Effects of Exercise on Iron Transfer in the Body
Effects of Exercise on Iron Transfer in the Body
Ryunosuke Takahashi¹ and Takako Fujii²
- The Institute of Health and Sports Science, Chuo University, Tokyo, Japan
- Department of Sports and Medical Science, Graduate School of Emergency Medical System, Kokushikan University, Tokyo, Japan.
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Takahashi, R. and Fujii, T., 2025. Effects of Exercise on Iron Transfer in the Body. Medical Research Archives, Ioneline 13(6). https://doi.org/10.18103/mra.v13i6.6673
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6673
ISSN 2375-1924
ABSTRACT
Although iron is found in trace amounts in the body, it plays an important role in oxygen transport and energy metabolism. Iron is therefore very important for athletes with high oxygen requirements, especially endurance athletes. Despite its importance, many athletes are diagnosed with iron deficiency. The relationship between iron regulation and exercise has been a focus of research, as it has been suggested that athletic performance is related to iron regulation. In particular, the discovery of hepcidin, an iron-regulating hormone synthesised in the liver, has received much attention. Hepcidin is known to inhibit iron absorption from the intestinal tract and promote iron excretion, and is closely related to iron nutritional status in vivo. In addition, the inflammatory cytokine interleukin-6 (IL-6) has been found to be an inducer of hepcidin expression. Much research has been conducted on the effects of training and diet (nutrients) on iron status in athletes. However, the detailed mechanisms of exercise-induced iron deficiency in athletes remain unclear. This paper summarizes the less commonly reported effects of exercise on iron movement and diet on iron status.
In particular, we will focus on the impact of exercise on iron levels in the body, and the impact of meal timing and composition on iron levels.
Keywords: Athlete, Exercise, Iron deficiency, Sports nutrition
Introduction
Iron deficiency is one of the most common nutritional problems worldwide. It is defined as insufficient iron reaching the body’s stores or various tissues. Biological iron metabolism is a semi-closed circuit whose basic functions are the absorption, storage, and reuse of iron. However, depending on the imbalance between iron intake, storage and requirements, deficiencies can develop either rapidly or very slowly. This may be due to inadequate iron intake, certain diseases or, in women, menstrual bleeding. The rate at which iron deficiency develops in individual tissues and intracellular organelles also depends on iron recycling within cells and the metabolic turnover rate of iron-containing proteins.
Iron plays a role in many metabolic processes, including electron transfer in mitochondria, neurotransmitter and protein synthesis, and organ formation. Therefore, when iron is depleted in vivo, many biological functions, such as immune function, sympathetic nerve function, endocrine metabolism and thermoregulation, are impaired. Additionally, iron deficiency significantly reduces the content of iron-sulphur and cytochromes in mitochondria, as well as total oxidative capacity. Furthermore, iron deficiency restricts haematopoiesis in haemopoietic tissues, resulting in decreased hemoglobin concentration and iron deficiency anemia. Decreased hemoglobin concentration significantly reduces physical work capacity by decreasing the supply of oxygen to exercising muscles. In other words, iron deficiency reduces exercise capacity due to decreased oxygen-carrying and diffusion capacities in athletic tissues, as well as decreased muscle oxidative capacity⁵.
In a recent human study, women with low serum ferritin and iron deficiency, but normal hemoglobin concentrations, were asked to take iron supplements or a placebo alongside four weeks of aerobic exercise for six weeks. The placebo group showed a decrease in serum ferritin and transferrin saturation, while the iron supplement group showed an increase. Both groups showed improvement in VO₂ max, but the iron supplement group showed greater improvement. These results suggest that tissue iron sufficiency, as well as hemoglobin, is important for increasing maximum oxygen uptake. Many studies have investigated the effects of exercise and nutrition on iron status, and it is well established that exercise itself can alter this. Recently, it has been reported to be associated with the development of symptoms of apathy and negative mood disorders⁷. However, the detailed mechanisms of exercise-induced iron deficiencies in athletes remain unclear. In this paper, we discuss the impact of exercise on iron levels in the body, as well as the influence of meal timing and composition on iron levels.
