Conservation of Stingless Bees: Biodiversity & Health Benefits
One biodiversity: Conservation of stingless bee bibliometrics and legal progress protecting pollinators; thus, their nest materials, microbiome, and active biomolecules
Patricia Vit, MSc, PhD¹²*, María Arague, MD, PhD³, Qibi Wang, MSc⁴, Flor Mora, MSc, PhD⁵, Cleofas Cervancia, MSc, PhD⁶, Ariel A Céspedes-Llave, MSc⁷, Carmen L Yurrita-Obiols, MSc, PhD⁸, Christopher A Mduda, MSc, PhD⁹, Mario A Gallardo, MSc¹⁰, Ingrid Aguilar, PhD¹⁰, Sadao Takahashi, MSc¹¹, Silvio Loayza, Eng¹²,¹³, Breno M Freitas MSc, PhD¹⁴, Mary-Michelle Moore, MSc¹⁵, Pierre Noiset, MSc, PhD¹⁶, Jason E Stajich, PhD¹⁷, David W Roubik, MSc, PhD¹⁸, Michael S Engel, PhD¹⁹
- Apitherapy and Bioactivity (APIBA), Food Science Department, Faculty of Pharmacy and Bioanalysis, Universidad de Los Andes, Mérida, Venezuela.
- Temporarily based at: Department of Microbiology and Plant Pathology, Institute for Integrative Genome Biology, University of California Riverside, Riverside, CA, USA.
- Laboratory of Molecular Microbiology, Department of Microbiology and Parasitology, Faculty of Pharmacy and Bioanalysis, Universidad de Los Andes, Mérida 5101,Venezuela.
- School of Ecology and Environment, Yunnan University, Kunming 650500, China.
- Department of Pharmacognosy and Organic Medicaments, Faculty of Pharmacy and Bioanalysis, Universidad de Los Andes, Mérida, Venezuela.
- Bee Program, University of the Philippines Los Baños, Laguna, Philippines.
- Instituto Experimental de Biología “Luis Adam Brignoni”, Universidad San Francisco Xavier de Chuquisaca, Dalence 51, Sucre, Bolivia.
- Instituto de Investigaciones Químicas, Biológicas, Biomédicas y Biofísica, Universidad Mariano Gálvez de Guatemala, Guatemala.
- Department of Crop Sciences and Beekeeping Technology, University of Dar es Salaam, Dar es Salaam, Tanzania.
- Centro de Investigaciones Apícolas Tropicales, Universidad Nacional (CINAT-UNA), Heredia, Costa Rica.
- Creando Valor Agregado para El Salvador (CREVAS), Japan International Cooperation Agency (JICA), Universidad de El Salvador (UES), San Salvador, El Salvador.
- Unidad de Gestión de Desarrollo Rural, Ministerio de Agricultura y Ganadería, Piñas, El Oro, Ecuador.
- Ministerio de Agricultura y Ganadería, Piñas, El Oro, Ecuador.
- Bee Unit, Department of Animal Science, Universidade Federal do Ceará, Fortaleza, Brazil.
- Teaching and Learning Department, Tomas Rivera Library, University of California Riverside, Riverside, CA, 92521, USA.
- Cellular and Organismic Networks, Faculty of Biology, Ludwig-Maximilians-Universität München, Großhaderner Str. 2-4, 82152 Planegg-Martinsried, Germany.
- Department of Microbiology and Plant Pathology, Institute for Integrative Genome Biology, University of California Riverside, Riverside, CA 92521, USA.
- Smithsonian Tropical Research Institute, Ancón, Panama.
- Museum of Natural History, New York, United States
-
P. Vit. Email [email protected]
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Vit, P., Arague, M., et al., 2026. One biodiversity: Conservation of stingless bee bibliometrics and legal progress protecting pollinators; thus, their nest materials, microbiome, and active biomolecules. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i2.7146
ISSN 2375-1924
Abstract
Stingless bees (Hymenoptera, Apoidea, Meliponini) are the most biodiverse group of social bees with more than 605 formally described species inhabiting the tropics. Protecting their biodiversity sounds challenging. A team work for humans in close contact with stingless bees: Those who take care of them in meliponaries, and dedicated multidisciplinary scientists, merging the best ancestral to innovative knowledge. The Scopus database was used for the first bibliometric study on conservation of stingless bees for most productive authors, institutions, countries, and sources, since 2000. A total of 359 documents were retrieved from 141 sources, published by 1463 authors in English (348), Spanish (10), Portuguese (6), Chinese, Indonesian and Thai (1), from 58 countries, no single-authored, citing 2350 references. Most productive authors and affiliations are from Brazil, Mexico, and Argentina. With its participation in 11 and 12 documents and impacting the theme with the most globally cited document, the United States and Brazil emerged as the leading producer of scholarly works. A steady growth on scientific publications on conservation of stingless bees reached maximum of 46 documents in 2024 and 44 in 2025. Bibliometrix was used to visualize our dataset in graphics on most cited documents, collaborative map, cloud of authors’ keywords, the most cited countries, multivariate analysis topic dendrogram plot which classified authors’ keywords in four clusters on: 1. Biodiversity loss (deforestation and climate change), 2. Stingless bee and honey, plant diversity for melissopalynology, distribution, pollen, and Heterotrigona itama, 3. Bee diversity, and 4. Pollination and pollinators’ ecological service needed to maintain biodiversity, including the most biodiverse stingless bee genus Melipona. Our review underscores policy advances and prospects for sustainably harnessing stingless bees, their active biomaterials, and microbiomes alongside the legal frameworks that support their conservation in Bolivia, Brazil, Costa Rica, Ecuador, El Salvador, Guatemala, the Philippines, and Tanzania.
Keywords
active metabolites, bibliometrics, biodiversity, conservation, microbiome, stingless bees
1. Introduction
Bees are one of the most diverse groups, as more than 20,700 bee species have been described worldwide. The well-known social honey bees (genus Apis) account for less than ten recognized species globally. Other social bees include about 250 species of bumblebees and over 600 species of stingless bees. These bees have co-evolved with flowering plants, developing a mutualistic relationship since their appearance on earth more than 100 million years ago, becoming one of the main pollinators. Bees play a fundamental role in ecosystems, since they ensure the reproduction of numerous plants and the maintenance of biodiversity. The presence and activity of stingless bees reflect the availability of diverse foraged plant resources and microbiomes, so they are health indicators of the stingless bee colony, the habitat, and potential therapeutic materials of biotic (botanical, entomological, and microbial) origins, and less studied abiotic components limited to elements of the periodic table.
Stingless bees (Hymenoptera; Apidae; Apinae; Meliponini) host such a biodiversity among honey making bees. More than 605 global species evidence admirable adaptations along their natural life. Biological communities comprise microorganisms, plants, and animals. Biodiversity has been partitioned into genetic, species, ecosystem, and functional diversity. One biodiversity could be a concept embracing the variety of life in a given ecosystem –Amazonian forest, a marine coast, the Andes, or an urban habitat– having genetic descriptors, number of species, their interactions within communities and the environment. A pool of diverse genes –genetic diversity– favor adaptations to environmental changes and resistance to diseases. Richness of species inform speciation processes were needed for survival. A wide variety of genes within a species, provides the raw material for natural selection and evolution. This diversity is crucial for long-term survival and adaptability. Therefore, all ecological knowledge used to protect natural products is needed for availability of medical resources, such as the materials produced by stingless bees in their nests.
Tropical ecosystems are known to have the highest biodiversity in our planet, having warm climate, rainfall, and solar energy year-round, supporting a vast biodiversity of plants and animals. Corcovado National Park in Costa Rica, and Yasuní National Park in the Ecuadorian Amazon are known to host the higher biodiversity in the planet. Threatening vegetation reduces availability of food and nesting sites, causing inbreeding and genetic loss of stingless bees. Particularly, a deforestation rate of 12.2 km2/year is not permitted in environmental agreements to overcome climate change, as well as invasions and mining on native people lands, and conversion to pasture increasing deforestation rates. Preliminary studies on the Brazilian Amazon have assessed a decline of 95 % in meliponines species for the Carajás National Forest. Expansion of protected areas is recommended for Meliponines visiting 756 species of plants having economic and ecological importance in Legal Amazon, mainly concentrated along the largest rivers of the Amazon basin. Biodiversity loss has a tremendous impact on humanity, and loosing pollinators reflects a risk in food security, because they feed populations and change landscapes for global crops. Additionally, their ancestral medicinal uses can be affected if they become not available in a region.
A bibliometric review was done using the Scopus database, covering research published in the period 2000 to 2025. Top ten prominent authors, institutions, countries, sources, subject areas and financial sponsors were ranked according to the number of documents retrieved. Diverse visualizations of the dataset were selected using Bibliometrix, to show authors’ keywords word cloud, tree map, topic dendrogram, most cited authors, sources, and countries, collaborative worldwide map, single or multiple country publications of corresponding author’s countries, and a three-field-plot for country, source, and author. This paper presents the legal frameworks relevant to stingless bee conservation in Bolivia, Brazil, Costa Rica, Ecuador, El Salvador, Guatemala, the Philippines, and Tanzania. Policy developments and opportunities for sustainable utilization of stingless are highlighted. Therefore, the purpose of this study is to perform the first bibliometric analysis on the conservation and sustainable use of stingless bees, aiming to identify the most productive authors, institutions, and countries in the field. The scope of the article comprises publications from 2000 to 2025 addressing stingless bee conservation, including their use in human health. Safeguarding biomaterials of phytochemical origin and biotransformations by microbiomes associated with stingless bees, as well as highlighting potential threats and protective legislation for stingless bees found in the tropics worldwide, is our call in this contribution.
