Use of near-infrared spectroscopy to probe occlusion severity in patients diagnosed with carotid atherosclerotic disease
Main Article Content
Abstract
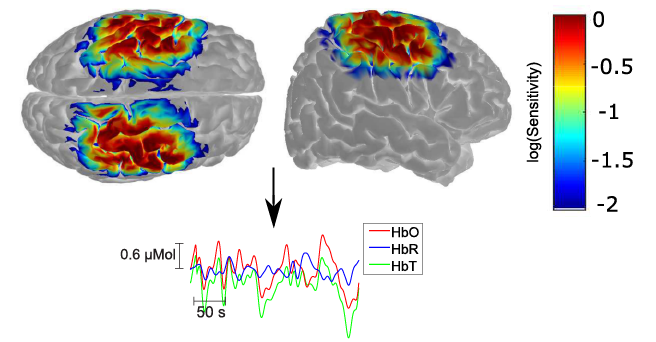
Atherosclerotic disease has been associated with increased risk of severe neurovascular consequences including transient ischemic attacks, ischemic strokes, and even mortality. Following the onset of carotid stenosis, the brain undergoes different compensatory mechanisms to provide adequate perfusion in order to maintain brain metabolism. In this study, we employed near-infrared spectroscopy (NIRS) to better comprehend how carotid stenosis affects cerebral hemodynamics, both at rest and during activity. Eighteen patients diagnosed with atherosclerotic disease were recruited for a protocol consisting of resting state and vasoreactivity testing performed with breath holding. Although the breath holding challenge induced global vasodilation in all patients, the extent of dilation varied according to the level of stenosis. Patients diagnosed with carotid stenosis have impaired hemodynamic response, with a median 77% vasoreactivity in the hemisphere ipsilateral to the stenotic vessel when compared to the healthy hemisphere. Hemodynamics of these patients differed at rest, with 33% fewer network links in the hemisphere ipsilateral to the stenosis than the healthy hemisphere. On the other hand, hemodynamic patterns were more heterogeneous with patients diagnosed with a carotid occlusion, which correlates with the opening of collateral circulation. Overall, our results suggest that NIRS can open new directions to the investigation of the effects of cerebrovascular atherosclerotic disease.
Article Details
The Medical Research Archives grants authors the right to publish and reproduce the unrevised contribution in whole or in part at any time and in any form for any scholarly non-commercial purpose with the condition that all publications of the contribution include a full citation to the journal as published by the Medical Research Archives.
References
Barrat, A., Barthélemy, M., Pastor-Satorras, R., Vespignani, A., 2004. The architecture of complex weighted networks. Proc. Natl. Acad. Sci. U. S. A. 101, 3747–3752. doi:10.1073/pnas.0400087101
Brigadoi, S., Ceccherini, L., Cutini, S., Scarpa, F., Scatturin, P., Selb, J., Gagnon, L., Boas, D.A., Cooper, R.J., 2014. Motion artifacts in functional near-infrared spectroscopy: A comparison of motion correction techniques applied to real cognitive data. Neuroimage 85, 181–191. doi:10.1016/j.neuroimage.2013.04.082
Carbonell, F., Bellec, P., Shmuel, A., 2011. Global and system-specific resting-state fMRI fluctuations are uncorrelated: principal component analysis reveals anti-correlated networks. Brain Connect. 1, 496–510. doi:10.1089/brain.2011.0065
Carr, S., Farb, A., Pearce, W.H., Virmani, R., Yao, J.S.T., 1996. Atherosclerotic plaque rupture in symptomatic carotid artery stenosis. J. Vasc. Surg. 23, 755–766. doi:http://dx.doi.org/10.1016/S0741-5214(96)70237-9
Carroll, B.A., 1991. Carotid sonography. Radiology 178, 303–313.
de Weerd, M., Greving, J.P., Hedblad, B., Lorenz, M.W., Mathiesen, E.B., O'Leary, D.H., Rosvall, M., Sitzer, M., Buskens, E., Bots, M.L., 2010. Prevalence of Asymptomatic Carotid Artery Stenosis in the General Population. Stroke 41, 1294 LP-1297.
