Inflammatory Markers and Cardiotoxicity in HER2 Therapy
Markers of inflammation as predictors of cardiotoxicity in HER2 directed therapy in breast cancer patients
Emma Nikoline Wennberg, MD 1, Cecilie Friis Hansen, MD 1, Anna Lings Kjeldgaard, MD 2, Peter Frederiksen, MD,
PhD 2, Jordi S. Dahl MD, DMS 3,4, 5, Anni Young Lundgaard, MD, PhD 5, Lars Videbæk, MD, PhD 2, Morten Schou,
MD, PhD 5 , Jacob E. Møller, MD, DMSC g h i , Ann Banke, MD, PhD 1,2**
* Shared first authorship between Emma
Nikoline Wennberg and Cecilie Friis Hansen
- Institute of Clinical Research, University of Southern Denmark
- Department of Cardiology, OdenseUniversity Hospital
- Department of Cardiovascular Medicine, Mayo Clinic, Rochester, Minnesota, USA
- Department of Oncology, Odense University Hospital
- Department of Cardiology, Herlev and Gentofte University Hospital
- Department of Cardiology, Rigshospitalet
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Wennberg, EN., Hansen, CF., et al., 2025. Markers of inflammation as predictors of cardiotoxicity in HER2 directed therapy in breast cancer patients. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7119
DOI https://doi.org/10.18103/mra.v13i11.7119
ISSN 2375-1924
ABSTRACT
Background: Early indicators of cardiotoxicity during trastuzumab treatment are desirable for risk assessment. Oxidative stress and inflammation are potential contributors to the cardiotoxic process, but early inflammatory markers at treatment initiation are not well investigated.
Aims: To investigate whether high-sensitivity C-reactive protein, myeloperoxidase and interleukin-6 measured at trastuzumab initiation are associated with deterioration in global longitudinal strain, left ventricular ejection fraction or the development of cardiotoxicity.
Methods: A post hoc analysis of blood samples from a single-center prospective cohort involving 45 human epidermal growth factor receptor 2 (HER2) – positive breast cancer patients treated with trastuzumab. Blood samples were collected before trastuzumab initiation and on days 3, 7 and 14, and evaluated in mixed model for repeated measures and logistic regression model.
Results: No significant association was found between elevated combined biomarker levels and global longitudinal strain (p = 0.17) or left ventricular ejection fraction (p = 0.18), nor for individual biomarkers. Results from the regression analysis were divergent, with high-sensitivity C-reactive protein negatively associated with mild cardiotoxicity at visit 3 (OR = 0.92, p = 0.041), interleukin-6 negatively associated with moderate and clinical cardiotoxicity at visit 3 (OR = 0.95, p = 0.041 and OR = 0.96, p = 0.036, respectively), but positively associated with moderate and clinical cardiotoxicity at visit 4 (OR = 1.14, p = 0.047 and OR = 1.08, p = 0.032, respectively). Myeloperoxidase showed no significant associations.
Conclusion: High-sensitivity C-reactive protein, myeloperoxidase and interleukin-6 are not reliable early predictors of cardiotoxicity during trastuzumab treatment in this cohort. Larger studies are needed to clarify their relevance in cardiotoxicity risk assessment.
Keywords: Biomarker, Inflammation, Trastuzumab, Left ventricular function, Global longitudinal strain, Cardiotoxicity
1. Introduction
Breast cancer is the most common cancer and the leading cause of cancer-related death among women. Approximately 20-25% of breast cancer tumors overexpress the human epidermal growth factor receptor 2 (HER2) protein, which is associated with a more aggressive course of disease behavior and a poorer prognosis. Trastuzumab, a monoclonal antibody targeting the HER2 receptor, has significantly improved survival in both localized and metastatic HER2-positive breast cancer. However, trastuzumab treatment has been associated with cardiotoxicity, with reported incidences of 7.5% for left ventricular ejection fraction (LVEF) reduction and 1.9% for heart failure. Due to the risk of cardiotoxicity, regular LVEF monitoring is recommended during trastuzumab treatment. However, LVEF assessments have limitations, including variability in the acoustic window, in techniques, a considerable interobserver variability and the fact that it often reflects myocardial dysfunction at a later stage. Deterioration in global longitudinal strain (GLS) has been suggested as an early predictor of later myocardial dysfunction, as it may detect changes that occur prior to declines in LVEF in trastuzumab-treated patients. It could however also be of clinical relevance to identify biomarkers, which can contribute to risk assessment of myocardial dysfunction at an earlier stage. An advantage of biomarkers is that they are more accessible and cost-effective compared to echocardiographic measurements. The exact mechanism of trastuzumab-induced cardiotoxicity is not fully understood, however trastuzumab appears to affect intracellular processes in cardiomyocytes, potentially leading to oxidative stress and inflammation, which are likely contributors to cardiotoxicity. Cardiac biomarkers such as troponins and natriuretic peptides have been suggested as potential risk markers of subsequent myocardial dysfunction and are the most extensively investigated, but their clinical relevance not fully established. Other biomarkers reflecting different pathophysiological pathways, such as inflammation and oxidative stress, have only been explored to a limited extent. High-sensitivity C-reactive protein (hs-CRP), myeloperoxidase (MPO) and interleukin-6 (IL-6) are inflammatory biomarkers and candidates in this context. High-sensitivity C-reactive protein (hs-CRP) is an acute-phase protein primarily synthesized in the liver. It is regulated by proinflammatory cytokines especially interleukin-6 (IL-6) and to a lesser extent by IL-2 and tumor necrosis factor-alpha (TNF-α). Newer research suggest that hs-CRP besides being is a key inflammation marker may also play an active role in the inflammatory process, and it is considered a cardiovascular prognostic indicator. Myeloperoxidase (MPO) is a heme-containing enzyme stored in the granules of neutrophils and released in relation to oxidative stress and inflammation. As an antibacterial action it is involved in the production of reactive oxygen and nitrogen species, but can therefore also enhance the oxidative stress and inflammation process and, it has been linked to trastuzumab-induced cardiotoxicity. Interleukin-6 (IL-6), is a cytokine produced in macrophages, T cells, endothelial cells and fibroblasts in response to tissue damage and is central in inflammatory responses, since it activates signaling pathways, which regulate immune responses, hematopoiesis and acute-phase protein synthesis in the liver. Further it is associated with heart failure risk and may predict cardiotoxicity. These three biomarkers may indicate early cardiotoxicity through immune response and oxidative stress, but most studies have not assessed them at an early stage. The aim of this study was to describe the acute inflammatory response at trastuzumab initiation and to explore whether an early increase in hs-CRP, MPO and IL-6 is associated with deterioration in GLS or LVEF or development of clinical cardiotoxicity in HER2-positive breast cancer patients treated with trastuzumab.