2. Methodology of the bibliometric study
2.1 Scopus database
The bibliometric search was carried out in the Scopus database, in the “TITLE-ABS-KEY” field using the operator AND, on November 8, 2025. TITLE-ABS-KEY ( conservation AND stingless AND bee ) A total of 359 documents were retrieved from 2000 to 2025, 299 articles, 25 reviews, 19 book chapters, 6 conference papers, and 1 books. The documents of published research in our dataset were ranked in Scopus for authors, affiliations, countries, sources, subject areas, and funding sponsors. The absolute number of documents was exported in a CSV Excel file for the Bibliometrix statistical analysis and visualization.
2.2 Bibliometrix statistical analysis and visualization
The CSV Excel file containing the Scopus search results was downloaded in the Bibliometrix Bibliometrix R package created by Aria and Cuccurullo (2017) in R Version 4.2.1 and the Biblioshiny App. We investigated correlations and classifications of authors’ keywords using multivariate factorial analysis for topical dendrograms by Hierarchical Cluster Analysis (HCA). Plots and Excel files were saved in files for the selected visualizations presented here.
3. Bibliometric analysis
3.1 Annual publication trends
The annual publication trend on conservation of stingless bee research is visualized in
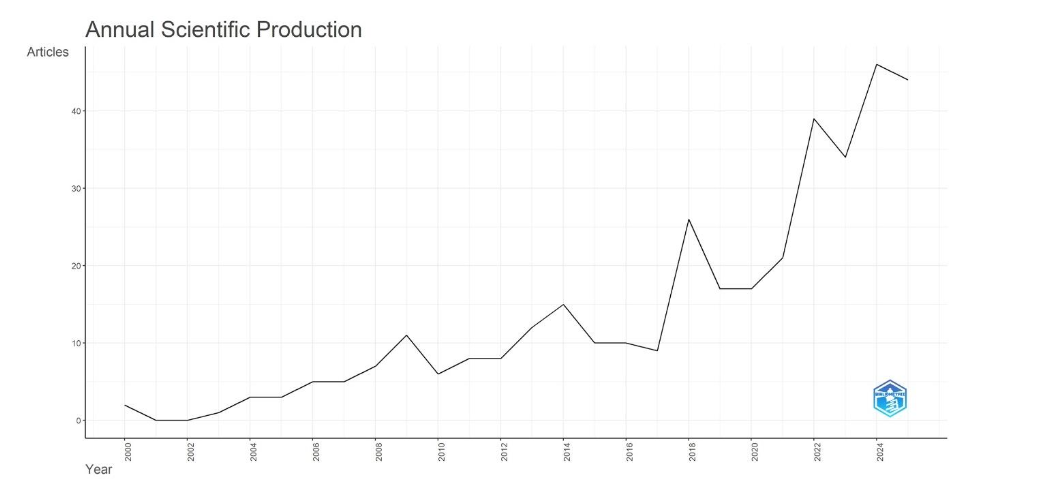
. The first document was published in 2000, showing a growing pattern up to 2025 with 44 documents.
3.2 Main information of the retrieved documents
The major information of our dataset is presented in
| Main information of the dataset bibliometric descriptors | Counts of all documents |
|---|---|
| Timespan | 2000:2025 |
| Sources (Journals, Books, etc) | 141 |
| Documents | 359 |
| Annual Growth Rate % | 13.6 |
| Document Average Age | 6.23 |
| Average Citations per Document | 30.77 |
| References | 2350 |
| Document Contents | |
| Keywords Plus (ID) | 1541 |
| Author’s Keywords (DE) | 1054 |
| Authors | 1463 |
| Authors of Single-Authored Documents | 0 |
| Authors Collaboration | |
| Single-Authored Documents | 0 |
| Multi-Authored Documents | 1463 |
| Co-Authors per Document | 9.57 |
| International Co-Authorships % | 29.53 |
| Document Types | |
| Article | 299 |
| Review | 25 |
| Book chapter | 19 |
| Conference paper | 6 |
| Note | 2 |
| Book | 2 |
| Editorial | 2 |
| Note | 2 |
| Conference review | 1 |
| Erratum | 1 |
| No. of languages | |
| Chinese | 1 |
| English | 348 |
| Indonesian | 1 |
| Portuguese | 6 |
| Spanish | 10 |
| Thai | 1 |
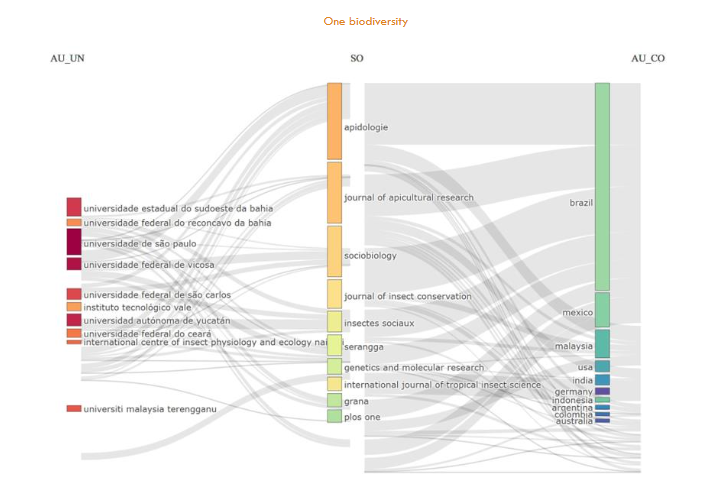
3.3 Most productive authors
| Ranking | NP | Microbiome of stingless bees and nest materials | Author | Affiliation, city | Country |
|---|---|---|---|---|---|
| 1 | 18 | Imperatriz-Fonseca VL | Universidade de São Paulo, São Paulo | Brazil | |
| 2 | 16 | Quezada-Euan JJG | Autonomous University of Yucatan UADY, Merida | Mexico | |
| 3 | 12 | Woldschmidt AM | Universidade Estadual do Sudoeste da Bahia | Brazil | |
| 4 | 11 | Francoy TM | Universidade de São Paulo, São Paulo | Brazil | |
| 5 | 9 | Giannini TC | Instituto Tecnológico Vale, Belem | Brazil | |
| 6 | 8 | Blochtein B | Pontifícia Universidade Católica do Rio Grande do Sul | Brazil | |
| 7 | 8 | Nunes LA | Faculdade Tecnologia e Ciencias, Jequie | Brazil | |
| 8 | 7 | Arias MC | Faculdade de Tecnologia e Ciências-FTC, Jequie | Brazil | |
| 9 | 7 | Freitas BM | Universidade Federal do Ceara, Fortaleza | Brazil | |
| 10 | 6 | Alves DA | Universidade de São Paulo, São Paulo | Brazil |
3.4 Most Productive Affiliations
Brazil has 8/10 of the most productive top ten affiliations ranked in
| Ranking | NP | Institution, city | Country |
|---|---|---|---|
| 1 | 47 | Universidade de São Paulo, São Paulo | Brazil |
| 2 | 23 | Universidad Autonoma de Mexico, Mexico DF | Mexico |
| 3 | 17 | Universidade Federal de Vicosa | Brazil |
| 4 | 16 | Instituto Tecnologico Vale | Brazil |
| 5 | 13 | Empresa Brasileira de Pesquisa Agropecuária – Embrapa | Brazil |
| 6 | 13 | Universidade Estadual do Sudoeste da Bahia | Brazil |
| 7 | 12 | Universidade Federal do Ceará | Brazil |
| 8 | 11 | Consejo Nacional de de Investigaciones Científicas y Técnicas | Argentina |
| 9 | 11 | Universidade Federal do Reconcavo da Bahia | Brazil |
| 10 | 11 | Universidade Federal do São Carlos | Brazil |
3.5 Most productive countries and collaborative map
The most prolific countries for conservation of stingless bee publications ranked in the Scopus database (
| Ranking | NP | Country |
|---|---|---|
| 1 | 154 | Brazil |
| 2 | 52 | Mexico |
| 3 | 31 | United States |
| 4 | 21 | Malaysia |
| 5 | 19 | Germany |
| 6 | 17 | Indonesia |
| 7 | 15 | Belgium |
| 8 | 15 | Colombia |
| 9 | 15 | India |
| 10 | 15 | United Kingdom |
) were Brazil (154), Mexico (52), USA (31), Malaysia (21), Germany 19), Indonesia (17), and 15 documents from Belgium, Colombia, India, and the United Kingdom. A total of 58 countries participated in our dataset, including Argentina (14), Australia (13), Spain (12), Kenya (11), Thailand and Costa Rica (9), Netherlands and Canada (8), Panama and China (6), Tanzania, Portugal, Guatemala, and France (5), Uganda, South Africa, Philippines, Bolivia and Austria (3), Vietnam, Sweden, Peru, Italy, Ghana, Ethiopia, Ecuador, Congo, Chile, and Bulgaria (2), and one document for Zimbabwe, Venezuela, United Arab Emirates, Turkey, Switzerland, Sri Lanka, South Korea, Singapore, Pakistan, Japan, Iran, Hong Kong, Democratic Republic Congo, Cameroon, Burundi, Burkina Faso, Brunei Darussalam, and Botswana.
The most globally ten cited countries for conservation of stingless bee in the Bibliometrix plot (Fig. 2) are Germany (5412), Brazil (1985), USA (646), Mexico (595), United Kingdom (498), Australia (189), Malaysia (122), France (106), Japan (88), and Panama (76).