Regulators of iron in the body
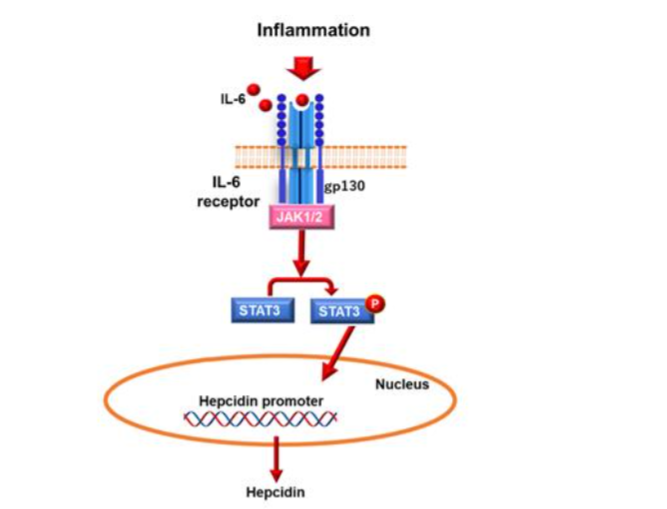
Many studies have been conducted on the effects of training and nutrition (nutrients) on athletes. In particular, hepcidin, an iron-regulating hormone synthesized in the liver, has attracted considerable attention. Hepcidin plays an important role in iron homeostasis⁹. Hepcidin, a peptide hormone, is secreted by hepatocytes¹⁰. The main iron flux of hepcidin is shown in Figure 1¹¹.
(1) HEPCIDIN EXPRESSION
Pigeon et al. found that iron overload causes hepcidin overexpression, suggesting that hepcidin has roles other than antibacterial properties. Hepcidin plays an important role in iron metabolism in the body. The basolateral membrane contains an iron transporter called ferroportin, which removes iron from the cells. Hepcidin regulates iron levels by binding to ferroportin. In other words, hepcidin produced in the liver binds to ferroportin, and moves from the cell membrane to inside the cell, where it is degraded in lysosomes. When iron is not needed, hepcidin levels increase, leading to a decrease in ferroportin and suppression of iron transport. Conversely, when iron is needed, hepcidin expression decreases, allowing ferroportin to promote iron transport. Under normal conditions, blood iron levels are regulated. However, when excess iron is administered, or during inflammation, hepcidin is overproduced, leading to a state of functional iron deficiency where stored iron cannot be utilized. Measuring blood hepcidin concentration is important for determining whether iron metabolism is normal¹².
(3) INFLUENCE OF EXERCISE AND DIET TIMING ON IRON STATUS IN THE BODY
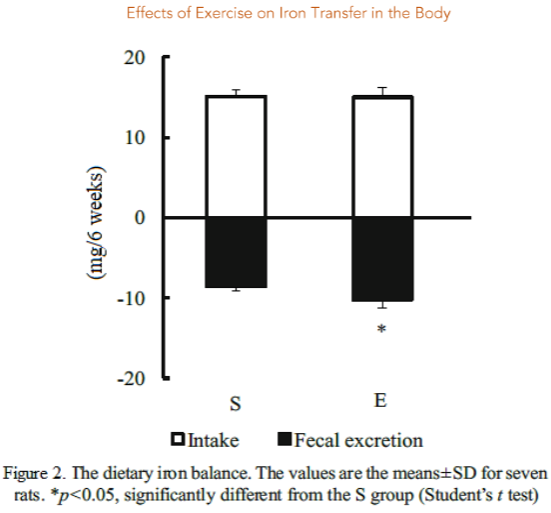
Fujii et al. investigated how meal timing affects the impact of resistance exercise on improving iron nutritional status in iron-deficient rats³⁵. Rats were subjected to resistance exercise and maintained for 3 weeks in two groups: one group received a meal immediately after exercise; the other received a meal 4 h after exercise. The results showed an immediate increase in plasma iron after exercise, with no increase due to food intake. However, both plasma iron levels and bone marrow ALAD activity increased after exercise. Reports indicate that anemic subjects subjected to resistance exercise show a significant increase in hemoglobin concentration in comparison to resting rats³⁶. Plasma iron is reported to be used for hemoglobin synthesis³⁷. The concurrent increase in bone marrow ALAD activity and plasma iron levels suggest that resistance exercise promotes hemoglobin synthesis and increases hemoglobin concentration³³,³⁴.