Debrey, S.M., Yu, H., Lynch, J.K., Lövblad, K.-O., Wright, V.L., Janket, S.-J.D., Baird, A.E., 2008. Diagnostic Accuracy of Magnetic Resonance Angiography for Internal Carotid Artery Disease. Stroke 39, 2237 LP-2248.
Delcker, A., Diener, H.C., Wilhelm, H., 1997. Source of cerebral microembolic signals in occlusion of the internal carotid artery. J. Neurol. 244, 312–317. doi:10.1007/s004150050093
Demchuk, a M., Christou, I., Wein, T.H., Felberg, R. a, Malkoff, M., Grotta, J.C., Alexandrov, a V, 2000. Specific transcranial Doppler flow findings related to the presence and site of arterial occlusion. Stroke. 31, 140–146. doi:10.1161/01.STR.31.1.140
Duncan, G.W., Pessin, M.S., Mohr, J.P., Adams, R.D., 1976. Transient cerebral ischemic attacks. Adv. Intern. Med. 21, 1—20.
Favilla, C.G., Mesquita, R.C., Mullen, M., Durduran, T., Lu, X., Kim, M.N., Minkoff, D.L., Kasner, S.E., Greenberg, J.H., Yodh, A.G., Detre, J.A., 2014. Optical bedside monitoring of cerebral blood flow in acute ischemic stroke patients during head-of-bed manipulation. Stroke 45, 1269–1274. doi:10.1161/STROKEAHA.113.004116
Ferrari, M., Quaresima, V., 2012. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage 63, 921–935. doi:10.1016/j.neuroimage.2012.03.049
Fisher, C.M., 1962. Concerning Recurrent Transient Cerebral Ischemic Attacks. Can. Med. Assoc. J. 86, 1091–1099.
Gagnon, L., Cooper, R.J., Yücel, M.A., Perdue, K.L., Greve, D.N., Boas, D.A., 2012. Short separation channel location impacts the performance of short channel regression in NIRS. Neuroimage 59, 2518–2528. doi:10.1016/j.neuroimage.2011.08.095
Gagnon, L., Yücel, M.A., Boas, D.A., Cooper, R.J., 2014. Further improvement in reducing superficial contamination in NIRS using double short separation measurements. Neuroimage 85, 127–135. doi:10.1016/j.neuroimage.2013.01.073
Garrity, A.G., Pearlson, G.D., McKiernan, K., Lloyd, D., Kiehl, K.A., Calhoun, V.D., 2007. Aberrant “default mode” functional connectivity in schizophrenia. Am. J. Psychiatry 164, 450–457. doi:10.1176/appi.ajp.164.3.450
Greicius, M.D., Supekar, K., Menon, V., Dougherty, R.F., 2009. Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb. Cortex 19, 72–78. doi:10.1093/cercor/bhn059
Hankey, G.J., Warlow, C.P., Sellar, R.J., 1990. Cerebral angiographic risk in mild cerebrovascular disease. Stroke 21, 209 LP-222.
Huppert, T.J., Diamond, S.G., Franceschini, M.A., Boas, D.A., 2009. HomER: a review of time-series analysis methods for near-infrared spectroscopy of the brain. Appl. Opt. 48, 280–298. doi:10.1016/j.drugalcdep.2008.02.002.A
Larsen, F.S., Olsen, K.S., Hansen, B. a, Paulson, O.B., Knudsen, G.M., 1994. Transcranial Doppler is valid for determination of the lower limit of cerebral blood flow autoregulation. Stroke 25, 1985–8. doi:10.1161/01.STR.25.10.1985
Li, Z., Liu, H., Liao, X., Xu, J., Liu, W., Tian, F., He, Y., Niu, H., 2015. Dynamic functional connectivity revealed by resting-state functional near-infrared spectroscopy 6, 15757–15767. doi:10.1364/BOE.6.002337
Lu, C.M., Zhang, Y.J., Biswal, B.B., Zang, Y.F., Peng, D.L., Zhu, C.Z., 2010. Use of fNIRS to assess resting state functional connectivity. J. Neurosci. Methods 186, 242–249. doi:10.1016/j.jneumeth.2009.11.010
Markus, H.S., Harrison, M.J., 1992. Estimation of cerebrovascular reactivity using transcranial Doppler, including the use of breath-holding as the vasodilatory stimulus. Stroke 23, 668 LP-673.
