Metalloestrogens and Breast Cancer: Endocrine Disruption Insights
Metalloestrogens and Endocrine Disruption in Breast Cancer
Ashlyn Burt1, Keyara Walters1, Gavin Cullinan1, Hilary Gaudet1
- Wheaton College, Norton, MA, 02766, USA
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Burt, A., Walters, K., et al., 2025. Metalloestrogens and Endocrine Disruption in Breast Cancer. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7116
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7116
ISSN 2375-1924
Abstract
Metalloestrogens, metals and metalloids capable of mimicking or interfering with estrogen signaling, are emerging environmental contributors to breast cancer risk and endocrine therapy resistance. Compounds such as cadmium, arsenic, aluminum, and chromium bind to estrogen receptors or activate non-genomic pathways such as G protein-coupled estrogen receptor and epidermal growth factor receptor signaling, leading to aberrant proliferation, survival, and oxidative stress. These mechanisms promote epithelial–mesenchymal transition, genomic instability, and therapy resistance in estrogen receptor-positive breast cancer. Cadmium and arsenic can sustain estrogenic signaling and activate MAPK/ERK and PI3K/Akt pathways even under tamoxifen treatment, while arsenic-mediated BRCA1 methylation further reduces drug responsiveness. Limited studies suggest metalloestrogens may also attenuate the efficacy of aromatase inhibitors and selective estrogen receptor degraders by maintaining G protein-coupled estrogen receptor and oxidative stress signaling. Epidemiological evidence correlates elevated cadmium, nickel, and aluminum exposure with increased estrogen receptor-positive breast cancer incidence, yet clinical translation remains incomplete. Understanding metalloestrogen-mediated crosstalk between estrogen receptor-dependent and estrogen receptor-independent pathways will be essential for designing effective combination therapies and integrating environmental toxicology into precision breast cancer management.
Keywords
metalloestrogens, breast cancer, endocrine disruption, estrogen receptor, therapy resistance
1. Introduction
Metalloestrogens are a class of metals and metalloids capable of mimicking or interfering with estrogenic signaling, thus functioning as endocrine-disrupting compounds. These elements are increasingly recognized for their ability to imitate natural hormones at the molecular level, altering gene expression and cellular behavior even at low environmental concentrations.
Common examples include cadmium (Cd), arsenic (As), aluminum (Al), and chromium (Cr), often found in cosmetics, diet, environmental pollution, and tobacco smoke. Human exposure to these metals occurs through multiple routes, including contaminated food, air, and water. Their bioaccumulation in tissues such as the liver, kidneys, and breast raises concerns about long-term hormonal and carcinogenic effects.
These compounds can interact directly with estrogen receptors or alter cellular signaling pathways, promoting proliferation, survival, and genomic instability. By binding to estrogen receptor alpha (ERα) or triggering non-genomic pathways such as those mediated by the G protein–coupled estrogen receptor (GPER), metalloestrogens can activate proliferative cascades independent of endogenous estrogen.
Metalloestrogens have been linked to an increased risk of estrogen receptor-positive (ER+) breast cancer and may contribute to tumor progression and endocrine therapy resistance. For instance, cadmium can function as a “false estrogen,” maintaining ER signaling even when estrogen levels are low, while arsenic has been shown to induce epigenetic modifications that silence tumor suppressor genes.
Despite growing mechanistic evidence, the clinical relevance of metalloestrogen exposure remains underexplored. Although in vitro and animal studies have mapped out receptor interactions and signaling effects, few human studies have quantified exposure-response relationships or linked metal burden directly to treatment outcomes. Understanding how these metals influence estrogen signaling, cancer progression, and therapeutic outcomes is essential for clarifying the environmental dimension of breast cancer and developing strategies to mitigate their effects. Recognizing metalloestrogens as both environmental toxins and potential modulators of endocrine therapy response could provide new insight into prevention strategies and precision treatment approaches for ER+ breast cancer.
2. Mechanistic Diversity of Metalloestrogens
A. ESTROGEN RECEPTOR BINDING AND MODULATION
Cadmium (Cd) is one of the best-characterized metalloestrogens and is often described as a “false estrogen.” It binds to estrogen receptor alpha (ERα) with high affinity, inducing conformational changes that mimic 17β-estradiol (E2) and activating transcription of estrogen-responsive genes in the absence of natural estrogen. This ligand-independent activation promotes proliferation and survival of hormone-responsive cells, supporting tumor growth and progression. Cadmium’s effects depend on the hormonal context; for instance, in ovariectomized or hormone-depleted animal models, it fails to activate ERα.
Beyond direct receptor binding, Cd also stimulates rapid non-genomic signaling. Together, these findings illustrate how Cd functions both as a direct estrogen receptor (ER) agonist and as a modulator of alternative estrogenic pathways.