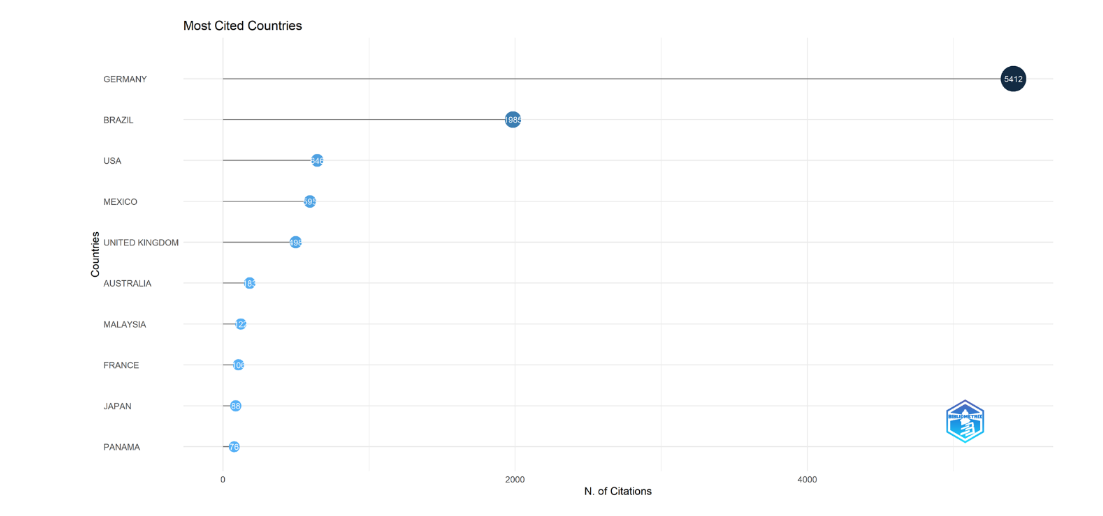
) are Germany (5412), Brazil (1985), USA (646), Mexico (595), United Kingdom (498), Australia (189), Malaysia (122), France (106), Japan (88), and Panama (76). The bubble size is proportional to the number of documents, and the blue color shade corresponds to the total citations per year, darker blue for more citations than lighter blue.
The collaboration between countries sharing publications on conservation of stingless bee research in the period 2000 to 2025 was visualized in the country collaborative map of
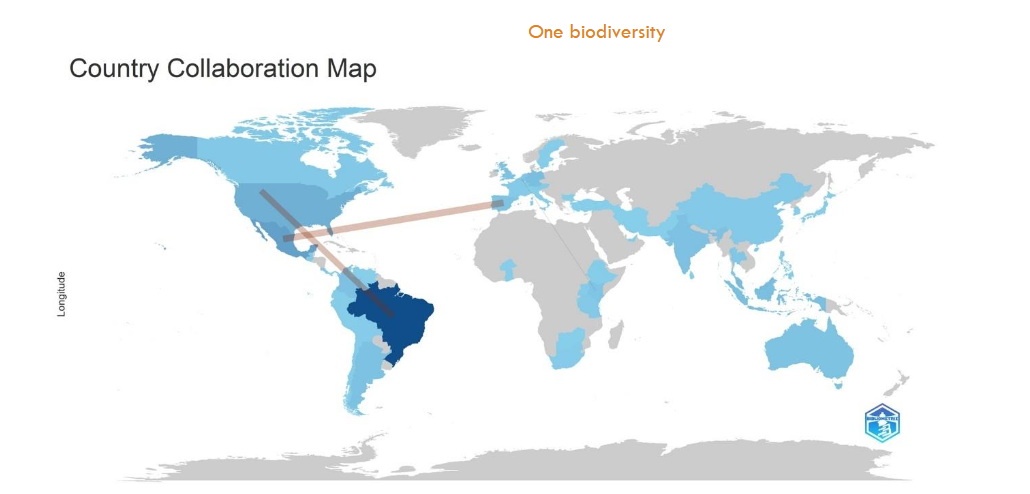
. The highest collaborative frequency was between Brazil and USA, and for Spain and Mexico.
3.6 Most productive sources
In
| Ranking | NP | Source | (h index, Quartile, impact score) | Editorial, Country |
|---|---|---|---|---|
| 1 | 115 | Apidologie | (h 105, Q1, 2.23) | Springer-Verlag Italia s.r.l., Italy |
| 2 | 92 | Journal of Apicultural Research | (h 81, Q1, 2.08) | Taylor and Francis Ltd., United Kingdom |
| 3 | 64 | Sociobiology | (h 44, Q4, 0.65) | Universidade Estadual de Feira de Santana, Brazil |
| 4 | 56 | Journal of Insect Conservation | (h 76, Q1, 2.46) | Springer Nature, Switzerland |
| 5 | 54 | International Journal of Tropical Insect Science | (h 35, Q2, 1.45) | Springer Nature, Switzerland |
| 6 | 49 | Stingless Bee Nest Cerumen and Propolis | Springer Nature, Switzerland | |
| 7 | 46 | Serangga | (h 11, Q3, 0.64) | Penerbit Universiti Kebangsaan Malaysia, Malaysia |
| 8 | 44 | Insectes Sociaux | (h 70, Q2, 1.18) | Springer Science and Business Media Deutschland GmbH. Switzerland |
| 9 | 44 | Plos One | (h 487, Q1, 2.67) | Public Library of Science, United States |
| 10 | 40 | Grana | (h 47, Q3, 1.02) | Taylor and Francis Ltd., United Kingdom |
3.7 Most productive Scopus subject areas on conservation and stingless bee research since 2000
The documents on conservation of stingless bees were most frequently published in eight subject areas of the Scopus database, covering the following percentages: 51.8% Agricultural and Biological Sciences, 20.2% Environmental Science, 8.2% Biochemistry, Genetics and Molecular Biology, 3.8% Multidisciplinary, 3.4% Social Sciences, 2.9% Medicine, 2.7% Earth and Planetary Sciences, 2.1% Engineering, and 3.2% Others.
3.6.1 Author’s keywords
A selection of the top 25 authors’ keywords used in the scientific literature on conservation of stingless bees’ research (2000–2025) was done. In

we show a word cloud using authors’ keywords, with words of higher frequencies occupying the center of the cloud with larger letters, and words with lower frequencies in the periphery or between lines with smaller letters: stingless bees (83), conservation (63), stingless bee (55), meliponini (54), meliponiculture (38), pollination (22), pollinators (21), biodiversity (17), honey (13), Apidae (11), bees (11), genetic diversity (11), melipona (11), melissopalynology (10), pollen (10), pollinator (10), diversity (9), geometric morphometrics (9), heterotrigona itama (9), microsatellites (9), climate change (8), deforestation (8), hymenoptera (8), distribution (7), and mexico (7).
The word cloud is free shape but the tree map is a box arranged into contrasting colored boxes sized according to the frequency of author’s keywords in descending order. The tree map in
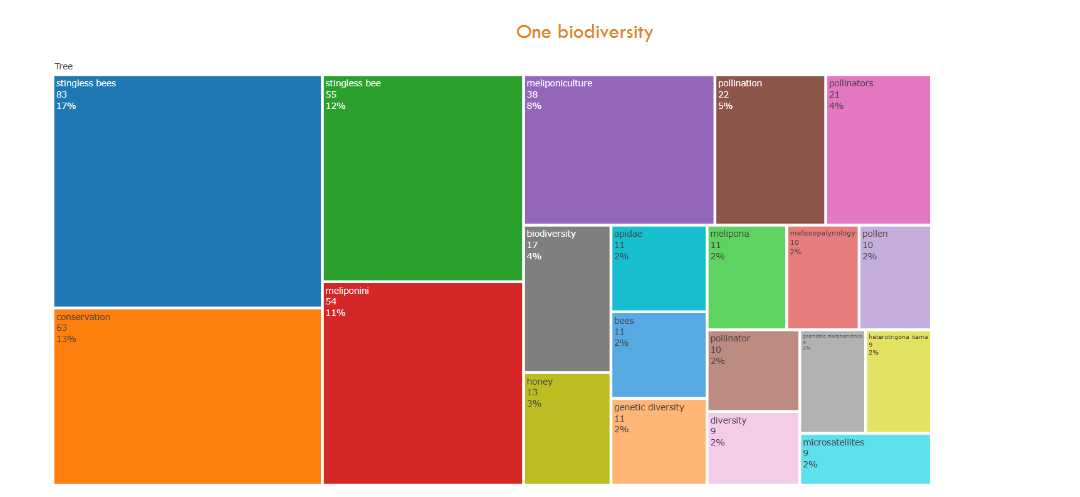
shows the top 20 higher frequencies of the authors’ keywords in the dataset, and informs their corresponding percentages. In the left half of the table, four words represent more than 50% frequency, stingless bee (83, 17%), conservation (63, 13%), stingless bee (55, 12%), and meliponini (54,11%). In the right side of the table, 16 words were used less frequently, meliponiculture (38, 8%), pollination (22, 5%), pollinators (21, 4%), biodiversity (17, 4%), honey (13, 3%), apidae (11, 2%), bees (11, 2%), genetic diversity (11, 2%), Melipona (11, 2%), melissopalynology (10, 2%), pollen (10, 2%), pollinator (10, 2%), diversity (9, 2%), geometric morphometrics (9, 2%), heterotrigona itama (9, 2%), microsatellites (9, 2%).