2. Methods
2.1 STUDY POPULATION
This study was a post hoc analysis of blood samples collected from a single-center prospective cohort study. The cohort has previously been described in detail, but briefly it included patients with HER2-positive non-metastatic breast cancer, who received adjuvant trastuzumab following an anthracycline-based chemotherapy regimen. The patients were treated between September 2014 and February 2017. Exclusion criteria included an inadequate acoustic window on echocardiography, LVEF < 50 %, permanent atrial fibrillation, or a history of moderate or severe valvular heart disease. 45 patients met the inclusion criteria and were included in the study.
2.2 ANTINEOPLASTIC TREATMENT
Prior to trastuzumab treatment, the patients received antineoplastic therapy consisting of three cycles of epirubicin (90 mg/m2) and cyclophosphamide (600 mg/m2). Thereafter the patients received either three cycles of docetaxel (100 mg/m2) administered every 3 weeks or nine weekly cycles of paclitaxel (80 mg/m2), both given concomitantly with trastuzumab. Trastuzumab was divided into 17 individual doses and administered over a 12-month period. If necessary and indicated according to guidelines, patients also received additional treatment as endocrine therapy and radiotherapy.
2.3 DATA COLLECTION
Prior to initiating trastuzumab treatment, baseline transthoracic echocardiography (TTE), electrocardiogram (ECG), blood test and clinical assessment were conducted at day 0. These procedures were repeated on day 3, 7 and 14 following the first trastuzumab treatment (visit 2-4). Transthoracic echocardiography (TTE), ECG and clinical assessments were repeated at 3-, 6- and 9-months follow-up (visit 5-7). Left ventricular ejection fraction (LVEF) was estimated by Simpson’s biplane method and GLS was analyzed using Philips Healthcare software, QLAB 10.3. An extended description of the echocardiographic procedure and measurements has previously been described. Data was stored in the REDCap electronic database managed by the OPEN Patient Data Explorative Network (OPEN), Region of Southern Denmark. Samples intended for post hoc research were centrifuged and stored at -80 degrees in the OPEN biobank. For this study, blood samples were analyzed for the biomarkers hs-CRP, MPO, and IL-6 at OPENLab, University of Southern Denmark (SDU).
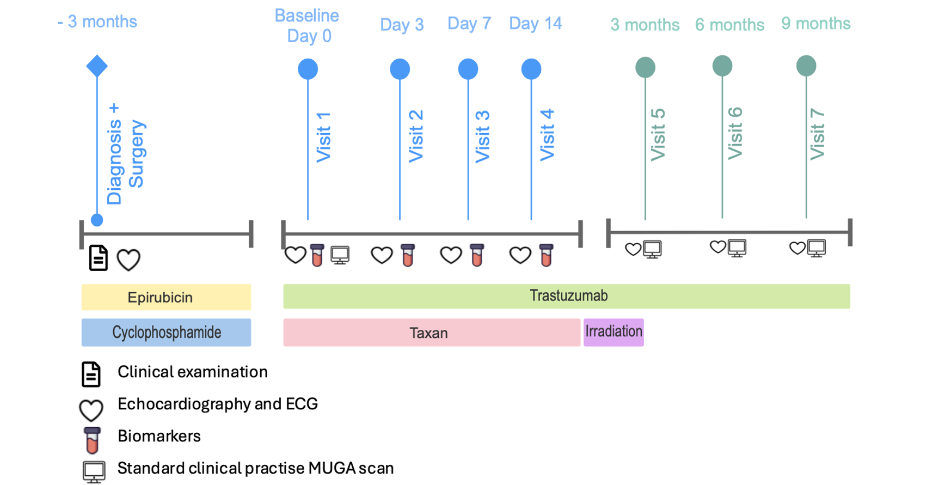
2.4 BIOMARKERS
Myeloperoxidase (MPO) and interleukin-6 (IL-6) levels were determined by enzyme-linked immunosorbent assay (ELISA) using the Duo Set ELISA kit (R&D Systems) according to the manufacturer’s protocol. (Human Myeloperoxidase DuoSet ELISA, R&D Systems, #DY3174 and Human IL-6 DuoSet ELISA, R&D Systems, #DY206). High-sensitivity C-reactive protein (hs-CRP) levels were determined by an in-house sandwich ELISA, developed by OPEN LAB, using an antibody from Sino Biological as the coating antibody and an antibody from RnD system as the detection antibody. The patients were divided into groups with and without an inflammatory response. In this study, an inflammatory response was defined using biomarker-specific thresholds. For hs-CRP, an inflammatory response was defined as an increase >20 mg/L from baseline. For MPO the threshold was based on its dichotomous pattern and set as a change of >100 pg/mL from baseline, allowing the inclusion of relevant patients in the inflammation group. Similarly, for IL-6, an inflammatory response was defined as an increase in >100 pg/mL from baseline, thereby excluding patients with pre-existing elevated baseline levels. A sensitivity analysis was performed using both higher and lower cut-off values for the biomarkers, which did not significantly affect the outcome.