B. NON-GENOMIC SIGNALING AND CROSSTALK VIA G PROTEIN-ESTROGEN RECEPTOR
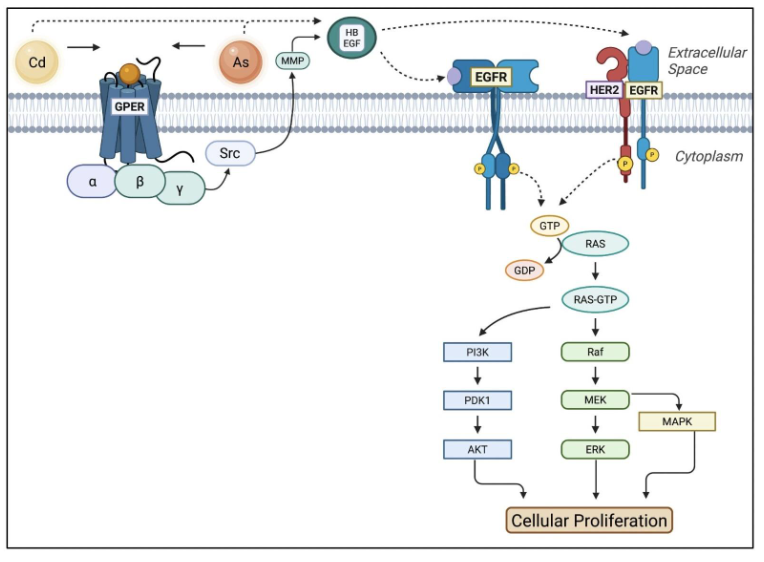
Metalloestrogens such as Cd and As have demonstrated the ability to initiate rapid, non-genomic signaling through the G protein-coupled estrogen receptor (GPER), as shown in Figure 1. Activation of GPER stimulates downstream PI3K/Akt and MAPK/ERK1/2 pathways, leading to proliferation and survival even in ER-negative cells. Cadmium-induced phosphorylation of Akt and ERK has been linked to GPER binding, and studies confirm that GPER interference suppresses this signaling.
G protein-coupled estrogen receptor crosstalk extends to other pathways, including EGFR, Notch, and Hippo/YAP, thereby amplifying proliferative signaling. Cadmium activates EGFR and MAPK pathways in uterine and breast cells, while inhibitors such as AG1478 (EGFR) or PD98059 (MEK) suppress Cd-induced proliferation. Cadmium can also interact with ERα36, creating a feedback loop with EGFR and HER2 that sustains MAPK/ERK signaling and contributes to tamoxifen resistance.
C. OXIDATIVE STRESS AND REDOX IMBALANCE
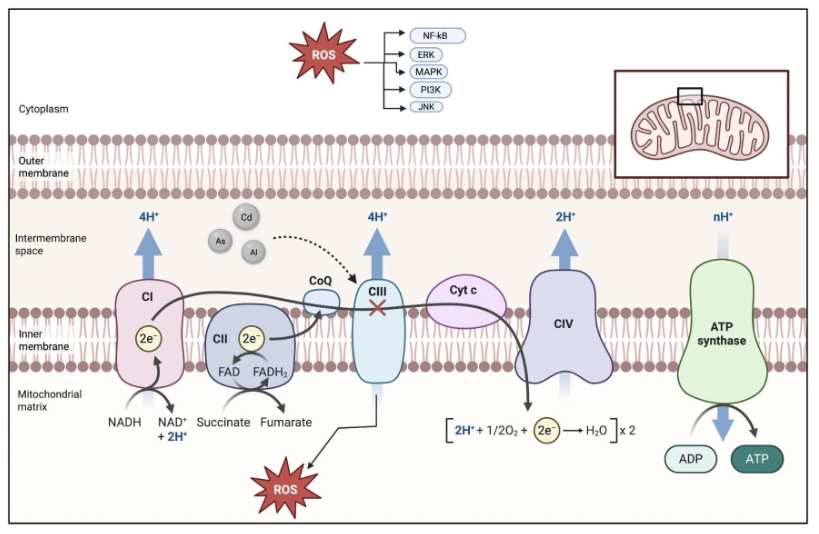
Redox-active metalloestrogens such as Cd, As, and Al contribute to carcinogenesis primarily by disrupting the cellular redox balance. Rather than directly mutating DNA, they promote oxidative stress by generating reactive oxygen species (ROS) and impairing antioxidant defenses. Mitochondrial dysfunction, glutathione depletion, and inhibition of antioxidant enzymes such as superoxide dismutase (SOD) and catalase lead to ROS accumulation and oxidative DNA damage.
Sustained oxidative stress activates oncogenic pathways such as MAPK, PI3K/Akt, and NF-κB, enhancing proliferation and survival under oxidative conditions. Chronic exposure to Cd and As maintains tumor-promoting signaling and can reduce sensitivity to endocrine therapies like tamoxifen.
3. Metalloestrogens and Breast Cancer Progression
A. PROLIFERATION AND APOPTOSIS
Metalloestrogens promote proliferation primarily through GPER and ERK1/2 activation, which regulate cell cycle proteins and drive uncontrolled cell division. Studies in breast cancer cells demonstrate that Cd and As mimic E2, binding GPER and inducing ERK phosphorylation, as shown in Figure 1. These studies found Cd-induced ERK activation to occur in ER+ MCF-7 and GPER-positive SKBR3 cells, but not in ERβ-only MDA-MB-231 cells, suggesting dependence on GPER and membrane-associated ERα.