A dendrogram is a tree diagram showing hierarchies (order, ranks, categories, similarities and dissimilarities) in clusters (branches grouping variables to be interpreted intellectually). This plot uses the analogy of a tree with branches and a common shared root, recently represented upside down (like a fork), with the variable descriptors in the X axis, and the height (distance) in the Y axis. In
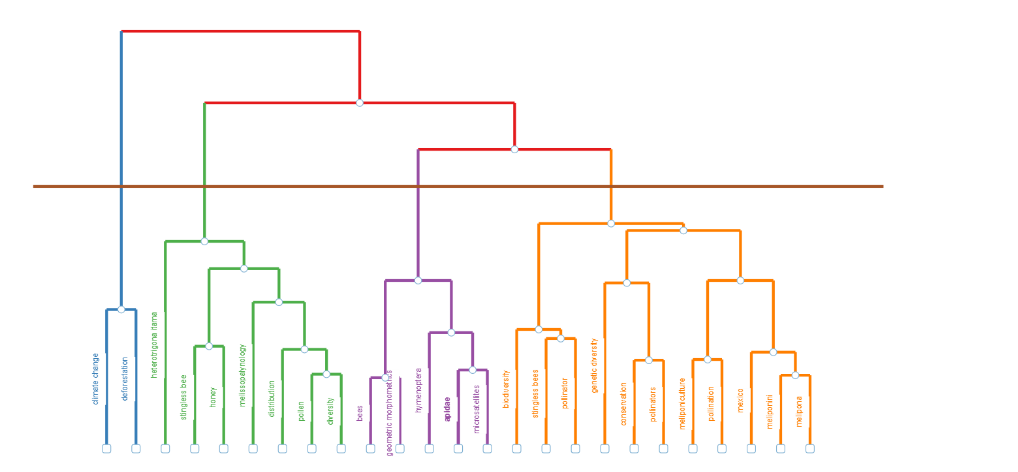
a topic dendrogram for authors’ keywords shows four clusters for 25 terms, obtained after factorial analysis using hierarchical cluster analysis (HCA). The blue cluster consisted in two related keywords for biodiversity loss, such as deforestation and climate change. The green cluster has two subclusters, one for stingless bee and honey, the other related with plant diversity for melissopalynology, distribution, pollen, and diversity. The stingless bee Heterotrigona itama is also included in the green cluster. The violet cluster is related to bee diversity, with two subclusters for bees, geometric morphometrics, and hymenoptera, Apidae, microsattelites. Finally, the orange cluster has three subclusters about pollination and pollinators ecological service needed to maintain biodiversity 1. biodiversity, stingless bee, and pollinator, 2. genetic diversity, conservation, pollinators, and 3. meliponiculture, pollination, mexico, meliponini, and Melipona; having one country and the most biodiverse genus included here.
3.6.2 Most globally cited documents
The plot of most globally cited documents in
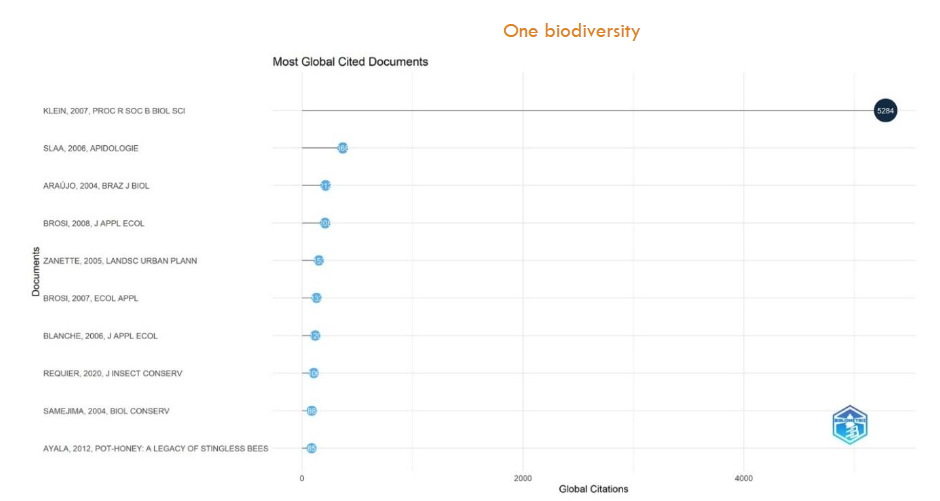
shows the top ten documents most cited in our Scopus dataset. Klein (2007) has been cited 5284 times and the second most cited Slaa (2006) has been cited 368 times.
In
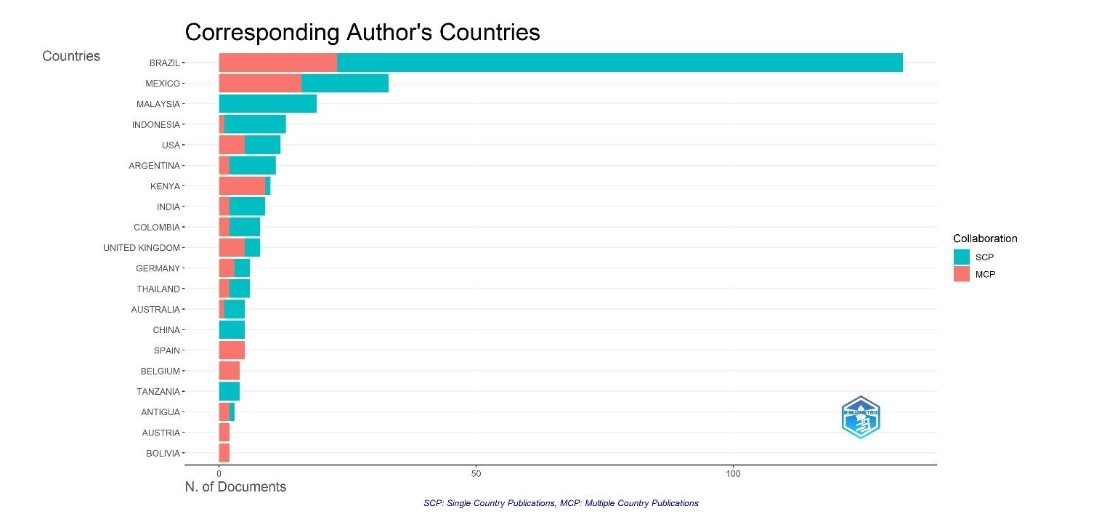
is visualized the composition of documents regarding the single or multiple country publications of corresponding author’s countries. At a glance, Brazil, the most productive country, has devoted 4.5 times to single country publications over multiple countries. The next country, Mexico, reaches a third of the publications of Brazil, but only half of the publications are single country and the other half for multiple country, like the United States. Malaysia only published single country, as well as China and Tanzania. On the contrary, Spain, Belgium, Tanzania, and Bolivia have selected multiple country publications. Indonesia, USA, Argentina, India, Colombia, Thailand, and Australia have more single than multiple country, whereas Kenya, the United Kingdom, and Antigua have more multi country teams of authors than single country.
are visualized four compartments of niche themes, motor themes, basic themes, and emerging or declining themes, showing the final stage on thematic evolution maps on publications of stingless bee conservation (2000–2025). Theme maps employ co-occurrence networks to group elements of Key Words Plus based on their relevance and development stage on publications of a theme. A theme’s significance is assessed by the centrality of its clusters and its thematic status, visualized by plots on the X-axis measuring the importance of a theme, and the Y-axis measuring cluster connection density to indicate the strength of the cluster networks, to foster the evolution of a theme. According to their position on the map, the themes are classified in motor, niche, emerging or declining, and basic themes. The basic theme of our dataset was represented by a unique cluster on conservation, stingless bees, and pollination; as well as the motor theme with one cluster about meliponiculture, stingless bees, and Heterotrigona itama –a conspicuous Malaysian stingless bee in the scientific literature, promoted to develop pot-honey for clinical use– Similarly, a unique cluster for the niche theme about well documented melissopalynology, native bees, and pollen, but somehow isolated in this analysis. Two clusters with one component on urbanization and mtdna resulted emerging themes.
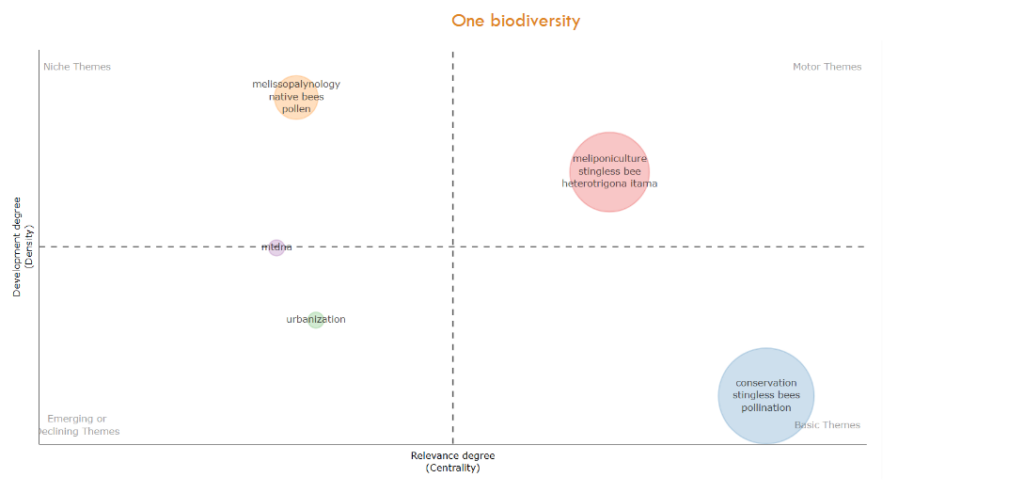
Fig. 10 Final slice for trends in thematic evolution maps on publications of stingless bee conservation (2000–2025).