2.5 END POINTS
The inflammatory responses were characterized by the three biomarkers and evaluated by their associations with deterioration in GLS and LVEF during the acute phase and treatment period. Endpoints included asymptomatic cardiotoxicity, which was classified into three severity levels according to the ESC guidelines. Mild cardiotoxicity was defined as an LVEF > 50 % with a >15 % decline in GLS from baseline, Moderate cardiotoxicity was defined either as a >10 % points reduction in LVEF to <50 % from baseline, or as an LVEF <50 % with a reduction of <10 % points, combined with a >15 % decline in GLS. Severe cardiotoxicity was defined by an LVEF reduction to <40 %. In addition, clinical cardiotoxicity was defined as treatment interruption based on MUGA scan results (LVEF <50 %) and clinical decision-making.
2.6 STATISTICAL ANALYSIS
Baseline characteristics were described using proportions for categorical variables. For continuous variables normally distributed data were summarized as means (± standard deviation, SD), while non-normally distributed data were summarized as medians (interquartile ranges, IQR). Normally distributed data were summarized as mean (SD), while non-normally distributed data were summarized as median (IQR). Normal distribution was assessed graphically using histograms and QQ plots, as well as the Shapiro-Wilk test. Comparisons of baseline characteristics and differences between groups were conducted using two-sample t-tests, Wilcoxon two-sample tests, and chi-square tests, as appropriate. When cell counts were less than five, Fisher’s exact test was applied. For comparisons across three groups, in a 2×3 table, chi-square tests were used, even when cell counts were below five. A mixed model for repeated measures was applied to analyze variations in GLS and LVEF throughout the study period, with a positive inflammatory response included as a fixed effect. Graphs for mixed models present least squares means. Statistical significance was defined as a two-sided p-value of <0.05. Logistic regression was used to evaluate the relationship between biomarkers measured at different time points as independent variables and specific cardiotoxicity groups (mild, moderate and clinical cardiotoxicity). Separate logistic models were estimated for each group. Odds ratios (ORs) and 95% profile-likelihood confidence intervals were calculated for the biomarkers across visits within each model. All statistical analyses and data processing were conducted using SAS version 9.4 (SAS Institute, Gary, NC, USA).
2.7 ETHICS
The study was conducted in accordance with the Helsinki Declaration. Informed consent was obtained for the study procedures and biobank storage, allowing the use of material in Denmark after the completion of the main study. Ethics Committee reference number: ID: S-20140090. Registered with research approval under reference number: 24/9844 at OUH data.
3. Results
3.1 STUDY POPULATION
A total of 45 patients were enrolled in the study. Baseline characteristics, divided into groups with (n=15) and without (n=30) inflammatory response, are presented in Table 1 a-c. The mean age was 53.8 years (SD ± 11.2) and the median BMI was 27.3 (IQR: 23.3-30.19) for the total cohort. P-values indicate no statistically significant differences between the groups for most variables, except for pro-B-type natriuretic peptide (proBNP), which was higher in the group without inflammatory response (p = 0.003).
