiopia, including parts of South Sudan, the authors showed that RDT’s missed approximately 10% of those plasmodia infections that were detected by blood films. Though quite different designs, the two studies suggest that RDT for malaria is substantially less sensitive than blood films.
One of my first assignments upon arriving in South Sudan in 2009 was to dump the contents of all our supplies of Chloroquine into the latrine. Chloroquine had been the backbone of therapy for malaria for decades, but the plasmodia had grown resistant, so the government outlawed Chloroquine. If Chloroquine was found in our pharmacy, we would have been fined.
That vignette leads to another controversy: what is the best drug for acute severe malaria? Quinine has been an effective treatment of malaria for centuries. But early in the 21st century, Artesunate was introduced for cerebral malaria and the results were overwhelming. This chart comes from a review in 2012.
There was a new sheriff in town. Two predictable things occurred. One was that the use of Artesunate shot up dramatically. The other was that clinicians were not particularly diligent in making sure Artesunate was followed up with ACT (Artemisin Combination Therapy—a group of medications that are artemisin based, but that have another drug working through a different, complementary mechanism). Artesunate rapidly cleared plasmodia from the blood, but is not as effective in eradicating the organisms completely. ACT provided a deterrent to resistance. The next predictable thing was that sensitivity to Artesunate declined. A study in Lancet of over 5000 patients enrolled from 2005-10 showed a significant difference in mortality, but much smaller than previous studies. In a study where data was collected from 2008 to 2012, Artesunate again as the preferable drug, but there was no statistically significant difference, though admittedly the numbers were small (92). A bench study showed emergence of Artesunate resistance with the potential for absolute resistance. By 2021, we started seeing Artesunate treatment failures, but almost never in patients treated with Quinine. Now Uganda Clinical Guidelines has both Quinine and Artesunate as first line drugs for severe malaria.
In dosing Quinine, I used the same method we used in Gidel, Nuba Mountains, Sudan. We did not use a loading dose. Though a loading dose does result in quicker clearance of the plasmodia, it greatly increases the complexity of the regimen, and that is a significant concern in African hospitals that are often inundated with patients during the rainy (malaria) season. In MIH in Mapuordit, the children’s ward often quadruples in size and becomes the most highly populated ward in the hospital. Moreover, electronic monitoring does not exist and the acuity of patients is high. Therefore, the more complex the regimen is, the greater the chance for errors. The Gidel method takes advantage of concentration of IV quinine and the desired daily dose. Quinine comes in a 2 ml vial, 300 mg/ml. The desired dosing is 30 mg/kg/day. Therefore, to calculate the ml of Quinine to put into the drip, multiply the weight in kg by 0.1. Put more simply, shift the decimal point to the left one space. We give that dose over 16 hours. A 36-kilogram woman gets 3.6 ml of Quinine (1080 mg) into a litter of dextrose over 16 hours. If there are no liter bags of Dextrose, split the dose into 1.8 ml in 500 ml Dextrose over 8 hours times two. For children 20 kg and below, I use 500 ml of Dextrose over 16 hours. The simplicity of calculations makes it easier for Clinical officers to properly dose, and makes it easier for nurses to pick up any errors of calculation. Following the Quinine drip, if the patient is substantially recovered, I switch to ACT if the patient has not taken a full course (3 days) in the last month.
Which leads into the final controversy, the ultimate solution. Why does Potomac Fever mean political intrigue and indoctrination instead of malaria? Why is the study of malaria more of an academic than practical concern in the first world? Because Anopheles mosquitoes have been virtually eliminated in America and Europe without causing any notable environmental concerns. Why not in Africa? Because Africa is the dark, and in many ways, forgotten continent, so attempts to clear the breeding grounds of the Anopheles mosquito were late coming. Then DDT was banned, based upon data of environmental impact. Some experts have reviewed the data upon which those recommendations were made and found means of using DDT with greater safety, but in fact, there is no political will to resume DDT use in Africa. In a letter to the editor of South Sudan Med J, I maintain that if there was a disease resulting in the death of 50,000 American or European children every year, there would be a tremendous outcry for a solution, and there would be little patience. There is a potential solution, but it is controversial. CRISPR technology has been used to insert functional genes into the genome of patients born with dysfunctional genes. The results have not been overwhelming, but they have been positive. Experience suggests that the downside of CRISPR technology is the inserted gene tends to lose expression over time. Scientists have come up with a gene that could be inserted into the DNA of male Anopheles mosquitoes that does not harm males but is fatal for female embryos. As we know, it is the female mosquito that bites. The result would be more and more males carrying the lethal gene and seeking to breed with fewer and fewer females until Anopheles mosquitoes became extinct. A more likely scenario, given our experience with CRISPR, is that for several years, there would need to be reintroduction of the gene because of the likelihood of reduced expression over time. It would also need introduction in multiple areas because all of sub-Saharan Africa is infested with the Anopheles mosquito. Environmental advocates have held up this potential solution by stating, quite truthfully, that once the gene is released, we will not be able to withdraw it. But the chance that the inserted gene could somehow cross over into other mosquito species, much less other animals, is miniscule. The far more likely problem is that reintroduction of the gene will need to be frequent and widespread, and will present its own technical challenges. Still, it is probably the most reliable possibility of ridding Africa of the Anopheles mosquito.