While research on metalloestrogen-induced apoptosis is limited, evidence from E2 studies indicates that E2 upregulates ER stress-related apoptotic genes such as BIM and CASP4. It is plausible that metalloestrogens exert similar effects through estrogen-mimetic mechanisms, although this remains to be fully confirmed by the literature. In fact, previous research found high apoptosis rates among chromium (VI) treated MCF-7 and MDA-MB-175-VII cells.
B. METASTASIS AND MICROENVIRONMENT
Although underexplored, Cd exposure has been linked to epithelial-mesenchymal transition (EMT) characterized by E-cadherin, Twist, and Slug suppression, and MMP, N-cadherin, and vimentin upregulation. Further studies found that Cd treatment induces T-cell factor/lymphoid enhancer factor transcription and significantly decreases the expression of genes responsible for antioxidant activity, detoxification enzymes, and xenobiotic transporters. Together, these mechanisms promote cell motility, invasion, and metastatic potential.
4. Influence on Endocrine Therapy Outcomes
A. TAMOXIFEN RESISTANCE
Tamoxifen is a selective estrogen receptor modulator (SERM) that inhibits ERα-mediated transcription in ER+ breast cancer. Metalloestrogens such as cadmium (Cd) and arsenic (As) compromise tamoxifen efficacy through both receptor-dependent and epigenetic mechanisms. Cadmium directly activates ERα even in the presence of tamoxifen, preventing full receptor antagonism and sustaining transcription of estrogen-responsive genes, as demonstrated in Figure 3. It also triggers non-genomic signaling through GPER and ERα36, forming a feedback loop with EGFR and HER2 that maintains MAPK and Akt activation despite drug treatment.
Arsenic exposure induces BRCA1 promoter methylation, silencing ERα expression, and diminishing tamoxifen sensitivity. Furthermore, chronic Cd exposure further promotes metabolic reprogramming and glycolytic adaptation, consistent with endocrine-resistant phenotypes. Together, these mechanisms illustrate how metalloestrogens enable tumor cells to evade tamoxifen suppression, either by reactivating ERα signaling or by shifting reliance toward alternative growth and survival pathways. Targeting ERα36 and downstream kinases with inhibitors such as lapatinib or AG1478 has been shown to restore tamoxifen responsiveness, emphasizing the therapeutic potential of combined hormonal and growth-factor blockade in overcoming metal-induced resistance.
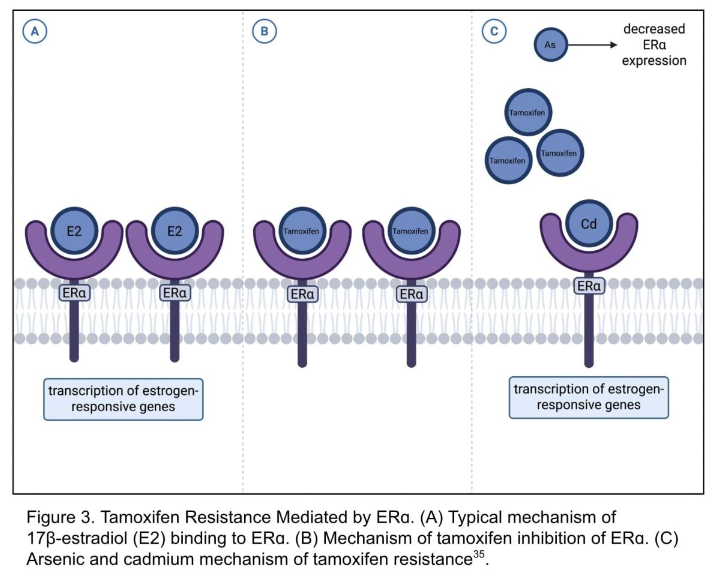
Figure 3. Tamoxifen Resistance Mediated by ERα. (A) Typical mechanism of 17β-estradiol (E2) binding to ERα. (B) Mechanism of tamoxifen inhibition of ERα. (C) Arsenic and cadmium mechanism of tamoxifen resistance³⁵.
Arsenic exposure induces BRCA1 promoter methylation, silencing ERα expression, and diminishing tamoxifen sensitivity¹¹. Furthermore, chronic Cd exposure further promotes metabolic reprogramming and glycolytic adaptation, consistent with endocrine-resistant phenotypes¹².
Together, these mechanisms illustrate how metalloestrogens enable tumor cells to evade tamoxifen suppression, either by reactivating ERα signaling or by shifting reliance toward alternative growth and survival pathways. Targeting ERα36 and downstream kinases with inhibitors such as lapatinib or AG1478 has been shown to restore tamoxifen responsiveness¹², emphasizing the therapeutic potential of combined hormonal and growth-factor blockade in overcoming metal-induced resistance.
B. AROMATASE INHIBITORS
Aromatase inhibitors (AIs) are primarily used by postmenopausal women diagnosed with early-stage ER+ breast cancer. AIs target the aromatase enzyme [cytochrome P450 19A1 (CYP19A1)], which is responsible for catalyzing the transformation of C19 androgens (TST and ASD) into C18 estrogens (E1 and E2) via aromatization in the endoplasmic reticulum, as illustrated in Figure 4. CYP19A1 is expressed in various tissue types, including the placenta, gonads (granulosa cells), blood vessels, skin, bone, adipose, and breast cancer tissues. ER+ breast cancer cells depend on aromatase activity to proliferate because these tumors need estrogen to develop and survive.