McCormick et al. found that despite increased hepcidin concentrations after exercise, more iron is absorbed when a meal is eaten after morning exercise than when fasting at rest or from an evening meal³⁸. While the physiological mechanisms promoting iron absorption after exercise remain unclear, this study suggests that overall iron absorption is influenced by cumulative responses to inflammation and hepcidin, in addition to transient physiological changes after exercise. Thus, they recommend that exercisers take or supplement iron immediately after morning exercise to enhance iron absorption. This recommendation is based on the fact that hepcidin levels increase 3 hours after exercise, regardless of whether it is performed in the morning or afternoon³⁹.
Conclusion
In this paper, we present evidence of the impact of exercise and diet on biological iron. Exercise induces an inflammatory response. The iron status of athletes should be monitored regularly throughout their training. It should be noted that exercise promotes iron excretion regardless of the type of training. Early detection of a decrease in biological iron stores may help maintain and improve athletic performance. However, further research is needed to reduce the burden of iron deficiency in athletes.
Acknowledgements:
The authors contributed equally to this work.
Disclosure statement:
No potential conflicts of interest were disclosed.
References:
2. Deldicque L, Francaux M. Recommendations for Healthy Nutrition in Female Endurance Runners: An Update. Front Nutr. 2015;2:17. Published 2015 May 26. doi:10.3389/fnut.2015.00017
3. Ganz T, Nemeth E. Iron metabolism: interactions with normal and disordered erythropoiesis. Cold Spring Harb Perspect Med. 2012;2(5):a011668. doi:10.1101/cshperspect.a011668
4. Varga E, Pap R, Jánosa G, Sipos K, Pandur E. IL-6 Regulates Hepcidin Expression Via the BMP/SMAD Pathway by Altering BMP6, TMPRSS6 and TfR2 Expressions at Normal and Inflammatory Conditions in BV2 Microglia. Neurochem Res. 2021;46(5): 1224-1238. doi:10.1007/s11064-021-03322-0
5. Beard JL, Connor JR. Iron status and neural functioning. Annu Rev Nutr. 2003;23:41-58. doi:10.1146/annurev.nutr.23.020102.075739
6. Sawada T, Konomi A, Yokoi K. Iron deficiency without anemia is associated with anger and fatigue in young Japanese women [published correction appears in Biol Trace Elem Res. 2015 Dec;168(2): 520-1. doi: 10.1007/s12011-015-0531-0.]. Biol Trace Elem Res. 2014;159(1-3):22-31. doi:10.100 7/s12011-014-9963-1
7. Sim M, Garvican-Lewis LA, Cox GR, et al. Iron considerations for the athlete: a narrative review. Eur J Appl Physiol. 2019;119(7):1463-1478. doi:10.1007/s00421-019-04157-y
8. Ganz T. Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation. Blood. 2003;102(3):783-788. doi:10.1182/blood-2003-03-0672
9. Lee PL, Beutler E. Regulation of hepcidin and iron-overload disease. Annu Rev Pathol. 2009;4:489-515. doi:10.1146/annurev.pathol.4.110807.092205
10. Nemeth E, Ganz T. Hepcidin and Iron in Health and Disease. Annu Rev Med. 2023;74:261-277. doi:10.1146/annurev-med-043021-032816
11. Fujii T, Kobayashi K, Kaneko M, Hata K. RGM Family Involved in the Regulation of Hepcidin Expression in Anemia of Chronic Disease. Immuno 20044(3):266-285. DOI: 10.3390/immuno4030017
12. Badenhorst CE, Dawson B, Cox GR, Laarakkers CM, Swinkels DW, Peeling P. Acute dietary carbohydrate manipulation and the subsequent inflammatory and hepcidin responses to exercise. Eur J Appl Physiol. 2015;115(12):2521-2530. doi:10.1007/s00421-015-3252-3