Do we need newer and more effective drugs for malaria? Absolutely. Do we need better vaccines for malaria? Of course we do. But the solution of the dilemma in the building of the Panama Canal and elimination of malaria from Washington, DC involved the elimination of Anopheles mosquitoes, and ultimately that will be the case in South Sudan and Africa. And Africa without malaria might surprise the world.
Conclusion:
The reasons for the persistence of HIV, TB, and malaria as the leading causes of death in Africa are multi-factorial. Regarding HIV, PEPFAR has been one of the most successful intervention programs from the US or Europe, but other attempted interventions have not fared as well, and improvements in PEPFAR’s success will depend on interventions that understand the attitudes of the various tribes and peoples. With respect to TB, elimination will probably require improved living conditions, but better diagnostic measures, more reliable means of supplying drugs, and intervention programs aimed at better than 85% cure rates are more readily attainable goals. To reduce the morbidity and mortality from malaria will require new vaccines, more effective drugs, but ultimately needs eradication of Anopheles mosquitoes.
References
- Nachega JB, Serwadda D, Abimiku A, Sikazwe I, Karim QA. PEPFAR at 20 — A Game-Changing Impact on HIV in Africa. Published July 1, 2023 N Engl J Med 2023;389:1-4. https://orcid.org/0000-0002-0985-477X DOI: 10.1056/NEJMp2304600.
- Vithalani J, Herreros-Villanueva M. HIV Epidemiology in Uganda: survey based on age, gender, number of sexual partners and frequency of testing. Afr Health Sci. 2018 Sep;18(3):523-530. doi: 10.4314/ahs.v18i3.8. PMID: 30602983; PMCID: PMC6307011.
- Setiabudiawan TP, Reurink RK, Hill PC, Netea MG, van Crevel R, Koeken VACM, Protection against tuberculosis by Bacillus Calmette-Guérin (BCG) vaccination: A historical perspective, Med, Volume 3, Issue 1, 2022, Pages 6-24, ISSN 2666-6340, https://doi.org/10.1016/j.medj.2021.11.006.
- WHO bulletin on Tuberculosis March 2025.
- Snider GL. Tuberculosis then and now: a personal perspective on the last 50 years. Ann Intern Med. 1997 Feb 1;126(3):237-43. doi: 10.7326/0003-4819-126-3-199702010-00011. PMID: 9027277.
- Apiu GK, Sala L, Iannetti R, McIntosh JC. One year’s experience of extra-pulmonary TB in a county/regional hospital in South Sudan. South Sudan Medical Journal 2022;15(1):12-15
- Daryoush Samim, Guy Muula, Nicolas Banholzer, Douglas Chibomba, Sihle Xulu, Carolyn Bolton, Denise Evans, Lisa Perrig, Stefano De Marchi, Gunar Günther, Matthias Egger, Thomas Pilgrim, Lukas Fenner, Cardiovascular Involvement in Tuberculosis Patients Treated in Southern Africa, JACC: Advances, Volume 4, Issue 1, 2025, 101427, ISSN 2772-963X, https://doi.org/10.1016/j.jacadv.2024.101427.
- Barnett TC, Bowen AC, Carapetis JR. The fall and rise of Group A Streptococcus diseases. Epidemiol Infect. 2018 Aug 15;147:e4. doi: 10.1017/S0950268818002285. PMID: 30109840; PMCID: PMC6518539.
- Kaplan EL, Markowitz M. The fall and rise of rheumatic fever in the United States: a commentary. Internat J Cardio. 1988; 21 (1): 3-10 ISSN 0167-5273
- Liang Y, Yu D, Lu Q, Zheng Y, Yang Y. The rise and fall of acute rheumatic fever and rheumatic heart disease: a mini review. Front Cardiovasc Med. 2023 May 23;10:1183606. doi: 10.3389/fcvm.2023.1183606. PMID: 37288267; PMCID: PMC10242100.
- de Loizaga SR, et al. Rheumatic Heart Disease in the US: Forgotten but not Gone. Results of a 10 Year Multicenter Study. Am J Heart Assoc. 2021 17 (10): 1-12.
- The Decline of Tuberculosis Mortality in the United States and the Influence of the Influenza Epidemic of 1918. American Review of Tuberculosis, 1925; 13(4), pp. 385–391
- Tuberculosis (TB) Disease in the United States 1993–2023 (CDC slide set, July 2024) Division of Tuberculosis Elimination, National Center for HIV, Viral Hepatitis, STD, and TB Prevention National Tuberculosis Surveillance System
- Tuberculosis Treatment Shortening, Singh V Drug Discovery Today, 2024: 29 (5) 1-8.
- Treatment of Tuberculosis Guidelines for National Programmes, WHO 2003.