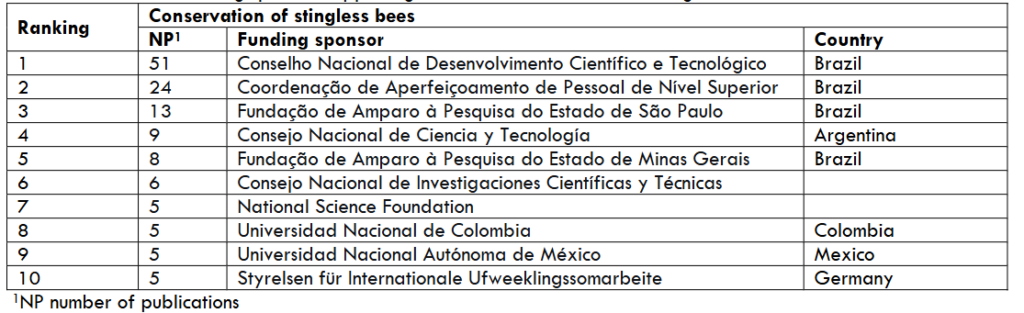
Funding sponsors of research are vital for conservation of stingless bees. In Table 7 it is evident that Brazil has more funding sponsors than any other country supporting conservation of the most biodiverse country with 259 species of stingless bees, distributed in the five regions of this country: 1. North, including the Amazon (197 species), 2. Central-West (99), 3. Northeast (97), 4. Southeast (70), and 5. South (36).¹⁴
Table 7. Most active funding sponsors supporting research on conservation of stingless bee since 2000
4. Repercussions of the environment in stingless bee nest materials
Stingless bees (Meliponini) exhibit profound sensitivity to environmental factors, which makes them excellent bioindicators of ecosystem health. A thriving population reflects ecological balance, whereas their decline serves as an early warning of environmental degradation. Their vulnerability to climatic variability, habitat loss, agrochemicals, pollution, pathogens, and competition underscores the urgent need to understand and mitigate the pressures threatening their survival.
4.1 Impact of Climate and Habitat Alteration
As tropical and subtropical species, stingless bees are highly susceptible to climatic shifts. Elevated temperatures can cause brood comb melting, larval mortality, and reduced foraging activity while extreme cold impairs brood development due to limited thermoregulation capacity. Humidity extremes further destabilize colonies, either through desiccation or fungal proliferation. Drought reduces floral resources, leading to malnutrition and decreased brood rearing, whereas heavy rains hinder foraging and flood ground-nesting species. Beyond climate, habitat loss and fragmentation represent the most critical threats worldwide. Deforestation eliminates nesting sites, monocultures create seasonal food deserts, and urbanization reduces floral diversity while increasing pollutant exposure.
4.2 Anthropogenic Stressors: Agrochemicals and Pollution
Stingless bees are particularly vulnerable to pesticides due to their smaller size and reduced pilosity compared to honeybees. Contaminated nectar and pollen can cause direct mortality, while sublethal effects—such as impaired navigation, reduced memory, and diminished queen fecundity—undermine colony viability. Pollution compounds these risks: air pollutants disrupt floral scent trails, impairing foraging efficiency, while soil and water contamination introduce heavy metals and toxins into nectar and pollen, generating chronic toxicity within nests.
4.3 Biotic Pressures and Invasive Species
Environmental imbalance also increases susceptibility to pathogens and parasites. Fungi such as Aspergillus spp. thrive in poorly ventilated nests, mites like Pyemotes spp. kill pupae in stressed colonies, and phorid flies consume brood and nest structures. Additionally, stingless bees face competition from invasive species. Honey bees often outcompete them for floral resources, while Africanized honeybees may directly usurp nesting cavities. Collectively, these pressures highlight the fragility of stingless bee populations and reinforce their role as sentinel species for monitoring ecosystem integrity. The significant geographical and seasonal variation in metabolite profiles are reported in studies. A comprehensive understanding of both botanical and microbial origins is therefore essential for standardizing these natural products for medicinal use and for developing conservation strategies that protect the diverse ecosystems sustaining these complex chemical factories.
The key classes of bioactive metabolites in stingless bee nest materials and their roles are summarized and integrated in
| Source / Origin | Key Metabolite Classes | Reported Bioactivity / Role | |
|---|---|---|---|
| Botanical Sources (External Environment) | Dalbergia spp. (Propolis) | Prenylated benzophenones, flavonoids | Potent antibacterial, antifungal (Campos et al., 2014). |
| Tualang, rainforest flora (Honey) | Flavonoids, phenolic acids, galangin | Antioxidant, Antibacterial (Abdullah et al., 2025). | |
| Microbial Sources (Internal Nest Environment) | Lactic acid bacteria (LAB) in pot-pollen | Aliphatic organic acids (acetic, lactic), bacteriocins | pH regulation, pathogen inhibition, preservative (Schwarz et al., 2023). |
| Yeasts and fungi in fermented pot-pollen | Ethanol, bioactive peptides, enzymes | Prebiotic, nutrient enhancement (Schwarz et al., 2023). |
5. Secondary metabolites in stingless bee materials of the nest, microbial diversity, and medicinal applications
Stingless bees collect nectar, pollen, plant resin and oils from plants and process them into pot-honey, pot-pollen, cerumen, and propolis. Phytochemicals such as flavonoids, phenolic acids, polyphenols, and terpenoids are secondary plant metabolites affected by environmental changes. Stingless bees and the materials derived from their nests have traditionally been studied in various tropical regions for their potential beneficial properties for human health. In recent years, scientific research has delved deeper into the analysis of the microbiome that resides in these bees and the materials they collect and process, such as honey, propolis, pollen, and cerumen. This research reveals that the associated microbial richness and diversity not only contribute to the health and survival of bee communities but are also a natural source of bioactive compounds with potent antimicrobial, antioxidant, anti-inflammatory, and immunomodulatory effects, giving these products beneficial and healing properties.
The microbiome plays a fundamental role in enriching these products with microbial secondary metabolites. When these metabolites combine with the bees’ own compounds, they generate unique biochemical synergies. This complexity has generated interest in the potential use of stingless bee products in integrative medicine, as well as in the development of innovative new therapeutic options capable of contributing to the fight against infectious diseases — especially those caused by multidrug-resistant pathogens — and of alleviate chronic inflammation, preventing oxidative stress and controlling metabolic dysfunctions. However, these medicinal applications must be considered in the context of biodiversity preservation, since the sustainability of stingless bee populations and the integrity of their microbiome are essential for ensuring the production and quality of these natural resources. The interplay between human health, functional microbiology, and ecological conservation is a promising interdisciplinary frontier that could improve biomedicine and environmental protection.
5.1 Antimicrobial activity
The microbiome of stingless bees and the materials in their nests constitute a reservoir of beneficial microorganisms and metabolites that exhibit a wide range of antimicrobial activities against bacteria, fungi, and viruses. In vitro studies and experimental models have supported this hypothesis, reaffirming the therapeutic potential of these products in combating infections, particularly in the context of bacterial resistance. The microbiome of Meliponini bees contains bacteria such as Lactobacillus, Bifidobacterium, and Bacillus, which produce bacteriocins, organic acids, and enzymes that directly inhibit pathogens. These microbial communities benefit the health of the colonies and enrich the nest materials with multifunctional bioactive compounds. The hydrogen peroxide, phenolic metabolites, and microbial enzymes found in honey from stingless bees act synergistically to inhibit both Gram-positive and Gram-negative bacteria, as well as pathogenic fungi. This spectrum includes multidrug-resistant strains, making these products important natural antimicrobial agents. Similarly, propolis and cerumen, which are rich in phenolic compounds and flavonoids, exhibit significant bactericidal and fungicidal properties. Their effectiveness increases when they are combined with metabolites produced by the associated microbiota, forming a biochemical complex with a multipurpose antimicrobial action.
Recent research has highlighted the synergistic effect that occurs when honey and other bee products are associated with antibiotics. Some studies suggest that these natural products can increase the effectiveness of antibiotics, reducing the minimum inhibitory concentrations needed to eliminate resistant bacterial strains such as methicillin-resistant Staphylococcus aureus (MRSA) and carbapenemase-producing Pseudomonas aeruginosa. This is because the compounds in honey and propolis affect different microbial targets, complementing the mechanisms of action of conventional antibiotics. For instance, honey can alter bacterial membranes and biofilms, thereby facilitating antibiotic penetration and efficacy, while its phenolic metabolites can inhibit enzymes involved in antimicrobial resistance. In this context, honey and other bee products, working synergistically with antibiotics, also reduce the required dose of these drugs, which may decrease adverse effects and help contain the spread of antimicrobial resistance. Therefore, the integration of products from stingless bee nests into antimicrobial therapies could optimize treatments, especially for difficult-to-treat infections caused by multidrug-resistant pathogens.
It is important to note that the set of bioactive metabolites, which includes hydrogen peroxide, organic acids, flavonoids, terpenoids, and microbial enzymes, acts through various mechanisms. These include perforation and destabilization of the bacterial cell membrane, inhibition of protein and nucleic acid synthesis, alteration of intracellular metabolism, and generation of oxidative stress. They also include disruption and prevention of biofilm formation. These mechanisms reduce the likelihood of resistance development in pathogens and emphasize the importance of preserving microbial biodiversity in bee products to ensure and maintain these therapeutic effects.