13. Varga E, Pap R, Jánosa G, Sipos K, Pandur E. IL-6 Regulates Hepcidin Expression Via the BMP/ SMAD Pathway by Altering BMP6, TMPRSS6 and TfR2 Expressions at Normal and Inflammatory Conditions in BV2 Microglia. Neurochem Res. 2021; 46(5):1224-1238. doi:10.1007/s11064-021-03322-0
14. Peeling P, Dawson B, Goodman C, et al. Effects of exercise on hepcidin response and iron metabolism during recovery. Int J Sport Nutr Exerc Metab. 2009;19(6):583-597. doi:10.1123/ijsnem.1 9.6.583.
15. Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014;6(10):a016295. Published 2014 Sep 4. doi:10.1101/cshperspect.a016295
16. Solberg A, Reikvam H. Iron Status and Physical Performance in Athletes. Life (Basel). 2023;13(10): 2007. Published 2023 Oct 2. doi:10.3390/life13102007
17. Solberg A, Reikvam H. Iron Status and Physical Performance in Athletes. Life (Basel). 2023;13(10): 2007. Published 2023 Oct 2. doi:10.3390/life131 02007
18. McKay AKA, Pyne DB, Burke LM, Peeling P. Iron Metabolism: Interactions with Energy and Carbohydrate Availability. Nutrients. 2020;12(12): 3692. Published 2020 Nov 30. doi:10.3390/nu121 23692
19. Henderson SA, Dallman PR, Brooks GA. Glucose turnover and oxidation are increased in the iron-deficient anemic rat. Am J Physiol. 1986; 250(4 Pt 1):E414-E421. doi:10.1152/ajpendo.198 6.250.4.E414
20. Hayashi N, Ishibashi A, Iwata A, Yatsutani H, Badenhorst C, Goto K. Influence of an energy deficient and low carbohydrate acute dietary manipulation on iron regulation in young females. Physiol Rep. 2022;10(13):e15351. doi:10.14814/ph y2.15351
21. Statuta SM, Asif IM, Drezner JA. Relative energy deficiency in sport (RED-S). Br J Sports Med. 2017;51 (21):1570-1571. doi:10.1136/bjsports-2017-097700
22. Marzuki MIH, Mohamad MI, Chai WJ, et al. Prevalence of Relative Energy Deficiency in Sport (RED-S) among National Athletes in Malaysia. Nutrients. 2023;15(7):1697. Published 2023 Mar 30. doi:10.3390/nu15071697
23. Loucks AB. Low energy availability in the marathon and other endurance sports. Sports Med. 2007;37(4-5):348-352. doi:10.2165/00007256-200 737040-00019
24. Nemeth E, Valore EV, Territo M, Schiller G, Lichtenstein A, Ganz T. Hepcidin, a putative mediator of anemia of inflammation, is a type II acute-phase protein. Blood. 2003;101(7):2461-2463. doi:10.118 2/blood-2002-10-3235
25. Steensberg A, Febbraio MA, Osada T, et al. Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol. 2001;537(Pt 2):633-639. doi:10.1111/j.1469-7793.2001.00633.x
26. Solberg A, Reikvam H. Iron Status and Physical Performance in Athletes. Life (Basel). 2023;13(10): 2007. Published 2023 Oct 2. doi:10.3390/life13102007
27. Hennigar SR, McClung JP, Hatch-McChesney A, et al. Energy deficit increases hepcidin and exacerbates declines in dietary iron absorption following strenuous physical activity: a randomized-controlled cross-over trial. Am J Clin Nutr. 2021; 113(2):359-369. doi:10.1093/ajcn/nqaa289
28. Barney DE, Ippolito JR, Berryman CE, Hennigar SR. A Prolonged Bout of Running Increases Hepcidin and Decreases Dietary Iron Absorption in Trained Female and Male Runners. J Nutr. 2022; 152(9):2039-2047. doi:10.1093/jn/nxac129
29. Gagne, C.M.; Walberg-Rankin, J.L.; Ritchey, S.J. Effects of exercise on iron status in mature female rats. Nutr. Res. 1994 14:211-219. DOI:10.1016/S0271-5317(05)80380-X.