Mesquita, R.C., Franceschini, M.A., Boas, D.A., 2010. Resting state functional connectivity of the whole head with near infrared spectroscopy. Biomed. Opt. Express 324–336. doi:10.1364/BOE.1.000324
Mesquita, R.C., Han, S.W., Miller, J., Schenkel, S.S., Pole, A., Esipova, T. V., Vinogradov, S.A., Putt, M.E., Yodh, A.G., Busch, T.M., 2012. Tumor blood flow differs between mouse strains: Consequences for vasoresponse to photodynamic therapy. PLoS One 7, 1–10. doi:10.1371/journal.pone.0037322
Mesquita, R.C., Huppert, T.J., Boas, D. a, 2009. Exploring neuro-vascular and neuro-metabolic coupling in rat somatosensory cortex. Phys. Med. Biol. 54, 175–85. doi:10.1088/0031-9155/54/2/001
Mesquita, R.C., Putt, M., Chandra, M., Yu, G., Xing, X., Han, S.W., Lech, G., Shang, Y., Durduran, T., Zhou, C., Yodh, A.G., Mohler, E.R., 2013. Diffuse optical characterization of an exercising patient group with peripheral artery disease. J. Biomed. Opt. 18, 57007. doi:10.1117/1.JBO.18.5.057007
Mesquita, R.C., Yodh, A.G., 2011. Diffuse Optics : Fundamentals & Tissue Applications. Astronomy 51–74. doi:10.3254/978-1-60750-755-0-51
Minati, L., Kress, I.U., Visani, E., Medford, N., Critchley, H.D., 2011. Intra- and extra-cranial effects of transient blood pressure changes on brain near-infrared spectroscopy (NIRS) measurements. J. Neurosci. Methods 197, 283–288. doi:10.1016/j.jneumeth.2011.02.029
Müller, M., Schimrigk, K., 1996. Vasomotor Reactivity and Pattern of Collateral Blood Flow in Severe Occlusive Carotid Artery Disease. Stroke 27, 296 LP-299.
Newman, M.E.J., 2004. Analysis of weighted networks. Phys. Rev. E - Stat. Nonlinear, Soft Matter Phys. 70. doi:10.1103/PhysRevE.70.056131
Norrving, B., Nilsson, B., Risberg, J., 2015. rCBF in patients with carotid occlusion. Resting and hypercapnic flow related to collateral pattern. Stroke. 13, 155–162. doi:10.1161/01.STR.13.2.155
North American Symptomatic Carotid Endarterectomy Trial(NASCET) Steering Committee, 1991. Original Contributions North American Symptomatic Carotid Endarterectomy Trial.
Novi, S.L., Rodrigues, R.B.M.L., Mesquita, R.C., 2016. Resting state connectivity patterns with near- infrared spectroscopy data of the whole head. Biomed Opt Express 7, 2524–2537. doi:10.1364/BOE.7.002524
O’Leary, D.H., Polak, J.F., Kronmal, R.A., Kittner, S.J., Bond, M.G., Wolfson, S.K., Bommer, W., Price, T.R., Gardin, J.M., Savage, P.J., 1992. Distribution and correlates of sonographically detected carotid artery disease in the Cardiovascular Health Study. The CHS Collaborative Research Group. Stroke 23, 1752 LP-1760.
Obrig, H., Neufang, M., Wenzel, R., Kohl, M., Steinbrink, J., Einhäupl, K., Villringer, A., 2000. Spontaneous Low Frequency Oscillations of Cerebral Hemodynamics and Metabolism in Human Adults. Neuroimage 12, 623–639. doi:10.1006/nimg.2000.0657
Oscillators, C., 2010. Europe PMC Funders Group Coupled Oscillators : 26, 25–29.