There are currently multiple AIs under investigation for clinical use, including anastrozole and letrozole, which competitively inhibit aromatase, and exemestane, which irreversibly covalently binds to aromatase’s substrate pocket. Additionally, studies have found that aromatase activity can be increased through PI3K/Akt/IGFR signaling in endocrine-resistant cell. While studies investigating the direct effect of metalloestrogens on AI efficacy are limited, a 2023 study by Boszkiewicz et al. found that decreased concentrations of exemestane and increased concentrations of letrozole, when combined with metalloestrogens, led to a decrease in AI effectiveness. The investigators found a significant difference in cell viability between only AI-treated cells and cells treated with AIs and Al or chromium (III) (Cr3). The cells treated with AIs and Al or Cr3 survived at a greater rate than the cells only treated with AIs. Furthermore, investigators found that in MCF-7/DOX cells, the combination of metalloestrogens and AIs leads to a decrease in the drug’s effectiveness due to an increase in the viability of breast cancer cells. This lack of therapeutic efficacy is observed most likely because metalloestrogens are still able to exert their effects by interacting with GPER and/or EGFR, as demonstrated in Figure 1.
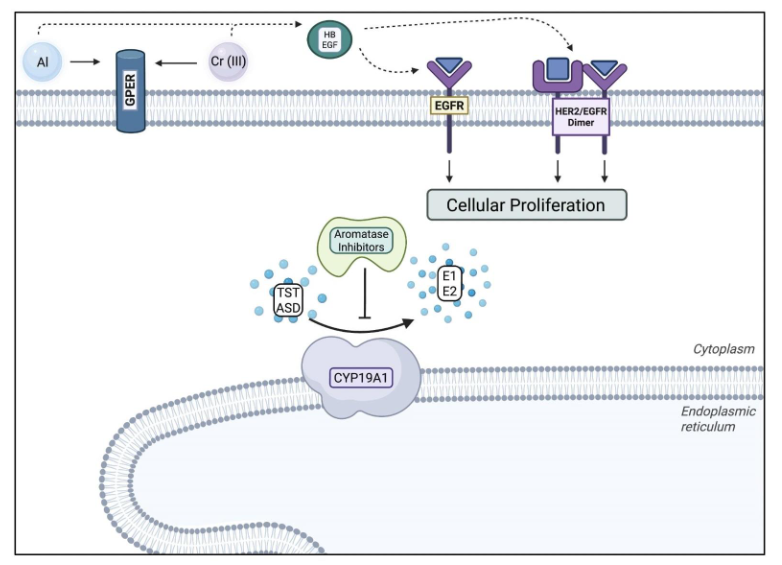
Figure 4. Aromatase Inhibitor Mechanism of Action and Resistance. Aromatase inhibitors prevent aromatase (CYP19A1) from transforming testosterone (TST) and androstenedione (ASD) into estrone (E1) and 17-β-estradiol (E2). Aluminium and chromium (III) bypass CYP19A1 by mimicking E2, and binding to subsequent receptors to promote cellular proliferation³⁹.
There are currently multiple AIs under investigation for clinical use, including anastrozole and letrozole, which competitively inhibit aromatase, and exemestane, which irreversibly covalently binds to aromatase’s substrate pocket³⁷,⁴⁰. Additionally, studies have found that aromatase activity can be increased through PI3K/Akt/IGFR signaling in endocrine-resistant cell³⁷.
While studies investigating the direct effect of metalloestrogens on AI efficacy are limited, a 2023 study by Boszkiewicz et al. found that decreased concentrations of exemestane and increased concentrations of letrozole, when combined with metalloestrogens, led to a decrease in AI effectiveness⁹. The investigators found a significant difference in cell viability between only AI-treated control cells and cells treated with AIs and Al or chromium (III) (Cr3). The cells treated with AIs and Al or Cr3 survived to a greater extent than the cells only treated with AIs. Furthermore, the investigators found that in MCF-7/DOX cells, the combination of metalloestrogens and AIs leads to a decrease in the drug’s effectiveness due to an increase in the viability of breast cancer cells⁹. This lack of therapeutic efficacy is observed most likely because metalloestrogens are still able to exert their effects by interacting with GPER and/or EGFR, as demonstrated in Figure 1.
C. SELECTIVE ESTROGEN RECEPTOR DEGRADERS
Selective estrogen receptor degraders (SERDs) such as fulvestrant and next-generation oral agents (elacestrant, camizestrant, giredestrant) promote ER degradation by inducing conformational changes in the receptors. SERDs are most commonly used in ER+ metastatic breast cancer; Figure 5 demonstrates the mechanism SERDs deploy to function as an endocrine therapy agent. While SERDs can block ERα-mediated transcription induced by metalloestrogens, they fail to inhibit ER-independent pathways such as GPER and oxidative stress signaling. This incomplete antagonism underscores the need for combination approaches addressing non-genomic resistance mechanisms.