| All (n=45) | Inflammatory response (n=15) | No inflammatory response (n=30) | p-value | ||
|---|---|---|---|---|---|
| Mean age 53.8 (SD ±11.2) | 50.8 (SD±11.1) | 55.4 (SD±11.0) | 0.27 | ||
| Body mass index (BMI (kg/m2) | 27.3 (IQR 23.3-30.1) | 25.5 (IQR 23.3-31.1) | 28.1 (IQR 23.2-30.1) | 0.43 | |
| eGFR (ml/min/1.73m2) | 89 (IQR 77 -90) | 86 (IQR 81-96) | 89 (IQR 75 -90) | 0.38 | |
| Smoking status: | Current | Previous | Never | 0.0 % (0) | |
| 8.9% (4) | 44.4 % (20) | 46.7 % (21) | 0.23 | ||
| Post menopausal | 51.2% (22) | 46.7 % (7) | 53.6 % (15) | 0.67 | |
| Comorbidity | Hypertension | 40.0 % (18) | 33.3 % (5) | 43.3 % (13) | 0.52 |
| Hypercholesterolemia | 15.6% (7) | 6.7 % (1) | 20.0 % (6) | 0.40 | |
| Previous heart disease | 6.7% (3) | 0 % (0) | 10 % (3) | 0.54 | |
| Type 2 diabetes mellitus | 4.4% (2) | 6.7 % (1) | 3.3 % (1) | 1.0 | |
| Other chronic diseases | 26.7 % (12) | 13.3 % (2) | 33.3 % (10) | 0.28 |
3.2 INFLAMMATORY RESPONSES
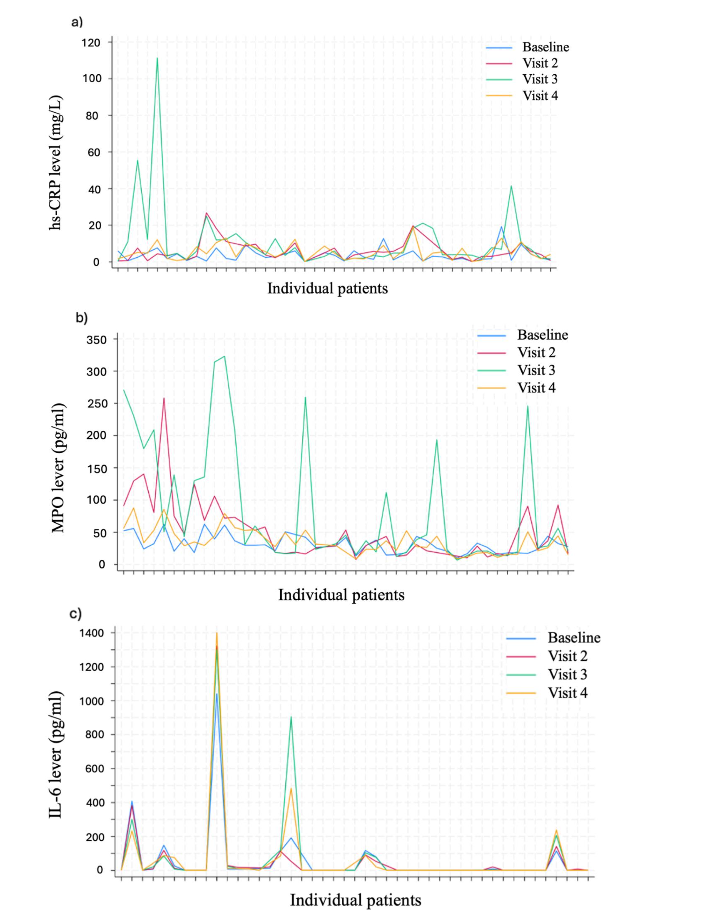
Figures 2 illustrates changes in hs-CRP, MPO, and IL-6 levels across the four initial visits, presenting the course of these biomarkers over time at the individual patient level. Figure 2a of hs-CRP shows that most patients maintained stable hs-CRP levels with low values throughout the study. However, a few patients demonstrated elevated hs-CRP levels at visit 3, indicating a potential inflammatory response. Importantly, none of these patients reported any clinical symptoms of illness, excluding underlying infections as the cause of the elevated hs-CRP. Figure 2b of MPO shows, that the majority of patients had a relatively homogeneous response with low and consistent MPO levels ranging between 20-80 pg/mL, particularly at baseline, visit 2, and 4. However, a few patients showed notable fluctuations and increases, particularly at visit 3. These variations highlight both temporal and interpatient variation in MPO levels compared to hs-CRP and IL-6. Figure 2c of IL-6 indicates that the majority of patients had a low and stable IL-6 response. However, six patients showed consistently high IL-6 levels across all visits, including elevated baseline levels.
3.3 ASSOCIATION BETWEEN INFLAMMATORY RESPONSES AND GLS
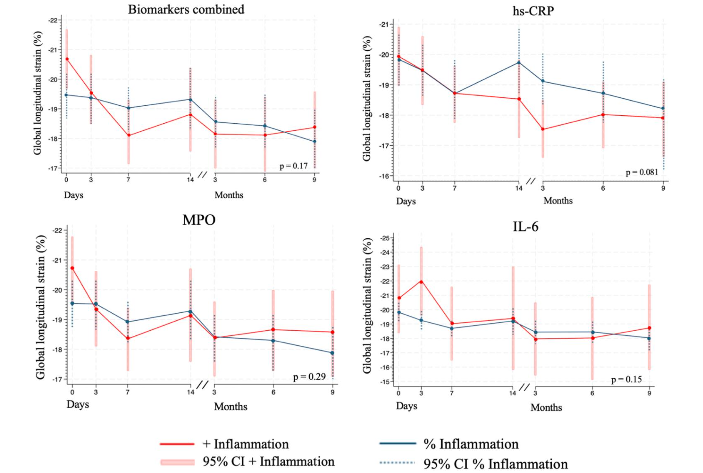
Figure 3 illustrates changes in GLS over time, stratified by inflammation status, for both combined and individual biomarkers. The combined biomarker classification refers to a composite grouping, in which patients were included in the inflammation group if they met the criteria for an inflammatory response in at least one of the three biomarkers. Across all biomarkers, an overall deterioration in GLS was observed in both groups over time, although GLS values remained within the normal range. For the combined biomarkers (3a), the differences between groups were small and not statistically significant (p = 0.17). For hs-CRP (3b), there was a slight trend toward differences between the groups, particularly on day 14, but these differences were not statistically significant (p = 0.081). For MPO (3c), the patterns between the groups were very similar, with no significant differences observed (p = 0.29). For IL-6 (3d), an initial difference was observed between the groups, but it diminished over time and did not reach statistical significance (p = 0.15). Overall, the differences in GLS across the inflammation groups were minor and not statistically significant.