- Malaria vaccines (RTS,S and R21) WHO bulletin 8 April 2025
- Mawazo A, Bwire GM, Matee MIN. Performance of Widal test and stool culture in the diagnosis of typhoid fever among suspected patients in Dar es Salaam, Tanzania. BMC Res Notes. 2019 Jun 5;12(1):316. doi: 10.1186/s13104-019-4340-y. PMID: 31167646; PMCID: PMC6551910.
- Goswami D, Patel H, Betz W, Armstrong J, Camargo N, Patil A, Chakravarty S, Murphy SC, Sim BKL, Vaughan AM, Hoffman SL, Kappe SH. A replication competent Plasmodium falciparum parasite completely attenuated by dual gene deletion. EMBO Mol Med. 2024 Apr;16(4):723-754. doi: 10.1038/s44321-024-00057-7. Epub 2024 Mar 21. PMID: 38514791; PMCID: PMC11018819.
- Berry, A.A., Richie, T.L., Church, L.W.P. et al. Safety, tolerability and immunogenicity of a condensed, multi-dose prime regimen of PfSPZ Vaccine for the prevention of Plasmodium falciparum malaria infection. Malar J 24, 88 (2025). https://doi.org/10.1186/s12936-025-05299-5
- Malaria in South Sudan—Past, Present, and Future, WHO bulletin March 2025.
- Obonyoh K, James A, McIntosh JC. A review of one year malaria blood film data from a hospital in Yei, South Sudan, South Sudan Medical Journal, 2024;17(4):198-1201 © 2024 The Author (s) License: This is an open access article under CC BY-NC Doi: https://dx.doi.org/10.4314/ssmj.v17i4.9
- Feleke, S.M., Reichert, E.N., Mohammed, H. et al. Plasmodium falciparum is evolving to escape malaria rapid diagnostic tests in Ethiopia. Nat Microbiol 6, 1289–1299 (2021). https://doi.org/10.1038/s41564-021-00962-4
- Sinclair D, Donegan S, Isba R, Lalloo DG. Artesunate versus quinine for treating severe malaria. Cochrane Database Syst Rev. 2012 Jun 13;2012(6):CD005967. doi: 10.1002/14651858.CD005967.pub4. PMID: 22696354; PMCID: PMC6532684.
- Dondorp AM, Fanello CI, Hendriksen IC, Gomes E, Seni A, Chhaganlal KD, Bojang K, Olaosebikan R, Anunobi N, Maitland K, Kivaya E, Agbenyega T, Nguah SB, Evans J, Gesase S, Kahabuka C, Mtove G, Nadjm B, Deen J, Mwanga-Amumpaire J, Nansumba M, Karema C, Umulisa N, Uwimana A, Mokuolu OA, Adedoyin OT, Johnson WB, Tshefu AK, Onyamboko MA, Sakulthaew T, Ngum WP, Silamut K, Stepniewska K, Woodrow CJ, Bethell D, Wills B, Oneko M, Peto TE, von Seidlein L, Day NP, White NJ; AQUAMAT group. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet. 2010 Nov 13;376(9753):1647-57. doi: 10.1016/S0140-6736(10)61924-1. Epub 2010 Nov 7. Erratum in: Lancet. 2011 Jan 8;377(9760):126. PMID: 21062666; PMCID: PMC3033534.
- Mathiba RM, Mathivha LR, Nethathe GD. Artesunate compared with quinine for the treatment of severe malaria in adult patients managed in an intensive care unit: a retrospective observational study. S Afr J Crit Care 2019: 35 (1): 13-18.
- Samal KK, Sahu CS. Malaria and Widal reaction. J Assoc Physicians India. 1991;10:745–747.
- Tyagi RK, Gleeson PJ, Arnold L, Tahar R, Prieur E, Decosterd L, Pérignon JL, Olliaro P, Druilhe P. High-level artemisinin-resistance with quinine co-resistance emerges in P. falciparum malaria under in vivo artesunate pressure. BMC Med. 2018 Oct 1;16(1):181. doi: 10.1186/s12916-018-1156-x. PMID: 30269689; PMCID: PMC6166299.
- Lesi AFE, Meremikwu MM. High dose Quinine regime for treating severe malaria. Cochrane Database of Systemic Reviews 2004 (3) Art No: Cd003341. Doi:10:1002/14651858. SD003341.pub2
- Mansouri, A., Cregut, M., Abbes, C. et al. The Environmental Issues of DDT Pollution and Bioremediation: a Multidisciplinary Review. Appl Biochem Biotechnol 181,309–339 (2017). https://doi.org/10.1007/s12010-016-2214-5
- McIntosh JC Letter to the Editor: Concerns about malaria in South Sudan South Sudan Med J; 2023; 16: 79
- The Mosquito: A Human History of Our Deadliest Predator. Timothy C. Winegard. Dutton, Penguin Random House, New York, NY, USA, 2019. ISBN (hardcover): 9781524743413; ISBN (ebook): 9781524743437; ISBN (export): 9781524745608