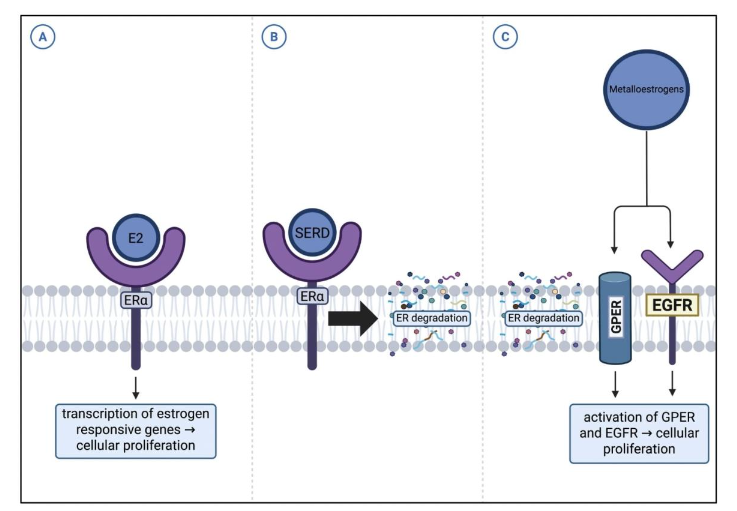
Figure 5. SERD Resistance Pathway. (A) Typical mechanism of 17β-estradiol (E2) binding to ERα. (B) Interaction of selective estrogen receptor degraders (SERDs) with ERα. (C) Mechanism of SERD resistance mediated by GPER and EGFR⁴².
The first clinically approved SERD, fulvestrant, has demonstrated efficacy in first-line and post AI therapy settings. A 500 mg monthly dosing regimen demonstrated greater progression-free survival relative to other endocrine therapies in two trials, CONFIRM and FALCON. However, fulvestrant exhibits limitations, including poor solubility, low bioavailability, and reliance on intramuscular injection. These limitations can complicate dose escalation in patients with resistant tumors or ESR1 mutations. These challenges have driven the development of oral SERDs, including elacestrant, camizestrant, and giredestrant.
Currently under preclinical studies is the oral SERD, G-5b, which binds ER and induces cytoplasmic translocation followed by proteasome-mediated degradation. Acting post-translationally, it avoids altering ER mRNA levels. This process both enhances ubiquitination and reduces the activity of ER target genes such as TFF1, PgR, and GREB1. Moreover, G-5b has demonstrated a favorable therapeutic index in preclinical tamoxifen-resistant models by minimizing uterine stimulation and inducing dose-dependent tumor regression.
The interaction between SERDs and metalloestrogens, such as Cd and arsenite (As3+), highlights both the potential and limitations of SERDs in modulating non-classical ER activation. SERDs, in combination with kinase inhibitors such as abemaciclib, can antagonize ERα-mediated transcriptional responses induced by metalloestrogens, as evidenced by reversal of canonical ER target genes (e.g., PgR, GREB1, c-fos) in vitro and in ovariectomized rat models. However, a substantial proportion of metalloestrogen-regulated genes, over 3,000 in the case of As3+, remain unaffected by SERD treatment, suggesting that metalloestrogens activate ER-independent or non-genomic pathways, including G-protein-coupled estrogen receptor (GPER) signaling and oxidative stress pathways.
Collectively, these observations indicate that while SERDs can mitigate classical ERα-mediated effects of metalloestrogens, their ability to fully neutralize metalloestrogen-driven signaling is incomplete.
D. COMBINATION THERAPIES AND TARGETED AGENTS
Targeted therapies against PI3K, HER2, and CDK4/6 are emerging adjuncts to endocrine therapy. While there is limited research on the interaction between metalloestrogens and targeted therapies, there have been a few studies specifically investigating cadmium chloride (CdCl2). Former studies have found CdCl2 treatment in MCF-10A cells to cause anchorage-independent colony growth, higher expression of oncogenic proteins, and an increase in PI3K–AKT–NFκB protein expression. An increase in expression of PI3K–AKT–NFκB was also observed in mice xenografts.
Moreover, evidence demonstrates that certain flavonoids exert anticancer effects, for example, decreasing phospho-AKT, in breast cancer cells by arresting the cell cycle at the G2/M phase and therefore initiating apoptosis. This mechanism potentially operates through PI3K signaling inhibition via targeting of PI3Kγ.
5. Clinical and Translational Evidence and Biomarkers
Epidemiological studies link elevated blood and breast tissue Cd levels with increased risk of ER+ cancer, yet human data remain limited. Although preclinical research has clarified how metalloestrogens influence estrogen signaling, clinical translation remains incomplete. No trials have directly tested metalloestrogen effects on endocrine therapy outcomes, and current biomarker studies primarily focus on Cd-induced transformation in MCF-10A cells and oxidative stress markers.
A recent French E3N cohort study found that greater dietary exposure to metalloestrogen profiles, including Cd, Ni, and Al, significantly correlated with ER+ breast cancer incidence. These findings align with earlier reports of Cd accumulation in breast tissue and its ability to sustain estrogenic signaling.
At the molecular level, chronic Cd exposure disrupts DNA repair, induces oxidative stress, and activates pro-inflammatory and estrogen-responsive pathways, mechanisms consistent with endocrine resistance. Experimental models also show that combined Cd and As3+ exposure promotes mammary tumor formation through both ER-dependent and independent mechanisms.