Philip, K.J., H., R.A., C., H.R., 1984. Therapy of Ischemic Cerebral Vascular Disease Due to Atherothrombosis. N. Engl. J. Med. 311, 100–105. doi:10.1056/NEJM198407123110206
Ratnatunga, C., Adiseshiah, M., 1990. Increase in middle cerebral artery velocity on breath holding: A simplified test of cerebral perfusion reserve. Eur. J. Vasc. Surg. 4, 519–523. doi:10.1016/S0950-821X(05)80795-9
Ringelstein, E.B., Sievers, C., Ecker, S., Schneider, P.A., Otis, S.M., 1988. Noninvasive assessment of CO2-induced cerebral vasomotor response in normal individuals and patients with internal carotid artery occlusions. Stroke 19, 963 LP-969.
Rothwell, P.M., Gibson, R.J., Slattery, J., Sellar, R.J., Warlow, C.P., 1994. Equivalence of measurements of carotid stenosis. A comparison of three methods on 1001 angiograms. European Carotid Surgery Trialists' Collaborative Group. Stroke 25, 2435 LP-2439.
Sabeti, S., Schillinger, M., Mlekusch, W., Willfort, A., Haumer, M., Nachtmann, T., Müllner, M., Lang, W., Ahmadi, R., Minar, E., 2004. Quantification of Internal Carotid Artery Stenosis with Duplex US: Comparative Analysis of Different Flow Velocity Criteria. Radiology 232, 431–439. doi:10.1148/radiol.2321030791
Sakakibara, E., Homae, F., Kawasaki, S., Nishimura, Y., Takizawa, R., Koike, S., Kinoshita, A., Sakurada, H., Yamagishi, M., Nishimura, F., Yoshikawa, A., Inai, A., Nishioka, M., Eriguchi, Y., Matsuoka, J., Satomura, Y., Okada, N., Kakiuchi, C., Araki, T., Kan, C., Umeda, M., Shimazu, A., Uga, M., Dan, I., Hashimoto, H., Kawakami, N., Kasai, K., 2016. Detection of resting state functional connectivity using partial correlation analysis: A study using multi-distance and whole-head probe near-infrared spectroscopy. Neuroimage 142, 590–601. doi:10.1016/j.neuroimage.2016.08.011
Sanborn, M.R., Edsell, M.E., Kim, M.N., Mesquita, R., Putt, M.E., Imray, C., Yow, H., Wilson, M.H., Yodh, A.G., Grocott, M., Martin, D.S., 2015. Cerebral Hemodynamics at Altitude: Effects of Hyperventilation and Acclimatization on Cerebral Blood Flow and Oxygenation. Wilderness Environ. Med. 26, 133–141. doi:10.1016/j.wem.2014.10.001
Sasai, S., Homae, F., Watanabe, H., Sasaki, A.T., Tanabe, H.C., Sadato, N., Taga, G., 2012. A NIRS–fMRI study of resting state network. Neuroimage 63, 179–193. doi:10.1016/j.neuroimage.2012.06.011
Scholkmann, F., Kleiser, S., Metz, A.J., Zimmermann, R., Mata Pavia, J., Wolf, U., Wolf, M., 2014. A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. Neuroimage 85, 6–27. doi:10.1016/j.neuroimage.2013.05.004
Silvestrini, M., Paolino, I., Vernieri, F., Pedone, C., Baruffaldi, R., Gobbi, B., Cagnetti, C., Provinciali, L., Bartolini, M., 2009. Cerebral hemodynamics and cognitive performance in patients with asymptomatic carotid stenosis. Neurology 72, 1062–1068. doi:10.1212/01.wnl.0000345015.35520.52
Silvestrini, M., Troisi, E., Matteis, M., Cupini, L.M., Caltagirone, C., 1996. Transcranial Doppler Assessment of Cerebrovascular Reactivity in Symptomatic and Asymptomatic Severe Carotid Stenosis. Stroke 27, 1970 LP-1973.
Soinne, L., Helenius, J., Tatlisumak, T., Saimanen, E., Salonen, O., Lindsberg, P.J., Kaste, M., 2003. Cerebral Hemodynamics in Asymptomatic and Symptomatic Patients With High-Grade Carotid Stenosis Undergoing Carotid Endarterectomy. Stroke 34, 1655 LP-1661.