3.4 ASSOCIATION BETWEEN INFLAMMATORY RESPONSES AND LVEF
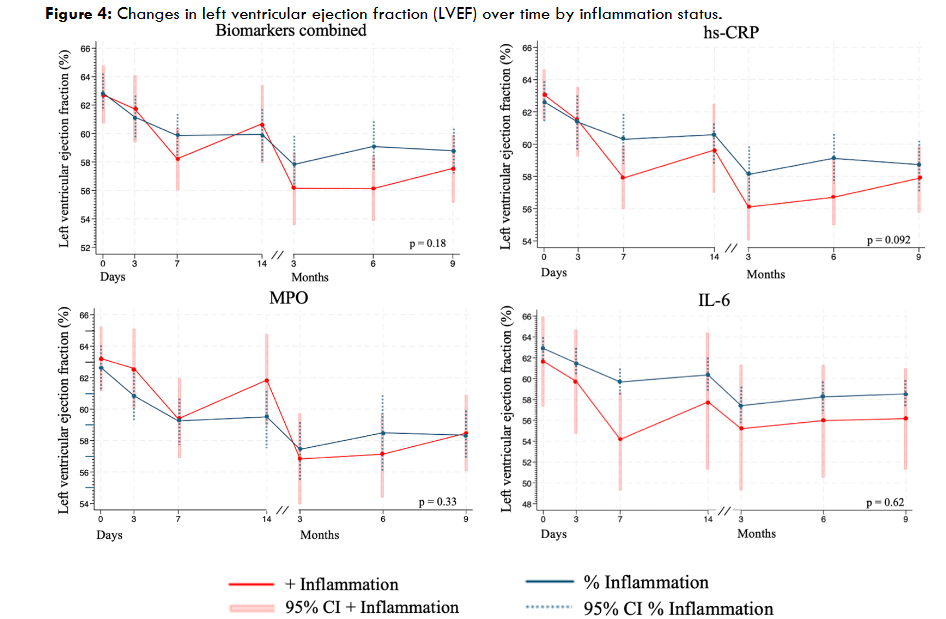
Figure 4 illustrates the change in LVEF over time, categorized by inflammation status, for both combined and individual biomarkers. For all biomarkers, a decline in LVEF in both groups was observed over time, but it remained within the normal range. For the combined biomarkers (4a), a decline in LVEF was observed in both groups, with a tendency toward lower values in the inflammation group. However, the difference was not statistically significant (p=0.18). For hs-CRP (4b), a similar pattern was observed, with lower LVEF in the inflammation group, though this difference was not significant (p=0.092). For MPO (4c), only minor differences were observed between the groups, and these were not statistically significant (p=0.33). For IL-6 (4d), an early decline in LVEF was noticeable in the inflammation group, but the difference diminished over time and remained statistically insignificant (p=0.62). Overall, LVEF tended to be lower in the inflammation group, but no statistically significant differences were observed.
3.5 ASSOCIATION BETWEEN INFLAMMATORY RESPONSES AND CARDIOTOXICITY
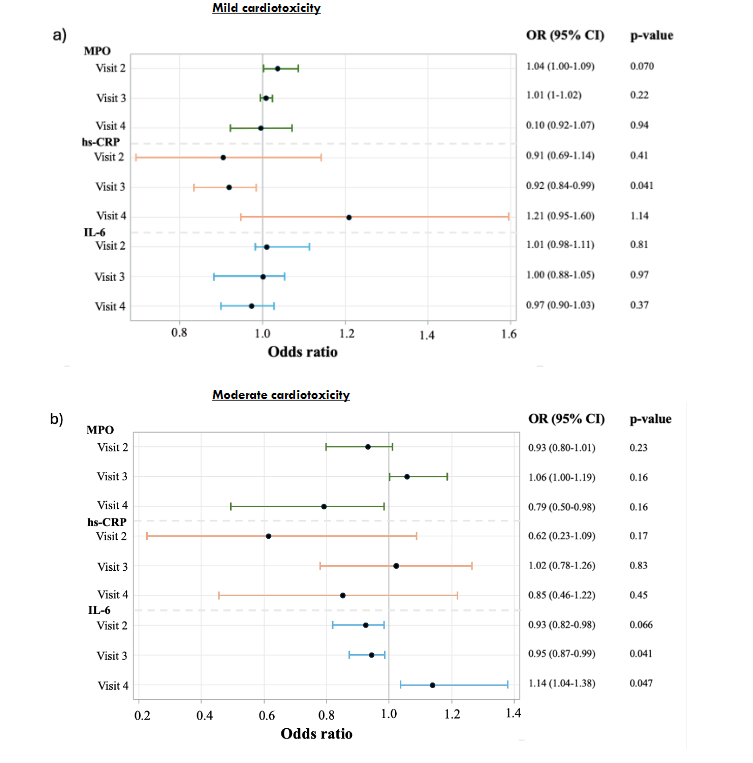
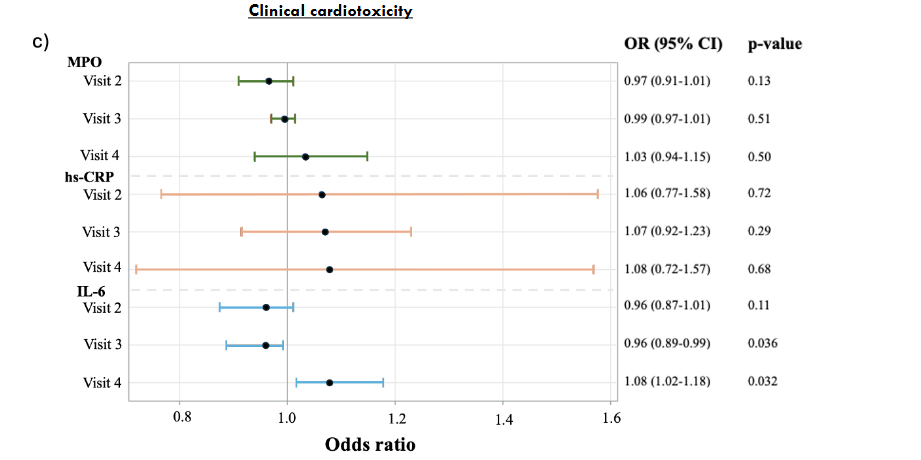
Figure 5: Odds ratios across cardiotoxicity categories.