5.2 Antioxidant and anti-inflammatory potential
The bioactive factors present in the microbiome of stingless bees and their nests constitute a multifaceted source of compounds with remarkable antioxidant and anti-inflammatory properties. These mainly result from high levels of phenolic substances and flavonoids, which act as powerful free radical scavengers and protect cells against oxidative damage, a key factor in the development of various chronic diseases. Recent studies have shown that the antioxidant capacity of stingless bee honey is superior to that of honey from other bee species, thanks to the symbiotic microbiome’s production of antioxidant enzymes such as catalase and superoxide dismutase, which enhances the protective effect. Furthermore, the anti-inflammatory potential of these products is proved by their ability to reduce the production of pro-inflammatory mediators, such as certain cytokines and enzymes related to chronic inflammation. In vitro trials and experimental models have shown that extracts of honey and propolis from stingless bees decrease the expression of cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), and modulate the innate immune response, which promotes the resolution of inflammatory processes and prevents chronic tissue damage. This action is attributed to the synergy between microbial metabolites and plant compounds present in bee products, which act together to regulate inflammatory processes.
The microbiome plays a critical role in this biochemical synergy by contributing to the production of metabolites that enhance these antioxidant and anti-inflammatory properties, resulting in multifactorial effects that would not be achieved by individual compounds alone. This microbial cooperation improves therapeutic efficacy and highlights the importance of conserving biodiversity to maintain the quality and functionality of these products. These antioxidant and anti-inflammatory properties suggest the potential for products derived from stingless bees to be used in the development of natural therapies for chronic inflammatory diseases and nutraceuticals to promote overall health. Incorporating these products into integrative medicine enables various pathologies to be prevented or treated within a framework, strengthening sustainability and environmental protection.
5.3 Immune modulation and its application to metabolic diseases
In the broader context of the microbiome of stingless bees and their nest materials, immune modulation plays a vital role in complementing and amplifying the recognized antimicrobial, antioxidant, and anti-inflammatory properties. Bioactive metabolites derived from the symbiotic microbiota and phytochemical compounds found in honey, propolis, cerumen, and pollen have an impressive capacity to interact with the host’s immune system, promoting precise regulation that favours homeostasis and an effective response to pathogens without causing harmful chronic inflammation. This immunomodulatory action is particularly relevant in metabolic diseases, where immune dysfunction plays a central pathogenic role. Recent research shows that these bee products can influence the activation and proliferation of immune cells, cytokine production, and the expression of key receptors, modulating both innate and adaptive responses. This balanced regulation helps to mitigate the systemic inflammation and oxidative stress that are characteristic of diseases such as type 2 diabetes mellitus, obesity, and metabolic syndrome. These pathologies are highly prevalent worldwide and require safe and effective therapeutic alternatives. Experimental models have shown that microbial and phytochemical extracts can improve insulin sensitivity, regulate lipid and carbohydrate metabolism, and reduce the expression of inflammatory markers. This implies a dual mechanism of action that addresses the immunological and metabolic origins of these diseases.
In patients with type 2 diabetes mellitus, systemic inflammation and oxidative stress are important factors in the development of insulin resistance and progressive tissue damage. Clinical and experimental studies have revealed that the topical application or oral administration of stingless bee honey can accelerate the healing of diabetic ulcers thanks to its combined antimicrobial, anti-inflammatory, and local immune-stimulating effects. Conversely, animal models have demonstrated that supplementation with these products enhances the activity of antioxidant enzymes, reduces inflammatory markers, and promotes tissue regeneration in areas affected by hyperglycaemia. The ability of the bee microbiome to modulate immune responses is also reflected in the regulation of T cell activation and the expression of immunomodulatory receptors, which allows the balance between inflammation and tissue repair to be regulated. This phenomenon is essential for delaying the progression of diabetes and its micro- and macrovascular complications. Studies indicate that these mechanisms contribute to improving both insulin sensitivity and glycolipid metabolism in experimental models, underscoring the multifunctionality of these products as adjuvant therapies. Therefore, preserving the microbiome and biodiversity of stingless bees is presented as an integrative conservation strategy that not only protects a valuable biological resource but also ensures the continued production of these natural therapeutic compounds that may have a decisive role in the management of chronic non-communicable diseases such as diabetes. The interaction between microbiota, inflammation, and metabolism is a field of growing interest, and these bee products offer a promising approach that integrates human health with environmental sustainability. This integrative approach contributes to our understanding of the therapeutic value of stingless bee products and opens up prospects for developing nutraceuticals and adjuvants for use in conventional therapies. Furthermore, preserving biodiversity and the health of the bee microbiome is essential for maintaining the quality and availability of these medicinal products. This establishes an indispensable link between biomedical innovation and ecological conservation. Therefore, the immunological modulation exerted by the microbiome of stingless bees and their nest materials is a prime example of how biodiversity can provide innovative, sustainable solutions for contemporary medicine.
6. Experts on stingless bee conservation inform endeavors in their countries
We were interested to know what actions have governments taken in a few countries. What government agencies? A brief outline of historical progress on conservation of stingless bees, and especially the current status on any legislation.
6.1 Legal and conservation framework for Philippine stingless bees
Stingless bees have great potential for the future of Philippine agriculture and the economy due to their sustainability, vital role in pollinating both wild and domesticated plant species, and the value of their hive products. To date, 12 species of stingless bees have been documented in the Philippines, of which three have shown strong potential for domestication and for the production of honey, pollen, and propolis. Their use in managed pollination is also expanding, particularly for high-value crops such as mango, rambutan, avocado, and various vegetables. Wild stingless bee populations, found primarily in forests, provide sources of genetic material for natural breeding and conservation. Their hive products also show therapeutic properties. Although stingless bees are not yet listed as threatened species under existing wildlife protection laws, their collection and utilization must be carefully regulated to avoid the depletion of wild populations.
6.1.1 Laws relevant to stingless bee conservation
The Philippines has several national laws, standards, and ordinances that regulate wildlife collection, protect forest habitats, and promote sustainable agricultural practices. However, these policies also present gaps that limit their effectiveness for species-specific conservation—particularly for stingless bees whose biology, nesting patterns, and ecological roles differ from those of honey bees.
6.1.2 Republic Act 9147 – Wildlife Resources Conservation and Protection Act
The Wildlife Resources Conservation and Protection Republic Act 9147 provides the basic legal foundation for the conservation of wildlife species and their habitats, promoting ecological balance and biological diversity in the Philippines. The law regulates the collection, possession, transport, and trade of wildlife—including insects such as stingless bees—and imposes penalties for unauthorized activities. Its provisions guide national wildlife management and support the development of science-based conservation strategies (Office of the President of the Philippines, 1995). A key component of RA 9147 is its regulation of bioprospecting, defined as the research, collection, and utilization of biological and genetic resources for scientific or commercial purposes. Bioprospecting activities involving wildlife require a permit from DENR (for terrestrial species) or DA-BFAR (for aquatic species), as well as Prior Informed Consent (PIC) from Indigenous Peoples, local communities, or protected area managers when applicable. A bioprospecting undertaking ensures equitable access and benefit-sharing, values traditional knowledge, and prevents the exploitation of native species. These provisions are highly relevant to stingless bees, whose wild colonies represent valuable genetic and biochemical resources that must be accessed responsibly.
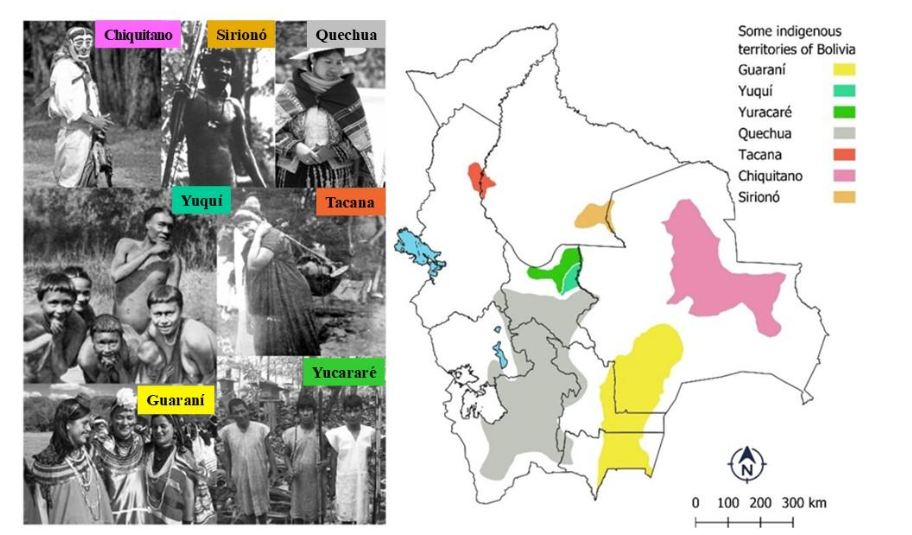
6.2 Stingless bee conservation in Bolivia: Current advances in governance, research, knowledge indigenous, collective and individual initiatives
In Bolivia, stingless bees (Meliponini) have acquired significant conservation and socioeconomic relevance through the progressive development of meliponiculture. Some species have demonstrated their capacity for breeding and management in box nests, thereby enabling the sustainable production particularly honey and propolis. These hive products exhibit distinctive nutraceutical properties, and well-documented therapeutic and nutritional benefits offering particularly valuable livelihood opportunities, and food-security options for indigenous and local communities.