Despite these advances, no clinical trials have directly examined metalloestrogen burden in relation to endocrine therapy outcomes. Incorporating biomarkers such as 8-hydroxy-2′-deoxyguanosine (8-OHdG), ERα36, and GPER into ongoing breast cancer studies could help determine whether chronic metal exposure contributes to resistance in anti-estrogen-treated patients.
6. Conclusion
Metalloestrogens represent a class of environmental endocrine disruptors with significant implications for breast cancer biology and treatment response. Through their ability to bind ERα, activate GPER-mediated non-genomic signaling, and induce oxidative stress, these compounds recreate or amplify estrogenic stimuli in ways that promote proliferation, survival, genomic instability, and metastatic behavior. Their mechanistic diversity enables them to influence both classical hormone signaling and alternative oncogenic pathways, positioning them as modulators of tumor progression and potential contributors to endocrine therapy resistance.
Evidence drawn from cellular and animal models demonstrates that metalloestrogens such as Cd, As, Al, and Cr³⁺ compromise the effectiveness of tamoxifen, AIs, and SERDs by sustaining ERα signaling, activating alternative kinase pathways, and inducing epigenetic changes that alter hormone responsiveness. These findings highlight the need for therapeutic approaches that address both receptor-dependent and receptor-independent mechanisms, including combination strategies integrating kinase inhibition, ERα36 targeting, and next-generation SERDs.
Despite substantial preclinical insight, the clinical impact of metalloestrogen exposure remains insufficiently understood. Epidemiologic study findings linking dietary and environmental metalloestrogen burden to ER+ breast cancer risk underscore the necessity of integrating exposure assessment, molecular biomarkers, and treatment outcome data into clinical research. Incorporating indicators such as ERα36, GPER expression, and oxidative stress markers into breast cancer trials may help to clarify how chronic exposure shapes tumor behavior and treatment efficacy.
While studies investigating Cd and As are plentiful, in vivo research efforts focusing on other metalloestrogens such as to include metals such as Al, Ni, and Cr³⁺ is also critical. Moreover, the combined effects of multiple metalloestrogens, reflecting real-world environmental conditions, remain insufficiently explored and warrant further investigation.
Overall, recognizing metalloestrogens as both environmental carcinogens and modulators of endocrine resistance expands our understanding of breast cancer pathogenesis and provides new opportunities for prevention and precision therapy. A deeper integration of mechanistic, epidemiologic, and translational data will be essential for determining how environmental metal exposure influences the clinical trajectory of ER+ breast cancer and for developing strategies to mitigate its impact on patient outcomes.
Conflicts of Interest
The authors have no conflicts of interest to declare.
AI Statement
The authors used Undermind to assist in the initial literature search and identification of relevant publications. ChatGPT (OpenAI) was employed to improve clarity, organization, and flow during the drafting and revision process; all content was reviewed and verified by the authors for accuracy and originality. BioRender was used to create figures and schematic illustrations. No generative tools were used to generate data, interpret results, or draw scientific conclusions.
References
- Byrne C, Divekar SD, Storchan GB, Parodi DA, Martin MB. Metals and Breast Cancer. J Mammary Gland Biol Neoplasia. 2013;18(1):63-73. doi:10.1007/s10911-013-9273-9
- Wallace DR. Nanotoxicology and Metalloestrogens: Possible Involvement in Breast Cancer. Toxics. 2015;3(4):390-413. doi:10.3390/toxics3040390
- Darbre PD. Environmental oestrogens, cosmetics and breast cancer. Best Practice & Research Clinical Endocrinology & Metabolism. 2006;20(1):121-143. doi:10.1016/j.beem.2005.09.007
- Jurkowska K, Kratz EM, Sawicka E, Piwowar A. The impact of metalloestrogens on the physiology of male reproductive health as a current problem of the XXI century. Journal of Physiology and Pharmacology. 2019; 70(3): 337-355. doi:10.26402/jpp.2019.3.02
- Hofer N, Diel P, Wittsiepe J, Wilhelm M and Degen GH. Dose- and Route-Dependent Hormonal Activity of the Metalloestrogen Cadmium in the Rat Uterus. Toxicology Letters. 2009;191:123-131. Doi: 10.1016/j.toxlet.2009.08.014