Figure 5 presents the results of the logistic regression for the biomarkers odds ratios (OR) and 95% confidence intervals across three cardiotoxicity groups: mild, moderate and clinical. In the cohort, 5 patients either paused or discontinued trastuzumab treatment due to a decline in LVEF on MUGA scan and clinical decision making and were therefore defined as having clinically cardiotoxicity. Additionally, 14 patients met the definition of asymptomatic mild cardiotoxicity, while 6 patients met the definition of asymptomatic moderate cardiotoxicity. No patients met the criteria for asymptomatic severe cardiotoxicity. For mild cardiotoxicity (Figure 5a) the analysis identified no statistically significant association for MPO or IL-6. At visit 3, hs-CRP demonstrated a statistically significant negative association (OR = 0.92, 95% CI: 0.84–0.99, p = 0.041), while no significant associations were observed for hs-CRP at the remaining visits. For moderate cardiotoxicity (Figure 5b), the analysis showed opposing odds ratios for MPO and hs-CRP across the different visits, with a weak positive association at visit 3 and weak negative associations at visit 2 and 4, though none were statistically significant. At visit 2 IL-6 indicated a possible weak negative trend, though it was not significant. Additionally, IL-6 at visit 3 demonstrated a statistically significant negative association (OR = 0.95, 95% CI: 0.87–0.99, p = 0.041), while at visit 4, it showed a statistically significant positive association (OR = 1.14, 95% CI: 1.04–1.38, p = 0.047). For clinical cardiotoxicity (Figure 5c), the analysis showed no statistically significant associations for MPO and hs-CRP at any of the visits. For IL-6 at visit 2, no statistically significant association was observed. In contrast, IL-6 demonstrated statistically significant associations at visit 3 and 4, with negative association at visit 3 (OR = 0.96, 95% CI: 0.89–0.99, p = 0.036) and positive association for visit 4 (OR = 1.08, 95% CI: 1.02–1.18, p = 0.032).
4. Discussion
In this study, we investigated hs-CRP, MPO, and IL-6 as early biomarkers for cardiotoxicity in breast cancer patients undergoing trastuzumab treatment. No significant correlation was found between early elevated biomarker levels and deterioration in GLS or LVEF during the treatment period. Additionally, no clear association was identified between elevated biomarkers and subsequent cardiotoxicity. However, IL-6 measured at visit 4 was significantly associated with both moderate and clinical cardiotoxicity, suggesting a possible link between elevated IL-6 levels and the development of cardiotoxicity. Previous studies have investigated biomarkers for cardiotoxicity in patients undergoing HER2 target therapy. Onitilo et al. conducted blood samples every three weeks for up to one year after trastuzumab initiation in a prospective cohort study of 54 patients. Their findings indicated that normal hs-CRP levels might be associated with a reduced risk of LVEF decline, suggesting a high negative predictive value for hs-CRP. However, among patients with elevated hs-CRP levels, both specificity and positive predictive value were low, precluding a definitive association between elevated hs-CRP and cardiotoxicity. Additionally, Putt et al. assessed hs-CRP levels every three months after trastuzumab initiation in 78 patients in a prospective cohort study, and found no significant correlation with cardiotoxicity, which is consistent with our results. The absence of a clear association between hs-CRP levels and cardiotoxicity highlights the limited effectiveness of hs-CRP as a predictive biomarker for trastuzumab-induced cardiotoxicity. A prospective cohort study of Demissei et. al. with 323 patients found an association between increases in MPO and cardiac dysfunction and suggest that MPO potentially could serve as a biomarker for predicting cardiotoxicity in treatment regimens involving anthracycline and trastuzumab. However, their study differed in timing, with blood samples collected six weeks after trastuzumab initiation, thereby not focusing on the acute response. Additionally Putt et. al. reported that increases in MPO were associated with cardiotoxicity. However, as mentioned, this study measured biomarkers every 3. months after the initiation of trastuzumab, meaning they did not evaluate the acute response. In contrast, our study did not find an association between increased MPO levels and reductions in GLS, LVEF or development of cardiotoxicity. It is possible that MPO levels do not increase during the acute phase but rise at a later stage when myocardial changes begin to occur. To our knowledge, no other studies have attempted to investigate an association between IL-6 and cardiotoxicity. Comparisons between studies are complicated due to the lack of consensus on the definition of cardiotoxicity. Additionally, studies use different reference values to define an inflammatory response. Furthermore, this study investigates biomarkers within the first 14 days after initiating trastuzumab treatment, whereas other studies focus on later inflammatory responses. These considerations suggest the need to evaluate biomarkers at both acute and late stages of treatment to better understand their role in cardiotoxicity. It is possible that the timing of blood sample collection in this study was too early to detect clinically meaningful associations. Acute inflammation might represent a transient physiological response, and may not inherently contribute to cardiac damage. In contrast, prolonged inflammation could have a more significant impact on cardiac function over time. Furthermore, the 9-month follow-up period may not be sufficient to capture the potential long-term effects of inflammation on cardiotoxicity, highlighting the importance of extended monitoring in future studies.
4.2 ANTHRACYCLINE TREATMENT PRIOR TO TRASTUZUMAB
Prior to trastuzumab treatment, patients in this cohort received anthracycline therapy, a known cardiotoxic agent with potential delayed cardiotoxic effects that can manifest years after exposure. Prior studies indicate that combining trastuzumab with prior anthracycline therapy significantly increases the risk of cardiotoxicity. Thus, patients in this study may have been predisposed to a higher risk of developing cardiotoxicity, including declines in LVEF and GLS, due to prior anthracycline exposure. In this study five patients were identified as having clinical cardiotoxicity, with onset ranging from 3 to 11 months post-treatment initiation. This observation supports the idea that clinically relevant decline in LVEF associated with trastuzumab is progressive and develops over time rather than occurring acutely. However, it remains unclear whether this is due to anthracycline therapy, trastuzumab therapy, or their combined effects.