6.2.1 Richness species of stingless bees in Bolivia
To understand the advances in stingless bee conservation in Bolivia, it is first necessary to review knowledge of their diversity. Early foundational contributions of meliponine bees were published by Moure (1950) and Kempff-Mercado (1968). Subsequently, the catalogue of bees (Hymenoptera, Apoidea) in the Neotropical region, recorded 21 genera and 88 species for Bolivia (Camargo et al. 2023). The most recent review documented 121 species distributed across 19 valid genera, with the added description of a new species, Melipona rasmusseni, bringing the current total to 122. However, it is important to note the possible addition of 15 species described near the borders with Brazil and Argentina.
6.2.2 Meliponiculture
Of all stingless bees known in Bolivia, at least eight species are managed in stingless bee keeping, which has led to various projects developed since the 1990s. Among them are various projects set up by non-governmental organizations (NGOs), such as the initiatives carried out in the department of Beni, specifically within the Sirionó indigenous communities of Ibiato, promoted by the Biodiversity Support Program WWF/USAID, the Nature Conservancy, and The Wood Resources Institute. Currently, efforts are underway in the Ibare-Mamoré municipal protected area of Trinidad, Beni, promoted through CIBIOMA of the Technical University of Beni Mariscal José Ballivián. Likewise, in Santa Cruz, projects were implemented starting in 2000 in Guaraní indigenous communities with the support of the Wildlife Conservation Society (WCS), as documented in the works of Martínez and Cuéllar (2004), and in settler communities within Amboró National Park supported by the eastern ecology association (ASEO, Asociación Ecología del Oriente). Additionally, Chuquisaca NGO Lider and the PASOS Foundation formed associations of meliponiculture producers, becoming a benchmark in stingless bee breeding and production since 2015. NGOs such as the Foundation for the Conservation of the Chiquitano Forest (FCBC, Fundación para la Conservación del Bosque Chiquitano), with support from indigenous farmers in eastern Bolivia (APCOB, Apoyo para el Campesino-Indígena del Oriente Boliviano), have been promoting research and development of meliponiculture in the Chiquitania region since 2020. Other locality such as Vallegrande are in their nascent stages of meliponiculture. Other valuable experiences took place in the Tacana indigenous communities in La Paz, supported by WCS. During 2010 in Tarija, in regions of the Chaco, they worked with the association of beekeepers of the Gran Chaco (ADACHACO, Asociación de Apicultores del Gran Chaco) – PUMA Foundation. In Cochabamba, the Yuracaré indigenous communities have promoted meliponiculture.
6.2.3 Law, Statutes and Programs (Governance)
National policies that attempt to include stingless bees arise through the national program for the strengthening and support of beekeeping production (PNFyAPA, Programa Nacional de Fortalecimiento y Apoyo a la Producción Apicola). Under the criterion of resilience to climate change, a variety of studies have been carried out that include preferentially the introduced honey bees (Apis mellifera) and also native bees (Meliponini) of economic importance. The following are important aspects of PNFyAPA:
- Bolivia promulgated the Supreme Decree 4632 (1 December 2021) governed by Law 144 (26 June 2011), establishing that better and greater production performance will be promoted within the framework of a plural economy, for traditional, organic, ecological, agricultural, and forestry production intended for domestic consumption, enabling food sovereignty to be achieved and surpluses to be generated, within the framework of local knowledge, practices, and technological innovation based on family, community, associative, and cooperative forms of production.
- Created the PNFyAPA, aim to consolidate and strengthen the national beekeeping sector. Although originally centred on Apis mellifera, the programme has progressively incorporated stingless bees.
- Execution of support for assigned to the national institute of agricultural and forestry innovation (INIAF, Instituto Nacional de Innovación Agropecuaria y Forestal), Bolivian food and derivatives company (EBA, Empresa Boliviana de Alimentos y Derivados), and national service of agricultural health and food safety (SENASAG, Servicio Nacional de Sanidad Agropecuaria e Inocuidad Alimentaria). SENASAG participates in the PNFyAPA, aimed at specific regulations for beekeepers and meliponiculturists. For example, they publish general animal health regulations (REGENSA, Reglamento General de Sanidad Animal) in the chapter 3.3 has regulated specific requirements for the registration of beekeeping and meliponiculturists in the production establishments.
Despite these advances, hitherto, Bolivia has passed no national law specifically for stingless bees, but departmental and municipal governments have generated regulations, the most notable being:
- The Law 366/2018 on the promotion, conservation, and development of beekeeping in the department of Chuquisaca, which in Article 5 (Chapter 1) includes definitions of meliponiculture and stingless bees. In addition, Chapter 5, Articles 18 and 19 deal with the conservation of the bee population in the department of Chuquisaca.
- The municipality of Machareti has issued two regulations
- Macharetí Municipal Law No. 15 Municipal Law on the Breeding, Management, Conservation, and Protection of Stinging Bees (Apis mellifera) and Native Stingless Bees (Meliponinos).
- Municipal Decree No. 3/2021 Regulations to Machareti Municipal Law No. 15. Municipal Law on the Breeding, Management, Conservation, and Protection of Stinging Bees (Apis mellifera) and Native Stingless Bees (Meliponinos).
6.2.4 Advances and lessons learned for stingless bee conservation
In December 2024, the first Bolivian discussion forum on native stingless bees was held, convened by the FAO together with the Ministry of Environment and Water, meliponiculture farmers, academics, and civil society. At that forum, the Bolivian organization for the protection of native bees (OBPAN, Organización Boliviana de Protección a las Abejas Nativas) was founded, the first national entity dedicated specifically to stingless bees. Among the issues that arose were: the need for technical, regulatory, and academic funding; the creation of networks among meliponiculture practitioners; the cataloging of species by ecoregion; and insurance for producers against fires or pesticides.
6.2.5 Scientific support to study biodiverse stingless bee nest materials and meliponiculture in Bolivia
The physicochemical characterization of pot-honey, its botanical origin, and uses of cerumen and other materials of the nest like pot-honey, pot-pollen and propolis, including women have attracted researchers, as well as the relationship between stingless bees and plants. The Bolivian standards for stingless bee honey are needed, and Bolivia has developed a multidisciplinary frame to appreciate their biodiversity and potential medicinal uses. Financial support for science and technology from the government will benefit quality control and discovery of rich stingless bee resources.
6.3 Stingless bee conservation in Guatemala
In Guatemala we currently recognize 33 species of stingless bees, and their presence in the region goes back to the time of the ancient Maya. From colonial records we also know that these bees were already understood and valued by Indigenous communities, not only for their biological traits but for their cultural and spiritual importance. Honey and pollen have been traditionally used as food supplements, medicinal resources, and are even considered as elements of family protection. Their role has become increasingly visible in recent years, especially as global attention to pollinator decline has intensified.
Since the early 2000s, the Universidad de San Carlos de Guatemala has taken the lead in studying stingless bees in a more systematic way. This includes documentation of species diversity, distribution, genetics, ecological roles, and also the characterization of their nest materials. Much of the national conversation about the importance of stingless bees for conservation and food security has grown directly from this body of work. Little by little, other institutions, both government agencies and private organizations, have joined these efforts. The Ministry of Agriculture, especially through the Mediterranean fruit fly control and eradication program, incorporated meliponiculture into its social extension activities, as part of the beekeeping module. Also, the National System of Protected Areas -Consejo Nacional de Áreas Protegidas (CONAP)- has contributed to biodiversity and public education initiatives related to these bees.
Over the last 25 years, the visibility of stingless bees in Guatemala has increased noticeably, and this is in large part due to the sustained work coming from the Universidad de San Carlos. Their research, outreach, and technical training programs have consistently brought Meliponini into public discussion, generating interest among local communities, policymakers, government agencies and private organizations. Their resemblance to the familiar honey bee, Apis mellifera, has helped the general public quickly link them to ecosystem services such as pollination and honey production. The improvement in public acceptance and conservation actions also stems from the biocultural appeal of stingless bees: people perceive them as gentle, harmless and culturally meaningful, and their condition as native species is especially valued because of their association with traditions of medicinal honey and family protection. These qualities make conservation efforts feel intuitive, relatable, and relevant to Guatemalan identity. Taken together, this indicates that meliponiculture cannot simply replicate the apiculture model: stingless bees embody a strong biocultural identity, and their management and conservation require approaches that integrate cultural, ecological, and social dimensions.
The protected-area agency -CONAP- has formally adhered to the use of global open biodiversity databases such as GBIF, recognizing their importance as essential tools for modern conservation planning. However, records of Meliponini, especially those published by Guatemalan institutions, are still scarce in these platforms. Strengthening national participation and increasing the availability of high-quality data will be crucial for identifying priority species and areas that need biodiversity protection.
In 2025, the first version of a “National Strategy for the Conservation and Sustainable Use of Native Stingless Bees in Guatemala” was released. Although it has not yet been officially launched, the document reflects a structured and collaborative effort to secure the future of these species. Its development involved the Universidad de San Carlos, several government institutions, including the protected area agency, meliponiculturists, and private organizations. The strategy represents the country’s first coordinated national framework aimed at promoting meliponiculture, protecting native species, and integrating cultural and social values into conservation planning.