- Ali I, Damdimopoulou P, Stenius U, Halldin K. Cadmium at nanomolar concentrations activates Raf-MEK-ERK1/2 MAPKs signaling via EGFR in human cancer cell lines. Chem-Biol Interact. 2015;231:44-52. doi:10.1016/j.cbi.2015.02.014
- Liu J, Yu L, Castro L, et al. A nongenomic mechanism for “metalloestrogenic” effects of cadmium in human uterine leiomyoma cells through G protein-coupled estrogen receptor. Arch Toxicol. 2019;93(10):2773-2785. doi:10.1007/s00204-019-02544-0
- Qie Y, Qin W, Zhao K, Liu C, Zhao L, Guo LH. Environmental Estrogens and Their Biological Effects through GPER Mediated Signal Pathways. Environmental Pollution. 2021;278:116826. doi:10.1016/j.envpol.2021.116826
- Boszkiewicz K, Moreira H, Sawicka E, Szyjka A, Piwowar A. The Effect of Metalloestrogens on the Effectiveness of Aromatase Inhibitors in a Hormone-Dependent Breast Cancer Cell Model. Cancers. 2023;15(2):457. doi:10.3390/cancers15020457
- Stoica A, Katzenellenbogen BS, Martin MB. Activation of estrogen receptor-α by the heavy metal cadmium. Mol Endocrinol. 2000;14(4):545-553. doi:10.1210/me.14.4.545
- Selmin O, Donovan M, Skovan B, Paine-Murieta G, Romagnolo D. Arsenic-induced BRCA1 CpG promoter methylation is associated with the downregulation of ERα and resistance to tamoxifen in MCF7 breast cancer cells and mouse mammary tumor xenografts. International Journal of Oncology. 2019;54(3):869-878. doi:10.3892/ijo.2019.4687
- Tarhonska K, Janasik B, Roszak J, et al. Environmental exposure to cadmium in breast cancer – association with the Warburg effect and sensitivity to tamoxifen. Biomedicine & Pharmacotherapy. 2023;161:114435. doi:10.1016/j.biopha.2023.114435
- Saint-Martin F, Marques C, Ren X, Lequy E, Mancini FR, Frénoy P. Associations between dietary exposure to profiles of metalloestrogens and estrogen-receptor positive breast cancer risk in the French E3N cohort. Environmental Health. 2025;24(1):22. doi:10.1186/s12940-025-01167-6
- Nail AN, Chavez AV, Bailey AN, et al. DNA damage response inhibition is an early event in cadmium-induced breast carcinogenesis. Toxicology and Applied Pharmacology. 2025;502:117439. doi:10.1016/j.taap.2025.117439
- Divekar SD, Li HH, Parodi DA, et al. Arsenite and cadmium promote the development of mammary tumors. Carcinogenesis. 2020;41(7):1005-1014. doi:10.1093/carcin/bgz176
- Hirao-Suzuki M, Takeda S, Kodama Y, Takiguchi M, Toda A, Ohara M. Metalloestrogenic effects of cadmium are absent in long-term estrogen-deprived MCF-7 cells: Evidence for the involvement of constitutively activated estrogen receptor α and very low expression of G protein-coupled estrogen receptor 1. Toxicology Letters. 2020;319:22-30. doi:10.1016/j.toxlet.2019.10.018
- Yu X, Filardo EJ, Shaikh ZA. The membrane estrogen receptor GPR30 mediates cadmium-induced proliferation of breast cancer cells. Toxicol Appl Pharmacol. 2010;245(1):83-90. doi:10.1016/j.taap.2010.02.005
- Liu Z, Yu X, Shaikh ZA. Rapid activation of ERK1/2 and AKT in human breast cancer cells by cadmium. Toxicol Appl Pharmacol. 2008;228(3):286-294. doi:10.1016/j.taap.2007.12.017
- Pupo M, Maggiolini M, Musti AM. GPER Mediates Non-Genomic Effects of Estrogen. In: Eyster KM, ed. Estrogen Receptors: Methods and Protocols. Springer; 2016:471-488. doi:10.1007/978-1-4939-3127-9_37
- Rasha F, Sharma M, Pruitt K. Mechanisms of endocrine therapy resistance in breast cancer. Molecular and Cellular Endocrinology. 2021;532:111322. doi:10.1016/j.mce.2021.111322
- Burt A. Cadmium and Arsenic Non-Genomic Mechanisms of Proliferation. BioRender. 2025. https://BioRender.com/pcjf33l
- Gao X, Yu L, Moore AB, Kissling GE, Waalkes MP, Dixon D. Cadmium and Proliferation in Human Uterine Leiomyoma Cells: Evidence of a Role for EGFR/MAPK Pathways but Not Classical Estrogen Receptor Pathways. Environmental Health Perspectives. 2014;123(4):331-336. doi:10.1289/ehp.1408234
- Li G, Zhang J, Xu Z, Li Z. ERα36 as a Potential Therapeutic Target for Tamoxifen-Resistant Breast Cancer Cell Line Through EGFR/ERK Signaling Pathway. CMAR. 2020;12:265-275. doi:10.2147/CMAR.S226410
- Yin L, Zhang XT, Bian XW, Guo YM, Wang ZY. Disruption of the ER-α36-EGFR/HER2 Positive Regulatory Loops Restores Tamoxifen Sensitivity in Tamoxifen Resistance Breast Cancer Cells. PLOS ONE. 2014;9(9):e107369. doi:10.1371/journal.pone.0107369
- Egiebor E, Tulu A, Abou-Zeid N, Oseji OF, Ishaque AB. Oxidative Stress Pathway Mechanisms Induced by Four Individual Heavy Metals (As, Hg, Cd and Pb) and Their Quaternary on MCF-7 Breast Cancer Cells. Journal of Advances in Medicine and Medical Research. 2016;17(7):1-11. doi:10.9734/BJMMR/2016/27687