4.3 RESULTS CONSIDERATION
A potential reason for the lack of an observed association between elevated biomarkers and declines in GLS or LVEF could be the method used to define the inflammatory response. The cohort was stratified into inflammation groups based on cut-off values established for each individual biomarker. These were established based on judgment, as no clearly defined or standardized reference values exist. However, this approach raises the possibility, that some patients may have been misclassified. To address this concern, we performed sensitivity analyses using both higher and lower cut-off values for the biomarkers, which did not significantly impact the outcome, thereby supporting the validity of the cut off value despite the potential limitations. Following this analysis, we also stratified the cohort based on cardiotoxicity categories, but no definitive association was observed. The odds ratios were close to 1 across all groups, with some biomarkers showing opposing trends and wide confidence intervals. This may be attributable to the limited cohort size or the possibility that the biomarkers alone are insufficient as predictive markers. It is noteworthy that there is a difference between patients who were clinically diagnosed with cardiotoxicity during the treatment period and those who, according to the ECS definition, met the criteria for cardiotoxicity. This difference arises in the basis of several factors. Clinical cardiotoxicity was assessed based on MUGA scans and clinical decision-making, whereas the classification of mild and moderate cardiotoxicity relied on echocardiographic measurements. Differences in these two methods, both in execution and analysis, can lead to varying results. A well-documented challenge of echocardiography is the high inter- and intra-observer variability in LVEF measurements, which can reduce the accuracy of the results. Another potential explanation is that clinical cardiotoxicity is based on local guidelines and a subjective clinical evaluation. Among the patients in this cohort, maintaining oncological treatment was highly prioritized, which may have influenced the classification of the group with clinical cardiotoxicity.
4.4 LIMITATIONS
A primary limitation of the study is the small cohort size, consisting of only 45 patients, which reduces statistical power and limits generalizability, especially given the few cases of cardiotoxicity. The single-center design further restricts applicability of findings to a broader population. The homogeneous study population limits applicability to other treatment regimens or diverse patient groups. Variability in LVEF measurements also weakens precision, which could be improved by using cardiac magnetic resonance imaging. Although the overall proportion of missing data is relatively low (4.5 % across all biomarkers, GLS and LVEF measures), it cannot be ruled out that this may have influenced the results. A substantial proportion (51.9 %) of the missing data relates to visit 2, likely reflecting the clinical impact of the initiation of trastuzumab and taxan treatment, as this time point occurs three days after the first administration, when patients may be more vulnerable to treatment-related effects. The results are therefore considering to be explorative, and the small sample size increases the risk of a type II error, potentially leading to false-negative findings.
4.5 CLINICAL RELEVANCE
Cardiotoxicity can lead to reduced cardiac function, resulting in reduced functional capacity and quality of life for patients. Currently, patients undergoing trastuzumab treatment are monitored every three months using imaging techniques to assess LVEF, which typically detect cardiac dysfunction after damage has occurred. Therefore, it is relevant to explore methods that can predict cardiac dysfunction at an earlier stage. HER2 targeted therapy is life-saving for patients, making it necessary to consider the clinical relevance and potential contributions of biomarkers in risk assessment for cardiotoxicity. Biomarkers alone are unlikely to serve as definitive indicators for treatment discontinuation, as the therapeutic benefits of life-saving interventions must be prioritized over the potential risk of cardiac dysfunction. However, in the early detection of cardiotoxicity biomarkers could be included in an integrated risk assessment. This may support the initiation of cardioprotective medication concomitant with HER2 therapy, lead to more frequent monitoring of the patient and prevent interruption and delays in cancer treatment. For future studies, it would be relevant to include a larger cohort, incorporate groups with different treatment regimens, and investigate the long-term consequences of the treatment in patients.
5. Conclusion
This study investigated the association between early changes in inflammatory biomarkers and cardiotoxicity in HER2-positive breast cancer patients treated with trastuzumab in a small single center cohort. None of the inflammatory biomarkers were significantly associated with reductions in global longitudinal strain (GLS) or left ventricular ejection fraction (LVEF). No clear associations were identified between biomarkers and grades of cardiotoxicity, though our results suggest that biomarkers alone during the acute phase, cannot serve as reliable early predictors or be effectively used for risk assessment of cardiotoxicity in this patient population. However, IL-6 levels after 14 days showed a potential association with an increased risk of cardiotoxicity. This trend could be of interest for further investigation in larger studies.
Disclosures: The authors have no conflicts of interest to declare.
Funding: The study was funded by the Danish Heart Foundation (grant number: 14-R97-A5188-22839 and 15-R99-A5940) and the Research Fond of the Region of Southern Denmark.
Acknowledgements: Odense Patient Explorative Network (OPEN) is acknowledged for their support. Biochemical analyses were performed by OPEN Lab, Odense University Hospital (OUH), financially supported by the Research Council at OUH, Odense, Denmark.