30. Ming Qian Z, Sheng Xiao D, Kui Liao Q, Ping Ho K. Effect of different durations of exercise on transferrin-bound iron uptake by rat erythroblast. J Nutr Biochem. 2002;13(1):47-54. doi:10.1016/s0955-2863(01)00194-2
31. Sawada T, Konomi A, Yokoi K. Iron deficiency without anemia is associated with anger and fatigue in young Japanese women [published correction appears in Biol Trace Elem Res. 2015 Dec;168(2): 520-1. doi: 10.1007/s12011-015-0531-0.]. Biol Trace Elem Res. 2014;159(1-3):22-31. doi:10.1007/s12011-014-9963-1
32. Sim M, Garvican-Lewis LA, Cox GR, et al. Iron considerations for the athlete: a narrative review. Eur J Appl Physiol. 2019;119(7):1463-1478. doi:10.1007/s00421-019-04157-y
33. Fujii T, Matsuo T, Okamura K. The effects of resistance exercise and post-exercise meal timing on the iron status in iron-deficient rats. Biol Trace Elem Res. 2012;147(1-3):200-205. doi:10.1007/s1 2011-011-9285-5
34. Fujii T, Matsuo T, Okamura K. Effects of resistance exercise on iron absorption and balance in iron-deficient rats. Biol Trace Elem Res. 2014;161 (1):101-106. doi:10.1007/s12011-014-0075-8
35. Silvestri L, Pagani A, Nai A, De Domenico I, Kaplan J, Camaschella C. The serine protease matriptase-2 (TMPRSS6) inhibits hepcidin activation by cleaving membrane hemojuvelin. Cell Metab. 2008;8(6):502-511. doi:10.1016/j.cmet.2008.09.012
36. Fujii T, Matsuo T, Okamura K. The effects of resistance exercise and post-exercise meal timing on the iron status in iron-deficient rats. Biol Trace Elem Res. 2012;147(1-3):200-205. doi:10.1007/s12 011-011-9285-5
37. Franzone PC, Paganuzzi A, Stefanelli M. A mathematical model of iron metabolism. J Math Biol. 1982;15(2):173-201. doi:10.1007/BF00275072
38. McCormick R, Moretti D, McKay AKA, et al. The Impact of Morning versus Afternoon Exercise on Iron Absorption in Athletes. Med Sci Sports Exerc. 2019;51(10):2147-2155. doi:10.1249/MSS.0 000000000002026
Most read articles by the same author(s)
- Ryunosuke Takahashi, Katsuhiko Hata, Koji Okamura, Takako Fujii, Iron deficiency in Athletes – Challenges and Future of Sports Nutrition , Medical Research Archives: Vol 13 No 2 (2025): Vol.13 issue 2 February 2025
- Ryunosuke Takahashi, Takako Fujii, The Effects of Different Athletic Performances on Dietary Intake , Medical Research Archives: Vol 13 No 9 (2025): Vol.13, Issue 9, September 2025
- Ryunosuke Takahashi, Takako Fujii, Iron deficiency of Sports Nutrition , Medical Research Archives: Vol 13 No 10 (2025): Vol.13, Issue 10, October 2025