- Wang Y, Fang J, Leonard SS, Krishna Rao KM. Cadmium inhibits the electron transfer chain and induces Reactive Oxygen Species. Free Radical Biology and Medicine. 2004;36(11):1434-1443. doi:10.1016/j.freeradbiomed.2004.03.010
- Burt A. Cadmium, Aluminum, and Arsenic Oxidative Stress Pathway. BioRender. 2025. https://BioRender.com/98v1qhi
- Mebratu Y, Tesfaigzi Y. How ERK1/2 Activation Controls Cell Proliferation and Cell Death Is Subcellular Localization the Answer? Cell Cycle. 2009;8(8):1168-1175. doi:10.4161/cc.8.8.8147
- Ariazi E, Cunliffe H, Lewis-Wambi J, et al. Estrogen induces apoptosis in estrogen deprivation-resistant breast cancer through stress responses as identified by global gene expression across time. Proceedings of the National Academy of Sciences of the United States of America. 2011. doi:10.1073/pnas.1115188108
- Sawicka E, Kulbacka J, Drąg-Zalesińska M, Woźniak A, Piwowar A. Effect of Interaction between Chromium(VI) with 17β-Estradiol and Its Metabolites on Breast Cancer Cell Lines MCF-7/WT and MDA-MB-175-VII: Preliminary Study. Molecules. 2023;28(6):2752. doi:10.3390/molecules28062752
- Shan Z, Wei Z, Shaikh ZA. Suppression of ferroportin expression by cadmium stimulates proliferation, EMT, and migration in triple-negative breast cancer cells. Toxicology and Applied Pharmacology. 2018;356:36-43. doi:10.1016/j.taap.2018.07.017
- Wei Z, Shan Z, Shaikh ZA. Epithelial-mesenchymal transition in breast epithelial cells treated with cadmium and the role of Snail. Toxicology and Applied Pharmacology. 2018;344:46-55. doi:10.1016/j.taap.2018.02.022
- Wei Z, Shaikh ZA. Cadmium stimulates metastasis-associated phenotype in triple-negative breast cancer cells through integrin and β-catenin signaling. Toxicology and Applied Pharmacology. 2017;328:70-80. doi:10.1016/j.taap.2017.05.017
- Darwish WS, Chen Z, Li Y, Wu Y, Chiba H, Hui SP. Identification of cadmium-produced lipid hydroperoxides, transcriptomic changes in antioxidant enzymes, xenobiotic transporters, and pro-inflammatory markers in human breast cancer cells (MCF7) and protection with fat-soluble vitamins. Environ Sci Pollut Res. 2020;27(2):1978-1990. doi:10.1007/s11356-019-06834-z
- Burt A. Tamoxifen Resistance Mediated by ERα. BioRender. 2025. https://BioRender.com/l3cer05
- Bartkowiak-Wieczorek J, Jaros A, Gajdzińska A, et al. The Dual Faces of Oestrogen: The Impact of Exogenous Oestrogen on the Physiological and Pathophysiological Functions of Tissues and Organs. International Journal of Molecular Sciences. 2024;25(15):8167. doi:10.3390/ijms25158167
- Bhutani K, Vishwakarma S, Yadav P, Yadav MK. The current landscape of aromatase inhibitors for the treatment of estrogen receptor-positive breast carcinoma. J Steroid Biochem Mol Biol. 2025;250:106729. doi:10.1016/j.jsbmb.2025.106729
- Lewoniewska S, Oscilowska I, Forlino A, Palka J. Understanding the Role of Estrogen Receptor Status in PRODH/POX-Dependent Apoptosis/Survival in Breast Cancer Cells. Biology. 2021;10(12):1314. doi:10.3390/biology10121314
- Burt A. Aromatase Inhibitor Mechanism of Action and Resistance. BioRender. 2025. https://BioRender.com/16djls2
- Amaral C, Borges M, Melo S, Silva ET da, Correia-da-Silva G, Teixeira N. Apoptosis and Autophagy in Breast Cancer Cells following Exemestane Treatment. PLOS ONE. 2012;7(8):e42398. doi:10.1371/journal.pone.0042398
- Lloyd MR, Wander SA, Hamilton E, Razavi P, Bardia A. Next-generation selective estrogen receptor degraders and other novel endocrine therapies for management of metastatic hormone receptor-positive breast cancer: current and emerging role. Ther Adv Med Oncol. 2022;14:17588359221113694. doi:10.1177/17588359221113694
- Burt A. SERD Resistance Pathway. BioRender. 2025. https://BioRender.com/sfcs2cf
- Wang B, Ma M, Dai Y, et al. A novel scaffold long-acting selective estrogen receptor antagonist and degrader with superior preclinical profile against ER+ breast cancer. European Journal of Medicinal Chemistry. 2024;264:115934. doi:10.1016/j.ejmech.2023.115934
- Mohapatra P, Preet R, Das D, et al. The contribution of heavy metals in cigarette smoke condensate to malignant transformation of breast epithelial cells and in vivo initiation of neoplasia through induction of a PI3K–AKT–NFκB cascade. Toxicology and Applied Pharmacology. 2014;274(1):168-179. doi:10.1016/j.taap.2013.09.028
- Zhang HW, Hu JJ, Fu RQ, et al. Flavonoids inhibit cell proliferation and induce apoptosis and autophagy through downregulation of PI3Kγ mediated PI3K/AKT/mTOR/p70S6K/ULK signaling pathway in human breast cancer cells. Sci Rep. 2018;8(1). doi:10.1038/s41598-018-29308-7