7. References
- Winters S, Martin C, Murphy D, Shokar NK. Breast Cancer Epidemiology, Prevention, and Screening. Prog Mol Biol Transl Sci. 2017;151:1-32. doi:10.1016/bs.pmbts.2017.07.002
- Slamon D, Eiermann W, Robert N, et al. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med. Oct 6 2011;365(14):1273-83. doi:10.1056/NEJMoa0910383
- Gusterson BA, Gelber RD, Goldhirsch A, et al. Prognostic importance of c-erbB-2 expression in breast cancer. International (Ludwig) Breast Cancer Study Group. J Clin Oncol. Jul 1992;10(7):1049-56. doi:10.1200/jco.1992.10.7.1049
- Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. Mar 15 2001;344(11):783-92. doi:10.1056/nejm200103153441101
- Trastuzumab for early-stage, HER2-positive breast cancer: a meta-analysis of 13 864 women in seven randomised trials. Lancet Oncol. Aug 2021;22(8):1139-1150. doi:10.1016/s1470-2045(21)00288-6
- Chen T, Xu T, Li Y, et al. Risk of cardiac dysfunction with trastuzumab in breast cancer patients: a meta-analysis. Cancer Treat Rev. Jun 2011;37(4):312-20. doi:10.1016/j.ctrv.2010.09.001
- Moja L, Tagliabue L, Balduzzi S, et al. Trastuzumab containing regimens for early breast cancer. Cochrane Database Syst Rev. Apr 18 2012;2012(4):Cd006243. doi:10.1002/14651858.CD006243.pub2
- Bouwer NI, Jager A, Liesting C, et al. Cardiac monitoring in HER2-positive patients on trastuzumab treatment: A review and implications for clinical practice. Breast. Aug 2020;52:33-44. doi:10.1016/j.breast.2020.04.005
- Murtagh G, Januzzi JL, Scherrer-Crosbie M, et al. Circulating Cardiovascular Biomarkers in Cancer Therapeutics-Related Cardiotoxicity: Review of Critical Challenges, Solutions, and Future Directions. J Am Heart Assoc. Nov 7 2023;12(21):e029574. doi:10.1161/jaha.123.029574
- Narang A, Addetia K. An introduction to left ventricular strain. Curr Opin Cardiol. Sep 2018;33(5):455-463. doi:10.1097/hco.0000000000000556
- Negishi K, Negishi T, Hare JL, Haluska BA, Plana JC, Marwick TH. Independent and incremental value of deformation indices for prediction of trastuzumab-induced cardiotoxicity. J Am Soc Echocardiogr. May 2013;26(5):493-8. doi:10.1016/j.echo.2013.02.008
- Eaton H, Timm KN. Mechanisms of trastuzumab induced cardiotoxicity – is exercise a potential treatment? Cardiooncology. Apr 25 2023;9(1):22. doi:10.1186/s40959-023-00172-3
- Ananthan K, Lyon AR. The Role of Biomarkers in Cardio-Oncology. J Cardiovasc Transl Res. Jun 2020;13(3):431-450. doi:10.1007/s12265-020-10042-3
- Ponde N, Bradbury I, Lambertini M, et al. Cardiac biomarkers for early detection and prediction of trastuzumab and/or lapatinib-induced cardiotoxicity in patients with HER2-positive early-stage breast cancer: a NeoALTTO sub-study (BIG 1-06). Breast Cancer Res Treat. Apr 2018;168(3):631-638. doi:10.1007/s10549-017-4628-3
- Sproston NR, Ashworth JJ. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front Immunol. 2018;9:754. doi:10.3389/fimmu.2018.00754
- Khan AA, Alsahli MA, Rahmani AH. Myeloperoxidase as an Active Disease Biomarker: Recent Biochemical and Pathological Perspectives. Med Sci (Basel). Apr 18 2018;6(2)doi:10.3390/medsci6020033
- Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. Sep 4 2014;6(10):a016295. doi:10.1101/cshperspect.a016295
- Chia YC, Kieneker LM, van Hassel G, et al. Interleukin 6 and Development of Heart Failure With Preserved Ejection Fraction in the General Population. J Am Heart Assoc. Jun 2021;10(11):e018549. doi:10.1161/jaha.120.018549
- Banke A, Schou M, Ewertz M, et al. Immediate evaluation of global longitudinal strain at initiation of trastuzumab treatment in breast cancer patients. Echocardiography. Oct 2021;38(10):1702-1710. doi:10.1111/echo.15190
- Senkus E, Kyriakides S, Ohno S, et al. Primary breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. Sep 2015;26 Suppl 5:v8-30. doi:10.1093/annonc/mdv298
- Lyon AR, López-Fernández T, Couch LS, et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J Cardiovasc Imaging. Sep 10 2022;23(10):e333-e465. doi:10.1093/ehjci/jeac106
- Onitilo AA, Engel JM, Stankowski RV, Liang H, Berg RL, Doi SA. High-sensitivity C-reactive protein (hs-CRP) as a biomarker for trastuzumab-induced cardiotoxicity in HER2-positive early-stage breast cancer: a pilot study. Breast Cancer Res Treat. Jul 2012;134(1):291-8. doi:10.1007/s10549-012-2039-z
- Putt M, Hahn VS, Januzzi JL, et al. Longitudinal Changes in Multiple Biomarkers Are Associated with Cardiotoxicity in Breast Cancer Patients Treated with Doxorubicin, Taxanes, and Trastuzumab. Clin Chem. Sep 2015;61(9):1164-72. doi:10.1373/clinchem.2015.241232
- Demissei BG, Hubbard RA, Zhang L, et al. Changes in Cardiovascular Biomarkers With Breast Cancer Therapy and Associations With Cardiac Dysfunction. J Am Heart Assoc. Jan 21 2020;9(2):e014708. doi:10.1161/jaha.119.014708
- Ades F, Zardavas D, Pinto AC, Criscitiello C, Aftimos P, de Azambuja E. Cardiotoxicity of systemic agents used in breast cancer. Breast. Aug 2014;23(4):317-28. doi:10.1016/j.breast.2014.04.002
- Cole GD, Dhutia NM, Shun-Shin MJ, et al. Defining the real-world reproducibility of visual grading of left ventricular function and visual estimation of left ventricular ejection fraction: impact of image quality, experience and accreditation. Int J Cardiovasc Imaging. Oct 2015;31(7):1303-14. doi:10.1007/s10554-015-0659-1