Stam, C.J., Jones, B.F., Nolte, G., Breakspear, M., Scheltens, P., 2007. Small-world networks and functional connectivity in Alzheimer’s disease. Cereb. Cortex 17, 92–99. doi:10.1093/cercor/bhj127
Störk, S., van den Beld, A.W., von Schacky, C., Angermann, C.E., Lamberts, S.W.J., Grobbee, D.E., Bots, M.L., 2004. Carotid Artery Plaque Burden, Stiffness, and Mortality Risk in Elderly Men. Circulation 110, 344 LP-348.
Tachtsidis, I., Leung, T.S., Devoto, L., Delpy, D.T., Elwell, C.E., 2008. Measurement of Frontal Lobe Functional Activation and Related Systemic Effects: ANear-InfraredSpectroscopy Investigation. Adv. Exp. Med. Biol. 614, 397–403. doi:10.1007/978-0-387-74911-2
Tian, L., Meng, C., Jiang, Y., Tang, Q., Wang, S., Xie, X., Fu, X., Jin, C., Zhang, F., Wang, J., 2016. Abnormal functional connectivity of brain network hubs associated with symptom severity in treatment-naive patients with obsessive-compulsive disorder: A resting-state functional MRI study. Prog. Neuro-Psychopharmacology Biol. Psychiatry 66, 104–111. doi:10.1016/j.pnpbp.2015.12.003
Trial, M.R.S.E.C.S., 1991. interim results for symptomatic patients with severe (70-99%) or with mild (0-29%) carotid stenosis: Europen Carotid Surgery Trialists Collaborative Group. Lancet 337, 1235–1243.
Tsivgoulis, G., Sharma, V.K., Lao, A.Y., Malkoff, M.D., Alexandrov, A. V., 2007. Validation of transcranial Doppler with computed tomography angiography in acute cerebral ischemia. Stroke 38, 1245–1249. doi:10.1161/01.STR.0000259712.64772.85
Vasdekis, S.N., Tsivgoulis, G., Athanasiadis, D., Andrikopoulou, A., Voumvourakis, K., Lazaris, A.M., Stamboulis, E., 2012. Cerebrovascular reacivity assessment in patients with carotid artery disease: A combined TCD and NIRS study. J. Neuroimaging 22, 261–265. doi:10.1111/j.1552-6569.2011.00595.x
Vernieri, F., Pasqualetti, P., Passarelli, F., Rossini, P.M., Silvestrini, M., 1999. Outcome of Carotid Artery Occlusion Is Predicted by Cerebrovascular Reactivity. Stroke 30, 593 LP-598.
Wardlaw, J.M., 2006. NHS Research and Development Health Technology Assessment Carotid Stenosis Imaging Group : Non-invasive imaging compared with intra-arteril angiography in the diagnosis of symptomatic carotid stenosis : A meta-analysis. Lancet 367, 1503–1512. doi:10.1016/S0140-6736(06)68650-9
Widder, B., Kleiser, B., Krapf, H., 1994. Course of cerebrovascular reactivity in patients with carotid artery occlusions. Stroke 25, 1963 LP-1967.
Wilterdink, J.L., Feldmann, E., Furie, K.L., Bragoni, M., Benavides, J.G., 1997. Transcranial Doppler Ultrasound Battery Reliably Identifies Severe Internal Carotid Artery Stenosis. Stroke 28, 133 LP-136.
Yodh, A.G., Boas, D. a, 2003. Functional imaging with diffusing light. Biomed. Photonics Handb. 1–45.
Zhang, Y., Wang, L., Yang, J., Yan, R., Zhang, J., Sang, L., Li, P., Liu, H., Qiu, M., 2017. Abnormal Functional Networks in Resting-State of the Sub-cortical Chronic Stroke Patients with Hemiplegia. Brain Res. doi:10.1016/j.brainres.2017.02.012
Zhu, G., Yuan, Q., Yang, J., Yeo, J.H., 2015. The role of the circle of Willis in internal carotid artery stenosis and anatomical variations: a computational study based on a patient-specific three-dimensional model. Biomed. Eng. Online 14, 107. doi:10.1186/s12938-015-0105-6
Zwiebel, W.J., 1992. Duplex sonography of the cerebral arteries: efficacy, limitations, and indications. Am. J. Roentgenol. 158, 29–36. doi:10.2214/ajr.158.1.1727355
 http://orcid.org/0000-0001-9336-9568
http://orcid.org/0000-0001-9336-9568