6.4 Legal and conservation framework for stingless bees in Tanzania
Stingless bees are highly regarded in Tanzania as a unique resource, contributing to income generation, pollination services, aesthetics (tourism) and biodiversity. The honey produced by stingless bees has long been appreciated for its distinct flavor and medicinal properties. Eleven species of stingless bees have been documented in Tanzania, representing seven genera of Axestotrigona, Hypotrigona, Plebeina, Meliplebeia, Dactylurina, Plebeiella, and Liotrigona. Among these, only few species in the genera Axestotrigona and Hypotrigona are actively managed for honey production. However, the utilization of stingless bee resources in the country raises sustainability concerns, particularly due to the reliance of wild stingless bee colonies for stocking managed hives and honey hunting practices that persist in some regions. Nonetheless, national policies and legislations recognize the importance of conserving these species, and effective implementation of these frameworks will be critical for ensuring their long-term sustainability.
6.4.1 Policies and legislations relevant to stingless bee conservation in Tanzania
In Tanzania, there are various legal and policy frameworks that govern the management of natural resources, and which directly or indirectly promote conservation of bees, including stingless bees. These include the National Beekeeping Policy, Beekeeping Act, Forest Act, Wildlife Conservation Act, and Guidelines for Management and Use of Honeybee Colonies for Pollination Services.
6.4.2 National Beekeeping Policy
The current National Beekeeping Policy of Tanzania was published in 1998 and it encompasses both stinging honey bees and stingless bees, including wild and domesticated colonies. The policy highlights the uniqueness of stingless bees due to their medicinal honey, and emphasizes the need for conservation of both types of bees amidst emerging threats such as pesticide poisoning, habitat destruction, and land clearing for agricultural expansion and industry development. One of the key policy objectives is to establish bee reserves, including for rare stingless bee species, to enable sustainable management of these species. The policy mandates joint management agreements between government and communities to ensure sustainable management of these bee reserves as breeding grounds for honey bees and stingless bees, and favorable areas for bee product production. Moreover, the policy advocates for environmental considerations to reduce risks which threaten stingless bees by requiring Environmental Impact Assessments (EIA) for projects near bee reserves to prevent habitat degradation, and promoting Integrated Pest Management (IPM) to minimize pesticide exposure.
6.4.3 Bee Keeping Act
The Bee Keeping Act of 2002 corroborates with the National Beekeeping Policy by promoting sustainable management and conservation of wild and domesticated bees, including stingless bees, through bee reserves. It delivers the mechanisms for establishing, managing, and protecting these reserves, and identifies prohibited activities to avoid disturbance or destruction of bee habitats. Additionally, the Act regulates trade related to bees and their products including importation, registration, and restrictions to control bee diseases and pests threatening bee populations. Furthermore, The Act mandates environmental impact assessments prior for developments near bee reserves to protect bee populations or habitats from planned activities. It also establishes the Beekeeping Development Fund, currently known as Tanzania Forest Fund (TaFF), which supports research and training, indirectly benefiting stingless bee conservation through sectoral capacity building and awareness creation.
6.4.4 Forest Act and Wildlife Conservation Act
The Forest Act of 2023 and Wildlife Conservation Act of 2022 both clearly recognizes bee ecosystems as targets for conservation under national laws. The Forest Act fosters biodiversity conservation within forest ecosystems which are the primary stingless bee breeding grounds through establishment and management of forest reserves from national to village levels. The Act promotes community involvement in forest management which supports protection of bee habitats through sustainable utilization and local stewardship.
The Wildlife Conservation Act places the bee ecosystems and related resources under the custody of a specialized paramilitary service which governs their conservation, management and utilization. Consequently, the Tanzania Forest Services Agency (TFS) converted into a paramilitary service under the Ministry of Natural Resources and Tourism which oversees the development and sustainable management of forest and bee resources. Additionally, TFS has legal powers to prevent, detain and investigate any crime related to forest and bee ecosystems and resources, creating a direct enforcement mechanism for the protection of stingless bee habitats.
The Forest and Wildlife Conservation Acts mandate environmental and wildlife impact assessments before approval and establishment of projects that are likely to affect forest and wildlife species, communities or habitats. This applies to projects involving logging, agricultural expansion, infrastructure development and mining which may affect stingless bee nesting sites.
6.5 Conservation of stingless bees in Costa Rica
A National Federation for Conservation (FECON) was created in Costa Rica 40 years ago. Ongoing proposals for conservation of bees wait for approval. We must recognize that bees are diverse, not just the European honey bee Apis mellifera, and that we must protect them and take advantage of their contributions without harming their populations. Conserving bee diversity means protecting native bees (including stingless bees, Meliponini) as well as other wild and solitary bees, protecting their habitats (native flowers, natural cavities, soils) and avoiding pesticides and diseases. Conserving means maintaining genetic and ecological diversity to ensure that they continue to play their fundamental role in pollination and ecosystem balance, and preserving the active biomolecules for medicinal uses. It is important to use them fairly and equitably. This refers to how we take advantage of the benefits that these bees generate. For example, pollination which enables agriculture and food production. Pot-honey, pot-pollen, cerumen, and propolis from the stingless bee nest. The traditional knowledge of communities that manage meliponines. Stingless bee keepers who conserve bees and their habitats receive real benefits (income, recognition, participation). The participation of farmers, indigenous communities, meliponiculture practitioners, and scientists in decision-making is promoted. In summary, conserving and using bee diversity in a fair and equitable manner means protecting both bees and their habitats and ensuring that the benefits of their existence and management are distributed in a responsible, ethical, and sustainable manner.
On the other hand, Costa Rica is very strict about collecting bees, and permits to export them take a while. There is a National Commission for Biodiversity Management, in Spanish Comisión Nacional para la Gestión de la Biodiversidad (CONAGEBIO), which is the permit office for samples that will be used for genetic analyses. The obligations of CONAGEBIO are primarily focused on the development of national policies pertaining to the conservation and sustainable utilization of biodiversity, as well as issues related to education, public awareness, research, and the transfer of technology. It serves as the authoritative entity responsible for establishing and overseeing adherence to regulations governing access to genetic and biochemical resources, regarded as assets within the public domain, thereby ensuring the preservation, conservation, and restoration of traditional knowledge linked to the utilization of biodiversity elements, framed in the Biodiversity Law of the Ministry of Agriculture and Livestock since 1998.
There are many causes that are affecting the biodiversity of different species of flora and fauna, but the fragmentation and loss of habitat is one of the main ones. Therefore, preservation, restoration, and connectivity of the different natural landscapes is one of the main challenges for the conservation of natural areas. Biological corridors arise from the needs to connect the different protected areas that were becoming islands, affecting the mobility of species. The use of stingless bees as indicators of connectivity in biological corridors is a valuable tool for the conservation and sustainable management of ecosystems, as it allows us to understand and mitigate the effects of habitat fragmentation and ensure the preservation of biodiversity.
6.6 Experience of El Salvador in Stingless Bee’s Conservation of Biodiversity
At present, El Salvador does not have a comprehensive unified national Biodiversity Law specifically dedicated to the conservation, sustainable use, and restoration of biological diversity. Although several legal instruments exist — including the Environmental Law (Ley de Medio Ambiente, 1998), the Protected Natural Areas Law (Ley de Áreas Naturales Protegidas, 2005), and provisions related to forest and wildlife management — these frameworks remain sectoral and fragmented, addressing biodiversity concerns in a dispersed manner rather than through an integrated, cohesive legal system. As a result, many native species and ecosystems lack comprehensive institutional safeguards.
In this context of limited formal protection, the CREVAS–JICA–UES Project has been working to safeguard the biodiversity of native stingless bees (Meliponini), which represent both ecologically essential pollinators and culturally significant species in Mesoamerica. Our approach combines scientific research, community-based management, environmental education, and territorial regeneration.
6.6.1 Key contributions of the CREVAS Project
- Strengthening community-led meliponiculture initiatives for species such as Melipona beecheii and Tetragonisca angustula, supporting biodiversity conservation, women and youth participation, and sustainable rural livelihoods.
- Developing improved hive systems and establishing a Meliponini (Stingless Bee) Colony Regeneration Center, contributing to species recovery, monitoring, and resilience against environmental threats; and ensuring the establishment of nurseries for native melliferous plants, through which seedlings are produced and distributed free of charge to restore habitats, expand forage availability, and reinforce local ecological networks.
- Promoting co-creation–based technical innovation, including improved hive technologies (e.g., INPA-style boxes), collaborative quality control and traceability, and the development of new stingless-bee-derived products.
- Training programs for farmers, students, technicians, and local residents, fostering mutual learning, knowledge transfer, entrepreneurship, and long-term stewardship of native pollinators.
- Environmental education programs co-created with schools, universities, botanical gardens, and local communities, cultivating a conservation-oriented culture and strengthening territorial pride and identity.
- Publication of an illustrated children’s book, co-created to introduce young audiences to the world of stingless bees, integrating traditional knowledge with scientific understanding and nurturing future environmental leaders.
- Installation of bee hotels in botanical gardens, primary schools, and public parks, contributing to ecological corridor formation and providing hands-on learning environments for children and local residents.
- Building a collaborative territorial platform that brings together producers, universities, public institutions, private companies, and civil society to integrate biodiversity conservation with local development strategies—aligned with CREVAS’s three pillars: 1. Territorial value creation, 2. Co-developed economic models, and 3. Embedding knowledge and technology in the territory.
Through these combined efforts, the project demonstrates that value is not extracted from nature but co-created with it, and that meliponiculture can serve as a flagship territorial strategy in El Salvador—not for profit, but as part of our responsibility to the Earth. This work highlights the experience of the JICA–UES CREVAS Project as a model of biodiversity conservation in a country where national biodiversity legislation remains limited. In addition, to advancing scientific understanding of native stingless bees, the project emphasizes that developing mission-driven, community-based enterprises is essential for addressing social challenges while promoting environmental stewardship.